Abstract
A lightweight, low-power instrument package to measure, in situ, both (1) the local gaseous environment and (2) the composition and microphysical properties of attendant venusian aerosols is presented. This Aerosol-Sampling Instrument Package (ASIP) would be used to explore cloud chemical and possibly biotic processes on future aerial missions such as multiweek balloon missions and on short-duration (<1 h) probes on Venus and potentially on other cloudy worlds such as Titan, the Ice Giants, and Saturn.
A quadrupole ion-trap mass spectrometer (QITMS; Madzunkov and Nikolić, J Am Soc Mass Spectrom 25:1841–1852, 2014) fed alternately by (1) an aerosol separator that injects only aerosols into a vaporizer and mass spectrometer and (2) the pure aerosol-filtered atmosphere, achieves the compositional measurements. Aerosols vaporized <600°C are measured over atomic mass ranges from 2 to 300 AMU at <0.02 AMU resolution, sufficient to measure trace materials, their isotopic ratios, and potential biogenic materials embedded within H2SO4 aerosols, to better than 20% in <300 s for H2SO4 -relative abundances of 2 × 10−9. An integrated lightweight, compact nephelometer/particle-counter determines the number density and particle sizes of the sampled aerosols.
1. Introduction
With benign Earth-like temperatures and pressures, abundant sunlight, and liquid water (albeit mixed with sulfuric acid), an outstanding issue is whether the clouds of Venus are potentially habitable (e.g., Schulze-Makuch and Irwin, 2002; Schulze-Makuch et al., 2004; Limaye et al., 2018; Seager et al., 2020). The recent detection of unexpected phosphine (PH3) vapor within the venusian cloud deck (Greaves et al., 2020a, 2020b; Mogul et al., 2020a, 2020b)—a disequilibrium species whose formation on Venus is currently unexplained by known chemical processes (Bains et al., 2020)—lends the most powerful evidence of a biogenic habitat to date.
Apart from potential biogenic processes, the clouds of Venus are host to a variety of photochemical and thermochemical processes within the atmosphere involving trace gases such as SO2, H2O, OCS, CO, as well as PH3 and possibly NH3 (Surkov et al., 1973). Within the cloud particles themselves, the chemistry involves gas-phase and solution-phase reactions and aerosol microphysical effects that may influence the composition and albedo of the cloud particles. Understanding both the size distribution and composition of venusian aerosols as a function of altitude/temperature, along with the composition of attendant atmospheric gases, provides essential information on (1) atmospheric chemistry, including halide/H2SO4 solution chemistry and the sulfur cycle, (2) chemical and potentially biogenic processes responsible for the UV absorption previously observed in the clouds, and (3) radiative balance and solar energy deposition within the clouds that likely play key roles in powering the planet's super-rotating zonal winds.
2. Aerosol Vertical Distribution
The number density and size characteristics of particles within the venusian cloud layers were analyzed by Knollenberg and Hunten (1980) from Pioneer probe LCPS data obtained during descent from approximately 64 km to 30 km altitude. Treating the aerosol as a single population of 2 μm diameter particles, the aerosol number density throughout the 46–66 km altitude range was found to be higher than 10 particles cm−3, exceeding 100 particles cm−3 in the 48–51 km and 57–66 km ranges. Throughout the middle and lower cloud regions (47–58 km altitude), the mean particle size was 2 μm, with a significant fraction of particles being >5 μm in size. For the 46–70 km altitude range, the rightmost two columns of Table 1 show the aerosol mass density per kilometer altitude and the integrated aerosol column mass above each altitude level as determined by Knollenberg and Hunten (1980). Over the entire cloud column from 46 to 70 km altitude, the aerosol burden is 22 mg/cm2 (0.22 kg/m2).
Predicted Abundances of HCl/H2SO4 inside H2SO4 Aerosol Particles in Venus
Aerosol structure from 46 to 70 km altitude. Rightmost two columns show aerosol mass density and overlying aerosol burden as calculated from Knollenberg and Hunten (1980). HCl/H2SO4 abundances predicted inside H2SO4 particles (fifth column). H2SO4 acid strength (concentration; second column) varies from 73% to 99% as local temperature varies from 216 to 374 K, resulting in a 7-orders-of-magnitude variation in HCl/H2SO4
3. Cloud Composition: Known and Unknown
The cloud particles found in the ∼40–70 km altitude range consist predominantly of liquid droplets containing solutions of sulfuric acid, H2SO4, in water at strengths between 70% and 100% by weight (James et al., 1997; Krasnopolsky, 2015), decreasing with increasing altitude. More specifically, as shown in column 2 of Table 1, the acid strength (or acidity) as weight percent decreases from close to 99% at 40 km to ∼97% at 50 km and to below 75% at 66 km. At the 70 km level, near the top of the clouds, the weight percent drops to ∼72% (Imamura and Hashimoto, 2001). We note that these estimates are calculated primarily from index of refraction results obtained from remote sensing polarization data (Hansen and Hovenier, 1974) combined with chemical and microphysical modelling. Direct in situ measurements of the water content of the aerosol particles are needed to (1) validate these estimates of H2SO4 acidity and, together with measurements of the water vapor in the surrounding gaseous environment, (2) observationally constrain the water activity level within the cloud deck, which is currently estimated at <0.02 (Zhang et al., 2012), equal to or lower than the driest terrestrial deserts (Kieft, 2003).
Beyond this significant component of H2SO4, the composition of the hazes and clouds within Venus's thick atmosphere is poorly understood. Recent analysis of Venus Express VMC observations of the glory observed reflected by upper-level clouds indicates that additional materials beyond H2SO4 are likely present in the clouds (Markiewicz et al., 2018). Of importance is the determination of the composition of the mysterious UV-absorbing layer found covering extensive areas of Venus (e.g., Esposito et al., 1983). Currently, due principally to its patchiness, the prime suspect is a UV-absorbing aerosol, perhaps composed of many components.
Using ground-based spectroscopic measurements as a primary constraint, investigators have proposed several potential UV-absorbing (0.2–0.4 μm wavelength) constituents in the clouds. Candidate absorbers include (1) SO2 gas (Barker et al., 1975); (2) stand-alone elemental sulfur particles, perhaps S8 together with small admixtures of the metastable allotropes S4 and S3 generated from OCS photochemistry at 55–60 km (Toon et al., 1982); (3) croconic acid C5O5H2 (Hartley et al., 1989); and (4) iron chloride, FeCl3 (Zasova et al., 1981; Krasnopolsky, 2017), either attached to or embedded within sulfuric acid droplets, perhaps as condensation nuclei. FeCl3 likely reacts with H2SO4 to form colorless ferric sulfate in about a week (Zasova et al., 1981); thus, a way is needed to continuously supply the 1% FeCl3 concentration required to produce the UV-absorbing features with lifetimes exceeding many weeks. While this appears difficult to accomplish for the stable >60 km level traditionally thought for the UV-absorbing aerosol layer, it may be achievable near the cloud base at ∼48 km altitude (Krasnopolsky, 2017).
Additional information on cloud composition was provided by the two VeGa landers, using a spectroscopic X-ray technique that induced fluorescent spectra indicative of their elemental composition and mass (Andreichikov et al., 1987). From 61 to 47 km altitude, the mass loading in each of three cloud layers was determined: Layer 1 (61–56.5 km) 1 mg/m3; Layer 2 (56.5–50.5 km) 7 mg/m3; and Layer 3 (50.5–47.5 km) 50 mg/m3. Above 50 km, these values are one-third to one-half of the Knollenberg and Hunten (1980) aerosol density profile shown in Table 1, column 7, likely due to evaporation loss within relatively clear sublayers in Layers 1 and 2 (Andreichikov et al., 1987).
The elemental composition of phosphorus, sulfur, and chlorine, with adjacent atomic numbers in the periodic table and adjoining fluorescent X-ray peaks, was determined. While Layers 1 and 2 contained a mixture of sulfur and chlorine containing aerosols, the lower and denser Layer 3 also contained a significant amount of phosphorus. However, since the fluorescent energy peaks of phosphorus and sulfur are only 0.30 keV apart, the ability to differentiate between them is questionable and not clearly described in the work of Andreichikov et al. (1987).
Additional evidence of cloud-level phosphorus comes from the recent identification of PH3 gas at the 56 km level (Greaves et al., 2020a, 2020b). Photochemical and radical chemistry processes then should produce a host of oxygenated phosphorus compounds, including phosphorus acids. As depicted in Table 2, these form as either liquids or solids throughout the cloud, but not as gases; thus, they cannot be measured readily via remote-sensing techniques.
Potential Phosphorus Constituents in the Clouds of Venus
While PH3 is insoluble in H2SO4 droplets, its interaction with the surface of H2SO4 droplets would likely induce the formation of phosphorous and phosphoric acids (H3PO3 and H3PO4, respectively). More complex species, such as pyrophosphoric acid (H4P2O7) may also be created. In so doing, sulfuric acid droplets may act as significant sinks for PH3 gas, accentuating the short lifetime (<1 Earth day; Bains et al., 2020) predicted from radical chemistry and photochemistry in gaseous portions of the atmosphere.
Given such a short lifetime, phosphine's observed existence at the ∼1–5 ppb level near the 56 km level (Encrenaz et al., 2020; Greaves et al., 2020a, 2020b) requires a source of production that is currently unknown. If microbes are present in the cloud layer, then they may act as cloud condensation nuclei for sulfuric acid vapor in the upper cloud layer (Bullock, 2018), an idea that has recently been expanded upon by Seager et al. (2020) to hypothesize a life cycle for venusian microbes. Since the H2SO4 acidity decreases with altitude (cf. Table 1), the H2SO4 water content increases, allowing the microbes to hydrate, germinate, and thrive inside H2SO4 droplets at higher altitudes. However, coagulation, sedimentation, and periodic atmospheric downdrafts force H2SO4 droplets to descend and sublimate, causing the microbes to desiccate and transform into small spores in the hotter atmosphere just below the H2SO4 cloud. Sometime later, riding convective updrafts or gravity or planetary waves, some of the spores are lifted back up into the cloud to become cloud condensation nuclei for H2SO4 vapor and to thrive again, completing the cycle. Thus, the Seager et al. (2020) hypothesis predicts that sizes and local densities of microbes should vary markedly with altitude within the H2SO4 cloud.
4. Expected Sulfuric Acid Cloud Chemistry
Several gaseous components of the venusian atmosphere have significant solubilities in sulfuric acid solutions, most notably the halides HCl, HF, and HBr (Sill, 1975; Krasnopolsky and Belyaev, 2017). The high solubilities allow gases to diffuse into the droplets, dissolve into solution, and form new chemical species (Baines and Delitsky, 2013; Delitsky and Baines, 2015, 2018), thus potentially producing a panoply of constituents within sulfuric acid droplets. The capture rate (known as “uptake”) and concentrations of these halides in H2SO4 droplets depend on the partial pressure of the constituent, acid strength, temperature, aerosol size, and other factors. In particular, as reported by Robinson et al. (1998), as the acidity of H2SO4 increases, the solubility of HCl decreases because more acidic H2SO4 reduces the dissociation of HCl into H+ and Cl-.
As discovered by Robinson et al. (1998) and confirmed by Davidovitz et al. (2011), at acid strengths above ∼69% the uptake of HCl increases by approximately a factor of 5 over that expected from simple uptake with no chemistry. This is because within the droplet HCl reacts with H2SO4 to form chlorosulfonic acid, HSO3Cl (aka ClSO3H; Robinson et al., 1998; Behr et al., 2001, 2009; Nathanson, 2004). Similarly, HF and HBr reacting with H2SO4 produce fluorosulfonic acid, FSO3H, and bromosulfonic acid, BrSO3H. In turn, these species may produce significant amounts of associated daughter products (e.g., SOCl2, SO2Cl2, SOF2, and SO2F2; Delitsky and Baines, 2015).
The initial calculations of Delitsky and Baines (2018) estimated the expected equilibrium abundance of HCl within sulfuric acid droplets for any altitude within the venusian cloud layer. Their estimates were based on extrapolating an array of measurements of the HCl equilibrium abundance for acid strengths of 39% to 69% obtained by Robinson et al. (1998). This calculation was done to calculate simple uptake as the lower limit and omitted secondary chemistry that converts HCl into chlorosulfonic acid at larger acid strengths. As such, the estimated abundance for a typical Venus balloon altitude of 55 km gives a useful conservative target for the desired measurement capability of an in situ aerosol composition experiment.
Table 1 shows results for the HCl/H2SO4 abundance throughout the venusian haze/cloud, from 70 km down to 50 km altitude. Due to the considerable variation in acid strength (or concentration; column 2, from James et al. [1997] for <60 km altitude and Imamura and Hashimoto [2001] for >60 km) and temperature (column 3), the HCl/H2SO4 abundance varies by 7 orders of magnitude through the cloud, from a high of 87 ppm at 70 km to 0.007 ppb at 50 km (column 5). At 55 km, the predicted abundance is 2 ppb (bold value in column 5). To measure this to ±20%, in situ measurements must be able to measure to ±0.4 ppb (Table 1, column 6, bold value).
5. Measurement Requirements for an In Situ Aerosol Composition Instrument
From the previous discussion, the requirements for an in situ aerosol instrument include the ability to measure mass ranges from 2 to 300 AMU at <0.02 AMU resolution to, for example, measure the component of iron chloride (FeCl3, 158 AMU; Krasnopolsky, 2017) and potential materials that could be the result of biotic processes occurring within sulfuric acid aerosols. Another requirement, based on the expected saturated equilibrium concentration of HCl in H2SO4 aerosols near the 55 km altitude level (Delitsky and Baines, 2018) is to measure HCl/H2SO4 with a mixing ratio of 2 × 10−9 to better than 20% in less than 300 s. Drawing on the experience with the VeGa 1 and 2 X-ray instruments, a required capability is to measure the composition locally and instantaneously within the cloud rather than having to derive it from the accumulation rate on a filter susceptible to particle evaporation, as suspected occurred on the VeGa missions.
6. Instrument Design
The Aerosol-Sampling Instrument Package (ASIP) currently under development at the Jet Propulsion Laboratory/Caltech consists of two primary components: the aerosol mass spectrometer (AMS, Fig. 1) and the nephelometer (LONSCAPE, Fig. 2), which is a separate device in-line with the AMS particle stream about midway through the AMS component (cf. Fig. 1). These two components are in an integrated pathway so that the same particles are measured by both components. The heart of the AMS is the quadrupole ion-trap mass spectrometer (QITMS; Madzunkov and Nikolić, 2014; Avice et al., 2019). The basic scheme of the AMS is depicted in Fig. 1. The instrument design uses an inlet aerodynamic lens (Schreiner et al., 1999; Cziczo et al. 2004) as an aerosol separator together with an adjustable piezoelectric aperture that allows only aerosols of a selectable size range within a broad range of 0.3–12.0 μm diameter into the QITMS. Upon entry into the instrument, the atmospheric flow is sequentially expanded through orifices from high-pressure segments of the aerodynamic lens into progressively lower pressure segments.
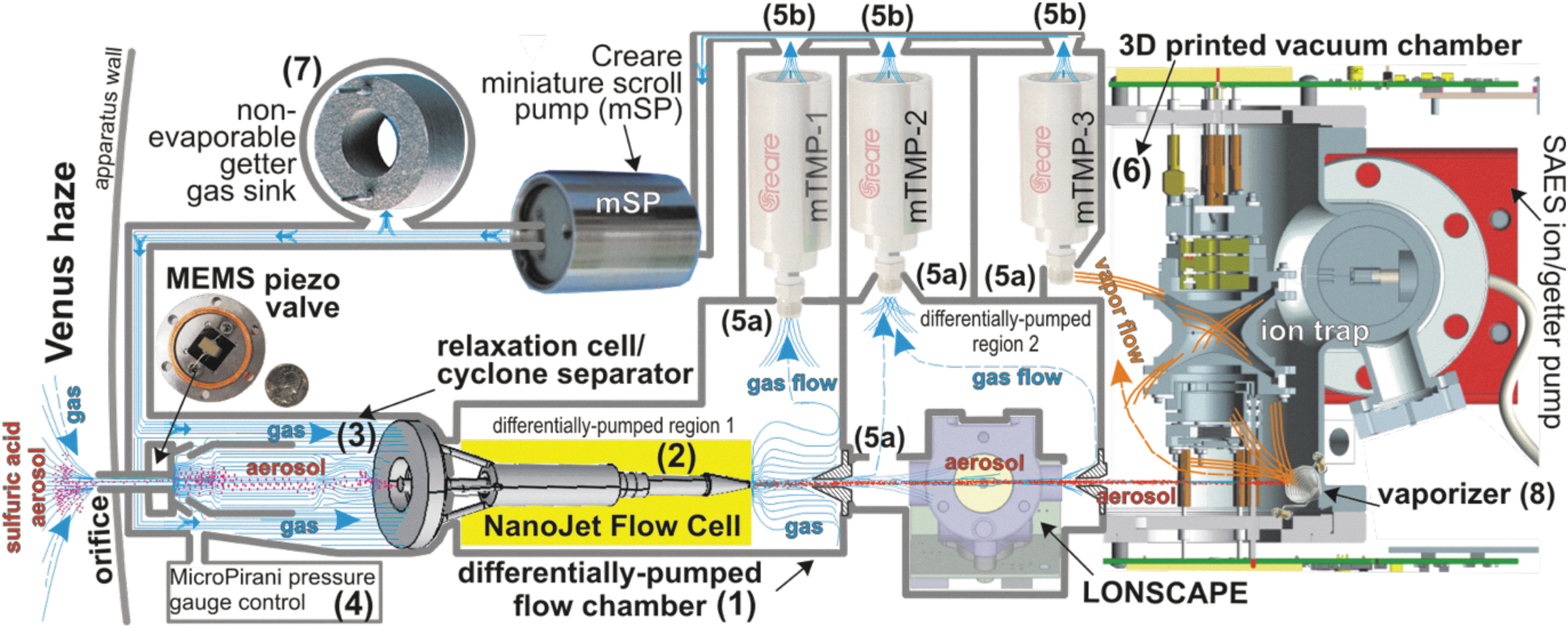
Schematic diagram of the aerosol mass spectrometer (AMS) component of the Aerosol-Sampling Instrument Package (ASIP) concept. The aerosol sample inlet is enclosed in a differentially pumped gas flow chamber (1) with two regions. Blue color streamlines represent the venusian gas flow, whereas red dots are suspended sulfuric acid aerosol particles. The NanoJet Flow Cell (2) is an aerodynamic lens (aerosol separator) being developed by Integrated Deposition Solutions Inc. (IDS). Specifically, under development are the relaxation cell with the cyclone separator (3), MicroPirani pressure gauge (4), internal (5a) and external (5b) vacuum ports, the noncorrosive 3D-printed vacuum chamber (6) with the alumina-coated tungsten flash vaporizer (8), and the St172 sintered porous getter (7), which serves as a static gas sink. Other components are the variable aperture JPL MEMS piezo valve; flight-grade Creare miniature turbo-molecular pumps (mTMP1–3), and the JPL quadrupole ion-trap mass spectrometer (QITMS) with supporting electronics. The leak-tight LONSCAPE optical particle counter and sizer currently being built by CNES is shown just downstream of the NanoJet Flow Cell and upstream of the vaporizer.
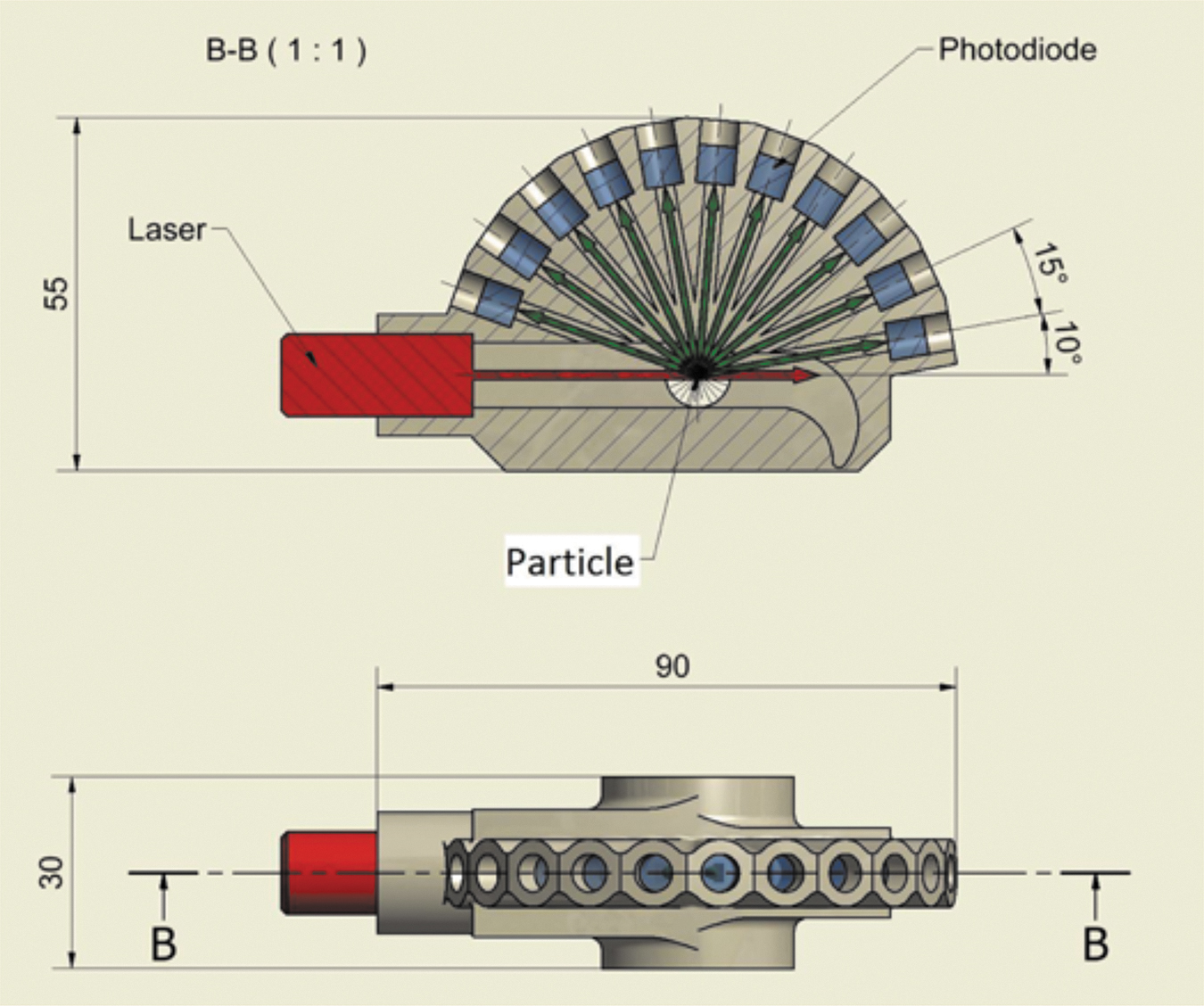
LONSCAPE nephelometer design (adopted from Renard et al., 2020). In the top panel, the aerosol/gas flow direction is into the page. Upon exiting LONSCAPE, aerosol/gas enters the vaporizer. Dimensions are shown in millimeters.
In each segment, gases are expanded away, while the much heavier aerosols remain collimated on the jet axis. Upon exiting the aerosol separator, the jet of aerosols mixed with the rarified gas passes through the LONSCAPE nephelometer and into a vaporizer that operates in two modes—one at 600°C and an energy-saving 300°C mode—converting the particles into their constituent gases which then flow into the QITMS and are analyzed to determine aerosol composition.
Notably, the elimination of CO2 and other gas species provided by the aerosol separator increases the trace aerosol species concentration by about 5 orders of magnitude. Thus, trace aerosol species with concentrations relative to the expected dominant H2SO4 component of 100 and 2 ppb (corresponding to about 1.0 and 0.02 ppt relative to the ambient atmospheric CO2) can be measured to 20% precision in <6 s and <5 min, respectively. Beyond pertaining to inorganic materials, such precisions would be attained as well for species of biogenic interest such as hydrocarbons, phosphorus compounds, and the isotopic ratios of carbon, nitrogen, sulfur, oxygen and hydrogen (i.e., the D/H ratio) found within both aerosol particles and their attendant gases. In particular, water and its isotopes can be measured to 5 ppb accuracy on a 1 min timescale, thus providing crucial information on H2SO4 acidity and water activity.
Three modes are used that measure (1) the pure aerosol as injected from the aerosol separator as described above, (2) gaseous constituents and their isotopes as measured from an air flow path that filters the air of aerosols, bypassing the aerosol separator, and (3) noble gases and their isotopes also using filtered and aerosol-separator-bypassed air. Comparison of the isotopic ratios of the aerosol versus atmospheric constituents may elucidate fractionation effects due to complex geochemical, organic, or biogenic processes. In the noble gas mode, our analysis shows that in 300 s 126Xe/130Xe can be determined to 12%, assuming 126Xe and 130Xe abundances of 0.006 and 0.29 ppb, while the isotopic ratio 80Kr/84Kr can be measured to 5%, assuming 3.6 and 24 ppb for 80Kr and 84Kr, respectively. These are more than adequate to resolve various hypotheses of Venus's origin and evolution, particularly regarding commonalities and differences with the origins and evolution of Earth and Mars (e.g., Baines et al., 2013).
The AMS component of the instrument package, including electronics, has an estimated mass of 9 kg and fits into a volume of 30 × 20 × 20 cm. The QITMS, including the vacuum chamber and vaporizer, fit into 20 × 20 × 10 cm; the nanojet aerosol separator uses 20 × 10 × 10 cm, and the electronics box occupies 30 × 10 × 10 cm.
To count the number of particles entering the QITMS and to characterize the particle size distribution, a lightweight, compact nephelometer/particle-counter device is installed after the aerosol separator. The design, known as LONSCAPE (Light Optical Nephelometer Sizer and Counter for Aerosols in Planetary Atmospheres; Renard et al., 2020), shown in Fig. 2, is evolved from designs previously flown on terrestrial balloons (Renard et al., 2016a, 2016b).
Via a red or infrared laser and a ring of 11 photodiodes, particle phase functions are measured at 11 angles, every 15 degrees from 20 to 170 degrees phase angle (10 to 160 degrees scattering angle). This allows (1) particle counting, (2) particle size determinations for 20 size classes in the 0.2–50 μm size range, (3) characterization of bulk particle composition and type (refractory vs. condensates, spherical vs. fractal) and (4) a measure of the particle albedo.
Such particle data, complementing the compositional information, enables ASIP to more comprehensively assess aerosol particles, including their potentially biological nature. For example, the altitudinal variability in microbe size predicted by the Seager et al. (2020) life cycle hypothesis would be testable via instrument measurements of aerosol size and composition. Such comprehensive instrument capabilities for detecting and characterizing biological materials have been given high priority for Venus exploration (e.g., Limaye et al., 2018, 2021; Gilmore et al., 2020).
As indicated in Fig. 2, LONSCAPE fits into a volume of less than 12 × 6 × 3 cm, which fits into the 30 × 20 × 20 cm instrument enclosure mentioned above. The entire ASIP is <11 kg, with the QITMS and ASIP electronics using 7 kg, the aerosol separator weighing 2 kg, and LONSCAPE using 1.5 kg. LONSCAPE uses less than 5 W; the QITMS and aerosol separator respectively use 30 and 17 W. With the vaporizer at 600°C, ASIP power is 71 W, decreasing to 59 W in 300°C vaporizer mode.
The Aerosol-Sampling Instrument Package has a flexible data volume collection rate. The AMS typically collects 5 min sample sets, with each set composed of three hundred 1 s measurements of the full mass spectrum that are co-added over 5 s intervals to provide 60 measurements per sample set. For an aerobot-based ∼100 day flight in the venusian clouds, a sample set would typically be acquired each hour on the solar-celled powered daylit hemisphere. On the nightside hemisphere, where rather limited battery power must be used, a sample set may only be acquired every ∼8 h.
The AMS inherently acquires 128 kbits (i.e., 16,000 AMU channels times 8 bits per channel) in obtaining a 1 s spectrum (which itself is the result of 20 spectra obtained every 0.05 s). Co-added over 5 s intervals during the 5 min sample set, 7.68 Mbits/h are acquired during dayside observations. Without co-adding, LONSCAPE acquires 90–240 kbytes during such 5 min observation periods. Thus, altogether ASIP typically collects about 8 Mbits/h, or 192 Mbits/day on the dayside and ∼24 Mbits/day at night. In daylight, ASIP data are uplinked during a ∼4 h satellite pass. Nightside data remains on board and then are uplinked once solar power becomes available.
On short-lived probe missions, continuous operations would be expected, resulting in 96 Mbits/h. Uplink to an orbiter/carrier spacecraft would be continuous during this time, resulting in 27.6 kbits/s uplink rate. In all of these scenarios, compression techniques can be used to reduce uplinked data volumes by a factor of 5–8.
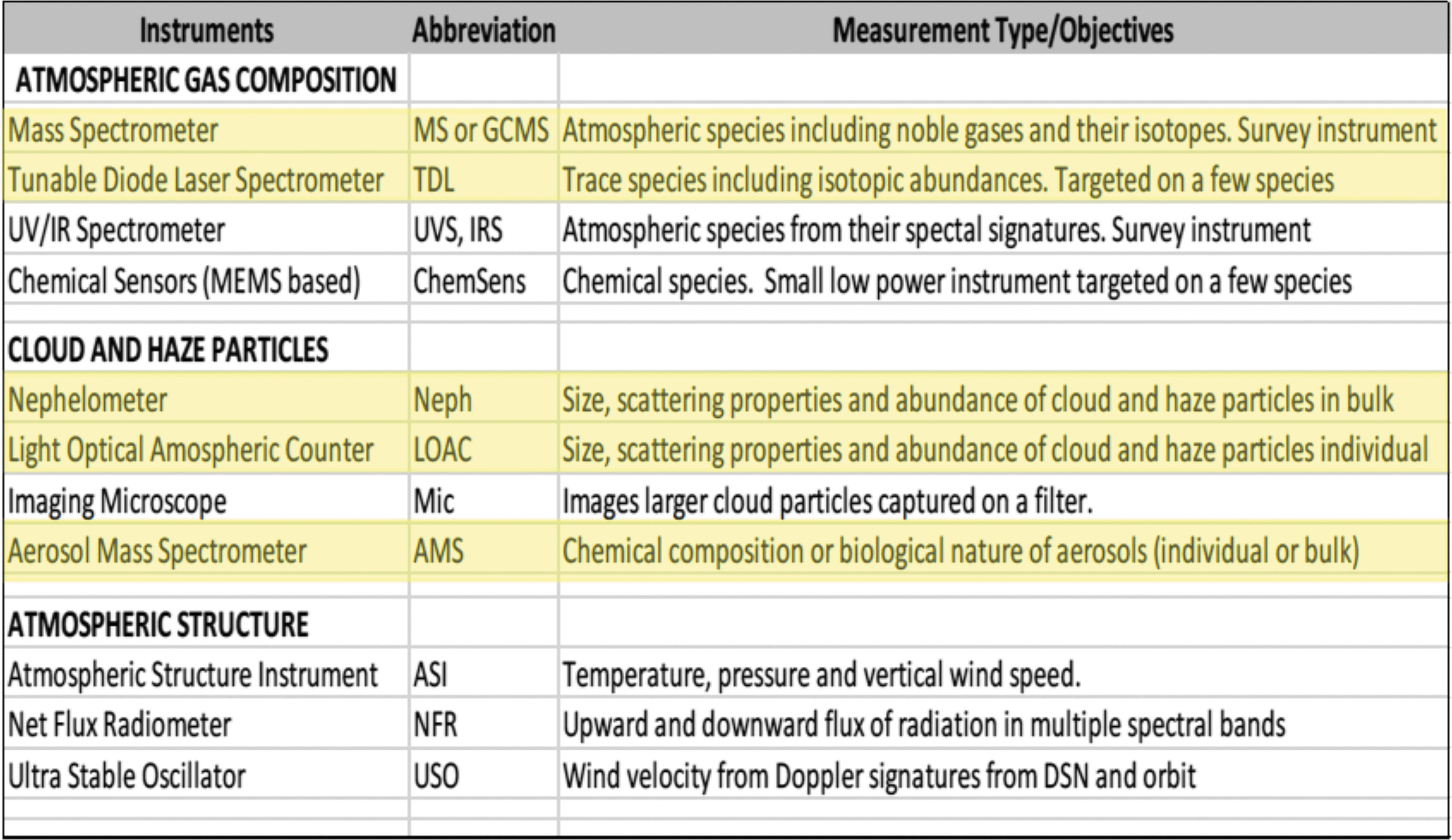
The Aerosol-Sampling Instrument Package satisfies five of the eleven instruments called for by Cutts et al. (Venus Aerial Platforms Study Team, 2018) in their analysis of most important in situ platform atmospheric instruments, as depicted in Table 3 (from the Venus Aerial Platforms Study Team, 2018). As such, ASIP provides a significant reduction in payload resource requirements. Indeed, due to its large science return at minimal resource cost, this instrument was selected as a key component of the aerobot payload proposed for the Venus Flagship Mission (Gilmore et al., 2020).
In Situ Platform Atmospheric Instruments and Measurement Objectives
7. Conclusion
The Aerosol-Sampling Instrument Package provides a comprehensive means to directly measure, in situ, the microphysical and compositional properties of atmospheric aerosols in planetary atmospheres. For Venus, such an instrument would determine the trace composition of both aerosols and the attendant atmosphere to the parts-per-billion level within 5 min. Such an instrument placed on a long-duration (several months) variable-altitude balloon repeatedly traversing the relatively benign 52–62 km altitude range would provide vertical profiles of aerosol and gaseous atmospheric constituents over a large range of latitudes and times of day, providing new insights into the planet's chemistry, radiative balance, and dynamics. As well, a comparison of the composition—including the isotopic abundances—of aerosols with the attendant gases and with other aerosols sampled during the mission could provide key data on the detection and characterization of any potentially biogenic materials and processes.
Footnotes
Acknowledgments
A portion of this research was carried out at the Jet Propulsion Laboratory, California Institute of Technology, under a contract with the National Aeronautics and Space Administration. J.-B.R., O.M., and N.V. acknowledge support from CNES. © 2020. All rights reserved.