Abstract
In the subsurface, the interplay between microbial communities and the surrounding mineral substrate, potentially used as an energy source, results in different mineralized structures. The molecular composition of such structures can record and preserve information about the metabolic pathways that have produced them. To characterize the molecular composition of the subsurface biosphere, we have analyzed some core samples by time-of-flight secondary ion mass spectrometry (ToF-SIMS) that were collected in the borehole BH8 during the operations of the Mars Analog and Technology Experiment (MARTE) project. The molecular analysis at a micron-scale mapped the occurrence of several inorganic complexes bearing PO3 -, SO x(2 to 4) -, NO x(2,3) -, FeO x(1,2) - , SiO2 -, and Cl-. Their distribution correlates with organic molecules that were tentatively assigned to saturated and monounsaturated fatty acids, polyunsaturated fatty acids, saccharides, phospholipids, sphingolipids, and potential peptide fragments. SO x - appear to be mineralizing some microstructures larger than 25 microns, which have branched morphologies, and that source SO3-bearing adducts. PO3-rich compounds occur in two different groups of microstructures which size, morphology, and composition are different. While a group of >40-micron sized circular micronodules lacks organic compounds, an ovoidal microstructure is associated with m/z of other lipids. The NO2 -/NO3 - and Cl- ions occur as small microstructure clusters (<20 microns), but their distribution is dissimilar to the mineralized microstructures bearing PO3 -, and SO3 -. However, they have a higher density in areas with more significant enrichment in iron oxides that are traced by different Fe-bearing anions like FeO2 -. The distribution of the organic and inorganic negative ions, which we suggest, resulted from the preservation of at least three microbial consortia (PO4 --, and NO2 --/NO3 --mineralizers PO4-lipid bearing microstructures), would have resulted from different metabolic and preservation pathways.
1. Introduction
The emergence of the terrestrial biosphere was propelled by the preexistence of geochemical cycles that likely were the original core of nascent metabolic pathways (Nealson and Rye, 2014). They probably involved early stages of energy transduction in prebiotic processes, which were inherited by the oldest microbial communities on Earth. Such processes have tied the evolving microbial communities to several different geochemical cycles occurring on Earth for more than 3.5 billion years, which have induced a strong imprint on Earth's mineral composition (Hazen et al., 2008; Hazen 2012). This is indeed observed in the underground communities where energy as well as CO2 and N2 fixation comes from the interplay with the mineral realm. From such interplay, subsurface microbial life not only obtains energy by uptaking inorganic compounds for its development but also has a strong impact on the geochemical composition of Earth's crust (Brantley et al., 2012; Lyons et al., 2014). If the geosphere has recorded unequivocal evidence of ancient biological activity, the geobiologist can reconstruct the ancient metabolic pathways through one of the fundamental basic pillars of geology which is the actualism. For such an approach, the researcher has a wide set of analytic instruments available whose use is essential to understand how the preservation processes have operated and which metabolic activity was involved in the alteration of Earth's crust.
In this paper, we show the geobiological record of ancient microbial metabolisms that have been involved in a complex interplay with the sulfide-rich Río Tinto Mars analog basement over the last 25 million years (Essalhi et al., 2011). Although this has been the main aim in several other papers (Fernández-Remolar et al., 2008, 2018), our recent research provides novel data obtained from the sample analysis by using state-of-the-art ToF-SIMS (time-of-flight secondary ion mass spectrometer) technology. It has also been supported by conventional imaging and geochemical techniques, which were the primary source of data in previous refereed works. As ToF-SIMS provides the compositional mapping of biological and nonbiological compounds at the microscale, it is possible to ascertain a direct relation between fabric and microstructures with biotic and nonbiotic by-products (Fernández-Remolar et al., 2015). As a result, we can trace direct association between microstructures and different inorganic (e.g., NO2 -/NO3 -, PO4 -, HS-, SO4 -) and biomolecular ions supporting a biological origin, in situ. Indeed, it is possible with our techniques to recognize a specific group of structures that are strictly associated with the mineralization of N-bearing anions, which provide essential information about the nitrogen cycle in the subsurface.
The results are relevant to a much broader framework, where the same techniques and procedures can be applied to understand how the different metabolic paths sustaining biogeochemical cycles have been operating in the subsurface on the long-term temporal scale, as well as how they could potentially be identified on other planetary bodies to demonstrate the emergence of non-terrestrial biospheres in the Solar System. This will come together with the careful analysis of materials from sample return missions, which will play an essential role in understanding whether life has arisen in other regions of the Universe.
2. Geological Settings
The geological record of ancient metabolism operating in the subsurface has been sampled through drilling the Río Tinto basement in the Peña de Hierro area (Fig. 1A). It is located in the Iberian Pyrite Belt (IPB), a 250 km long and 60 km wide unit of volcanogenic massive sulfide deposits that belong to the South Portuguese Zone, the southernmost terrane of the Variscan Belt of Iberia (Fig. 1B). Several geological units comprise the IPB; however, except for its late- to post-Variscan intrusive rocks and continental intermontane deposits, three different units define its general lithostratigraphy: the Phyllite-Quartzite (PQ) unit at the bottom, the intermediate Volcanic Sedimentary Complex (VSC) unit with massive and stockwork sulfides, and the Culm Group at the top. In the Río Tinto area, the sulfide bio-oxidation has led to the formation of a gossan reaching up to 10 m depth which formation has occurred in three different alteration episodes averaging 25, 7, and 2 Ma (Essalhi et al., 2011; Velasco et al., 2013).
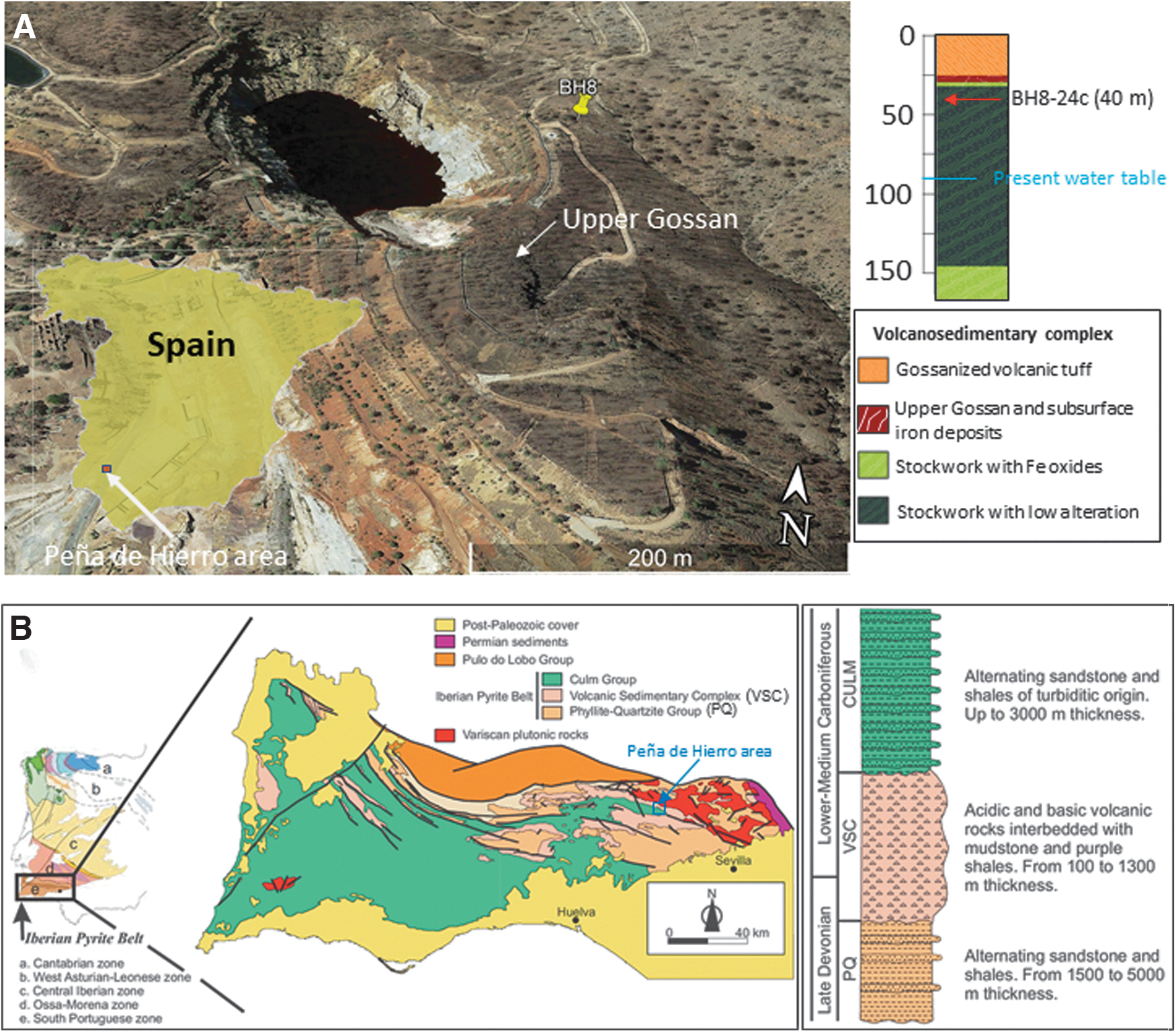
Composite image showing (
During the last 20 years, extensive exploration of the geological, geophysical, and microbiological conditions in the Río Tinto area, especially in Peña de Hierro, has provided a huge amount of data about both the surface and subsurface geology of the area (Fernández-Remolar et al., 2005, 2008; Gómez-Ortiz et al., 2014). The data comprises geophysical information and several boreholes (BH1, BH4, BH8, BH10, and BH11) ranging from 59 to 610 m depth, which has provided lithological information about the subsurface structure around the Peña de Hierro orebody (Fernández-Remolar et al., 2018).
Sample BH8-24c was collected at a depth of around 40 m along a 165 m deep borehole collared 50 m east of Peña de Hierro pit lake (Fig. 1A). It occurs north of the San Miguel-Minas del Castillo Shear Zone, mainly composed of shales, acidic tuffs, and volcanic materials (Fig. 1A). The upper unit is composed of 25.5 m thick orange acidic tuffs (Fig. 1A) affected by low-grade metamorphism. The acidic tuff unit is followed by an intermediate unit ranging from a 25.5 to 31 m thickness consisting of a gossan deposit with chert. In this zone, the characteristics of the gossan do not correspond to the oxidization of massive sulfides but hematite and goethite-rich gossanized tuffs. The mineralogical distribution of the gossan (Fernández-Remolar et al., 2008) agrees with the general vertical gossan profile previously defined. The upper part of the gossan is dominated by hematite and quartz with minor amounts of goethite. In contrast, at the bottom, the goethite is a dominant mineral together with quartz, where hematite is secondary.
The lower unit consists of a pyritic stockwork extending from 31 m depth to the bottom of the borehole. It is mainly composed of quartz and pyrite with lower contents of barite, feldspars, chlorite, mica, and siderite (Fernández-Remolar et al., 2008, 2018). The upper part (∼12 m), where sample BH8-24c was collected, shows iron oxides replacing pyrite associated with quartz veins. The rest of the unit is an unaltered stockwork excepting two slightly oxidized horizons extending from 66 to 92 m depth and 103.5 to 109 m depth. At the borehole bottom (151 to 166 m depth), the stockwork is affected by small (millimeter- to centimeter-sized) fractures filled by secondary sulfates, iron oxides, and chlorite.
3. Methods
3.1. Sample collection and preparation
As discussed above, core samples were collected at a depth of 40 m during the Mars Analog and Technology Experiment (MARTE) project drilling operations that were arranged to research the traces of the microbial communities living in the IPB basement. The procedures for sampling and preparing underground materials are described in detail elsewhere (Fernández-Remolar et al., 2008, 2018; Stoker et al., 2008) and will not be described in detail in this paper. In short, subsurface sample collection was performed with a Boart-Longyear (Salt Lake City, UT, USA) HQ wireline system that produced 60 mm diameter cores within a plastic liner. A chemical tracer (NaBr) was added into the drilling fluid to identify possible sample contamination during the drilling process. Core providing sample BH8-24c had no bromide traces in the interior, which evidences that it was not contaminated with the drilling fluid. Cores were retrieved in 1 m sections encased in plastic liners. These liners were flushed with N2 gas at the borehole, sealed, and then transported to a field laboratory within 1 h, approximately 6 km from the borehole.
The core was split into several subsamples and cut with a pneumatic rock cutter and a diamond saw blade. Thin sectioning was prepared through cutting and polishing a subsample set by using a 0.3 micron-sized alumina paste for both transmitted light microscope inspection and ToF-SIMS surface analysis. A second subsample set was split into small fragments (<1 cm) to expose fresh material to search for traces of microbial activity under a scanning electron microscope (SEM).
3.2. Light microscope and scanning electron microscope
Samples were scrutinized under both light microscope and SEM to find secondary ferruginous deposits mineralizing microbial structures or other mineral traces that could have resulted from ancient microbial activity. Thin sections were observed under a Leica stereomicroscope and an upright trinocular microscope Motic BA400 coupled to a MoticamPro 282a camera. Thin section examination was complemented with SEM analysis. For such purpose, the rock sample was coated with gold by way of an SC7620 Thermo VG Scientific sputter-coating device. Semiquantitative chemical image microanalysis was done under a JEOL JSM-5600V scanning electron microscope coupled to an Oxford INCA X-sight EDS (energy dispersive X-ray spectroscopy) Microanalysis Probe, which are emplaced in the Center of Astrobiology in Madrid.
3.3. Time-of-flight secondary ionization mass spectrometry analysis
Before analysis, thin sections were cleaned by surface sputtering with a 100 nA 3V oxygen ion beam for 3 s over a square area of 500 × 500 μm. The ToF-SIMS analyses were performed with a ToF-SIMS IV (ION-TOF, Münster, Germany) that was emplaced at the Nanotechnology Platform, Institute for Bioengineering of Catalonia in Barcelona.
Under an operational pressure of 5 × 10−9 mbar, the sample was bombarded with a pulsed bismuth liquid metal ion source (Bi3 ++), at energy of 25 keV. The gun was operated with a 20 ns pulse width, 0.3 pA pulsed ion current for a dosage lower than 5 × 1011 ions/cm2, well below the threshold level of 1 × 1013 ions/cm2 generally accepted for static SIMS conditions.
Secondary ions were detected with a reflector time-of-flight analyzer, a multichannel plate, and a time-to-digital converter. Charge neutralization was achieved with a low-energy (20 eV) electron flood gun. Secondary ion spectra were acquired from randomly rastered surface areas of 500 × 500 μm within the sample's surface. Secondary ions were extracted with 2 kV accelerating voltage and were postaccelerated to 10 keV kinetic energy before hitting the detector. Mass spectral acquisition and image analysis were performed within the ION-TOF Ion Spec and Ion image software (version 7.4).
Spectral analysis and molecular identification were assisted by the software Mmass (Strohalm et al., 2010) and the molecular database Metlin (Smith et al., 2005). Data calibration used different known inorganic anions (e.g., Cl-, SO2 -, PO3 -) that provided an accuracy of <90 ppm, which is sufficient in terms of ToF-SIMS analysis (Sjövall et al., 2008).
4. Results
4.1. Transmitted light microscope and scanning electron microscope
Visual inspection led to selecting a few core samples showing clear evidence of rock alteration and ferric mineralization in millimeter-sized cracks or microcavities left by pyrite dissolution. This is the case for samples BH8-22b, BH8-23c, BH8-24c, and BH8-25c occurring between 34 and 43 m below the surface. They consist of a weathered stockwork composed of cherty mass crosscut by sulfide-rich veins and cracks, which have been attacked, dissolved, and infilled with iron oxides (Fig. 2A, 2B) (Fernández-Remolar et al., 2008). The ferruginous infill consists of glassy iron oxide occurring as a massive cavity infill or several micron-scale thin laminae stacks growing at both crack faces. In some cases, the laminae stacks grow toward the crack or cavity interior to coalesce and form a unique deposit (Fig. 2B). Such laminations contain aggregates of dark particles and <1 micron thickness and up to 100 micron length isolated filaments with straight to curvilinear outline (Fig. 2C, 2D).
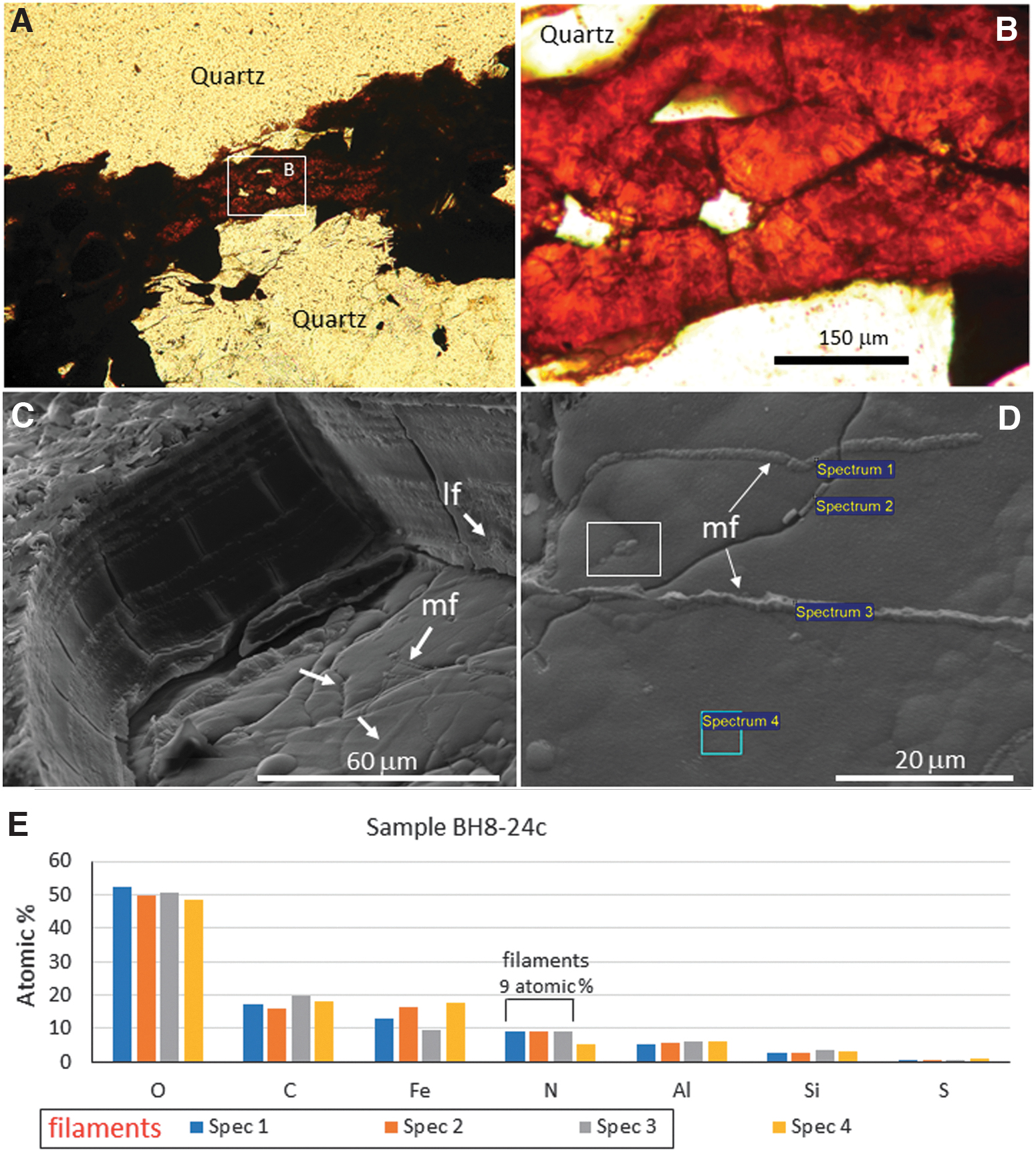
Microscope images showing different aspects of ferruginous laminations infilling the stockwork deposits in sample BH8-24c at a depth of ∼40 m. Panels (
SEM imaging of sample BH8-24c showed an abundance of filament structures that flourished, embedding the upper part of the lamination surface (Fig. 2C) with nanometer-scale mineralized particles (Fig. 2D). A comparative elemental analysis of the filamentous structures and the lamination surface through the energy dispersive X-ray spectroscopy (EDS) microanalysis system show that the N content in filaments (∼9 atomic %) doubles the N concentration in the lamina surface (Fig. 2E). In addition, both filamentous structures and laminae have a high enrichment in C (∼18 atomic %), which concentration is higher than Fe (∼12 atomic %).
The filament and lamination EDS analysis show a secondary concentration in Si and a minor concentration in S averaging 3 and 0.7 atomic %, respectively. The occurrence of C and Fe as major, Si as secondary, and S as minor elements suggests that the mineral matrix of the cracks and microcavity ferric infillings were initially composed of ferric oxysulfates that have experienced dehydration and sulfate loss during diagenesis (Fernández-Remolar et al., 2011). Furthermore, the occurrence of Si as a secondary element suggests that it plays a role in the mineralization and preservation of the biological remains in the Río Tinto materials (Fernández-Remolar and Knoll, 2008).
4.2. Time-of-flight secondary ion mass spectrometry analysis
The ToF-SIMS analysis has performed molecular mapping and spectral analysis of negative ions in sample BH8-24c. The target area was selected to have different structures of potential biological origin found inside the laminated ferruginous deposits that are infilling cracks and microcavities (Fig. 2C, 2D). The molecular mapping of positive and negative masses establishes a direct association between molecular composition and structure. At the same time, the spectral analysis provides additional information about the molecular composition recorded in the sample. The combination of physical properties and composition is essential to ascertain biogenicity in the preserved microstructures (Cady et al., 2003; Westall et al., 2015).
As the ToF-SIMS analysis provides direct information about the relation between the preserved biological structures and the mineral substrate, it is a reliable tool to distinguish mineralization from ancient metabolic activity in the Río Tinto subsurface (Fernández-Remolar et al., 2018).
4.2.1. Image analysis
The ToF-SIMS molecular mapping targeted a 500 × 500 micron sized area with ferruginous laminations occurring as crack fillings (Fig. 3). As a result, the more abundant fragment ions outline three main image patterns (Fig. 4), which are the following

Mosaic of images of sample BH8-24c including a thin section picture
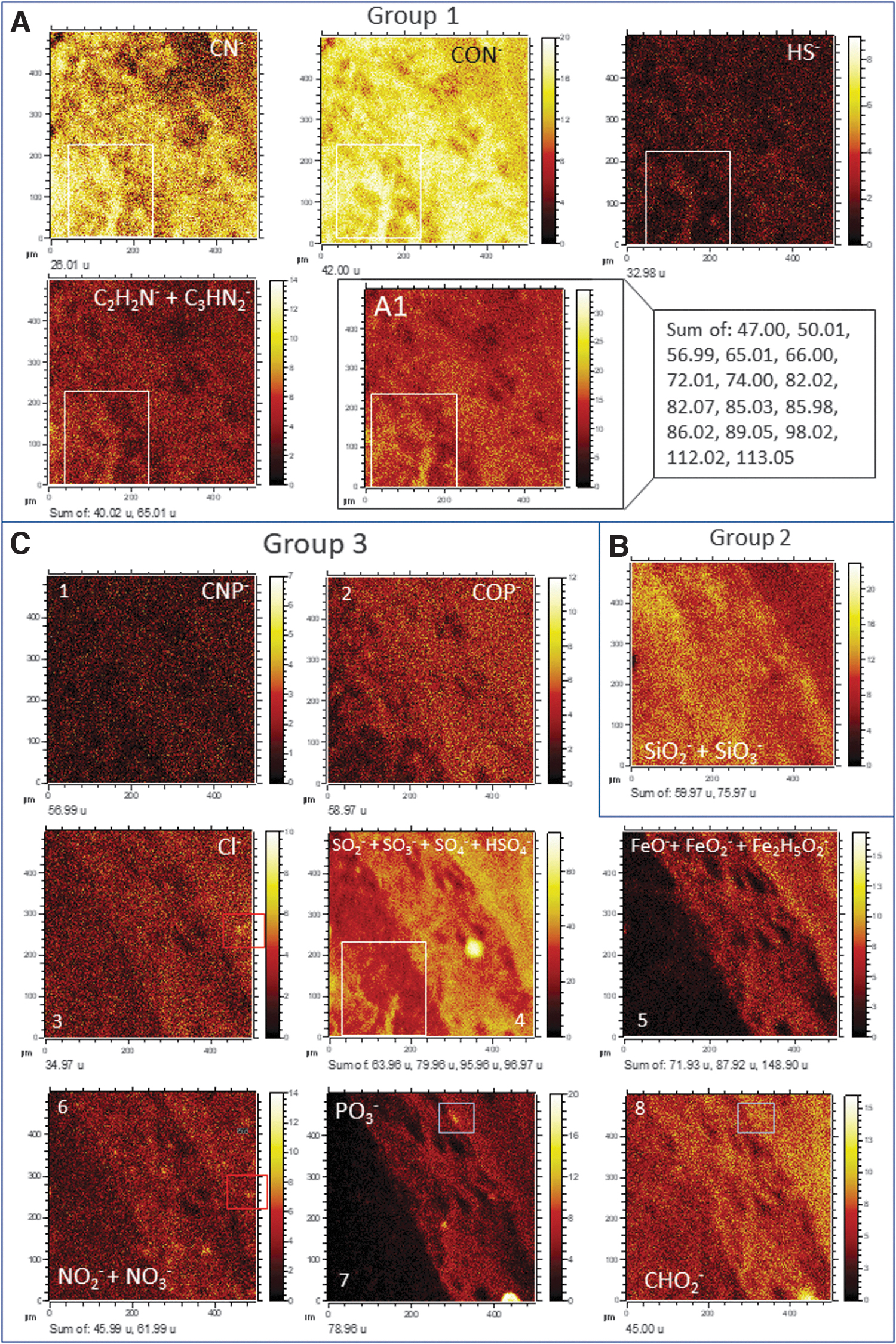
ToF-SIMS mapping of different anions obtained in the target area TA (Fig. 3A, B), which outline can be classified into three different main patterns. (
Mosaic of different ToF-SIMS ion images obtained from target area TA. (
4.2.2. Spectral analysis
The fragmentation pattern of the negative ions found in sample BH8-24c has provided the detection of different major groups of organic compounds and biomolecules, which can eventually be mapped (Figs. 5 and 6)
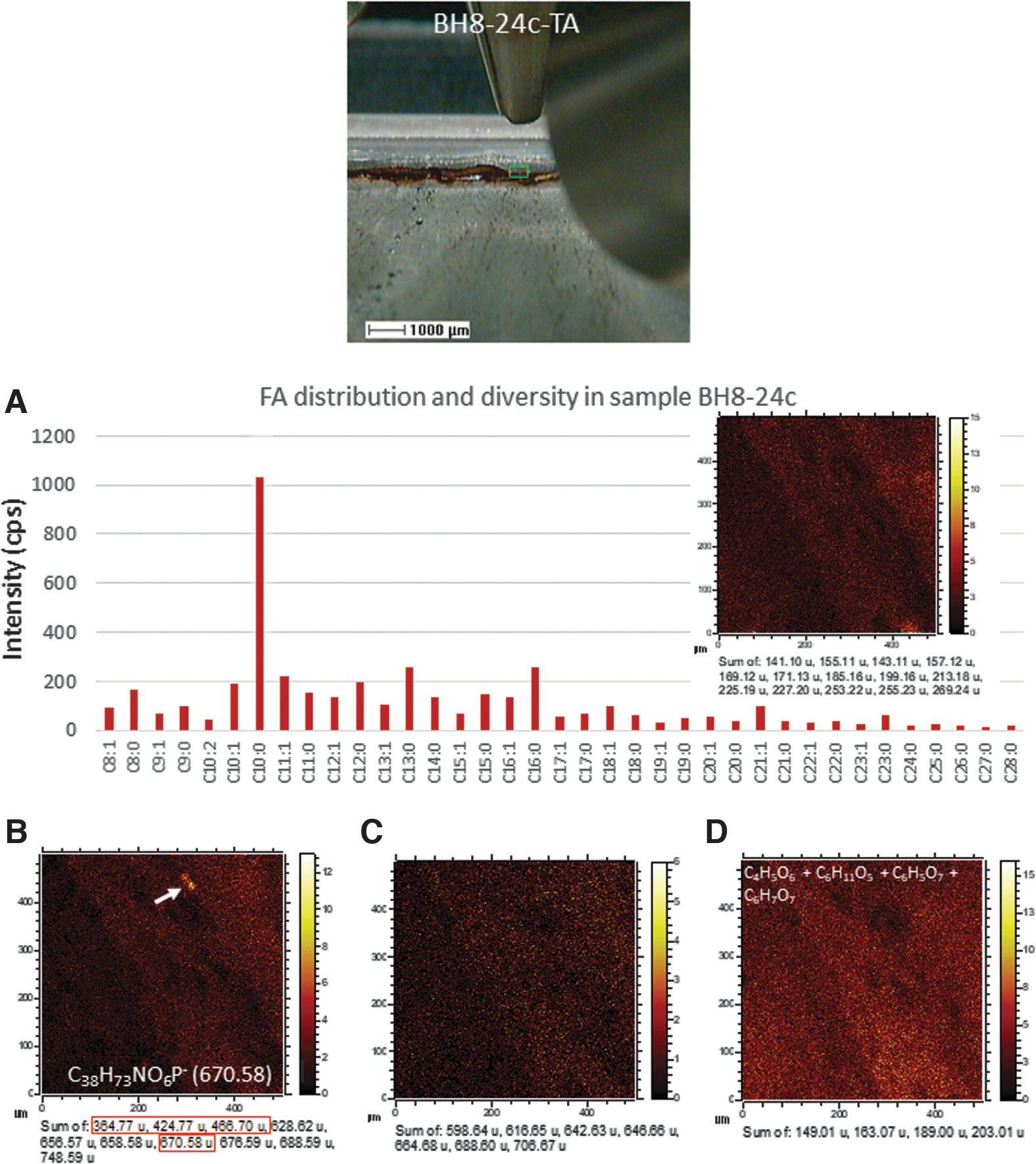
(
The ToF-SIMS mapping in sample BH8-24c shows that the FA distribution fairly outlines the main lamination orientation following the pattern found in Group 3 (Fig. 6), where the intensity decreases as the S- and Fe-bearing inorganic ions do (Fig. 4C). However, FAs are also occurring in areas with higher content in SiO2 - and Si2H4O- that is consistent with preservation with mineralization with silica.
Compounds larger than 600 Da which sum has been defined through the Metlin database (Smith et al., 2005) as C37H71NO6P- (656.57), C37H73NO6P- (658.58), C38H73NO6P- (670.58), C38H79NO6P- (676.59), and C44H79NO6P- (748.59), C40H69NO10P- (754.43), C40H71NO10P- (756.42), and C40H75NO10P- (760.40). Although the compound distribution follows the pattern shown in Group 3 (Fig. 4C), they show a higher intensity outlining the PO4-bearing micronodule (Fig. 6B). Such composition is similar in the mass range to some bacterial phospholipids characterized by ToF-SIMS and other mass spectrometry techniques (Mazzella et al., 2005; Heim et al., 2009; Bühring et al., 2012). The ToF-SIMS mapping of the compounds listed above shows the same distribution as the pattern, followed by a set of unknown anions including 364.77, 424.77, 466.70, 568.65, and 628.62 (Fig. 6B), which could be related to adducts of phosphates. A set of different even negative fragments fitting N-bearing compounds (Fig. 6C; Supplementary Fig. S1; Table S1) that can tentatively be assigned as C42H82NO- (616.63), C42H78NO2
-/C38H79NO3P- (628.62), C42H84NO2
- (634.64), C43H80NO2
- (642.62), C43H84NO2
- (646.64), C43H86NO3
- (664.68), C45H80NO3
-/C42H84NO5
- (682.63), C45H82NO3
- (684.62), C45H84NO3
- (688.62), C46H92NO3
- (706.67), C47H82NO3
- (708.63), and C47H82NO4- (724.64). They could be associated with the formation of [M – H2O – H]- or [M – H]- ions of some ceramides (Olsen and Jantzen, 2001) as shown in the Metlin database (Smith et al., 2005). The ToF-SIMS images resulting from the addition for each m/z (Fig. 6C) show that they distribute following the pattern of Group 3 but show a lower intensity.
The presence of the m/z peak at 657.53 could tentatively be assigned to C42H73O5 - (Supplementary Table S1), which corresponds to a diacylglyceride backbone. It could be sourced from different lipids as simple acylglycerides or phospholipids, if they come together with a polar group (Fahy et al., 2005). Although the peak intensity is low, it agrees with detecting a series of abundant cations separated by 14 Da (e.g., 495.44, 509.45, 523.47, 537.49, and 551.50) that evidences the presence of diacylglycerides in sample BH8-24c.
5. Discussion
The ToF-SIMS analysis of sample BH8-24c has combined molecular mapping and spectral analysis to recognize the mineral substrate that is the preserving matrix for biological information. It also shows the occurrence of several preserved entities that record ancient microbial activity in the form of mineralized structures. In this regard, the distribution of Fe-, S-, and Si-bearing inorganic anions, when using ToF-SIMS mapping capabilities, shows that there are three compositional areas with a characteristic mineral composition following the lamination fabric. Going from the right to the left side in the target area, such layered areas are the following (Fig. 7A): (1) a ferric and sulfate-rich layer depleted in silica, (2) an iron- and sulfate-rich intermixing intermediate layer intermixing with silica, and (3) a silica-rich layer with sulfate compounds and low ferric concentration. The co-occurrence of iron and sulfate anions is consistent with a mineral matrix formed by the precipitation of ferric oxysulfates. They precipitate out from acidic solutions (pH <3) through the microbial attack on the subsurface orebody, which releases high amounts of SO4 -, Fe3+, and H+ (Gómez-Ortiz et al., 2014; Fernández-Remolar et al., 2018). As acidic conditions usually inhibit silica precipitation, its presence as a component of some underground laminations likely occurred under different geochemical constraints.
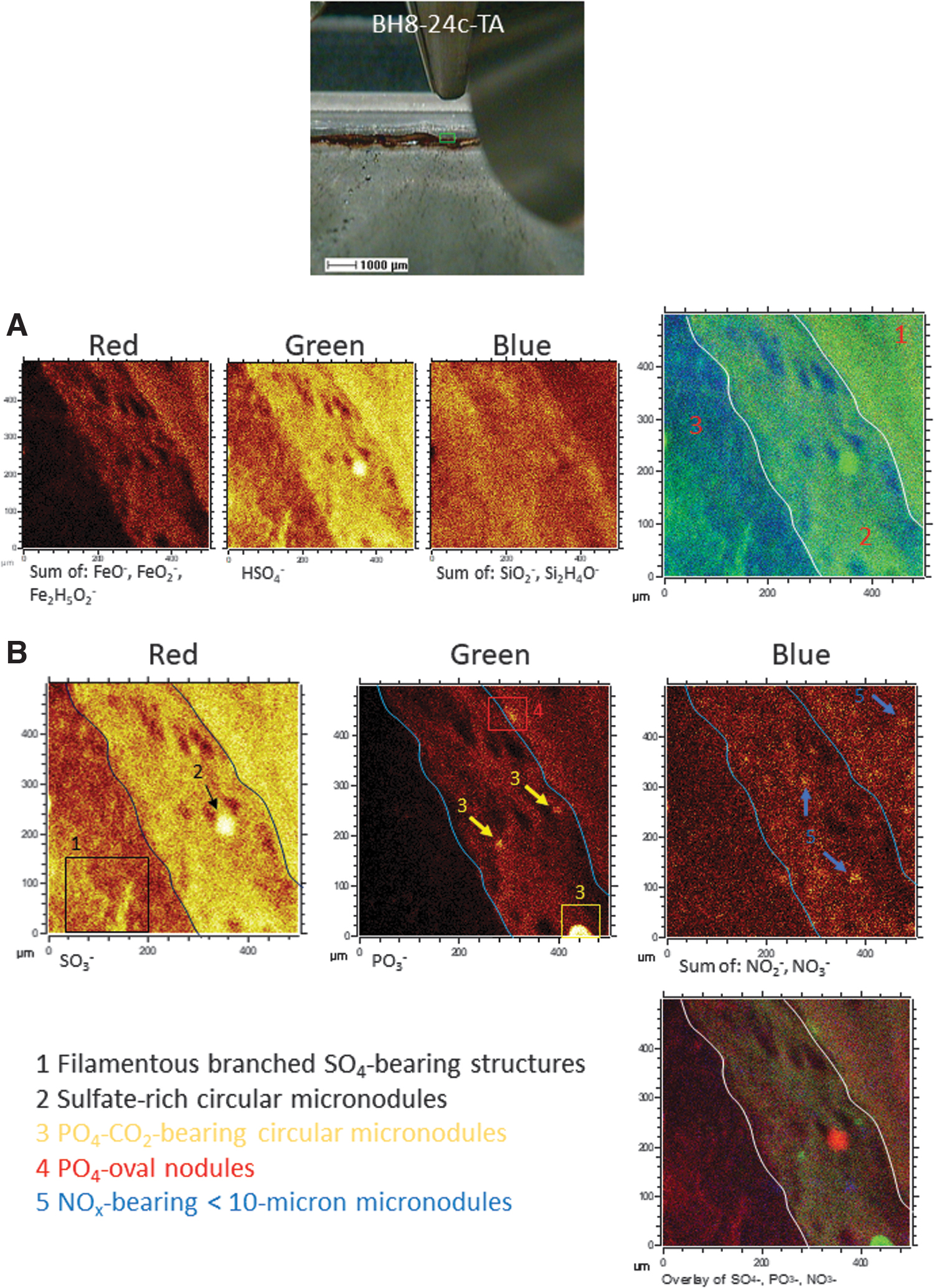
(
The alternation between ferric- and sulfate-rich and silica-bearing layers suggests seasonal or temporal chemical variations in pH and/or redox. The microbial activity has been claimed to trigger microenvironmental changes such as alkalinization, leading to silica precipitation in a sulfate-rich deposit (Birnbaum and Wireman, 1984, 1985). Neutralization of acidic solutions has been reported in Río Tinto through microbial reduction of the ferric ion and precipitation of carbonate (Fernández-Remolar et al., 2012). However, whether the microbial activity was involved in the silica precipitation is unclear since an increase in the pH through diagenesis could have also triggered the silica precipitation. In the Río Tinto acidic materials, neutralization has been associated with ferric oxysulfate exhumation and exposition to rainwater (Fernández-Remolar and Knoll, 2008). The percolating meteoric solutions would have increased the pH of the ferric deposits, washed down, and co-precipitated sulfates and silica. Consequently, the alternation between laminae of ferric oxysulfates and silica-rich layers (Fig. 7A) could have recorded in a few millimeters a full cycle of the Río Tinto gossan formation, including both acidic alteration and maturation stages.
In this mineral substrate, the ToF-SIMS mapping analysis has identified several microstructures bearing essential anions for life (Figs. 4C and 7B). They correspond to the following five elements recording potential metabolic information (Fig. 7B): (1) 40-micron size circular nodules enriched in sulfate ions; (2) >100-micron long sulfate-rich filamentous structures with straight to sinuous branched trajectory; (3) >40 micron-sized circular nodule mineralized by inorganic PO4-bearing compounds; (4) <20-micron sized oval nodule with PO4-bearing lipid traces; and (5) <15 micron-sized nodules with NO2 - and NO3 - anions. The occurrence of different microstructures that are identified by their anion composition suggests that they have been formed by other abiotic and/or microbial processes.
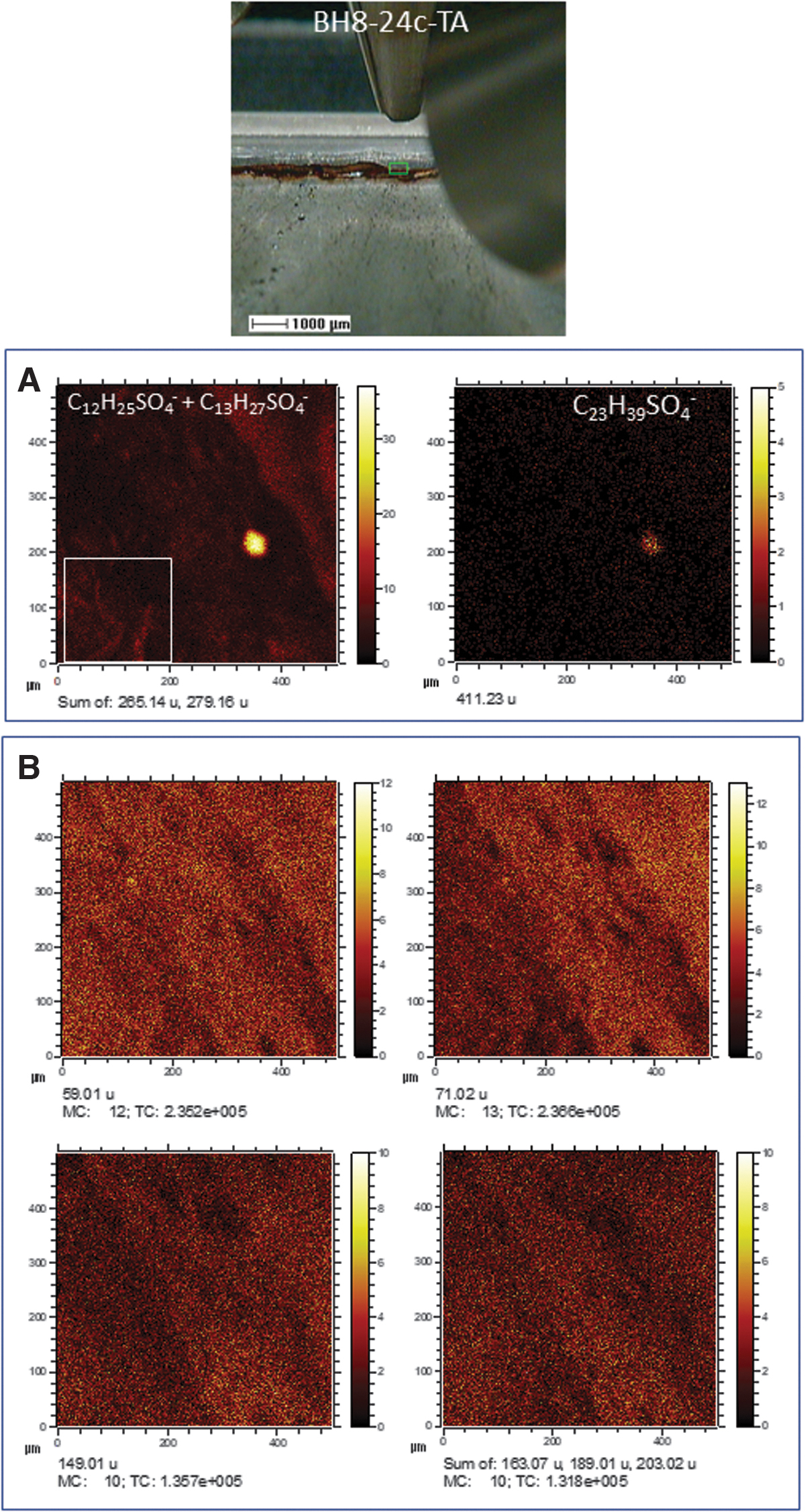
The abundance of sulfate in the subsurface (Fernández-Remolar et al., 2008) suggests that the mineralization of different structures by this anion has been the result of a protonation reaction on the microbial surface as a consequence of a low pH (Konhauser and Riding, 2012). The formation of a predominantly protonated microbial surface has likely favored a preferential binding of the available negative ions in the Río Tinto subsurface as SO4 - leading to the mineralization of the biological structures. The detection of different organic compounds bearing SO x (x = 2–7) groups strongly supports that the sulfate mineralization proceeded through a direct binding between the protonated biomolecular surface and sulfate anion that was highly available in the subsurface solutions. Acidic fungi and tolerant plants have been reported as active agents of biomineralization of jarosite (KFe3+ 3(OH)6(SO4)2) (Rodríguez et al., 2005; Oggerin et al., 2013), and the biological activity plays an essential role in the fluvial basin sedimentation by sulfate biomineralization in biofilms (Fernández-Remolar et al., 2010), where it has also been recognized in ancient Río Tinto sediments (Fernández-Remolar et al., 2005). The ToF-SIMS image analysis of terrace and gossan samples currently shows the presence of sulfate and cholesteryl sulfate associated with larger biological structures (Fig. 8). The occurrence of cholesteryl derivatives strongly supports that they correspond to different eukaryotic forms that inhabited the acidic basin more than 6 million years ago (Fernández-Remolar and Knoll, 2008). In this regard, the SO4-bearing branched filamentous structure (Figs. 2E, 4C2, and 7B) could represent a large prokaryotic filament as it is also composed of likely N-bearing fragments like amino acids (Fig. 4A1). The circular structure has, in turn, an unknown origin and could have been formed either through abiotic or biotic mineralization. In both cases, the sulfate mineralization does not depend on the metabolic process itself but on the high availability of sulfate that promoted the mineralization of biological and nonbiological structures in the Río Tinto subsurface.
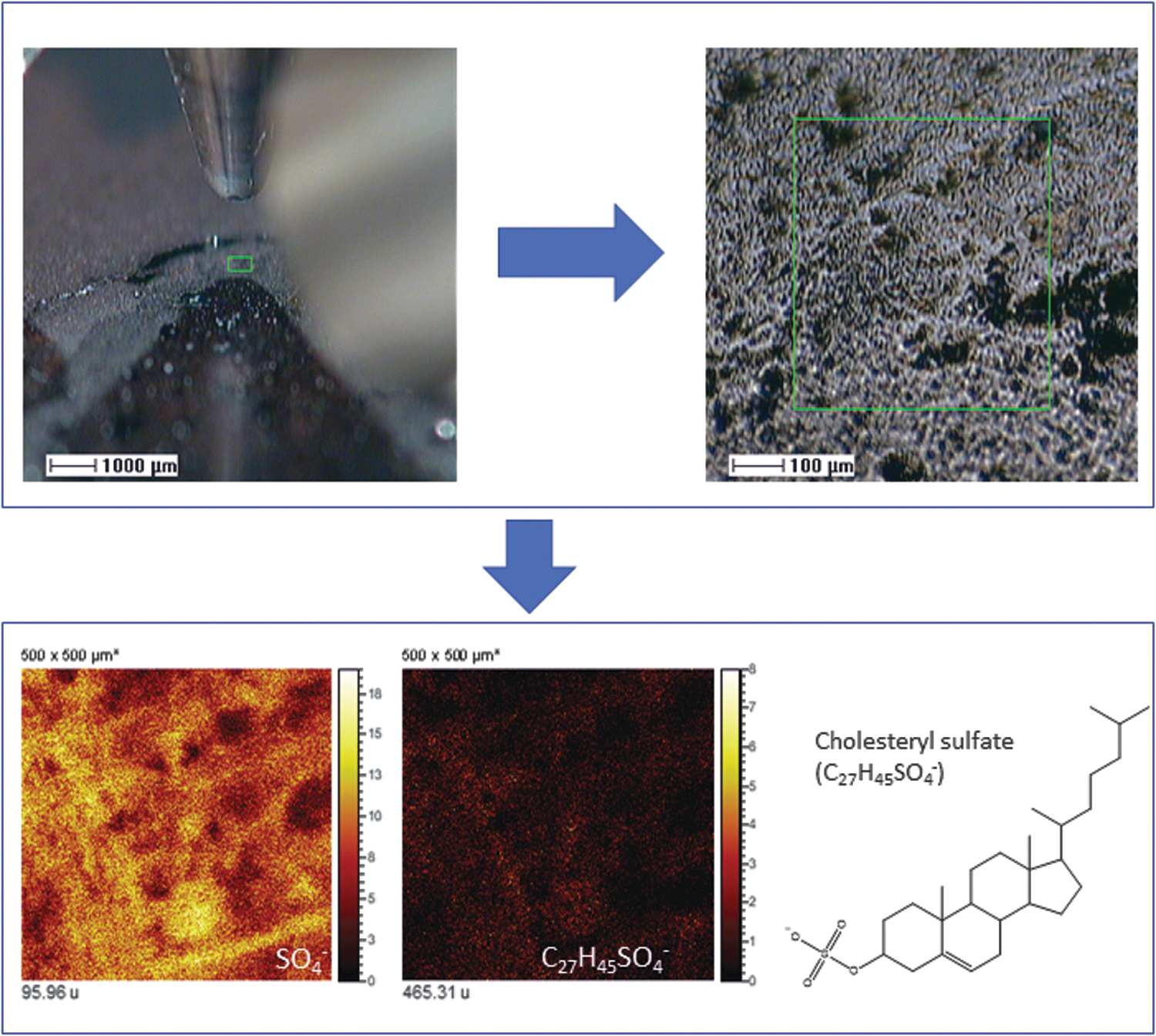
The occurrence of SO4 - and C27H45SO4 - (cholesteryl sulfate [M – H]- ion) in samples from the Upper Gossan in Pena de Hierro (∼25 Ma) mapping different remains of eukaryotic structures containing cholesterol. Cholesteryl sulfate has not been found in the BH8-24c sample occurring in the subsurface, supporting a negligible content in steroids, which is consistent with a sample preserving compounds produced by subsurface prokaryotes.
As discussed above, the second group of microstructures has been outlined by the occurrence of PO2 - and PO3 - anions. They correspond to two different morphologies that include both circular and ovoidal shapes, which are associated with different compounds. The circular morphology meets the distribution of anion CHO2 - which could be a major fragment of carbonates. In turn, the PO4-bearing ovoidal structure is associated with a set of several compounds that have been tentatively assigned to different lipid anions like phospholipids (Mazzella et al., 2005; Heim et al., 2009). Such a compositional difference suggests that while the circular microstructures preserve a biomineralized microstructure by carbonate and phosphate (Fernández-Remolar et al., 2012, 2018; Sánchez-Román et al., 2014, 2015), the smaller and ovoidal elements could correspond to the preservation of a microbial population that has been mineralized by oxysulfates. An alternative explanation for the different molecular composition in the phosphatic micronodules could also be related to the number of preserved cells. In this regard, fewer biomineralized cells will lower the concentration in lipids that would accordingly decrease the intensity below the intensity levels to be plotted as a ToF-SIMS molecular image. Although this could be a plausible explanation, we observed that the largest phosphatic nodule, which should have biomineralized a more significant number of cells, does not show any evidence of lipids, suggesting that such an explanation does not elucidate the compositional changes in the different phosphatic nodules. Consequently, it is more likely that differential composition in the PO3-bearing micronodules results from the metabolic activity by chemoheterotrophs that produced biomineralized structures which composition was mainly mineral.
Indeed, similar spheroidal microstructures composed of carbonate and phosphate (Fig. 9) have been reported in surface and subsurface areas of Río Tinto (Fernández-Remolar et al., 2012, 2018). The SEM-EDS analysis of samples collected in borehole BH4 (Fernández-Remolar et al., 2008) shows that they consist of >30–60 micronodular elements with discoidal to spherical morphology whose primary mineral composition varies from ankerite (CaFe2+(CO3)2) to siderite (FeCO3), and likely different amounts of phosphate. The carbonate and phosphate biomineralization could have been associated with chemoheterotrophic metabolism (e.g., Acidiphilium sp.), which would have oxidized organic matter by using ferric ion as an electron acceptor (Sánchez-Román et al., 2014; Fernández-Remolar et al., 2018). Recent experiments have shown that Tessaracoccus lapidicaptus, a heterotrophic bacteria isolated from the Río Tinto basement at 297 m deep (Sánchez-Román et al., 2015), precipitates siderite and vivianite (Fe3(PO4)2·8H2O) in cell walls and the EPS. Interestingly, T. lapidicaptus reduces NO3 - as it uses this anion as an electron acceptor, which means that it could recycle nitrogen in the subsurface of the IPB.
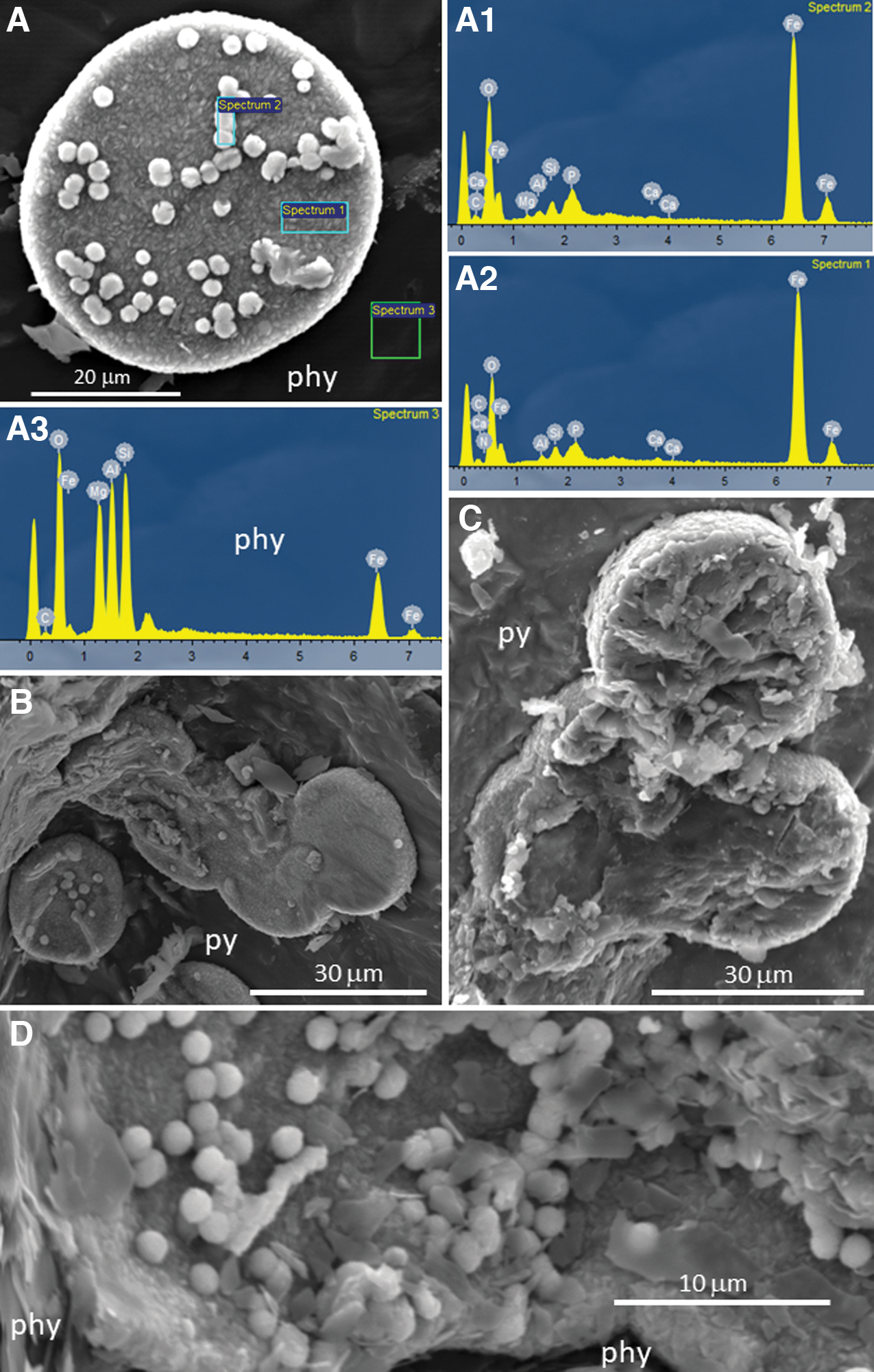
Image and compositional analysis by SEM-EDS of sample BH4-60a, collected at 151.5 m deep in borehole BH4 (Fernández-Remolar et al., 2008). It shows the occurrence of discoidal (
The ToF-SIMS mapping ability has also provided information to identify tiny micronodules (<15 microns), which are enriched in NO2 - and NO3 - (Fig. 7B). The microstructure proportions (<10 microns) fit well with the thickness of microfilament sections (Fig. 2C, 2D), which also have a high content in elemental N (∼9 atomic % ≈ 9 weight %). In addition, the microfilaments also show a primary elemental composition dominated by C, O, and Fe, suggesting a siderite-like mineral composition. The co-occurrence of elemental Si as a secondary composition supports a pH increment during the formation of carbonates in the filament interior. The SEM images show that the microfilaments are currently entrenching the lamination surface, suggesting that they likely grew through the mineral surface dissolution (Fig. 2C). Assuming that the outermost part of a layer is composed of iron oxyhydroxides and oxysulfates (Fig. 2C, 2D), the attack process could be related to the reduction of Fe3+ and/or SO4 -. The electron donors could have been different compounds like organic molecules such as CH4 (Puente-Sánchez et al., 2014), leading to the nucleation of siderite (Drake et al., 2017; Fernández-Remolar et al., 2018; Leandro et al., 2018) but also the formation of nitrate as a final member of the ammonia oxidation that uses the ferric cation as an electron acceptor as the Feammox bacteria (in ‘t Zandt et al., 2018). Feammox bacteria use the ferric cation as an electron acceptor through ammonia oxidation where dinitrogen gas, nitrite, or nitrate are released as a final by-product. In the microbial attack on the ferric surface, SO4 - ions could also be available as the electron acceptor for different metabolic pathways, including autotrophic and heterochemotrophic (Zhang et al., 2009; Drake et al., 2017). Furthermore, the oxidized nitrogen compounds could also be recycled by using ammonia as an electron donor (in ‘t Zandt et al., 2018). Under such metabolic activity, NO2 - and NO3 - are by-products that would accumulate inside the microfilament structure as it is evidenced by ToF-SIMS and SEM-EDS results (Figs. 2C–2E, 4C4, 7B, and 10).
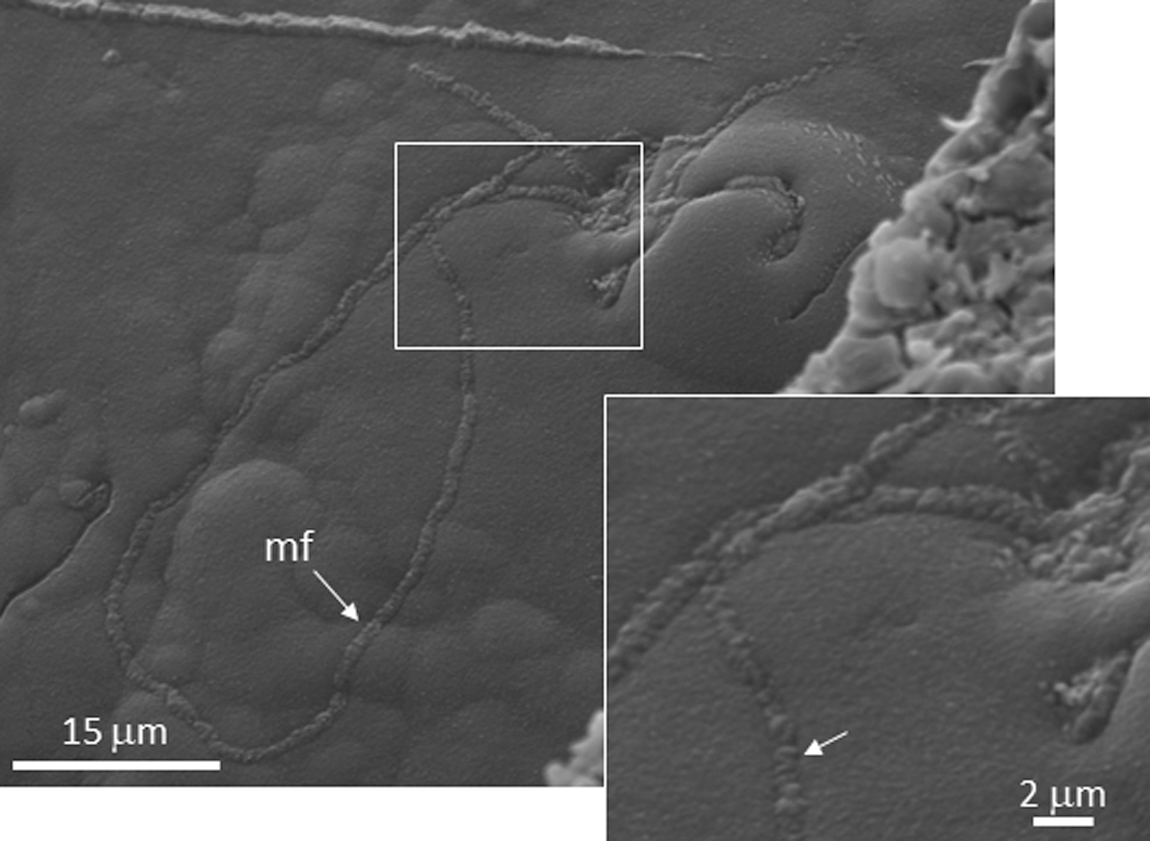
SEM image of the BH8-24c sample collected at ∼40 m deep. It shows the microfilament (mf) morphology and microstructure interior, which seems filled by different minor elements (<0.5 microns) with filamentous or rodlike shape.
The ToF-SIMS mapping in another target area of sample BH8-24c provides further information about the source of nitrogen in the subsurface. The imaging analysis of the positive ions at 18.04, corresponding to NH4 +, provides a full-frame of the nitrogen cycle if the NO2 - and NO3 - mapping is included in the analysis (Fig. 11). It shows the occurrence of the different nitrogen molecular species following an inverse distribution where the NH4 + and NO3 - ions occur in opposite areas, while the NO2 - co-occurs with areas with both ammonium and nitrate species. Such distribution suggests that NO2 - is an intermediate specie that could be formed through the microbial oxidation of NH4 + microlocations and recycled by microbial reduction of NO3 -. The ammonia distribution meets the occurrence of some amino acid fragments (e.g., C2H8N+ in Fig. 11), suggesting that the NH4 + production results from the ammonification of biological compounds like proteins (Herbert, 1999). While the ammonium can be recycled by ammonification of biological nitrogen, the primary source of nitrogen is a questionable matter. Although it could be expected the occurrence of organic compounds fitting the distribution of NO2 - and NO3 -, we have found no evidence of large molecular compounds associated with such distribution. While this could be a consequence of the mass spectral range limited up to 750 Da, the small size of the microstructures containing the N-bearing anions should also have a low concentration in lipids, as can be estimated from the SEM images. The ToF-SIMS images show that the NO2 - and NO3 - microstructures have size smaller than 10 microns (Fig. 11), which correspond to <50 cell bodies as recognized in the SEM images (Fig. 9). As the lipidic content should be low as the cellular remains have experienced mineralization and degradation, it is expected that the molecular concentration be low enough to have a clear signal above the spectral data background.
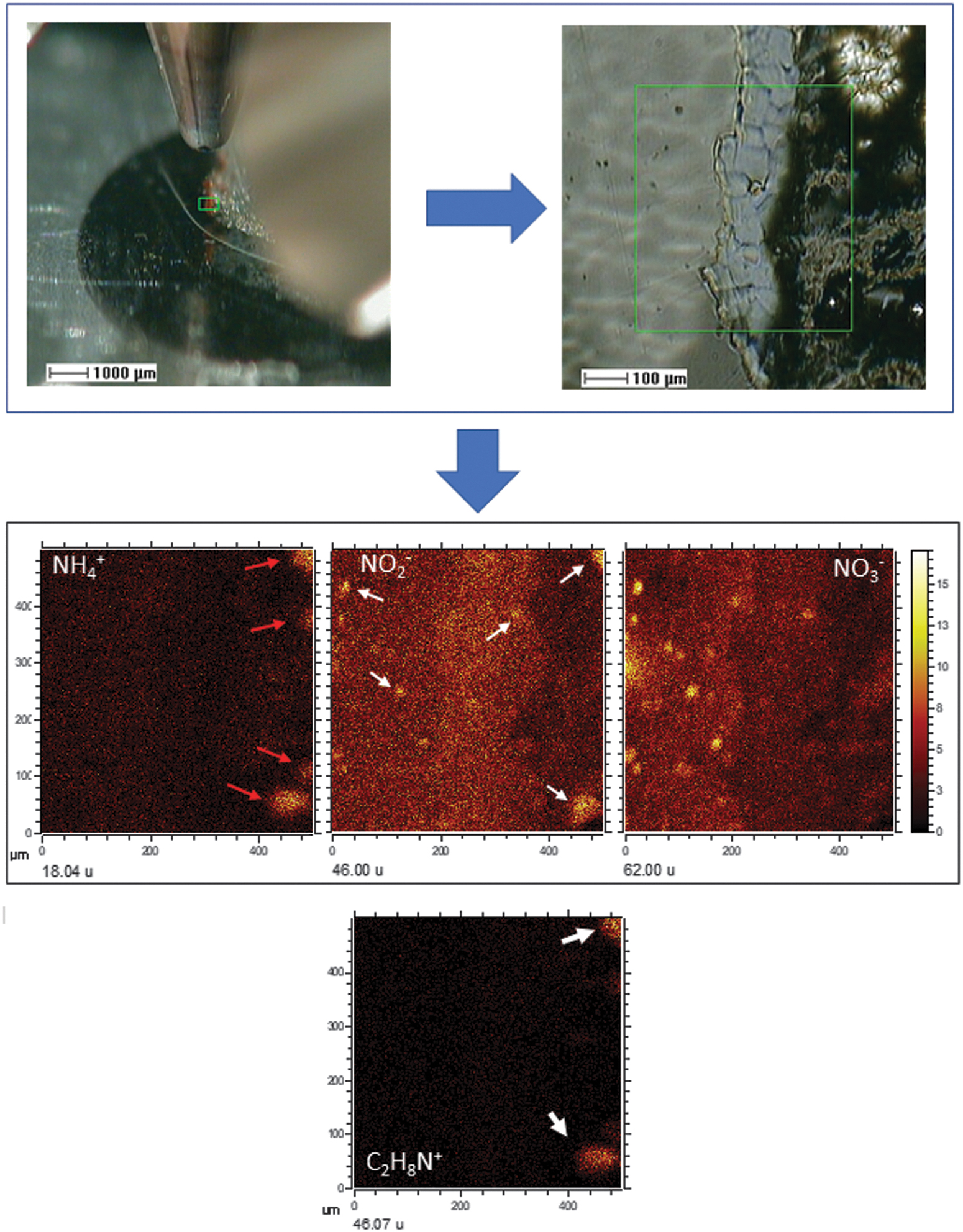
ToF-SIMS image of another target area in sample BH8-24c showing the distribution of positive and negative N-bearing ions as NH4 +, NO2 -, and NO3 - which provide some insight of how the nitrogen cycle could operate in the subsurface. This could be partly initiated by ammonification (red arrows) in areas enriched with peptidic chains, which would be released to other microlocations where the NH4 + will be oxidized to nitrite and nitrate by microbes that formed the microfilaments. The comparison between the three intensity distribution images shows that NO2 - (white arrows) appears as an intermediate compound that co-occurs in the NH4 +- and NO3 --rich micronodules.
Rock leachates of samples BH8 collected at 40 m deep corresponding to the weathered hydrothermal stockwork (Fernández-Remolar et al., 2008) have shown that it can release up to 0.2–2 ppm of NO3 -, while the NO2 - concentration was around 72 ppm. Rock samples of nonweathered stockwork materials (Fernández-Remolar et al., 2008) sampled below 140 m deep released up to 50 ppm NO2 - but, in turn, a NO3 - concentration lower than 0.02 ppm. We suggest that the primary source of nitrogen could be the nitrite ion produced during the Carboniferous hydrothermal activity (Leistel et al., 1998; Almodóvar and Sáez, 2004). Hydrothermal systems have been reported to be associated with the transfer and formation of nitrates by the volatilization of ammonia and dinitrogen gas from Earth's crust, as well as through nitrite and nitrate leaching of underlying sedimentary, igneous, and metamorphic rocks by hydrothermal fluids (Holloway and Dahlgren, 2002). The chemical analysis of subsurface fluids from multilevel diffusion samplers in BH8 (Fernández-Remolar et al., 2008) shows a NO3 - concentration lower than 4.2 ppm (∼67 μM), suggesting that it is a transitional reservoir that receives nitrogen from meteoric replenishment and the basement alteration. Such a weathering process could be accelerated during ancient thermal episodes that led to the formation of the Gossan and oldest terraces of Río Tinto during different thermal episodes (Fernández-Remolar et al., 2005; Essalhi et al., 2011; Velasco et al., 2013).
The ToF-SIMS surface analysis has also shown that other anions like Cl- follow the distribution pattern of NO2 - and NO3 - (Fig. 4C1), which occur inside the microfilament structures (Figs. 2C, 2D, and 4C4). Leachates of samples recovered at 40 m depth, where the weathered stockwork is found, gave a Cl- concentration of around 8.5 ppm. However, chloride was increased up to 72.5 ppm in pristine hydrothermal materials occurring below 140 m depth. The analysis of underground water samples from the multilevel diffusion samplers (Fernández-Remolar et al., 2008) in BH8 provided a chloride concentration of up to 290 ppm (8200 μM). Although the Cl- distribution in the subsurface suggests that it is released from the hydrothermal materials like the nitrogen anions, it experiences an increment in the subsurface fluids. This could have resulted from an additional supply from other sources (e.g., rainwater) or that it was not taken up by microbial activity at the same rate as nitrate.
The distribution of the main biomolecular groups that have been listed above provides additional information about the microbial communities occurring in the Río Tinto subsurface. In this regard, the N-bearing compounds associated with the preservation of amino acid, as well as the occurrence pattern of saccharides, are homogeneously distributed in the target area analyzed by ToF-SIMS. However, some slight variations depend on the anion type. The CN-, CON-, and the N-bearing anions interpreted as amino acid fragments, and likely N-bearing lipids, show a higher affinity to the distribution of silica anions (Fig. 4A). In turn, the occurrence of CP- and CNP- anions is more akin to the distribution pattern of the Fe- and S-bearing inorganic anions, as the P-bearing lipid fragments do (Fig. 6B). Both N- and P-bearing major anions, as well as the HS- anion, have a higher affinity for the filamentous structure enriched in sulfate that is present in the lower-left corner of the sample (Figs. 4A, 4C, and 7B). Such a complementary distribution suggests a change in the microbial community that would follow the increment of silica in the laminations (Fig. 7A) or alternation in the preservation conditions resulting from environmental and/or diagenesis changes. In the first case, such a variation will go from communities that would produce a higher content in P-bearing lipids during a ferric-rich episode (Figs. 6B and 7B) to an increment in prokaryotic forms synthesizing N-bearing biomolecules during the episode precipitating silica (Figs. 4A and 7A), which could be associated with an increment in the EPS formation by microbial communities (Escudero et al., 2018). Interestingly, the saturated and monounsaturated FAs follow the general pattern shown by lipids greater than 600 Da. This could be related to the environmental alternation from ferric-rich and acidic solutions to more neutral, where the silica formation was favored. It could come together with a change in the preservation or environmental conditions affecting the microbial composition.
The ToF-SIMS analytic capabilities have also mapped the distribution of different m/z fragments that are characteristic of other saccharides. Although the distribution is more homogeneous, it follows the main laminae fabric and shows a slight intensity increment in the iron-rich laminae (Figs. 4C and 6D). The occurrence of a regular distribution of saccharides in the target area suggests multiple origins. As discussed above, saccharides occur in head groups of membrane lipids, such as sphingolipids or saccharolipids, but are also found as polysaccharides in EPS. However, in terms of quantity, EPS produce larger amounts of biological compounds compared to membrane lipids and would be the primary source of saccharides in the subsurface. This is consistent with novel findings suggesting that biofilm growth in the subsurface is a common microbial process in the Río Tinto subsurface (Escudero et al., 2018).
6. Conclusions
The combination of the composition and distribution of different molecules assigned to an inorganic or organic origin provides a piece of evidence to distinguish microbial structures that have resulted from the metabolic cycling of elements in the Río Tinto subsurface. Based on such information, at least four groups of microstructures of microbial origin can be recognized as the following:
(A)
(B)
(C)
(D)
Here, we provided mineralogical and biomolecular evidence that different mineralized structures in the Río Tinto basement were formed by the interaction of the microbial communities with the geological substrate. Its proposed formation demonstrates how the biosphere has developed through the preexistent chemical cycles that have been maintained by the geological activity over billions of years. In the Río Tinto case, the microbial communities have produced some structures that can be unequivocally identified as by-products of the existence of biochemical cycles that have been powered by the underground metabolic activity of chemoheterotrophic and chemosynthetic life. Such biogeochemical cycling was accompanied by mineralization driven by the production of sulfate and ferric iron sourced in the biooxidation of the sulfide orebody by chemolithotrophic life. Such a process was intimately integrated into the transfer of matter and energy in the Río Tinto subsurface habitats and became the principal agent to preserve the biological information of the underground microbial communities.
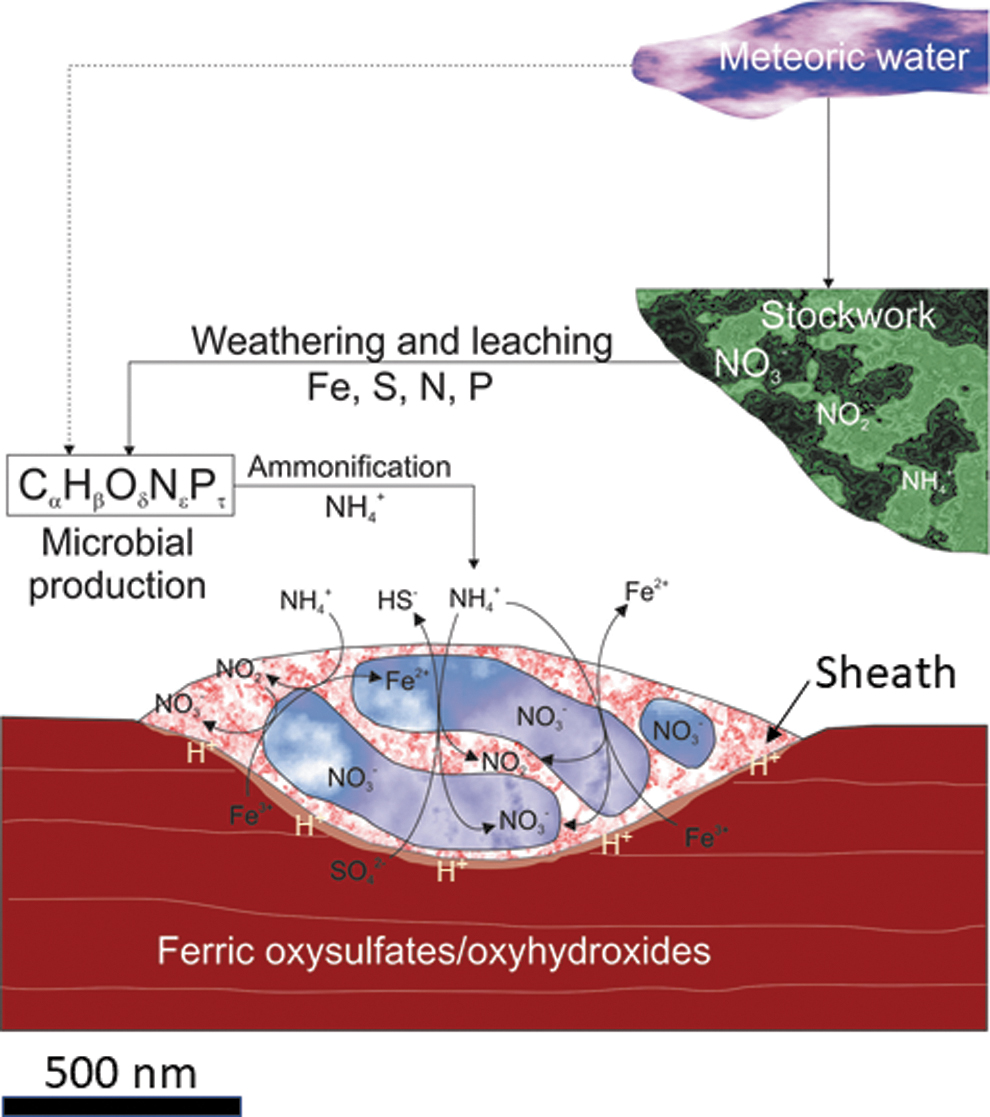
Image showing an idealized section of the microfilaments describing the biogeochemical pathways of NO2 - and NO3 -. The ammonia distribution in the sample suggests that it comes from the microbial ammonification of peptide-based compounds which could be eventually used as an electron donor to reduce the ferric iron found in the oxysulfates and/or oxyhydroxides. The ammonia would be oxidized to produce nitrite and nitrates. The microbial attack would release the ferric ion through using different organic acids (e.g., oxalic) and/or siderophores, which would dissolve the lamination surface. Although ammonification could be the transitional source of nitrogen, the high content of nitrogen in the stockwork suggests that it can be the primary source of nitrogen. However, the meteoric water could also act as a minor source as well. The occurrence of elemental C in the microfilaments brings up carbonate as the main mineralizing component with some remaining organic compounds.
Footnotes
Acknowledgments
The authors are grateful for the ideas provided by several reviewers who have contributed greatly to improve the manuscript. David Fernández-Remolar acknowledges the full support provided by the Director of the SKL Lunar and Planetary Sciences Keke Zhang.
Author Contribution
DCFR, DGO, and TH wrote the manuscript with input from RA, NR, NB, AA, and CE. DCFR, TH, and NB ran the ToF-SIMS analysis. DCFR and NR collected the SEM-EDS data. RA, NR, and CE provided microbial information for data interpretation. All authors contributed to the discussion and final manuscript.
Funding
This research has been supported by the Fundo de Desenvolvimento das Cientifico e da Tecnologia da RAE de Macau (proposals FDCT-0005-2020-A1 and FDCT-18-024-SSI), and the project on Civil Aerospace Technologies No. D020303 funded by China National Space Administration. Samples of borehole BH8 were collected during the ground drilling operations funded by the NRA-02-OSS-01 NASA-ASTEP “Mars Astrobiology Research and Technology Experiment (MARTE),” a Mars analog drilling project to search for subsurface life at Río Tinto. Y.S. acknowledges funding by FDCT (No. 0002/2019/APD). N.B. acknowledges funding from NSERC.
Author Disclosure Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary Material
Supplementary Table S1
Supplementary Table S2
Supplementary Figure S1
Abbreviations Used
Associate Editor: Victor Parro
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
