Abstract
Comparisons between the preservation potential of Mars-analog environments have historically been qualitative rather than quantitative. Recently, however, laboratory-based artificial maturation combined with kinetic modeling techniques have emerged as a potential means by which the preservation potential of solvent-soluble organic matter can be quantified in various Mars-analog environments. These methods consider how elevated temperatures, pressures, and organic–inorganic interactions influence the degradation of organic biomarkers post-burial. We used these techniques to investigate the preservation potential of deposits from a circumneutral iron-rich groundwater system. These deposits are composed of ferrihydrite (Fe5HO8 · 4H2O), an amorphous iron hydroxide mineral that is a common constituent of rocks found in ancient lacustrine environments on Mars, such as those observed in Gale Crater. Both natural and synthetic ferrihydrite samples were subjected to hydrous pyrolysis to observe the effects of long-term burial on the mineralogy and organic content of the samples. Our experiments revealed that organic–inorganic interactions in the samples are dominated by the transformation of iron minerals. As amorphous ferrihydrite transforms into more crystalline species, the decrease in surface area results in the desorption of organic matter, potentially rendering them more susceptible to degradation. We also find that circumneutral iron-rich deposits provide unfavorable conditions for the preservation of solvent-soluble organic matter. Quantitative comparisons between preservation potentials as calculated when using kinetic parameters show that circumneutral iron-rich deposits are ∼25 times less likely to preserve solvent-soluble organic matter compared with acidic, iron-rich environments. Our results suggest that circumneutral iron-rich deposits should be deprioritized in favor of acidic iron- and sulfur-rich deposits when searching for evidence of life with solvent extraction techniques.
1. Introduction
The successful search for life on Mars is dependent on the careful consideration of rocks that reflect deposition in environments that are likely to have supported life. Iron-rich, aqueous environments provide habitable niches on Mars due to the availability of dissolved Fe(II) that can drive both aerobic and anaerobic microbial metabolism (e.g., Widdel et al., 1993). Iron is an abundant element in martian regolith and is an especially powerful biogeochemical element due to its ability to act as both an electron donor for iron-oxidizing bacteria (Baker and Banfield, 2003; Emerson and Weiss, 2004), and an electron acceptor in dissimilatory iron reduction processes (Lovley, 1991). Subsurface or subaqueous chemolithotrophs may have existed on early Mars and taken advantage of the abundant iron present on the red planet.
Iron-rich environments have, historically, been considered deleterious to organic matter preservation due to the predisposition of iron to oxidizing reactions (Sumner, 2004; Klein, 2005), but they are becoming increasingly accepted as capable of preserving microbial biosignatures, either in the form of structural biosignatures due to iron oxide encrustation (Potter-McIntyre et al., 2014; Williams et al., 2015a, 2016) or molecular biomarkers in the form of lipids that survive the initial stages of diagenesis by being encrusted or adsorbed onto the surface of amorphous, high-surface-area iron oxides, and oxyhydroxides (Lalonde et al., 2012; Parenteau et al., 2014, 2016; Tan et al., 2018).
Low-temperature, circumneutral, iron-rich aqueous environments have recently been the subject of significant interest due to observations made by the Mars Science Laboratory (MSL) rover Curiosity that suggest the presence of a circumneutral, low-salinity, possibly redox-stratified iron-rich lacustrine environment that existed between 3.8 and 3.1 Ga at Gale Crater (Hurowitz et al., 2010, 2017; Grotzinger et al., 2014, 2015; McLennan et al., 2014; Vaniman et al., 2014; Bristow et al., 2015). The primary iron species found in similar terrestrial settings is ferrihydrite, a nanocrystalline iron oxide (Fe5HO8 · 4H2O) that exists in two forms, “2-line ferrihydrite” and “6-line ferrihydrite,” that are distinguished by the degree of crystallinity that it exhibits according to the presence of X-ray diffraction (XRD) lines (Cornell and Schwertmann, 2003).
Ferrihydrite comprises a majority of the X-ray amorphous component in the martian regolith, being present in proportions of up to 50 wt% in mudstones examined at Yellowknife Bay (Dehouck et al., 2017). Ferrihydrite is also found in a wide variety of martian environments, such as in the Rocknest, Cumberland, and Windjana samples as analyzed by CheMin on the MSL Curiosity rover (Bish et al., 2013; Vaniman et al., 2014; Rampe et al., 2016; Treiman et al., 2016; Dehouck et al., 2017), at Gusev Crater and Meridiani Planum as observed by the Mars Exploration Rover missions (Morris et al., 2006a, 2006b), and other locations such as McLaughlin Crater as observed by the Compact Reconnaissance Imaging Spectrometer for Mars (Michalski et al., 2019).
Due to the metastability of ferrihydrite, this mineral is often not found in terrestrial sediments and rocks older than the Holocene, and it is instead a precursor to more stable iron oxide species such as goethite (α-FeOOH) and hematite (α-Fe2O3) (Cornell and Schwertmann, 2003). On Mars, however, the low-temperature, rock-dominated environment inhibits the transformation of ferrihydrite even when subjected to short-term aqueous alteration (Dehouck et al., 2017). The widespread, long-term preservation of ferrihydrite at Gale Crater over geological time, its ubiquity in the martian surface and subsurface, and the possibility that it may have supported biological activity suggest that ferrihydrite has the potential to be a target of astrobiological interest.
Little is known, however, about the degradation reactions, mechanisms, and organic matter–mineral interactions associated with biomarker preservation in ferrihydrite, especially with respect to the processes and fluxes associated with the post-burial effects of diagenesis. In particular, rapid burial is relevant to biomarker preservation not only as a degradative process but also as a mechanism by which the organic biomarkers contained within these sediments and rocks may be shielded from deleterious radiation, such as solar ultraviolet radiation or galactic cosmic rays (Cockell et al., 2005; Dartnell et al., 2007; Dartnell, 2011; Cockell, 2014; Hassler et al., 2014). Planned missions to Mars such as ExoMars have been designed with the aim of collecting samples that have been shielded from surface radiation, where a drill will be used to access samples at depths of up to 2 m (Vago et al., 2016). Other strategies include targeting surfaces that have only recently been exhumed such as near escarpments or at sites of impact ejection (Farley et al., 2014; Montgomery et al., 2016). It is, therefore, necessary to elucidate the relationship between organic matter, minerals, and their interactions during diagenesis.
Recent geochemical work has focused on using hydrous pyrolysis to expound on the post-burial effects of diagenesis on the degradation of specific organic compounds in Mars-analog geochemical environments (e.g., Royle et al., 2018; Tan et al., 2018; Tan and Sephton, 2020). Hydrous pyrolysis has traditionally been used to study the diagenesis and maturation of organic compounds as it replicates the organic geochemical reactions that occur over geological timescales by simulating them with short-term, high-temperature, and high-pressure laboratory conditions; this technique has been utilized by several authors to study the effects of thermal maturity on specific biomarker compounds (e.g., Eglinton et al., 1986; Peters et al., 1990; Koopmans et al., 1995; Jaeschke et al., 2008; Rush et al., 2014; Royle et al., 2018).
By analyzing the products of artificial maturation experiments under laboratory conditions and deriving the kinetic parameters of organic matter breakdown, it is possible to determine the optimal conditions under which preservation is favored, and this can also help to quantitatively predict the effects of degrading reactions on organic biomarker concentrations over geological time. Here, we present the results of hydrous pyrolysis experiments performed on ferrihydrite deposits found flocculating in a circumneutral, iron-rich bog. We report on the complex interactions between the hosted organic matter and the ferrihydrite-rich mineralogy of the deposits, their response to the post-burial stresses of diagenesis, and the applications of our findings to future missions to Mars.
2. Materials and Methods
2.1. Sample collection
The geology of the Imperial College Silwood Park Campus is located stratigraphically at the base of the Group, a series of Eocene deposits (50–44 Ma) that comprise the Camberley, Windlesham, and Bagshot iron-rich sand horizons that, in turn, overlie the impermeable London Clay (Barrott et al., 2014). The downward permeation of groundwater in these sand horizons has resulted in the anaerobic leaching of Fe(III) from glauconite hosted within these deposits, leading to the widespread occurrence of circumneutral iron seeps rich in dissolved Fe(II) (Barrott et al., 2014). Fe-rich groundwater discharges from sand horizons are rare, as the depletion of reactive iron from ferruginous sands is a geologically rapid process. The Bracklesham Group is unique, as the groundwater in this region was relatively isolated from the surface until the last deglaciation; the iron seepage in this area is, thus, a geologically ephemeral process and is expected to continue only for another 0.1 Ma (Barrott et al., 2014).
This iron-rich groundwater system is manifested as a large iron bog ∼20 by 30 km in area, with springs rich in Fe2+ emerging from various locations depending on the topography, and it is defined by an ocherous color derived from the precipitation of iron oxides by iron-oxidizing bacteria (FeOB). Streamwater conditions were found to have a temperature of ∼10°C, with circumneutral pH and containing 10–20% dissolved oxygen, consistent with its provenance as groundwater flow (Barrott et al., 2014). The sampling area was limited to the lacustrine and stream environment accessible from the Imperial College Silwood Park campus owing to safety concerns (Fig. 1a, b).

Field and SEM images of the sampling sites.
Ocherous, highly amorphous, ferrihydrite flocs were precipitated in these environments by FeOB via the conversion of dissolved ferrous iron into insoluble ferric iron as part of their metabolic pathways (Chan et al., 2016). These flocs encrusted the bacteria that precipitated them, forming filamentous sheaths that could be clearly observed in scanning electron microscopy (SEM) images (Fig. 1c). These sheaths were identified as characteristic biostructures of Leptothrix ochracea, a chemolithoautotrophic FeOB (Emerson et al., 2010; Fleming et al., 2014; Chan et al., 2016). L. ochracea is capable of oxidizing Fe at a range of O2 concentrations, and it flourishes at microaerobic conditions down to 30 μM dissolved O2 (Chan et al., 2016; Fleming et al., 2018). The filamentous sheaths produced by L. ochracea and iron oxidation during metabolism are known to be tightly coupled, indicating a strong association between precipitated ferrihydrite and organic matter in these flocculent mats (Chan et al., 2016; Vesenka et al., 2018).
Samples of flocculent ferrihydrite were collected by filtering streamwater through VWR 12–15 μm filter paper and transferring the ocherous residue to falcon tubes pre-washed in deionised (DI) water. The residues were returned to the laboratory, freeze-dried, and powdered with a solvent-cleaned agate pestle and mortar.
2.2. Scanning electron microscopy
The filamentous sheaths present in the ferrihydrite flocs were imaged with a Phenom ProX Desktop SEM. The samples were loaded onto an SEM sample stage by using a double-sided carbon tape. The SEM was operated at 5 kV with a working distance of 5.9 mm. Images were taken at magnifications of ∼10,000, 25,000, 50,000, and 100,000.
2.3. Ferrihydrite synthesis
Synthetic 2-line ferrihydrite was made following the method detailed in Cornell and Schwertmann (2003). Eight grams of Fe(NO3) · 9H2O (Sigma-Aldrich, suitable for cell culture) was dissolved in 100 mL of DI water and stirred with a magnetic stirrer. Sixty-six milliliters of 1 M potassium hydroxide (KOH) solution was continuously added, and the pH of the resulting mixture was brought to 7.5. The final volume of KOH solution was added dropwise, and the pH was continuously monitored. Dissolved salts were removed by adding DI water, centrifuging, and decanting the supernatant until the conductivity was <10 μS/cm as measured with a total dissolved solids probe. The synthesized ferrihydrite was then freeze-dried and ground gently in a solvent-cleaned agate pestle and mortar. It was then analyzed by powder XRD to ensure that the products were amorphous and analogous to the mineral content of the natural ferrihydrite (NF) (Fig. 2). As ferrihydrite is known to recrystallize at room temperature, synthetic samples were wrapped in aluminum foil and stored at 4°C.
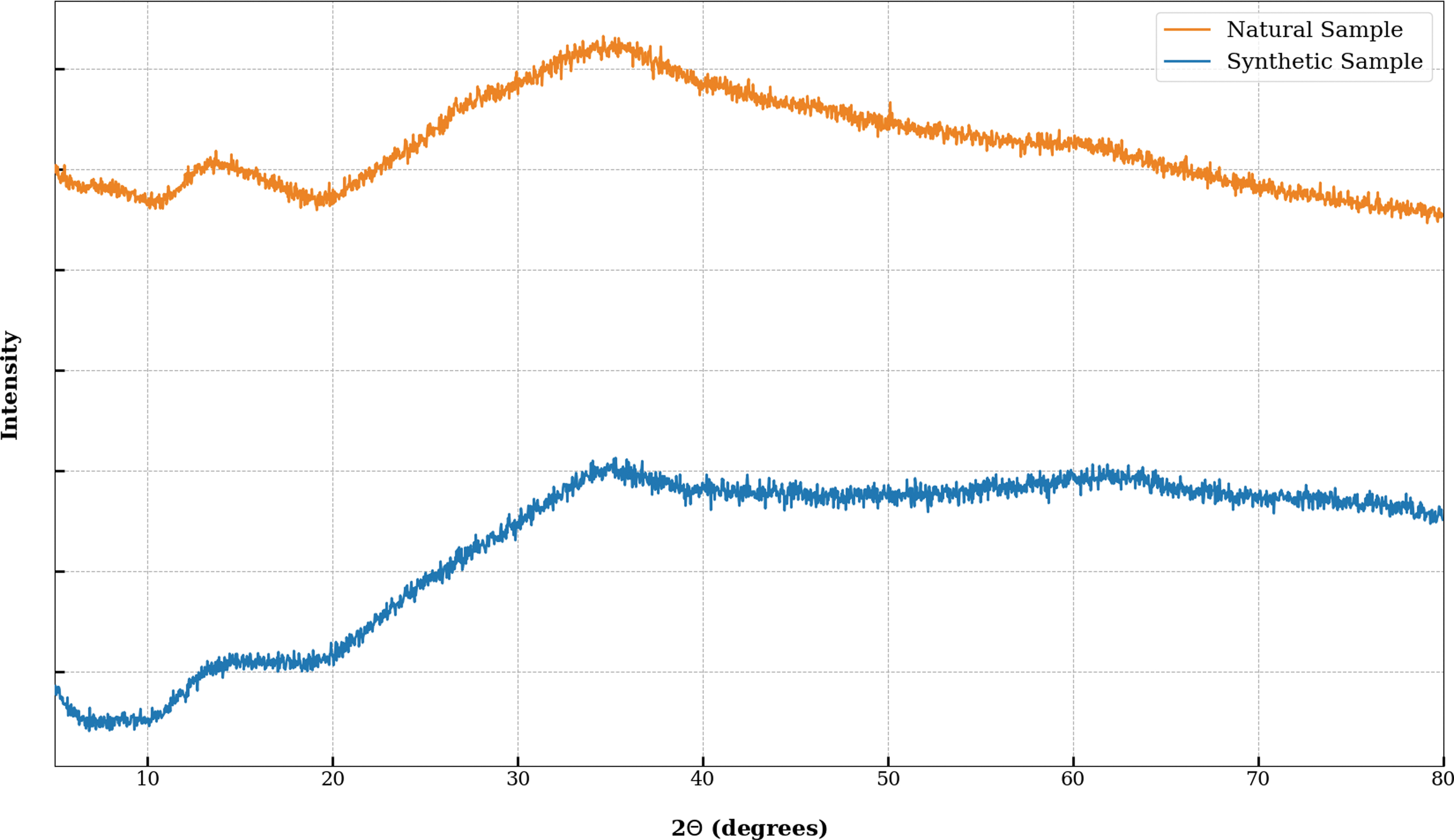
Comparison between XRD spectra of natural and synthetic ferrihydrite samples. XRD, X-ray diffraction. Color images are available online.
2.4. Artificial maturation by hydrous pyrolysis
NF samples from Silwood Site 6 (hereafter referred to as NF) and the synthetic ferrihydrite (SF) samples produced under laboratory conditions (hereafter referred to as SF) were chosen for artificial maturation by hydrous pyrolysis. The two samples were selected to distinguish the mineralogical differences associated with iron species transformation in the presence or absence of organic matter (NF and SF, respectively). A list of experimental conditions and sample names is presented in Table 1.
List of Samples Subjected to Hydrous Pyrolysis
Samples with the MO suffix were artificially matured for 1 month, and all other samples were matured for 72 h.
N/A = not applicable; NF = natural ferrihydrite; NFH = natural ferrihydrite pyrolyzed at high water-to-rock ratios; NFL = natural ferrihydrite pyrolyzed at low water-to-rock ratios; SF = synthetic ferrihydrite.
Hydrous pyrolysis experiments were set up as described in previously published artificial maturation studies (Royle et al., 2018; Tan and Sephton, 2020). A high-temperature and high-pressure stainless steel reactor (70 mL Model 4740; Parr Instruments) was used for these experiments. To accommodate the small sample sizes, small stainless steel “bomblets” were constructed by using T316 ½-inch stainless steel tube (35 × 9 mm o.d. × 6 mm i.d.) sealed at both ends with Swagelok end caps (SS-600-C) after deburring, with a final internal volume of ∼1.5 mL. Before each experiment, the bomblets were ultrasonically washed for 10 min in deionized (DI) water, methanol, and dichloromethane, and they were dried at 110°C.
Each sample was loaded into the bomblet, and either 0.1 or 0.5 mL of degassed DI water was added depending on the water-to-rock ratios to be studied. The bomblets were sealed in nitrogen gas to mimic an anoxic atmosphere and thoroughly shaken to ensure homogenisation. Up to two bomblets were inserted into the Parr reaction vessel per run. Twenty milliliters of degassed DI water was added to the vessel to ensure a reduced pressure differential across the bomblet walls. The reaction vessel was then heated to the appropriate temperatures in a muffle furnace for 72 h (Table 1). Samples hydrously pyrolyzed at 100°C and below were found to result in changes in lipid abundance that were smaller than the measurement errors associated with the solvent extraction process. Hence, these samples were pyrolyzed for a period of 1 month to ensure observable changes in lipid abundance before and after artificial maturation, a period that has historically been used in previous hydrous pyrolysis studies (Lewan, 1985; Lewan et al., 1986) and that is well beyond the time that changes in lipid abundance would have stabilized (Leif and Simoneit, 1995).
2.5. Pyrolysate extraction
The pyrolysates were extracted from the bomblets by using the method described in the work of Tan and Sephton (2020). A total of 5 μL of a 1.0 mg/mL 5β-cholanic acid and 0.5 mg/mL p-terphenyl mixture was added to the sample and used as an internal standard for quantification purposes. The sample was then concentrated in a rotary evaporator and dried by using anhydrous sodium sulfate.
The hydrous pyrolysates were then treated with boron trifluoride in methanol (BF3-methanol), as described in the work of Tan et al. (2018) to break any ester bonds present. This transesterification treatment liberated fatty acids and converted them to fatty acid methyl esters that were more amenable to gas chromatography-mass spectrometry (GC-MS) analysis. The hydrous pyrolysates were then further derivatized with 99-1 N,O-bis(trimethylsilyl) trifluoroacetamide-trimethylchlorosilane to silylate any remaining hydroxyl groups.
2.6. Gas chromatography-mass spectrometry
GC-MS analyses of the hydrous pyrolysates were conducted with an Agilent Technologies 7890 GC coupled to a 5973 MS. Injection (1 μL) was injected with a split ratio of 10:1, with helium carrier gas at a constant flow rate of 1.1 mL/min. Separation was performed on a J&W DB-5MS UI column (∼30 m in length, 0.25 mm internal diameter, and a film thickness of 0.25 μm). The GC oven temperature was held at 40°C for 2 min and then ramped at 5°C/min to 310°C, where it was held for 9 min. Mass spectra were acquired in the scan range (45–550 amu). Peak identification was based on retention time and mass spectra comparisons with authenticated standards and by reference to the NIST-08 mass spectral database.
2.7. Kinetic parameter modeling
The kinetic parameters of the degradation reaction can be derived from the ratio of lipid abundances before and after artificial maturation (Royle et al., 2018). Assuming first-order reaction kinetics, it is possible to derive the activation energy E (J/mol), and the Arrhenius constant A (s−1) from the linear form of the Arrhenius equation (Eq. 1), which relates the aforementioned parameters with the reaction rate constant, k (s−1), the absolute temperature T (K), and the gas constant R (J/mol/K).
The derived kinetic parameters of these organic degradation reactions include any additional effects promoted by the intersections between the organic matter and the mineral matrix. The preservation of organic biomarkers in these specific environments can then be modeled over geological time (Lewan, 1985; Hunt et al., 1991), and it can be specifically adjusted for martian geological time for any given Mars-relevant temperature profile (Royle et al., 2018).
2.8. X-ray diffraction
The mineralogy of both the NF and SF samples was characterized by XRD before and after pyrolysis. Samples were mounted in a flat holder, and powder XRD patterns were acquired by using a PANalytical X'Pert Pro MPD Alpha-1 X-Ray Diffractometer with an X'Celerator detector. Samples were analyzed between 5° and 80° 2θ under copper radiation with a step size of 0.033° at a speed of 5° per minute. Measurements were made at a current of 40 mA and a voltage of 40 kV. The diffraction patterns were analyzed with an X'Pert Highscore Plus program with references from the International Centre for Diffraction Data (ICDD) database.
3. Results
3.1. Lipid abundances
Figure 3a and b reveal that the solvent extracts of the unmatured ferrihydrite were found to be composed primarily of fatty acids, including saturated, unsaturated, and terminally branched species, consistent with their high abundance in bacterial cell membrane phospholipids (e.g., Harwood and Russel, 1984; Ratledge and Wilkinson, 1988; Killops and Killops, 2005). The saturated fatty acids were also observed to exhibit a clear even-over-odd predominance (EOP) pattern, which can be used to distinguish the observed lipid profile from non-biological organic matter. The suite of lipids found in these samples is consistent with the results of previous studies focused on lipid preservation in Mars-analog environments (e.g., Parenteau et al., 2014; Wilhelm et al., 2017; Tan et al., 2018).
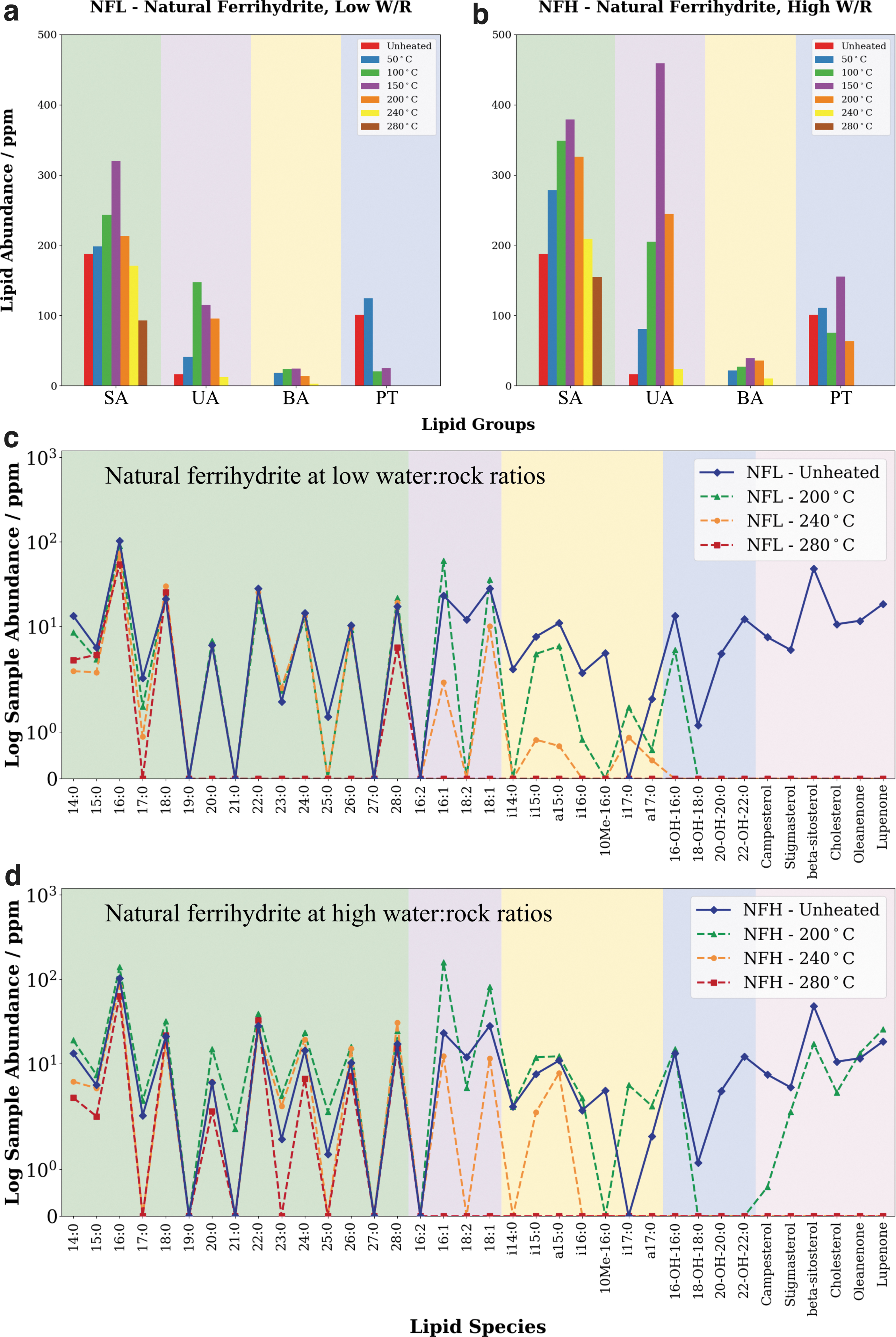
The variation of the lipid abundance per group of lipids for
Figure 3c and d indicate that the unheated NF sample (NF-Unheated) contains ω-hydroxy fatty acids. These molecules are key components of cutin (Kolattukudy and Walton, 1972; Soliday and Kolattukudy, 1977), and thus they are characteristic of higher order plant material. Cutin is formed from the inter-esterification of ω-hydroxy fatty acids; these esters bonds would have been cleaved during the derivatization process. Polycyclic terpenoids such as campesterol, stigmasterol, β-sitosterol, oleanone, and lupenone are also found in the ferrihydrite samples (Fig. 3c, d), and they are also characteristic of higher order plant material. Plants are unlikely to have evolved on Mars, but the observation of how these compounds are affected by simulated diagenesis can shed light on the evolution of other Mars-relevant polycyclic terpenes such as bacterial hopanoids during diagenesis in Mars-like conditions.
All the major lipid groups found in the solvent extracts of each sample were observed to respond strongly to changes in hydrous pyrolysis temperature (Fig. 3a, b). In general, solvent-extractable lipid abundances were found to increase with increasing hydrous pyrolysis temperatures until a peak abundance was reached at around 150°C, after which lipid abundance decreased rapidly with increasing hydrous pyrolysis temperatures. Individual lipid groups followed a similar pattern, with all major groups peaking in abundance between 100°C and 150°C. The exception to this was the polycyclic terpenoids observed in the lower water-to-rock ratio sample, which peaked in abundance at 50°C.
Changes in lipid abundance were investigated between 200°C and 280°C, as these are the temperatures at which the processes of diagenesis are most relevant (Eglinton and Douglas, 1988; Peters et al., 1990; Koopmans et al., 1995; Jaeschke et al., 2008). It was observed that almost all lipid species are destroyed in the presence of ferrihydrite during late-stage diagenesis (hydrous pyrolysis run at 280°C) regardless of lipid group or water-to-rock ratios (Fig. 3c, d).
Saturated fatty acids were observed to be the most resistant of all the major lipid groups. Saturated fatty acids that were retained also preserved their biogenic signatures in the form of an EOP pattern in carbon-chain lengths, consistent with previous hydrous pyrolysis studies of other Mars-analog samples (Tan and Sephton, 2020). In the 280°C hydrous pyrolysis run, only lipids remaining in the NF samples (natural ferrihydrite pyrolyzed at low water-to-rock ratios [NFL] and natural ferrihydrite pyrolyzed at high water-to-rock ratios [NFH]) were saturated fatty acids; 16:0, 18:0, and 28:0 were present in both samples, whereas 20:0, 22:0, 24:0, and 26:0 were present only in NFH. The lack of any of the other lipid groups at lower hydrous pyrolysis temperatures, and even some species of saturated fatty acids, highlights the deleterious effect of ferrihydrite.
Qualitatively, the preservation of lipids appeared to be slightly more favorable in water-dominated environments when pyrolyzed to temperatures between 200°C and 280°C (Fig. 4). This was most clearly seen in the unsaturated fatty acid fraction at 200°C, and the saturated fatty acid fraction at 280°C, where the high water-to-rock ratio samples (NFH) were found to have higher lipid abundances than the corresponding low water-to-rock ratio samples (NFL), and in some cases entire species of fatty acids were preserved compared with rock-dominated environments. The preservation bias of water-dominated environments lessened at higher pyrolysis temperatures, as all lipids were lost regardless of the degree of water activity.
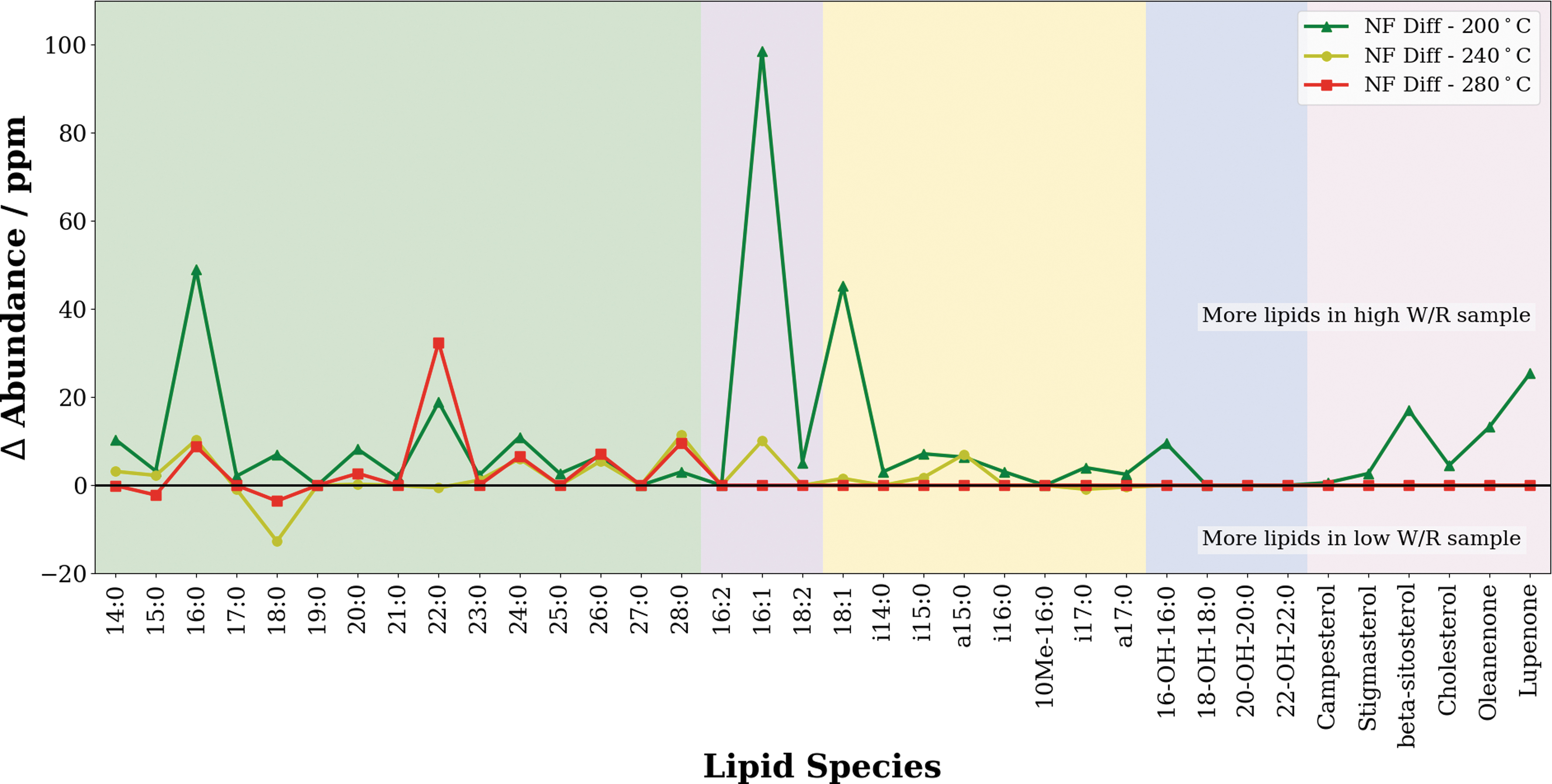
Graph showing the differences in abundance between individual lipid species in the lipid extracts when comparing the NFH and NFL across hydrous pyrolysis temperatures between 200°C and 280°C. A positive value indicates that more lipids of that species were present in NFH, whereas a negative value indicates that more lipids were present in NFL. Preservation appears to be slightly favored in circumneutral iron-rich environments with higher water availability. Note that most lipid species are lost by 280°C, and hence the difference between the two samples appears to be zero. Color images are available online.
3.2. Mineral transformation in the presence and absence of organic matter
It is well known that iron oxides undergo complex mineral transformations when exposed to increased temperatures and/or in the presence of organic matter (e.g., Cornell and Schwertmann, 2003). To investigate these changes, all samples were visually inspected for differences after each hydrous pyrolysis experiment. The XRD was also conducted on all samples.
The NF samples initially exhibited the characteristic ochre hue as observed at the sampling site at Silwood Park. After hydrous pyrolysis at temperatures ≤100°C, the color of the samples remained unchanged. At temperatures of 150°C or greater, the samples were observed to be pitch black on extraction from the bomblets, suggesting that a change in mineralogy had occurred between the temperatures of 100°C and 150°C.
The XRD analyses of the natural samples at both high and low water-to-rock ratios are shown in Fig. 5. The NF exposed to temperatures of 100°C or lower showed no observable changes compared with the initial samples, and it retained the two broad diffraction peaks that are indicative of poorly crystalline 2-line ferrihydrite (Cornell and Schwertmann, 2003). Diffraction peaks located at 30.14°, 35.50°, 43.15°, 53.54°, 57.04°, and 62.63° 2θ first appeared at 150°C, and they remained present up to 280°C. These peaks were identified as magnetite (Fe3O4), the appearance of which corresponded with the color change observed in the physical samples after extraction. Additional magnetite peaks at 18.30°, 37.11°, and 74.17° 2θ were observed in samples pyrolyzed to temperatures of 200°C and above.
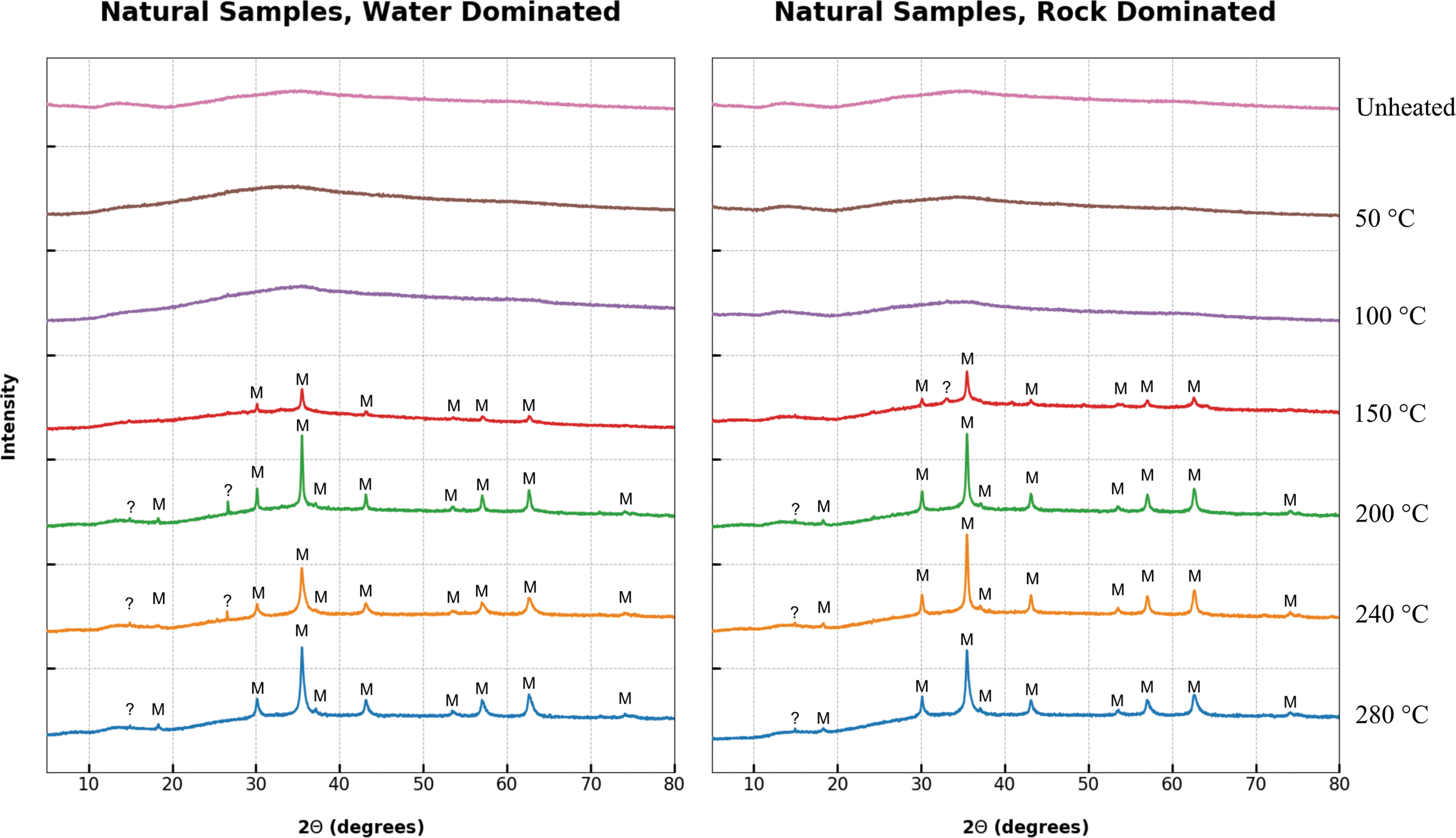
XRD spectra of unheated and pyrolyzed natural ferrihydrite samples under both rock- and water-dominated conditions. The data clearly show the transformation of ferrihydrite to magnetite (M) at 150°C regardless of water availability. Color images are available online.
The formation of well-defined peaks and the complementary disappearance of the two broad ferrihydrite diffraction peaks suggested that the ferrihydrite was transformed into crystalline magnetite. The appearance of additional magnetite peaks, as well as the sharpening and increasing intensity of pre-existing peaks, suggested that although magnetite transition commenced before 150°C, peak crystallinity was not achieved until 200°C, indicating a transitional temperature range between 100°C and 200°C. No further changes in mineralogy were observed at temperatures >200°C. Some unidentified peaks were present, but they did not correspond to other iron oxide or oxyhydroxide species.
A similar mineralogical change was observed in the SF samples, although this transformation occurred at a lower temperature. These dark brown precipitates were unchanged until hydrously pyrolyzed at 100°C, on which the samples were observed to have a bright orange coloration on extraction from the bomblets.
No magnetite was observed in the XRD spectra of the SF samples (Fig. 6). Instead, hematite (Fe2O3) diffraction peaks were observed at temperatures of 100°C and above, consistent with the color change observed in the hand specimen. Hematite was identified by clear diffraction peaks at 24.20°, 33.19°, 35.69°, 40.91°, 49.50°, 54.09°, 57.62°, 62.46°, 64.04°, 71.90°, and 75.49° 2θ. As with the natural samples, the loss of the broad diffraction peaks suggested that the ferrihydrite had been fully transformed into crystalline hematite—notably, this transformation had a lower transition temperature than the ferrihydrite–magnetite transition observed in natural samples. No goethite peaks were observed in either natural or synthetic samples. This suggested that organic matter not only provided a reducing environment in which magnetite transformation was favored, but it also inhibited the formation of hematite.
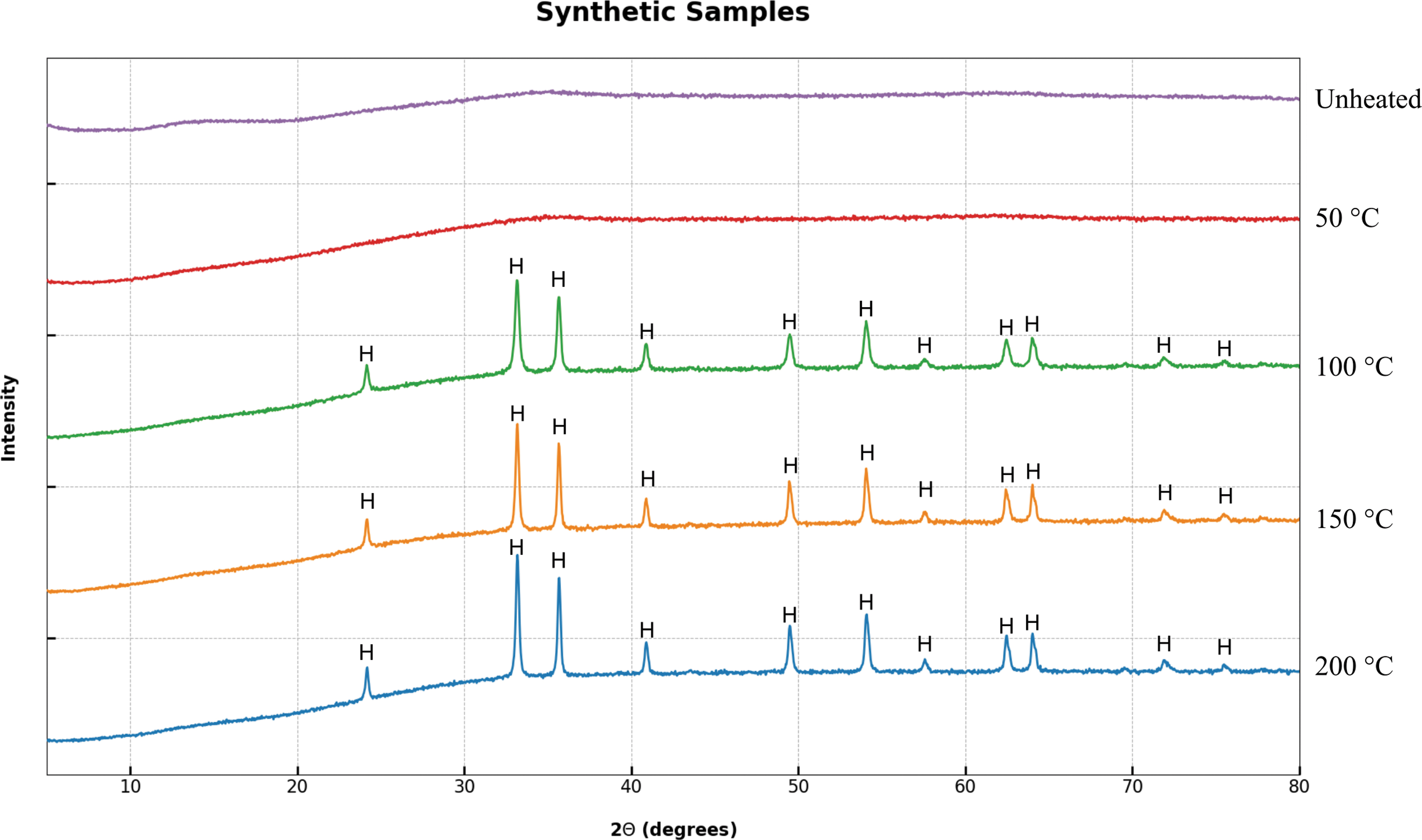
XRD spectra of unheated and pyrolyzed synthetic ferrihydrite samples. The data clearly show the transformation of ferrihydrite to crystalline hematite (H) at 100°C. Color images are available online.
3.3. Survivability of saturated fatty acids as predicted by kinetic modeling
Kinetic parameters related to lipid degradation were derived from the hydrous pyrolysis results. Specifically, the ratio of saturated fatty acid abundance before and after hydrous pyrolysis was chosen for analysis, as these lipids were observed to be the most resilient to degradation (Table 2). It was found that the lipid degradation reaction was well described with a pseudo-first-order approximation. Using the linear form of the Arrhenius equation, we derived the activation energy (E a) and the Arrhenius constant (also called the frequency factor) (A) for the NF under both high and low water activities (Fig. 7).
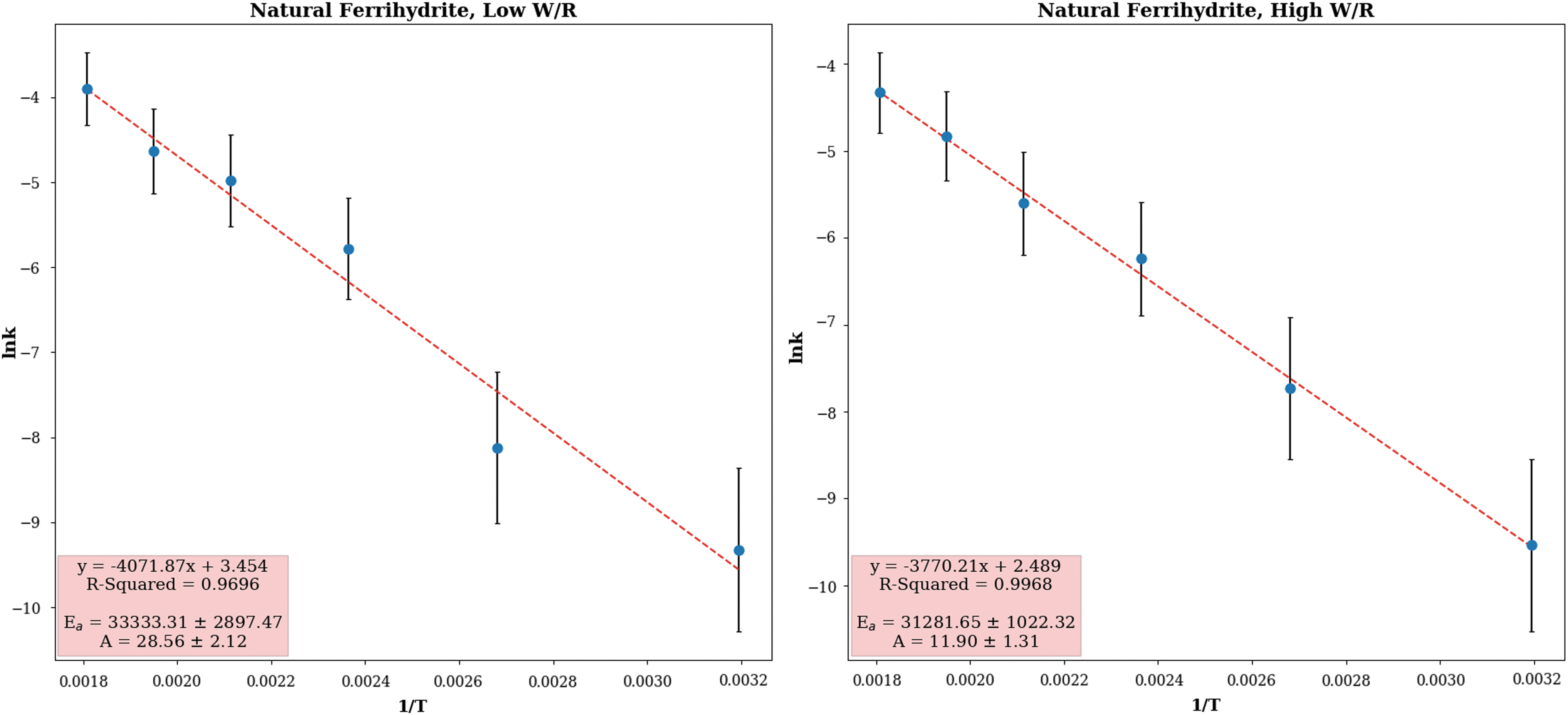
Kinetic parameters as determined by the linear form of the Arrhenius equation. All data points are well described with a linear trend with high confidence (R 2 > 0.95), suggesting that the pseudo-first-order approximation is valid for the lipid degradation reaction. k, rate constant; T, temperature. Color images are available online.
Data Used to Calculate the Kinetic Parameters Associated with Saturated Fatty Acid Degradation Under Varying Mineralogical and Water-To-Rock Conditions
C 0/C t refers to the ratio of initial and final concentrations of saturated fatty acids; k refers to the rate constant as calculated from C 0/C t assuming first-order reaction kinetics [k = ln(C 0/C t)/t, where t is the time of the experiment in hours]. Errors in ln k are from instrument uncertainty.
These kinetic parameters were used to model the loss of saturated fatty acids over geological time under Mars-relevant conditions (Royle et al., 2018; Tan and Sephton, 2020). The major assumptions of the model were constant geothermal gradients, surface temperatures, burial and exhumation rates, and a closed system. A temperature profile that represented the best-case scenario for the preservation of organic material was constructed and comprised a geothermal gradient of 0.008 K/m (present day geothermal gradient; Hoffman, 2001), a surface temperature of −50°C (present day average annual equatorial temperature), a burial rate of 10 m/Ma (average burial rates at Gale Crater; Borlina et al., 2015), and a maximum burial depth of 2 m. A more detailed description of how these values were chosen is found in the work of Royle et al. (2018).
The model data that represent saturated fatty acid degradation under ferrihydrite-rich Mars conditions are presented in Fig. 8, where t max is the time taken for all saturated fatty acids to be destroyed under the stated temperature and pressure constraints. A full description of how the model was constructed from kinetic parameter data is found in the work of Royle et al. (2018). In addition, the link to the freely accessible modeling code is available in the Supplementary Information. The results suggest that all saturated fatty acids would be destroyed within 1 ka under Mars-relevant conditions. Errors in the degradation profile were calculated with the non-linear least-squares method and plotted to show the uncertainties in degradation time, and they are presented as shaded areas in Fig. 8.
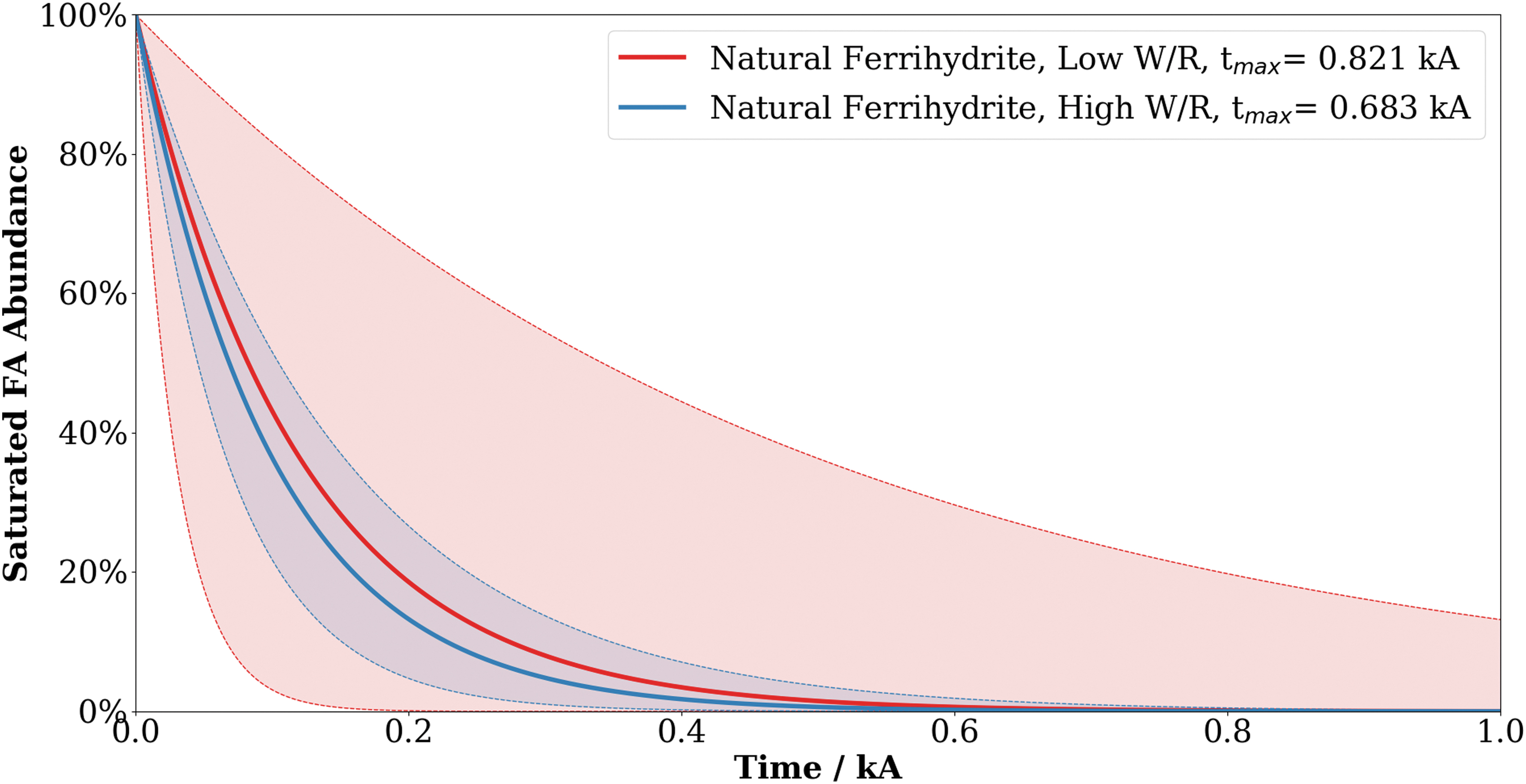
Model data showing the degradation of saturated fatty acids over geological time under martian surface conditions, with projections derived from the calculated activation energy and Arrhenius constant. Red and blue shaded areas show the uncertainty associated with the timing of degradation of organic matter under low and high water availabilities, respectively. t max values indicate the time at which all fatty acids are lost. At optimal conditions, all saturated fatty acids are modeled to be destroyed within 1 ka, and water availability has no significant effect on degradation rate. Color images are available online.
4. Discussion
4.1. Organic matter–mineral relationships
Our results suggest that changes in the mineralogy of the iron substrates strongly affect the organic matter–mineral interactions at the surface of the substrate. This is most clearly seen when visualizing the variation in free lipid abundance as the mineralogy of the iron substrate changes (Fig. 9). Between 25°C and 100°C, the ferrihydrite remains amorphous and undergoes no mineralogical changes, whereas the free lipid extract is observed to increase for all lipid groups except for polycyclic terpenoids, which are the most structurally complex and thus most susceptible to degradation. A possible explanation for this phenomenon is the breaking of adsorptive bonds between the sorbed fatty acids and the ferrihydrite substrate via hydrolysis as pyrolysis temperatures increase. This mechanism is likely primarily driven by temperature, hence the increasing abundance of solvent extractable organic matter as temperatures increase. This behavior was also reported in Tan and Sephton (2020), where increased abundances of lipids were observed post-maturation.
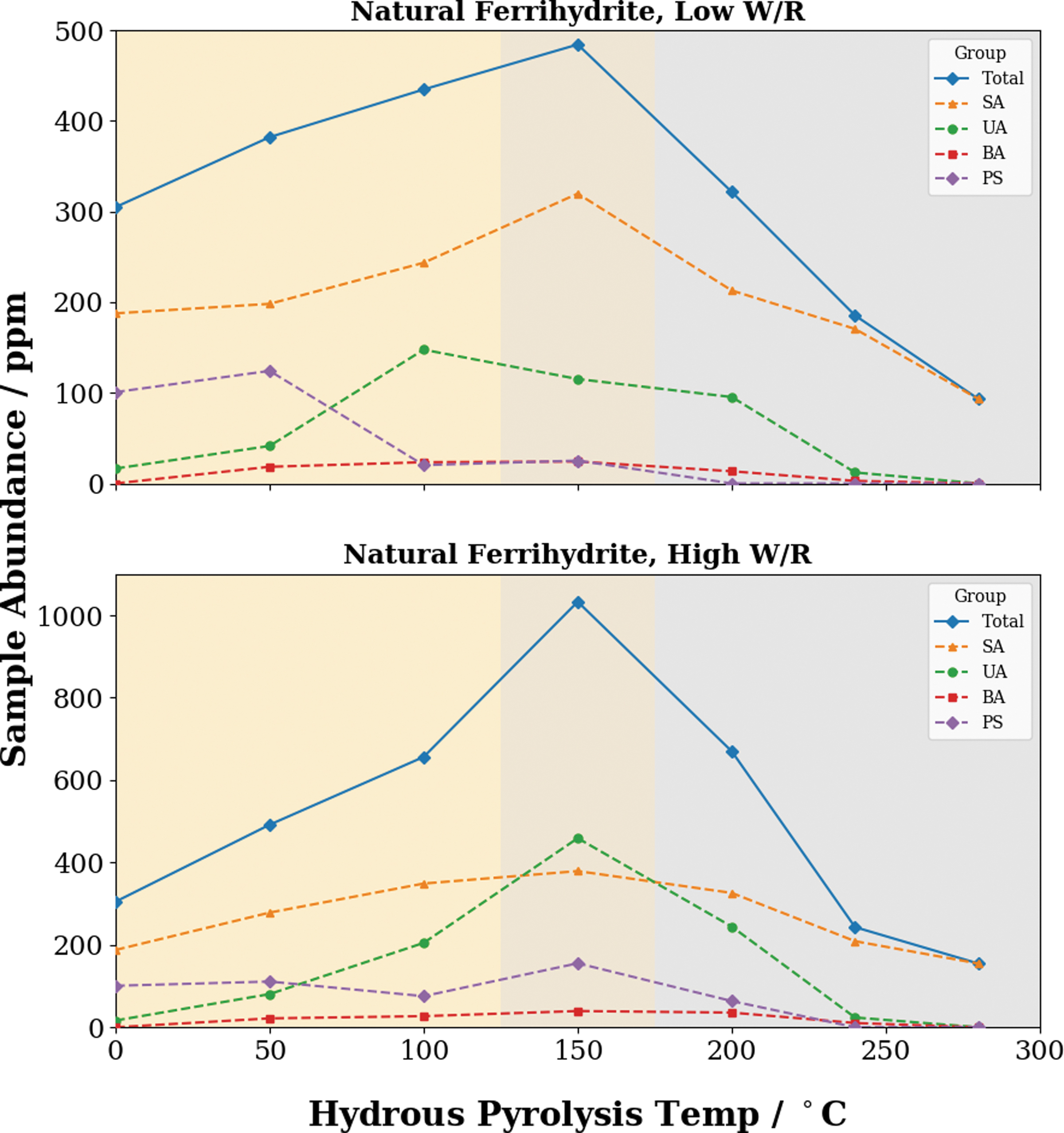
The variation of abundance of the free lipid extract with increasing pyrolysis temperature. The different-colored backgrounds indicate the mineralogy of the iron oxides; orange indicates ferrihydrite, whereas gray indicates magnetite. Recall that at 150°C magnetite had formed but not yet achieved full crystallinity and is colored slightly darker to reflect this. Color images are available online.
At 150°C, the ferrihydrite transforms into magnetite but it does not reach peak crystallinity (Fig. 5). This mineralogical change is accompanied by a sharp increase in the abundance of several lipid groups, including saturated and unsaturated fatty acids (Fig. 9). The release of sorbed materials from ferrihydrite as it undergoes phase transformations has been observed in other compounds such as arsenic (Pedersen et al., 2006), primarily as a result of the surface area of the ferrihydrite becoming too small to retain all the sorbed material (Schikorr, 1933; Booy and Swaddle, 1978; Cornell and Schwertmann, 2003). Organic material entombed within the iron precipitates represents another potential source of solvent-soluble organic matter. Phase transitions in the iron precipitates would likely have released any occluded organic material, rendering them susceptible to solvent extraction. These results suggest that lipids are not necessarily destroyed by the ferrihydrite–magnetite transition and may instead be merely desorbed.
At temperatures between 200°C and 280°C, the magnetite becomes fully crystalline and the abundance of all lipid groups decreases with increasing temperature until all lipids are lost except for the saturated fatty acids. The clear redox effect as ferric iron in ferrihydrite is transformed to ferrous iron in magnetite, complementary with the loss of organic matter as it is lost due to oxidation, suggests that the primary mechanism by which organic degradation occurs is an oxidative reaction. This is further supported by the lack of alkanes in both the unheated samples and the post-hydrous pyrolysis solvent extracts, suggesting that either decarboxylation is not a significant process during degradation, or that all alkane products were subsequently oxidized, or both.
Our data also suggest that, among the lipid groups, saturated fatty acids are the best targets for life detection missions, as they are the most resistant to degradation and can retain their biogenic EOP signatures, an important quality of potential biosignatures that help distinguish them from abiotic carbon. The suitability of saturated fatty acids as a lipid biosignature is a finding that is consistent with other studies (Tan et al., 2018).
4.2. Iron mineral phase transformations in the presence of organic matter
The presence or absence of organic matter results in significant mineralogical differences when ferrihydrite is heated at anoxic conditions and low water availabilities. In the absence of organic matter, hematite is the dominant iron oxide species, consistent with the results of ferrihydrite evolution at high temperatures as investigated by other authors (e.g., Cornell and Schwertmann, 2003; Dehouck et al., 2017).
In contrast, the natural samples containing a high concentration of organic matter were transformed from amorphous ferrihydrite to crystalline magnetite. Unlike most other iron oxide species, magnetite contains both divalent and trivalent iron cations. For this phase transformation to occur, organic matter must act as the reductant and be oxidized, resulting in the complementary partial reduction of ferrihydrite to magnetite (Booy and Swaddle, 1978; Blesa and Matijević, 1989). These results are consistent with previous work on the heating of ferrihydrite in the presence of organic matter, with magnetite being the sole iron oxide formed as long as excess reductant was present; in samples with insufficient reductant, ferrihydrite was initially reduced to magnetite, and subsequently the ferrous iron present in magnetite was fully oxidized to hematite or maghemite (Campbell et al., 1997; Bishop et al., 2006, 2008).
The lack of hematite or maghemite in the post-maturation NF samples indicates that the OM:Fe ratio was greater than the threshold at which magnetite transforms into either of those iron oxide phases. On Mars, where the biomass of any microbial community may have been limited, the final iron phase may instead be maghemite—the natural oxidation product of magnetite, sharing its structural form while possessing only Fe(III) ions (Fasiska, 1967; Cornell and Schwertmann, 2003; Bishop et al., 2008).
In addition to the reducing effect of organic matter, the results suggest that the presence of organic matter also inhibits the transformation of ferrihydrite into hematite. In the absence of organic matter, SF is transformed into hematite at 100°C. In the presence of organic matter, however, ferrihydrite remains stable past 100°C, and instead it is only transformed into magnetite at 150°C.
The inhibitory effect of organic acids on ferrihydrite crystallization has been noted earlier: Ferrihydrite formation in soils is stabilized by the presence of organic matter, and both the concentration and type of organic acids have been shown to have a strong effect on ferrihydrite crystallization (Schwertmann, 1966). Carboxylic acids reduce the rate of crystallization of the iron oxide products as well as inhibit the formation of goethite in favor of hematite, whereas hydroxy fatty acids completely inhibit the crystallization of ferrihydrite (Schwertmann, 1970; Cornell, 1979; Kandori et al., 1992). It has been suggested that the adsorption of organic acids onto the surface of the amorphous ferrihydrite prevents crystallization from occurring through the formation of organic “bridges” that link multiple ferrihydrite particles (Cornell, 1979).
On Mars, it is unlikely that ferrihydrite deposits would be transformed into other iron oxides by purely diagenetic processes in low-temperature conditions. The presence of stable ferrihydrite dating back to the deposition of the Sheepbed mudstone suggests that ferrihydrite is stable on the surface of Mars and does not oxidize to hematite or magnetite even under the highly oxidizing martian surface conditions (Vaniman et al., 2014; Treiman et al., 2016; Dehouck et al., 2017). A potential conclusion is that outcrops containing extensive hematite derived from ferrihydrite precursors may not be good targets for astrobiological missions, as this would indicate a lack of organic material that would have prevented hematite recrystallization.
4.3. Implications of kinetic modeling
The nature of organic degradation in the subsurface of these ferrihydrite-rich environments was investigated by kinetic modeling. Our data show that the preservation potential of solvent-extractable organic matter in these iron-rich sediments is extremely poor, with all saturated fatty acids lost within 1–3 ka, even under optimal conditions. Current understanding of the hydrological and geological history of Mars suggests that the latest period during which the martian surface was habitable was 3 Ga ago, during the Noachian and Hesperian (Cockell, 2014; Westall et al., 2015). The results of the kinetic modeling, thus, indicate that any ancient circumneutral iron-rich environments would not be able to preserve any deposited solvent-extractable molecular biosignatures to the present day.
The model also suggests that in iron-dominated environments, the kinetics of the oxidation reaction is purely thermally driven. Figure 7 shows that the same kinetic parameters can be applied at all temperatures, regardless of mineralogical transformations, iron mineralogy, and water availability. This was surprising, as current understanding would suggest that iron mineralogy and transformations should have an effect on degradation rate, as the amorphous ferrihydrite would confer the protective qualities associated with the formation of absorptive bonds and organo-metallic complexes (e.g., Lalonde et al., 2012). These protective qualities would eventually be lost at higher temperatures, as lipids are released from the surface of the ferrihydrite by thermal agitation, as well as by the restructuring of ferrihydrite as it is transformed into magnetite. Similarly, water availability should influence lipid degradation, as low water-to-rock ratios have been shown to heighten both protective and degradative effects of the mineral matrix in pyrolysis experiments (Tannenbaum and Kaplan, 1985; Koopmans et al., 1998).
Instead, our results suggest that the breaking of adsorptive bonds between the fatty acids and the iron surfaces via thermal agitation and/or the release of lipids during mineralogical transformations does not necessarily make the lipids more kinetically susceptible to oxidation. They also suggest that neither changes in oxidation state and surface chemistry between ferrihydrite and magnetite, nor differing water availabilities significantly contribute to the overall loss of organic matter in these samples (Fig. 8). Although this cannot be extrapolated to all iron oxide species, our results imply that the rate of degradation in environments dominated by iron oxides/oxyhydroxides is primarily a function of temperature rather than of organic matter–iron mineral interactions and/or water availabilities.
As with all models that attempt to simplify and predict the complexities of the natural environment, it is important to understand the full extent of assumptions made in the construction of this model (an exhaustive list is presented in Tan and Sephton, 2020). In brief, the model assumes a closed system environment, and thus does not account for any open system processes such as the influence of hydrothermal and diagenetic fluids, which are likely to have existed during early Mars (Duda et al., 2018). In the case of hydrothermal systems, other preservative mechanisms associated with the hydrothermal environment, such as permineralization, may allow for better survivability of biosignatures than would be expected from this study (e.g., Cady and Farmer, 1996; Farmer and Des Marais, 1999; Phoenix et al., 2000; Konhauser et al., 2001; Ferris and Magalhaes, 2008).
The model also makes no attempt to determine whether any surviving organic matter can be distinguished from abiotic sources of carbon such as meteoritic material, or organic matter produced from Fischer-Tropsch-type reactions; these materials and processes can produce saturated fatty acids (Chyba and Sagan, 1992; McCollom and Seewald, 2006; Summons et al., 2008; Sephton, 2012; Mißbach et al., 2018).
It is important to emphasize that this simulation only accounts for lipids that are extractable when using wet chemistry, that is, soluble organic matter, and does not preclude the survival of insoluble macromolecular organic matter such as kerogens, which have been shown to be present on Mars (Eigenbrode et al., 2018). Other studies (e.g., Watson and Sephton, 2015) have shown that iron oxides can aid the preservation of organic matter in the form of macromolecular material via catalytic oxidative polymerization. Recent experiments have also shown that macromolecular organic matter can survive artificial maturation and be detected by using thermal extraction techniques such as pyrolysis-gas chromatography-mass spectrometry (Tan et al., 2020). If preparative chemistry steps, such as alkali/acid leaching, are taken before analysis, the macromolecular organic matter detected by flash pyrolysis can also reveal diagnostic biogenic information (Tan et al., 2020).
Regardless of the process by which the saturated fatty acids were lost, the current study suggests that evidence of extinct life would not be preserved in a solvent-extractable form and advocates that purely iron-dominated environments should not be the targets of life detection methods that are reliant on solvent extraction techniques.
4.4. Quantitative comparisons to other Mars-analog environments
One of the primary benefits of kinetic modeling derived from hydrous pyrolysis experiments is that it provides an experimental standard by which the preservation potential of different Mars-analog environments can be quantified. This allows for a ranking of Mars-analog deposits in terms of which environment is the most likely to preserve any detectable evidence of organic matter.
We compared the preservation potential of iron-rich circumneutral deposits with iron-rich acid deposits by using previous work that utilized the same hydrous pyrolysis/kinetic modeling experiments (Table 3) (Tan and Sephton, 2020).
Quantitative Comparisons Between the Preservation Potentials of Different Mars-Analog Environments Based on Kinetic Data
The PPF was calculated by normalizing the time taken for saturated fatty acids to be fully degraded under martian surface conditions to a standard value of 1 ka. The PFF values for acidic deposits are taken from Tan and Sephton (2020).
PPF = preservation potential factor.
In Table 3, the preservation potential factor of each Mars-analog environment was based on the expected time it takes for all saturated fatty acids to be destroyed under best-case scenario martian surface conditions by normalizing the values to 1 ka. This allows us to gain quantitative insights into the relative preservation potential of various Mars-analog environments.
Under these parameters, the results suggest that the preservation potential of circumneutral iron-rich deposits is similar to that of iron-rich acid deposits that contain a minor clay component. However, it can be clearly seen that it is much more desirable to target acidic iron-rich deposits that do not contain a clay component for sample selection, as solvent-extractable organic matter is expected to survive between 20 and 100 times longer in those sediments compared with the other Mars analogues that have been studied.
These quantitative results are consistent with previous work, which has shown that clay content catalyzes the breakdown of solvent-soluble organic matter (Tan and Sephton, 2020 and references therein). In addition, circumneutral pH favors the precipitation of amorphous ferrihydrite compared with the more crystalline goethite or hematite that is favored in acidic conditions (Dehouck et al., 2017). The increased surface area of ferrihydrite compared with goethite or hematite leads to a more deleterious environment, resulting in a lower preservation potential.
4.5. Applications to Mars
This study has shown that sediments where ferrihydrite or magnetite are the only mineral phase, or where these iron oxides dominate the mineralogy, are poor targets for wet chemistry GC-MS experiments. Both ferrihydrite- and magnetite-rich sediments are found to be deleterious to organic matter due to the oxidizing power of ferric iron within their structures and they have the same oxidative effect on organic material regardless of provenance, organic matter–mineral interactions, and water availability.
Although our work highlights the poor long-term preservation potential for organic matter in iron-rich environments, it should be noted that iron oxides on Mars are not as widespread as previously believed; the mineralogical data collected by the Chemistry and Mineralogy (CheMin) instrument on the MSL Curiosity rover show that hematite abundance is lacking in a majority of the analyzed mudstone samples (e.g., Bish et al., 2013; Vaniman et al., 2014; Treiman et al., 2016; Rampe et al., 2017; Morris et al., 2019). However, even within the subset of iron-dominated environments on Mars, our results suggest that circumneutral iron-rich deposits should be deprioritized in favor of iron- and sulfur-rich acid deposits when searching for evidence of life with solvent extraction techniques.
The organic matter–mineral interactions revealed by this study can shed light on processes that may be occurring on the present-day surface of Mars, specifically in environments where iron phases contribute to, but do not dominate, the overall mineralogy of the rock. As indicated by the low-temperature maturation experiments, lipids are adsorbed to the surfaces of amorphous iron phases and are only liberated at higher temperatures. In environments such as those observed in Gale Crater, where ferrihydrite has been shown to be a stable, amorphous iron phase but does not dominate the mineralogy of the analyzed mudstones (Vaniman et al., 2014; Treiman et al., 2016; Dehouck et al., 2017), fossil lipids may still remain adsorbed to the amorphous iron phase, especially as organic matter desorption has been shown to be highly unfavorable over long timescales (Gu et al., 1995). Lipids preserved in this way may be more widespread than expected due to the surprising resilience of martian ferrihydrite to short-lived aqueous events, as observed in laboratory simulations (Dehouck et al., 2017).
In cases where ferrihydrite is buried to depths that are shallow enough to retain its stability, the slight increase in temperature would result in the desorption of lipids, rendering them extractable by wet chemistry experiments. This opens a “preservation window” for low-temperature ferrihydrite environments, where lipid biosignatures are preserved and are extractable before being oxidized during interactions with the iron oxide substrate.
The degradation of organic matter in magnetite-rich environments is also relevant to a wide variety of martian environments. Short-lived post-diagenetic hydrothermal fluids or events, such as impact-generated hydrothermal systems, can result in temperatures that are high enough to promote the ferrihydrite–magnetite phase transition (Ames et al., 1998; Osinski et al., 2013). A similar scenario can occur if a ferrihydrite deposit is buried to sufficient depth, where temperatures can be ≥150°C depending on the circumstances (Borlina et al., 2015). In these cases, ferrihydrite can be transformed to magnetite within an extremely short timeframe (within 72 h). Phase changes can also be caused by the effects of radiative oxidation (Cairns-Smith, 1978; Braterman et al., 1983). As evidenced by the poor preservation potential of magnetite regardless of provenance, these conditions are deleterious to extractable organic matter preservation and should not be considered as sites for future astrobiological missions aimed at using wet chemistry techniques to detect signs of life.
However, magnetite and maghemite derived from the reduction of ferrihydrite could be potential mineralogical biomarkers. Magnetite and/or maghemite formation via this pathway can only occur at elevated temperatures in the presence of water and a reductant such as organic matter. Maghemite has been previously proposed as an astrobiology indicator (Bishop et al., 2008), but these studies drew their conclusions from the heating of synthesized ferrihydrite in the presence of a reducing agent (Campbell et al., 1997; Bishop et al., 2008) rather than the more appropriate aqueous heating of natural terrestrial analogues as is the case in this study.
Although these minerals would contain little diagnostic information about the organic matter (as any organic material would have been consumed as the reductant in this redox reaction), there may be other biogenic signatures associated with the deposit, such as structural biomarkers formed by iron encrustations (Emerson et al., 2010; Williams et al., 2017). Potentially diagnostic insoluble macromolecular organic compounds have been shown to survive simulated diagenesis in iron- and sulfur-rich acid stream environments (Tan et al., 2020); such compounds are likely to survive diagenesis in circumneutral iron-rich deposits as well, and they would be a good target for thermal extraction techniques.
However, it must be emphasized that magnetite found in association with ferrihydrite may have formed via a variety of geochemical pathways that do not require an organic matter reductant; it is now widely accepted that magnetite in the Sheepbed mudstone is formed from the alteration of olivine during the authigenic formation of clay minerals: olivine + Al + oxidizing fluids → saponite + magnetite (McLennan et al., 2014; Vaniman et al., 2014). Magnetite observed in the Murray formation has been interpreted to be the result of the disproportionation of ferrous hydroxide [Fe(OH)2] precipitated under anoxic, alkaline conditions (Schrauzer and Guth, 1976; Hurowitz et al., 2017), or the reduction of ferric iron by dissolved inorganic ferrous-iron complexes (Stumm and Sulzberger, 1992; Hurowitz et al., 2017).
Similarly, maghemite may have been formed by the high-temperature oxidation of magnetite (Cornell and Schwertmann, 2003), the dehydroxylation of lepidocrocite (anhydrous heating at 200°C) (Morris et al., 1998; Cornell and Schwertmann, 2003), the oxidation of Fe(II) complexes in solution (Stumm and Sulzberger, 1992; Cornell and Schwertmann, 2003), the reduction of ferric oxide phases such as ferrihydrite (Cornell and Schwertmann, 2003), or the heating of phosphate-rich ferrihydrite (Barron and Torrent, 2002).
In summary, magnetite and maghemite can be potential mineralogical markers for locations that once contained organic material, especially if there is evidence that they were derived from ferrihydrite precursors and were exposed to elevated temperatures in the presence of water. However, the sheer variety of formative pathways for magnetite and maghemite makes their use as mineralogical biomarkers that are at best limited.
5. Conclusions
Circumneutral, anoxic, iron-rich bogs dominated by ferrihydrite precipitates can support microbial communities in settings that are geochemically, and potentially biologically, similar to Noachian Mars. In this study, we investigated the preservation of lipid biosignatures in one such environment to understand the factors affecting the preservation potential of lipids, as well as organic matter–mineral interactions that underpin the diagenetic processes that control preservation in these environments.
We find that the organic matter–mineral interactions in these iron-rich environments are dominated by the changes in the mineralogy of the iron substrate. As temperature increases, lipids are desorbed from the surface of the ferrihydrite substrate, with peak desorption occurring when ferrihydrite transforms into magnetite, and the surface area of the crystalline iron oxide cannot retain all sorbed material. At the same time, the presence of organic matter was found to influence the transformation products of ferrihydrite; synthetic, organic-poor ferrihydrite was transformed into hematite, whereas natural, organic-rich ferrihydrite inhibited this process and instead transformed into magnetite at higher temperatures. Inorganic–organic interactions showcase the potential for minerals to be indicators of rocks that once contained organic matter; maghemite may be one such residual indicator of an initial mixture of organic matter and iron minerals.
Our results also confirm that saturated fatty acids were the most resistant subset of lipids, consistent with other Mars-analog studies. However, it was also shown that iron-dominated environments are poor targets for wet chemistry-based life detection techniques on Mars, with kinetic modeling indicating that lipids are expected to be completely oxidized on thousand-year timescales, regardless of water availability or specific iron oxide mineralogy.
By comparing our results with previous work, we find that circumneutral iron-rich deposits are less favorable for the preservation of solvent-extractable organic matter compared with other iron-rich deposits, such as acid streams. Future work in which this technique is used will allow us to quantify the relative preservation potential of solvent-extractable organic matter in other Mars-relevant environments and expand the catalogue of potential targets for sample selection strategies on Mars.
Footnotes
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This work was supported by the United Kingdom Space Agency grant ST/N000560/1 and an Imperial College President's PhD scholarship granted to J.T. The code used for kinetic parameter modeling is freely available at:
Abbreviations Used
Associate Editor: Kathleen Campbell
