Abstract
The organic record of Earth older than ∼3.8 Ga has been effectively erased. Some insight is provided to us by meteorites as well as remote and direct observations of asteroids and comets left over from the formation of the Solar System. These primitive objects provide a record of early chemical evolution and a sample of material that has been delivered to Earth's surface throughout the past 4.5 billion years. Yet an effective chronicle of organic evolution on all Solar System objects, including that on planetary surfaces, is more difficult to find. Fortunately, early Earth would not have been the only recipient of organic matter–containing objects in the early Solar System. For example, a recently proposed model suggests the possibility that volatiles, including organic material, remain archived in buried paleoregolith deposits intercalated with lava flows on the Moon. Where asteroids and comets allow the study of processes before planet formation, the lunar record could extend that chronicle to early biological evolution on the planets. In this study, we use selected free and polymeric organic materials to assess the hypothesis that organic matter can survive the effects of heating in the lunar regolith by overlying lava flows. Results indicate that the presence of lunar regolith simulant appears to promote polymerization and, therefore, preservation of organic matter. Once polymerized, the mineral-hosted newly formed organic network is relatively protected from further thermal degradation. Our findings reveal the thermal conditions under which preservation of organic matter on the Moon is viable. Key Words: Moon—Regolith—Organic preservation—Biomarkers. Astrobiology 15, 154–168.
1. Introduction
1.1. Organic records in the Solar System
T
The Moon may provide us with this archive (Armstrong et al., 2002; Crawford et al., 2008; Joy et al., 2012; Burchell et al., 2014b). Major surface volcanic activity on the Moon ceased around 3.1 Ga (Wilhelms et al., 1987). The Moon has essentially no atmosphere and is largely anhydrous. It is also a relatively large body (3475 km diameter), causing it to readily attract material owing to its gravitational pull. These features make it a potentially ideal witness to the organic chemical processes occurring close to Earth in the early Solar System. The surface of the Moon is largely covered by a few meters to kilometers thickness of regolith, a fine-grained rock powder developed by impacting material and space weathering (Lucey et al., 2006). After the return of the Apollo lunar samples, a number of studies assessed the lunar materials for the presence of organic matter (e.g., Abell et al., 1970; Burlingame et al., 1970; Nagy et al., 1970). Small quantities of carbon were detected (<200 ppm total C, Burlingame et al., 1970; Ponnamperuma et al., 1970). The indigenous nature of detected organic compounds was uncertain. Measurements by Brinton and Bada (1996) quantified the level of free amino acids at around 15 ppb; however, some of the detected compounds were attributed to terrestrial contamination based on enantiomeric ratios. More recent studies in which highly sensitive modern instruments were used have indicated the presence of organic compounds in lunar samples that are not of terrestrial contamination origin but may have come from added meteoritic material (Thomas-Keprta et al., 2014). A lunar impact melt breccia returned from the Moon by Apollo 17 (sample 72255) has been shown to contain graphitic carbon that may have formed from condensation of vaporized C-rich impactors (Steele et al., 2010). Understanding how these carbonaceous materials were formed, and modified, would allow us to test proposed models of preservation. The presence of well-preserved records on the Moon may be crucial in illuminating the behavior and chemical evolution of organic matter in the early Solar System.
1.2. Sources of organic matter to the Moon
Organic matter is prevalent throughout the Solar System, and the possibility of the presence of indigenous organic matter on the Moon has been considered for some time (e.g., Sagan, 1960). In addition to exogenous delivery, organic compounds may be generated in situ by the action of radiation on ices (e.g., Moore et al., 1983; Bernstein et al., 1995; Lucey, 2000; Crites et al., 2013). Carbon may also be directly implanted in the regolith from the solar wind (Bibring et al., 1974; Pillinger and Eglinton 1977; Haskin and Warren, 1991). There would have been a number of sources that could have delivered organic and volatile material during the early history of the Earth–Moon system. Models have shown that enstatite and ordinary chondrite-type bodies would have been the dominant large impactor types during the Late Heavy Bombardment (Bottke et al., 2012; Morbidelli et al., 2012). These meteorite types are known to contain organic material (e.g., Alexander et al., 1998; Quirico et al., 2003; Sephton et al., 2004a). There would also have been a high flux of micrometeorites and dust particles at this time, dominated by carbonaceous material (Court and Sephton, 2014). CI carbonaceous chondrites can contain >3% carbon (Botta and Bada, 2002), and interplanetary dust particles can contain several times this amount (Thomas et al., 1994). Another potential source of organic matter would have been from the surfaces of Mars, Earth, and other planets, as large impacts would have ejected fragments of surface material into space at velocities sufficient to escape the gravitational pull of the planets (Armstrong et al., 2002; Armstrong, 2010). Ejected fragments of planetary surfaces could potentially have contained organic matter, including biological material. The potential for preservation of the earliest biotic and prebiotic material in terrestrial or martian meteorites is a key motivation for exploration of the Moon (Crawford et al., 2008; Burchell et al., 2014b). Recent experimental work has shown the ability of organic biomarkers to survive the impact shock conditions that would have ejected this material from the planetary surface (e.g., Parnell et al., 2010; Burchell et al., 2014a). The total amount of terrestrial material at the lunar surface today has been estimated as 1–2 ppm; however, the distribution of impacts will be non-uniform, and some areas of the surface may have received several times more or less than this amount (Armstrong, 2010). The proportion of organic matter in the ejected terrestrial material is unknown; however, assuming organic-poor rocks with 1% organic matter or less would result in parts-per-billion levels of terrestrial organic matter in the lunar regolith.
1.3. Survival of infalling material
1.3.1. Impact at the lunar surface
Organic material within an ejected object traveling toward the lunar surface must survive the initial impact in order to be preserved. With a negligible atmosphere to provide deceleration, impacting bodies strike the lunar surface at high velocity, producing high temperatures and great levels of shock. The lack of atmosphere means that a fusion crust will not be developed on the meteorite by ablation; however, it has been suggested that material ejected from the surface of Earth may develop a fusion crust on its passage out of the atmosphere (Gutiérrez, 2002). The high temperatures generated during atmospheric ablation of stony meteorites falling to Earth are restricted to the outermost portion of the meteorite and do not penetrate to significant depth. The detection of organic compounds in carbonaceous chondrite meteorites and experimental work with artificial meteorites have shown that organic compounds and biomarkers are capable of surviving infall to Earth (Sears, 1975; Parnell et al., 2011). It might therefore be expected that organic compounds would survive heating by ablation in materials ejected from Earth's surface.
Micrometeorites impacting the lunar surface are vaporized, and the vapor subsequently condenses onto grain surfaces (Keller and McKay, 1997). The high impact temperatures generate glass-bound agglutinates that are abundant in the lunar regolith (e.g., McKay et al., 1991). However, a fraction of impacting material can survive. Direct evidence for survival following impact comes from a small number of meteorites recovered from regolith samples collected during the Apollo missions (e.g., Joy et al., 2012 and references therein). One important example is the Bench Crater meteorite, recovered by Apollo 12. This millimeter-sized object was identified as a carbonaceous chondrite, and the hydrated mineral phases remain preserved (McSween, 1976; Zolensky et al., 1996; Zolensky, 1997). Bench Crater provides encouragement that carbonaceous materials in meteorites may be identifiable and characterizable in situ on the Moon (see also Joy et al., 2012). Terrestrial meteorites would have had a lower range of impact velocities than asteroidal or cometary fragments. Armstrong (2010) calculated terrestrial meteorite impact velocities as typically occurring at ∼3 km s−1, with a substantial proportion of impactors having relatively low vertical impact velocities. Computer modeling has shown that portions of larger impactors (0.125 m3) experience relatively moderate conditions upon impact within the range of 2.5–5 km s−1, well within the survivability window for many biomarkers (Crawford et al., 2008; Burchell et al., 2010).
1.3.2. The lunar regolith environment
Despite the absence of tectonics, biodegradation, and other destructive agents that are present on Earth, the lack of an atmosphere on the Moon means that its surface is subjected to space weathering (e.g., Lucey et al., 2006). Space weathering constitutes impacts from micrometeorites and larger impactors (comminution) and irradiation from the solar wind, energetic solar events, and galactic cosmic rays. The radiation causes sputtering on grain surfaces, and the more energetic particles can generate a cascade of particles within the regolith, which alters the mineral grains. It has been shown that ionizing radiation can change the structure of organic matter by formation of free radicals during radiolysis, causing aromatization, a reduction in the degree of alkylation of aromatic units, and polymerization of smaller organic molecules into larger aromatic networks (Court et al., 2006). For fragile materials to be preserved over billion-year timescales, some mechanism is required to protect them from the harsh radiation and impact environment; processes related to lunar lava flows may provide one such mechanism.
1.4. Preservation of the lunar regolith
The continual process of lunar regolith formation is slow (2–5 mm Myr−1 over the last 3.5 billion years; e.g., McKay et al., 1991) and subjects materials to repeated vertical and lateral reworking and direct surface exposure. Given the current rate of overturn, it would take a considerable length of time to build up a thickness of regolith sufficient to completely protect material from ionizing radiation and impacts. Therefore, a more rapid mechanism of burial is required. Burial under an ejecta blanket or an impact melt flow from a nearby large impact is one possible method, but identification of discrete regolith horizons within the layers of pulverized rock and mixed regolith material would be a challenging task. A recently proposed alternative method of rapid burial is by lava flows (Crawford et al., 2010; Fagents et al., 2010; Rumpf et al., 2013). In the lava flow model, a layer of regolith that has accumulated on an exposed rock surface is covered by lava, which quickly cools. Subsequent space weathering develops a new regolith layer on the lava surface, which in turn is covered by another flow. Repeated again, this would result in a succession of paleoregolith deposits separated and protected from the space environment by layers of igneous rock. Analogous processes occur on Earth, with the development of soils on lava flows that are in turn buried by later flows, causing thermal alteration of soil material (e.g., Olson and Nettleton, 1998; Sheldon, 2003; Marques et al., 2014). The heating profiles below terrestrial lava flows have been modeled (e.g., Fagents and Greeley, 2001; Kerr, 2001), and there is an existing literature on the analysis of thermal metamorphism of organic matter–containing rocks as a result of igneous activity (e.g., Bishop and Abbott 1993, 1995; Farrimond et al., 1996). However, terrestrial conditions differ markedly from those on the Moon, particularly with the absence of water in the lunar rock profile. The sequence of lunar regolith and lava flow layers would record changes in the types of meteorite infall over time, as well as changes in other parameters of interest such as the radiation environment and particle fluxes (Crawford et al., 2010; Fagents et al., 2010; Joy et al., 2014). Combined with radiometric dating of the lava units, a sequence of lava-encapsulated regolith layers could provide a tightly defined record of processes occurring at the lunar surface including the receipt of material from elsewhere in the Solar System such as Earth's surface.
1.5. The influence of volcanic heat on organic records
Having established a possible method of preserving regolith deposits by lava flows, consideration needs to be made of the effects of heating of the materials of interest within the buried regolith by the lava. Heating of organic materials can result in the loss of volatile compounds, degradation, and aromatization (e.g., Killops and Killops, 2005). The presence of minerals is known to affect the alteration of organic matter during heating (e.g., Espitalié et al., 1980, 1984; Huizinga et al., 1987). Space weathering would gradually break up an organic-bearing meteorite on the surface of the regolith, with the result that a portion of the organic material would become dispersed within the lunar regolith, with direct contact between organic material and lunar minerals. The minerals of the lunar regolith may have properties that accelerate the destruction or reconfiguration of organic materials when heated, or otherwise result in organic products different to those where the organic matter is heated in isolation, as has been investigated for the martian regolith (e.g., Iñiguez et al., 2009).
The composition of the lunar regolith depends upon the bedrock from which it is formed and tends to be predominantly basaltic in mare regions or anorthositic in highland areas (McKay et al., 1991). As such, the regolith is chiefly composed of a complex mixture of rock fragments and grains containing plagioclase feldspar, olivine, and pyroxene in variable amounts, as well as amorphous glass (Papike et al., 1982; McKay et al., 1991). There are also quantities of ilmenite and meteoritic iron. Elemental iron also occurs as a nanoparticle distribution within the amorphous glass rims of grains, and in agglutinates, formed as a result of micrometeorite impacts and redistribution of Fe from lunar silicates (Keller and McKay, 1997; Taylor et al., 2001).
In this work, we performed heating experiments on various organic materials under vacuum in the presence and absence of lunar regolith analogue Johnson Space Center 1 (JSC-1) to determine any alteration effects of the minerals on the organic materials. The aim was to assess the validity of the proposed mechanism of preservation of organic materials in lunar regolith capped by lava flows. Our results can be used to direct the search for organic records of chemical and early biological evolution in the Solar System that may exist on the Moon.
2. Materials and Methods
A variety of organic compounds and materials were heated under vacuum at a range of temperatures with and without lunar regolith simulant present. The products were then extracted with solvent, and these extracts were analyzed by gas chromatography–mass spectrometry (GC-MS). The solvent-extracted residues were analyzed by pyrolysis–gas chromatography–mass spectrometry (pyrolysis-GC-MS).
2.1. Lunar regolith simulant
JSC-1 is a simulant of the lunar mare regolith and was developed to fulfill a wide range of analog roles. It is a basaltic volcanic ash from Arizona, USA, and the main mineral constituents are plagioclase feldspar, olivine, pyroxene, and amorphous glass of basaltic composition, with a median particle size of approximately 100 μm (see McKay et al., 1994, and Willman et al., 1995, for a complete description of JSC-1). Although primarily intended as a geotechnical and engineering analogue, it nevertheless has a similar mineralogy and particle size distribution to lunar mare regolith. Key chemical differences include a greater abundance of water in JSC-1 as a consequence of the hydrous nature of terrestrial lavas, and a lack of Fe3+ in the lunar regolith (McKay et al., 1994). Analog materials such as JSC-1, which are available in significant quantities, are essential for exploratory work, allowing experimental insight without sacrificing valuable authentic samples.
2.2. Organic samples
Two types of organic materials were chosen for experimentation: free organic compounds and polymeric materials.
2.2.1. Free organic compounds
The free (solvent soluble) organic compounds selected for the heating experiments have been used before to test organic-detection instruments for life-detection missions (Table 1; Court et al., 2010). The suite of eight free compounds (hereafter termed “FC-8”), comprising atrazine, hexadecane, phytane, squalene, coprostane, stigmasterol, anthracene, and pyrene, represent a range of structures and include both biogenic compounds and compounds that can occur in the absence of life. Defining the range of temperatures that these compounds can withstand will provide insights into whether it is possible that biological organic material delivered to the Moon by terrestrial meteorites, and nonbiological material from asteroid and comet debris, could survive in the lunar surface in the lava flow trapping scenario.
2.2.2. Polymers
Both meteoritic and biotic carbonaceous materials contain a large proportion of macromolecular organic materials. Synthetic polymers were used as a simulant of this material. Poly(ɛ-caprolactone) (PCL) and poly[(R)-3-hydroxybutyrate] (PHB), which are both polyesters, were chosen. Although markedly different in structure to the meteoritic macromolecular organic material, the synthetic polymers have well-characterized mechanisms of thermal decomposition, allowing any deviations due to the presence of mineral matrix to be recognized.
2.3. Sample preparation
Glassware, including pyrolysis tubes, was prepared by baking in aluminum foil in air at 500°C for a minimum of 3 h, or by cleaning with solvent. JSC-1 was cleaned by exhaustive washing with dichloromethane to remove organic contaminants. The eight compounds of FC-8 were made up in 20 μL of methanol, which was spiked with a syringe onto JSC-1, which was preloaded into a pyrolysis tube to give 20 μg per compound. The methanol was then removed by thorough drying at 37°C. The spiking process resulted in a total organic content of 0.5–0.8 wt %. For those experiments where FC-8 was heated in the absence of JSC-1, the 20 μL of FC-8 methanol solution was spiked onto quartz wool in a quartz pyrolysis tube to give 20 μg per individual compound, before the methanol was evaporated by using a hot box at 37°C. The quartz wool was intended to act as a simple substrate to retain the compounds; therefore a precise ratio of compound mass to quartz wool mass was not applied. To check for contamination, aliquots of JSC-1 (∼25–40 mg each) containing no added organic material were heated alongside the other samples at each temperature step.
Finely powdered polymers were mixed with JSC-1 to give approximately 5 wt % organic content. Approximately 20–30 mg of this mixture was then loaded into quartz pyrolysis tubes and secured with quartz wool at both ends. For organic samples heated without JSC-1, variable masses from 2 to 12 mg were loaded into pyrolysis tubes in the same manner.
For each individual experiment, the quartz pyrolysis tube with loaded sample was placed into a 200 mm length of 3 mm internal diameter borosilicate glass tubing sealed at one end with a flame. The unsealed end of the tube was connected to a vacuum line and evacuated before the whole tube was flame sealed. Care was taken to ensure that the heat from the flame sealing process did not affect the loaded sample. The vacuum sealing process will have removed the majority of atmospheric and adsorbed water.
2.4. Heating experiments
Sample tubes were heated in a furnace from room temperature (∼20°C) up to the required final temperature at a rate of 1°C min−1. The final temperature was held for 80 min; then the furnace was allowed to cool. This temperature profile was intended to simulate the gradual heating of regolith material beneath a recently emplaced lava flow up to a maximum temperature. The four maximum temperatures chosen were 250°C, 400°C, 550°C, and 700°C. The chosen temperatures can be considered to correspond to a range of depths into the regolith beneath the lava flow. A thicker overlying lava flow will mean a greater temperature excursion at a given depth than for a thinner lava flow, and it will also remain hotter for longer owing to the greater volume of lava (Fagents et al., 2010; Rumpf et al., 2013). The minimum observed thickness of individual lava flow units on the Moon is ∼1 m (Vaniman et al., 1991). The maximum temperature isotherm of 700°C modeled by Rumpf et al. (2013) for a 1 m lava flow reaches a maximum depth of ∼0.1 m into the regolith beneath the lava flow after 10 days. The 300°C isotherm reaches a maximum depth of ∼0.2 m after 20 days. This temperature is slightly higher than the 250°C step used in our heating experiments but provides an indication that the peak temperatures chosen for our experiments correspond to the first few tens of centimeters depth below the surface of the regolith.
2.5. Sample extraction
After the sample tubes cooled, they were carefully broken open. Soluble organic compounds were then extracted from the sample and the internal glass surface of the tube by the addition of approximately 0.5 mL of 93:7 v/v dichloromethane:methanol solution. The solvent extraction step was repeated a further two times to ensure complete removal of organic compounds. Subsequently, the quartz pyrolysis tube containing the sample was removed from the borosilicate tubing and was dried at 37°C to remove solvent.
2.6. Analysis
The solvent extracts were made up to 1 mL with dichloromethane for analysis by GC-MS. Samples were run by using an Agilent 7890 N gas chromatograph coupled to an Agilent 5975C mass selective detector (MSD). The injected sample volume was 1 μL. The oven temperature was programmed for 50°C (1 min) then ramped at 4°C min−1 up to 310°C, and then held for 20 min. Separation was carried out on an Agilent HP-5MS capillary column (30 m length, 250 μm i.d., 0.25 μm film thickness) with helium carrier gas at a flow rate of 1.1 mL min−1. The inlet was operated in splitless mode and held at 250°C. The MSD scan range was m/z 50–550.
The solvent-extracted residue in the quartz pyrolysis tube was subjected to pyrolysis-GC-MS. Analysis was carried out with a Chemical Data Systems 5200 pyroprobe coupled to an Agilent 6890 gas chromatograph with a 5973 MSD. Pyrolysis was performed at 600°C for 15 s. Separation was achieved with an Agilent J&W DB-5MS UI column (30 m length, 250 μm i.d., 0.25 μm film thickness) with helium carrier gas at a flow rate of 1.1 mL min−1. The oven was held at 40°C for 2 min, before being ramped to 300°C at a rate of 5°C min−1, and then held at the final temperature for 8 min. The inlet was held at 270°C and operated in split mode (50:1). The MSD scan range was m/z 45–550.
Compound identification was performed based on retention times, comparison with standard compounds in the case of FC-8, and mass spectra referenced in the NIST-08 mass spectrum library.
3. Results
Analysis of the solvent extracts and the pyrolysis-GC-MS results are complementary; the solvent extracts reveal the unbound compounds that survive the heating process, as well as those that are generated by degradation of any polymers present in the experimental residue. Pyrolysis-GC-MS provides information about polymers that survive thermal processing but also newly formed polymeric structures that are created by cross-linking reactions. Solvent extraction of an 18.6 mg split of JSC-1 (i.e., for a typical mass used for heating experiments) showed no organic components. Aliquots of JSC-1, which contained no added organic material and were heated alongside the other samples at each temperature step, showed no substantial organic contamination that would affect the interpretation of results in either the solvent extracts or pyrolysis runs.
3.1. Free compounds (FC-8)
3.1.1. Solvent extracts
The pattern of survival of the free organic compounds (Table 1) for samples heated with JSC-1 present and those where JSC-1 was absent are similar. In both cases, at the lowest heating step of 250°C, stigmasterol and atrazine are lost (Table 2). Squalene has a very low response where JSC-1 is absent and was not detected when JSC-1 was present. At 550°C, only anthracene and pyrene remain in both the sample heated with JSC-1 and the one without. After the heating experiments at 700°C, none of the original compounds of FC-8 are detectable in the total ion chromatogram. Aromatization of the FC-8 compounds also occurred, as evidenced by a methylated 3-ringed (m/z 192) aromatic compound in the 550°C sample heated without JSC-1.
An, anthracene; At, atrazine; Cop, coprostane; DMN, dimethylnaphthalene; Hex, hexadecane; MN, methylnaphthalene; N, naphthalene; PAH m/z #, polycyclic aromatic hydrocarbon with m/z ratio for main response indicated; Phy, phytane; Py, pyrene; Sq, squalene; Sti, stigmasterol; TMN, trimethylnaphthalene; n.d., not determined.
3.1.2. Pyrolysis products
Up to 400°C, the key products detected by pyrolysis-GC-MS were hexadecane and phytane. It is likely that these compounds were incompletely removed by solvent extraction. At the 550°C temperature step, new compounds that were not initially present in the FC-8 were detected in the JSC-1 sample. Methyl-, dimethyl-, and trimethylnaphthalene are indicative that a temperature of 550°C is sufficient to cause aromatization of the constituent compounds of FC-8. This is reflected in the formation of a methylated aromatic compound (m/z 192), which was detected in the solvent extract of FC-8 heated at 550°C in the absence of JSC-1. At 700°C, in the sample heated with JSC-1, only naphthalene and a 3-ringed polycyclic aromatic hydrocarbon (PAH) (m/z 178) are detectable. The increased level of condensation at the higher temperature is indicated by the lack of alkylated aromatic compounds. No compounds with a response allowing definite identification were found where FC-8 had been heated in the absence of JSC-1 at 550°C or 700°C. It is also possible that the analytical pyrolysis temperature of 600°C was not sufficiently high to completely access the refractory carbonaceous phase formed by heating at 700°C.
3.2. Polymers: poly(ɛ-caprolactone)
3.2.1. Solvent extracts
The solvent extracts of PCL heated with and without JSC-1 at 250°C both show systematic breakdown products and do not display any major differences (Fig. 1a). Analysis of the solvent extracts from the experiments at 250°C is complicated by the solubility of the surviving polymer in solvent. Upon injection, the solubilized polymer can break down in the inlet of the gas chromatograph. This was confirmed by the generation of the same products where PCL mixed with JSC-1 was subject to solvent extraction without prior heating. Following heating at 400°C, a more complex assemblage of polymer fragments is produced. However, following heating at 550°C, the dominant response in the solvent extracts of both samples is pyrene (m/z 202). Polycyclic aromatic compounds up to m/z 290 are detectable in the solvent extract of PCL heated without JSC-1 (Fig. 1a). In the solvent extract following heating at 700°C without JSC-1, a range of aromatic compounds were detected including naphthalene, biphenyl, fluoranthene, pyrene, and an aromatic compound with m/z 252. There are no peaks in the total ion chromatogram of the solvent extract from the experiment where PCL was heated with JSC-1 at 700°C.
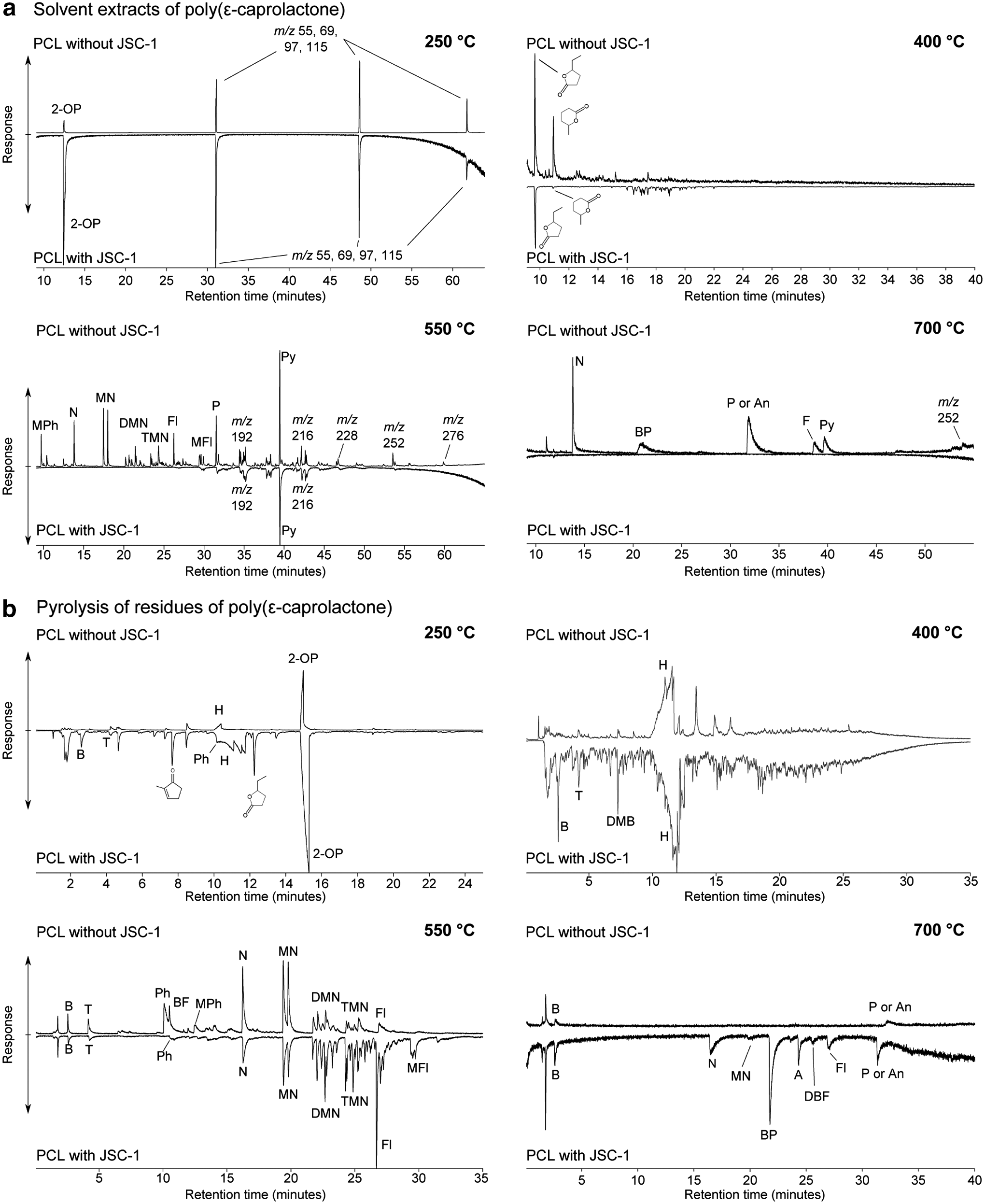
Chromatograms of (
3.2.2. Pyrolysis products of residues
Following heating at 250°C, 2-oxepanone and hexenoic acids indicate the presence of surviving PCL polymer. These are products of unzipping reactions and cis-elimination of the polymer (Persenaire et al., 2001; Aoyagi et al., 2002). Comparing the pyrolysis-GC-MS results of PCL heated with and without JSC-1 present at 250°C reveals that there is a slightly greater diversity of products released from the residual polymer when JSC-1 is present (Fig. 1b).
At 400°C, pyrolysis-GC-MS generates a much greater diversity of products from the residual polymer. The product with the greatest response for both the sample heated with JSC-1 and the one without was hexenoic acid. The broad “fingerprint” of the chromatograms was similar between the two; however, the sample heated with JSC-1 showed greater responses for benzene and alkylbenzenes relative to hexenoic acid than the sample without (Fig. 1b). This indicates that aromatization of the polymer is facilitated by the presence of the mineral matrix. At 550°C, aromatization of both samples is more complete. However, the proportion of di- and trimethylnaphthalenes and fluorene relative to naphthalene is much greater in the sample heated with JSC-1 than the one without (Fig. 1b). Responses at m/z 178 that correspond to the 3-ring PAH anthracene and phenanthrene are minor in the pyrolysis-GC-MS chromatograms for both samples, and 4-ring or larger aromatic PAHs were not detected, indicating that while relatively large free aromatic compounds are released during the heating experiments, the polymer residue is composed of smaller aromatic units.
After heating PCL without JSC-1 at 700°C, only benzene and a response at m/z 178 are detectable in the pyrolysis-GC-MS total ion chromatogram, indicating that the polymer residue has been further condensed relative to the previous temperature step. In the pyrolysis-GC-MS total ion chromatogram for the sample of PCL heated with JSC-1, benzene, naphthalene, biphenyl, and anthracene or phenanthrene are detectable, along with minor responses for alkylated aromatic compounds.
3.3. Polymers: polyhydroxybutyrate
3.3.1. Solvent extracts
The thermal decomposition of PHB follows a broadly similar pattern to that of PCL (Fig. 2a). After heating at 250°C, the solvent extracts of PHB with and without JSC-1 show the presence of crotonic acid and its oligomers, which are produced from the polymer as a result of cis-elimination reactions. Crotonic acid is recognized by characteristic m/z 68, 69, and 86 responses (Aoyagi et al., 2002). However, these products are again likely to be at least partly the result of solubilized polymer breaking down in the heated gas chromatograph inlet.
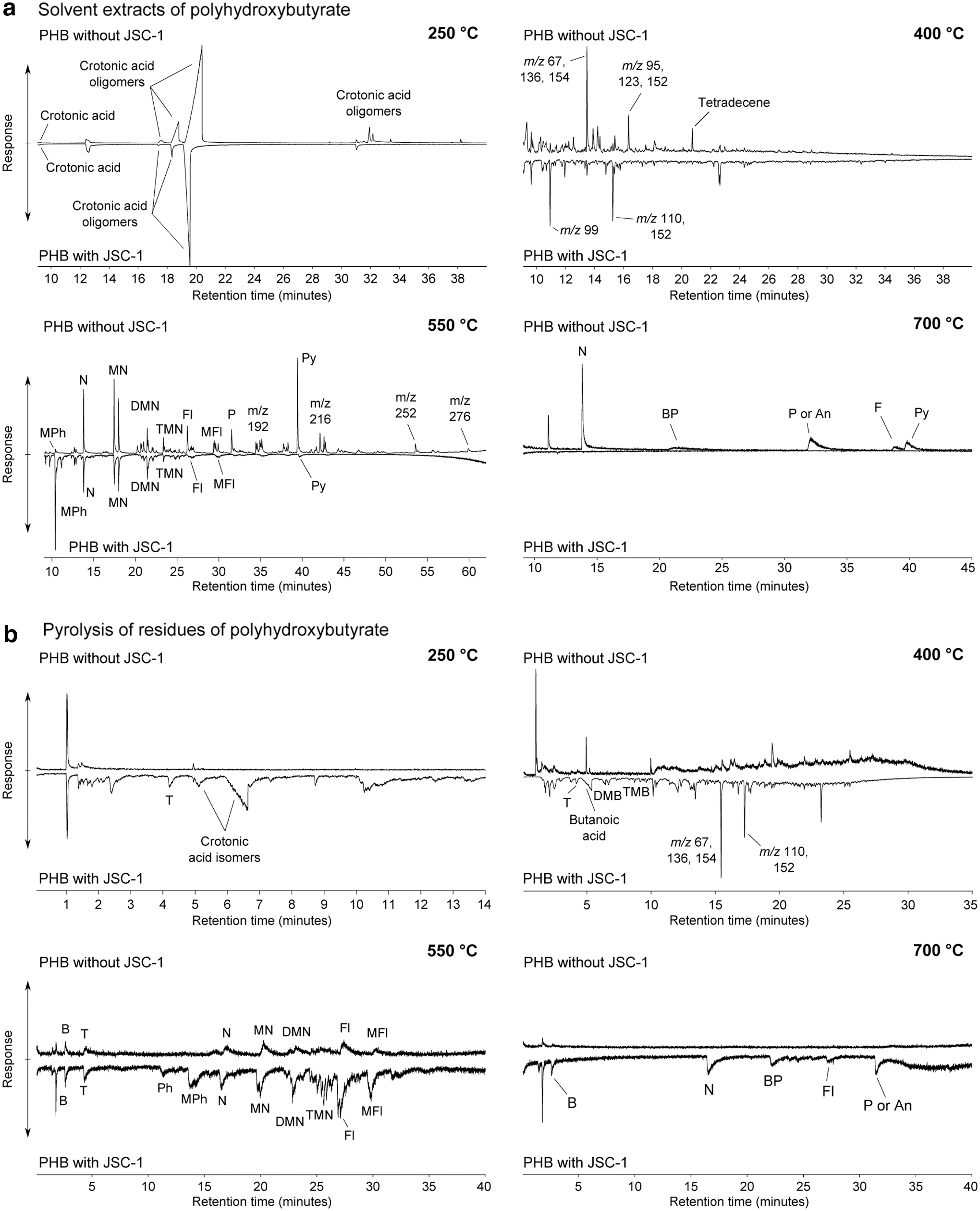
Chromatograms of (
At 400°C, both the pyrolysis-GC-MS and solvent extract chromatograms reveal a wide range of products with an unresolvable complex mixture (Fig. 2a, 2b). However, there are some differences in the responses of individual compounds in the solvent extracts. A peak with signals at m/z 67, 136, 154 gives a greater relative response in the PHB heated without JSC-1, and the compound gives a major response in the pyrolysis-GC-MS chromatogram where JSC-1 was present. This may be due to incomplete extraction of the compound from the minerals by solvent extraction. Another two peaks, one with signals at m/z 99 and the other at m/z 110 and 152, are prominent in the solvent extract of the sample heated with JSC-1 but are minor peaks in the PHB sample heated in isolation (Fig. 2a). After heating of PHB without JSC-1 at 550°C, the solvent extract is very similar to that of PCL heated in isolation at the same temperature, with a broad range of condensed and alkylated aromatic compounds present. The key responses are for naphthalene, methylnaphthalenes, and pyrene. However, the solvent extract of PHB heated with JSC-1 at 550°C shows a different pattern to that of PHB heated on its own. Where JSC-1 is present, pyrene is a weak response, and methylphenol is a strong response relative to naphthalene. Where JSC-1 is absent, pyrene is a strong response, and methylphenol is a weak response relative to naphthalene. The polymer has interacted with the mineral matrix to produce a different distribution of organic products. As with PCL, low responses of simple aromatic compounds are detected in the solvent extract chromatograms and pyrolysis-GC-MS chromatograms of PHB heated both with and without JSC-1 at 700°C.
3.3.2. Pyrolysis products of residues
Pyrolysis-GC-MS of the residue of PHB heated with JSC-1 at 250°C also shows crotonic acid; no crotonic acid was detected by pyrolysis-GC-MS where PHB was heated in isolation. The pyrolysis-GC-MS chromatograms of the residues of the PHB heated at 550°C also show a similar pattern to the PCL samples. After heating with JSC-1, the residual PHB polymer gives a greater pyrolysis-GC-MS response for di- and trimethylnaphthalenes and fluorene relative to naphthalene than when PHB is heated in isolation. There is also a strong response for methylphenol relative to naphthalene in the pyrolysate where JSC-1 is present, concordant with the strong methylphenol response in the corresponding solvent extract.
4. Discussion
4.1. Alteration of organic materials in a lunar regolith simulant
We performed heating experiments to determine the viability of preserving organic compounds in lunar regolith material beneath a lava flow. Particularly, we tested to see whether the lunar regolith, simulated by JSC-1 analog material, has any deleterious effects on the preservation of organic materials under heating.
For the free organic compounds, JSC-1 does not appear to have any dramatic destructive effect. As in the experiments without JSC-1, all compounds except atrazine, squalene, and stigmasterol are detectable after the 400°C experiment. FC-8 comprises a variety of terrestrial biomarker compounds; the fact that these compounds can survive at high temperature under vacuum in the presence of lunar regolith analogue lends credibility to the possibility that biomarkers in terrestrial meteorites from early Earth could remain preserved on the Moon in regolith at tens of centimeters depth under lava flow units.
For the polymers PCL and PHB, increasing the temperature of heating firstly causes the compounds to depolymerize into monomers and oligomers at 250°C, a process that is well characterized (Persenaire et al., 2001; Aoyagi et al., 2002). These systematic breakdown products can be seen in both the solvent extracts and the residues (Figs. 1 and 2). However, due to the nature of the analytical techniques employed, it is likely that some of the detected compounds are not primary products from the experiments but instead result from breakdown of the solubilized polymer in the hot (250°C) gas chromatograph inlet. At 400°C, breakdown of the polymers is more complete, with a wide variety of smaller molecules being generated. At 550°C, the polymers are charred; that is, they have been extensively aromatized by heating in the absence of oxygen, and aromatic hydrocarbons are the main responses from both the solvent extracts and pyrolysis of the residues. Charring is almost complete at 700°C, with low responses for simple aromatic hydrocarbons and a loss of compound diversity compared with the previous heating step.
It can be seen from the back-to-back comparisons of the chromatograms in Figs. 1 and 2 that the presence of JSC-1 influences the types and proportions of compounds produced at different temperatures. The pyrolysis of the residues where JSC-1 is present shows a greater variety of responses. In the pyrolysis residues at 400°C, JSC-1 appears to be promoting aromatization; benzene and alkylbenzenes are prominent relative to other responses in the chromatograms of the residues where JSC-1 was present. With both PCL and PHB, after heating with JSC-1 at 550°C, the residual polymer, when subject to pyrolysis-GC-MS, releases a greater proportion of alkylated napthalenes relative to naphthalene than when the polymers were heated in isolation. The distribution and responses of soluble aromatic compounds generated from PHB and PCL heated in isolation at 550°C are very similar; however, it can be seen that the presence of JSC-1 alters the distribution of responses of the generated compounds (Figs. 1a and 2a). It is apparent that the presence of the JSC-1 material affects the ability of the dichloromethane:methanol solvent to efficiently extract soluble organic compounds from the samples after the heating experiments, complicating interpretation of the results. However, particularly in the case of the aromatic compounds generated at the 550°C step for the synthetic polymers, a range of distributions of responses is seen that cannot be attributed to solvent extraction efficiencies and is instead a result of organic-mineral interactions during the heating experiments. The chromatograms of the PCL and PHB polymers at the 700°C step show similar behavior. Where JSC-1 was absent, there are readily detectable aromatic compounds in the solvent extracts; however, this is not the case where JSC-1 was present. Conversely, a wider variety of aromatic compounds are present in the pyrolyzed residues of the polymers where JSC-1 was present. The data indicate that the mineral and glass surfaces of JSC-1 are adsorbing and retaining the products of aromatization. The JSC-1 lunar analogue consists of a mixture of different mineral types, the main constituents being feldspar, pyroxene, and olivine. The catalytic properties of these minerals have been investigated in a number of industrial and natural settings (e.g., Rapagnà et al., 2000; Demiral and Şensöz, 2008; Tian et al., 2013). However, for this study we simply focused on the overall effects of the natural JSC-1 mineral assemblage as a representative of reactions that may occur in the lunar regolith.
4.2. General mechanism
The results from the heating experiments with free compounds and with polymers can be synthesized into a general mechanism (Fig. 3). The lunar regolith simulant appears to promote polymerization and cross linking (reflected by the production of more diverse pyrolysis products at lower heating experiment temperatures) and retard degradation and depolymerization (indicated by the continued production of pyrolysis products at high heating experiment temperatures; the mineral surface must be acting as a protective adsorbent). The lunar regolith with a similar basaltic chemistry, glass content, and grain size as the JSC-1 simulant may, therefore, aid in the preservation of its organic matter when encapsulated and heated by lava flows.
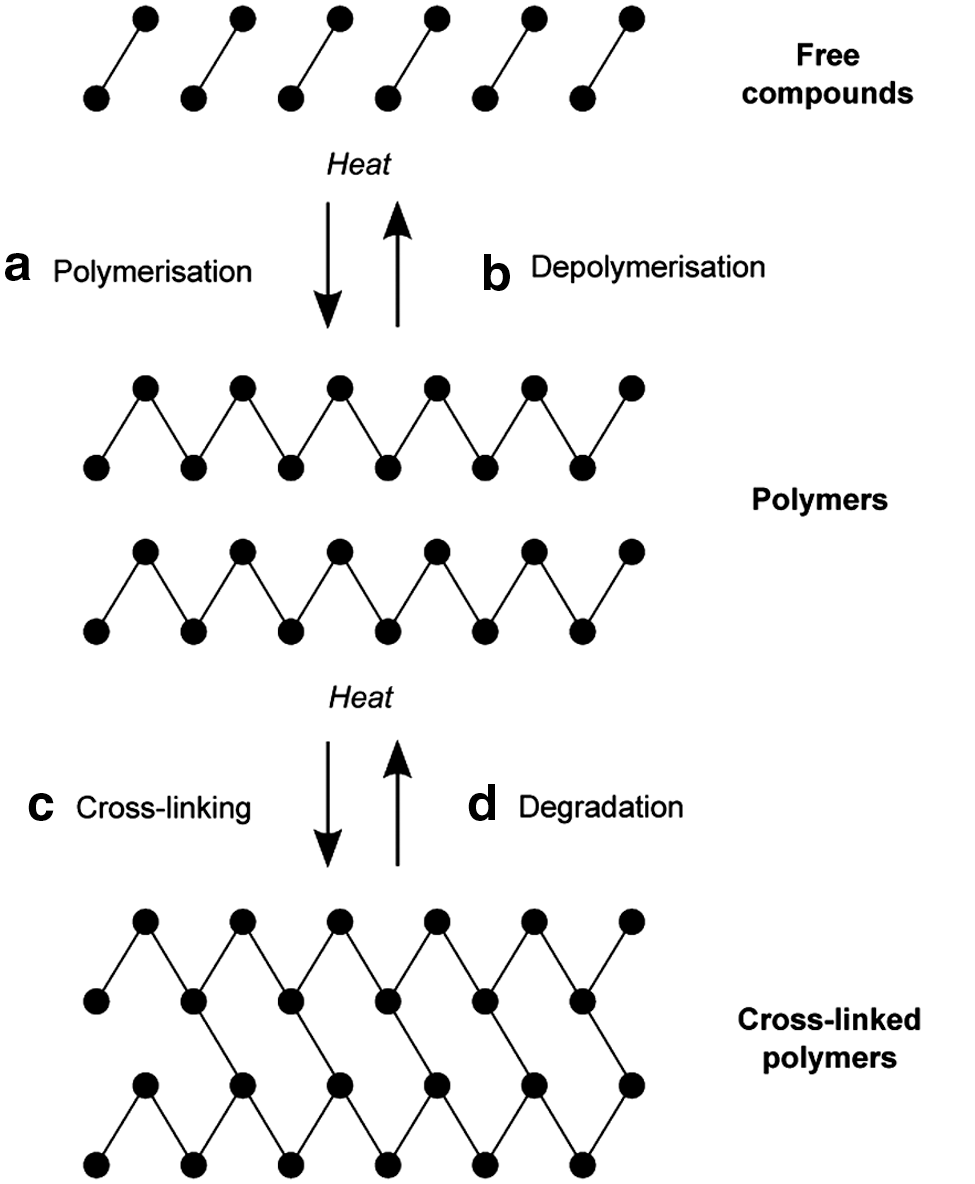
Schematic summarizing the processes occurring during heating of the organic compounds. The relevant chemical reactions can be divided into four processes: (
4.3. Preservation of organic materials in a lava-capped regolith layer
Thermal alteration of organic matter can be considered to have both positive and negative impacts in the context of preservation. Thermal alteration of an assemblage of organic molecules destroys information about the nature of the original material, but the process of aromatization and polymerization renders the organic material more resistant to damaging environmental processes. It may in fact be a requirement to have a moderate level of alteration of organic material to a more robust aromatic structure in order to ensure long-term survival in an energetic environment.
An ideal scenario for the preservation of organic materials within paleoregolith layers has a number of competing factors. While it would be advantageous to reduce the surface exposure time of organic-containing rock fragments to a minimum to protect from radiation and impacts, there nevertheless needs to be a sufficient elapse of time to build up a regolith layer thick enough to effectively insulate against the overlying lava flow (Fagents et al., 2010; Rumpf et al., 2013). There also needs to be time to allow the organic-containing material to build up in the regolith to a concentration sufficient that a drill core (or other sampling technique) will have a reasonable chance of intercepting a useful quantity of the material. Understanding the role of radiation in the alteration of organic matter will, therefore, be extremely informative in refining the model. It has been well established that ionizing radiation can have a variety of effects on organic materials (Lewan and Buchardt, 1989; Bernstein et al., 2003; Court et al., 2006; Kminek and Bada, 2006; Dartnell, 2011).
Duration of heating in the lava flow paleoregolith model is another key consideration. Modeling studies (Fagents et al., 2010; Rumpf et al., 2013) show that the regolith can remain at temperatures substantially elevated above ambient for timescales up to months, which is substantially longer than the durations employed in the experiments described here. The minimum lava flow thickness observed on the Moon is approximately 1 m (Vaniman et al., 1991); however, many lunar lava flows are known to be substantially thicker than this. Thicker lava flows would increase both the duration of heating and the depth of heating into the underlying regolith (Fagents et al., 2010; Rumpf et al., 2013). Thicker lava flows also have the potential to overprint the thermal profile of thinner lava flows and regolith horizons lying beneath. These factors would increase the degree of thermal alteration of the organic matter compared to a thinner flow. Although not explored in this work because of the generally accepted anhydrous nature of lunar surface materials in regoliths between latitudes <70°, water may also play a role in preservation. In terrestrial environments, the presence of water has a substantial effect on how organic matter is preserved (e.g., Killops and Killops, 2005). This also extends to space environments: the parent asteroidal bodies from which type 1 and 2 carbonaceous chondrites originated have experienced varying levels of aqueous alteration that has affected the composition and structure of the organic material (e.g., Sephton et al., 2004b). The Moon is essentially an anhydrous environment, although local sources of hydration may be provided by the hydrated silicate minerals of meteorites (Court and Sephton, 2014), hydrated volcanic pyroclastic glass beads (Saal et al., 2008, 2013; Hauri et al., 2011), indigenous minerals like apatite, and implanted solar wind (Pieters et al., 2009; for reviews see Anand et al., 2014, and Robinson and Taylor, 2014).
When subjected to heating by volcanic or other processes, water can be driven from these minerals. The released water can then interact with other materials within the host regolith. Recent observations have suggested the presence of water ice in permanently shadowed craters at the poles of the Moon (Colaprete et al., 2010). This water, which may be exogenous or endogenous in origin, could host organic compounds (Pierazzo and Chyba, 1999; Lucey, 2000). It is, therefore, important to take the role of water into account when considering the preservation of organic materials on the Moon. For the experiments in this study, although vacuum sealing of the samples before heating removed the majority of the water from the vessel, it is likely that minor amounts of atmospheric water remained adsorbed on mineral surfaces. Water may also have been released from clays in the JSC-1 analogue; however, since clay is a minor mineral phase (McKay et al., 1994), the quantity of water released from this source is assumed to be low. In the case of the synthetic polymers PCL and PHB, water would be eliminated from the polymer structure during thermal degradation (Persenaire et al., 2001) and would be available for reaction with other products or the polymer residue.
5. Conclusions
We have shown that organic matter is capable of surviving within a chemical and physical analogue of the lunar regolith, even when subjected to a demanding thermal regime intended to simulate the effects of an overlying flow of lava. However, we have seen that the aromatization of polymers is affected by the presence of the mineral matrix, producing a different distribution of responses for individual products than where compounds were heated on their own.
Using a range of heating temperatures intended to simulate different depths in the regolith beneath a lava flow, we have shown that for a 1 m thick flow, organic compounds have the potential to survive in regolith only a few tens of centimeters deep, due to the highly insulating properties of the lunar soil. These include compounds of biological origin, indicating the potential for biomarkers to be preserved in terrestrial meteorites on the Moon. Our work suggests that the paleoregolith of the lifeless Moon may be a valuable location to search for records of organic chemical evolution in the early Solar System and, perhaps, the transition from prebiotic to biotic processes on early Earth.
Footnotes
Acknowledgments
This work was supported by the Leverhulme Trust grant RPG-2012-581 to M.A.S., I.A.C., and A.P.J. R.W.C. was supported by a UK Space Agency Aurora fellowship. K.H.J. is supported by Leverhulme Trust grant ECF-2011-569. We would like to thank the reviewers for their helpful comments.
Author Disclosure Statement
The authors declare they have no competing interests.
