Abstract
Introduction:
Laboratory-acquired infections (LAIs) remain a significant occupational hazard worldwide, with the potential for public health risks beyond the laboratory. This study examined 2016 to 2023 data from the Laboratory Incident Notification Canada (LINC) surveillance system to identify risk factors associated with LAIs in Canadian laboratories.
Methods:
LINC incident reports, focusing on LAIs resulting from exposures to human pathogens or toxins, were analyzed. Potential risk factors contributing to LAIs were identified through univariate, bivariate, and multivariate analyses. Logistic regression was used to assess the association between potential risk factors and the incidence of LAIs.
Results:
Between 2016 and 2023, there were eight LAI exposure incidents that met the inclusion criteria and 354 non-LAI exposure incidents. Bivariate analyses between 10 potential risk factors and LAI occurrence only identified failure of or inadequate personal protective equipment (PPE) to be statistically significantly associated with LAIs (
Conclusion:
Failure of or inadequate PPE was a significant risk factor for LAIs in Canadian laboratories, thus reinforcing the importance of safety protocol adherence, ongoing training, and targeted interventions to reduce the risk of LAIs.
Keywords
Introduction
Laboratory-acquired infections (LAIs) pose a significant occupational risk to laboratory personnel worldwide. These infections, which result from exposure to human pathogens or toxins (HPTs), not only have an impact on the health and well-being of laboratory workers but also pose potential public health concerns due to the risk of transmission of pathogens beyond the perimeters of the laboratory. 1
Case reports from a variety of settings highlight distinct scenarios leading to exposure incidents.2,3 Needlestick injuries during experimental procedures, such as those involving viruses like the Zaire species of Ebola virus and Dengue virus, have been identified as a key risk factor.4,5 These incidents often resulted from breaches in standard laboratory procedures, including inappropriate handling of needles or failure to use adequate personal protective equipment (PPE).
Additionally, instances of contamination due to inadequate biosafety measures have been observed. These include non-adherence to glove use, improper hand hygiene, and conducting procedures with high contamination potential outside biosafety cabinets.
6
Such deviations from recommended protocols increase the risk of cross-contamination, leading to infections among laboratory workers, as seen in cases involving
Furthermore, specific manipulations of infectious agents outside of appropriate containment zones, particularly involving isolates capable of generating aerosols or droplets, significantly heighten the risk of exposure. This was evidenced in incidents related to laboratory-acquired meningococcal disease, emphasizing the criticality of performing manipulations within biosafety cabinets and employing adequate protective measures against aerosolized pathogens. 8
An overarching concern in these reported cases is the deviation from established safety guidelines, including inadequate PPE, lack of proper containment facilities, and oversight in handling potentially infectious materials. These cases stress the importance of rigorous compliance with biosafety protocols, routine risk assessments, continuous surveillance, and the imperative need for ongoing education and training of laboratory personnel to mitigate the risk of LAIs.2–5
To protect the health and safety of both facility staff and the general public in Canada, the
LINC’s surveillance system remains one of the only such systems in the world and has been fundamental in collecting and analyzing national data related to laboratory incidents, including LAIs. From 2016 to date, LINC has systematically recorded and monitored laboratory exposure incidents, providing valuable insight into the nature, frequency, and potential risk factors associated with such incidents and LAIs in Canada.11–18
The objective of this study is to examine laboratory exposure incident data to reveal potential risk factors and correlations that predispose laboratory workers to LAIs in Canada, the implications for workplace safety practices, and suggestions for targeted interventions to reduce the risk of LAIs in Canadian laboratories.
Materials and Methods
Data Source
The data utilized in this study were obtained from the LINC surveillance system, a comprehensive repository for recording and reporting laboratory incidents, including LAIs, across Canada. The LINC surveillance system systematically collects and documents incidents involving HPTs from laboratories across diverse sectors, including clinical, research, and educational settings, and serves as a valuable resource for understanding and mitigating laboratory-associated risks. The data are collected through a Microsoft Customer Relationship Management system called the integrated Suite of Tools for Operational Processes (iSTOP).
Inclusion Criteria
Incidents from 2016 to 2023 meeting the criteria for exposure report and exposure follow-up report, as defined by established biosafety guidelines 9 and as confirmed by reporters and LINC reporting protocols, were included in this analysis. Exposure incident reports are submitted when there is reason to believe infection or intoxication due to exposure to pathogens or toxins (i.e., via inoculation, ingestion, absorption, inhalation) may have occurred. 9 Follow-up reports are submitted within 15 to 30 days of the exposure to document the incident investigation findings. Incidents with “Unknown” or “Not applicable” values in one or both types of predictor variables (root causes or occurrence types) were not included in this study.
Study Variables
The dataset included information on reported exposure incidents, including LAIs, biological agent types, root causes of exposure, types of exposure, areas of activity, incident dates, sectors, and total number of affected persons, among others. The outcome of interest was the incidence of LAIs, which is infection/disease caused by exposure to pathogens or toxins handled in the containment zone. All other exposure incidents that did not result in an LAI were considered as non-LAI exposures. Descriptions and definitions of main activities, occurrence types, and root causes involved are provided in Appendix A, Tables A1, A2, and A3, respectively.
Data Analysis
All data analyses were conducted using SAS Enterprise Guide 7.15, with the descriptive statistics validated using Excel 2024.
Descriptive analysis
LAIs were described according to their containment level, sector, biological agent involved, date of occurrence, and main activity. To compare the characteristics of LAIs and non-LAI exposures, frequencies and percentages were used for all relevant variables, which were categorical in nature.
Identification of risk factors
To identify potential risk factors associated with LAIs in Canada, a comprehensive analysis encompassing univariate, bivariate, and multivariate approaches, was employed. The univariate analysis examined individual variables such as sector, occurrence types (e.g., incidents related to PPE, Procedures, and Sharps), the specific biological agents involved, main activities within the laboratory, and various root causes using frequency tables and/or chi-square goodness-of-fit tests. The associations between each of these variables and the occurrence of LAIs were assessed with Fisher’s exact test.
To quantify the association between the potential risk factors and the likelihood of LAIs, stepwise logistic regression with a significance level of 0.05 for entry and removal of predictors was used. Logistic regression examines the relationship between independent variables and a binary outcome, making it suitable for assessing the association between various potential risk factors and the probability of acquiring a laboratory infection. Implementing the stepwise selection procedure was useful in building a robust model to identify risk factors, as it incorporates both forward and backward selection, focuses on the most significant predictors, and provides a parsimonious model that could increase generalizability.
Factors showing potential significance in the univariate and bivariate analyses, or those with theoretical relevance, were considered in the multivariate analysis. The following independent variables were included in the model as potential risk factors: procedure (occurrence type), equipment (root cause), human factors (root cause), standard operating procedure (SOP) (root cause), PPE (occurrence type), and sharps (occurrence type). Some factors, such as certain root causes or occurrence types, could not be incorporated into the multivariate analysis due to multicollinearity, as reporters could select multiple occurrence types and root causes for each exposure incident. The number of affected individuals per incident was also not included to avoid overrepresentation. In addition, variables such as sector, main activity as well as spill and equipment-related occurrence types were not included in the regression model to avoid issues with separation and convergence.
Results
Descriptive Results
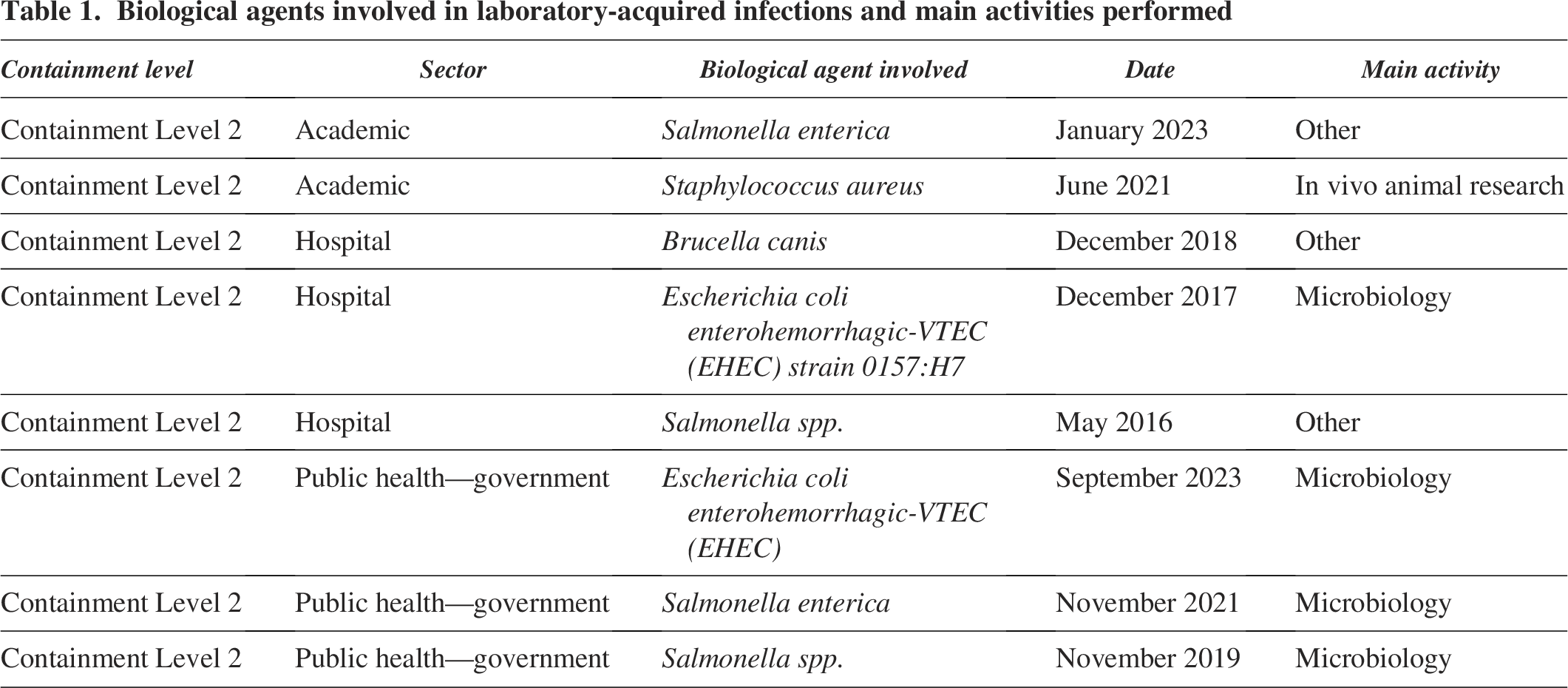
Between 2016 and 2023, there were 366 exposure incidents reported to LINC of which 354 were non-LAI exposures and 12 were confirmed LAIs. In accordance with this study’s inclusion criteria, out of the 12 confirmed LAI cases, 4 were excluded due to the presence of “unknown” or “not applicable” values in one or both of the predictive root cause or occurrence type variables. The final sample size included 362 exposure incidents. Table 1 describes the eight confirmed LAIs that met the inclusion criteria. All the confirmed LAIs occurred in Containment Level 2 facilities, with academic institutions accounting for 25%, while hospitals and the public health sectors each contributed 37.5% to the reported incidents. LAIs were observed across different laboratory activity areas, with microbiology showing the highest incidence, representing 50% of reported cases.
Biological agents involved in laboratory-acquired infections and main activities performed
Characteristics of confirmed LAIs from Containment Level 2 laboratories and non-LAI exposures
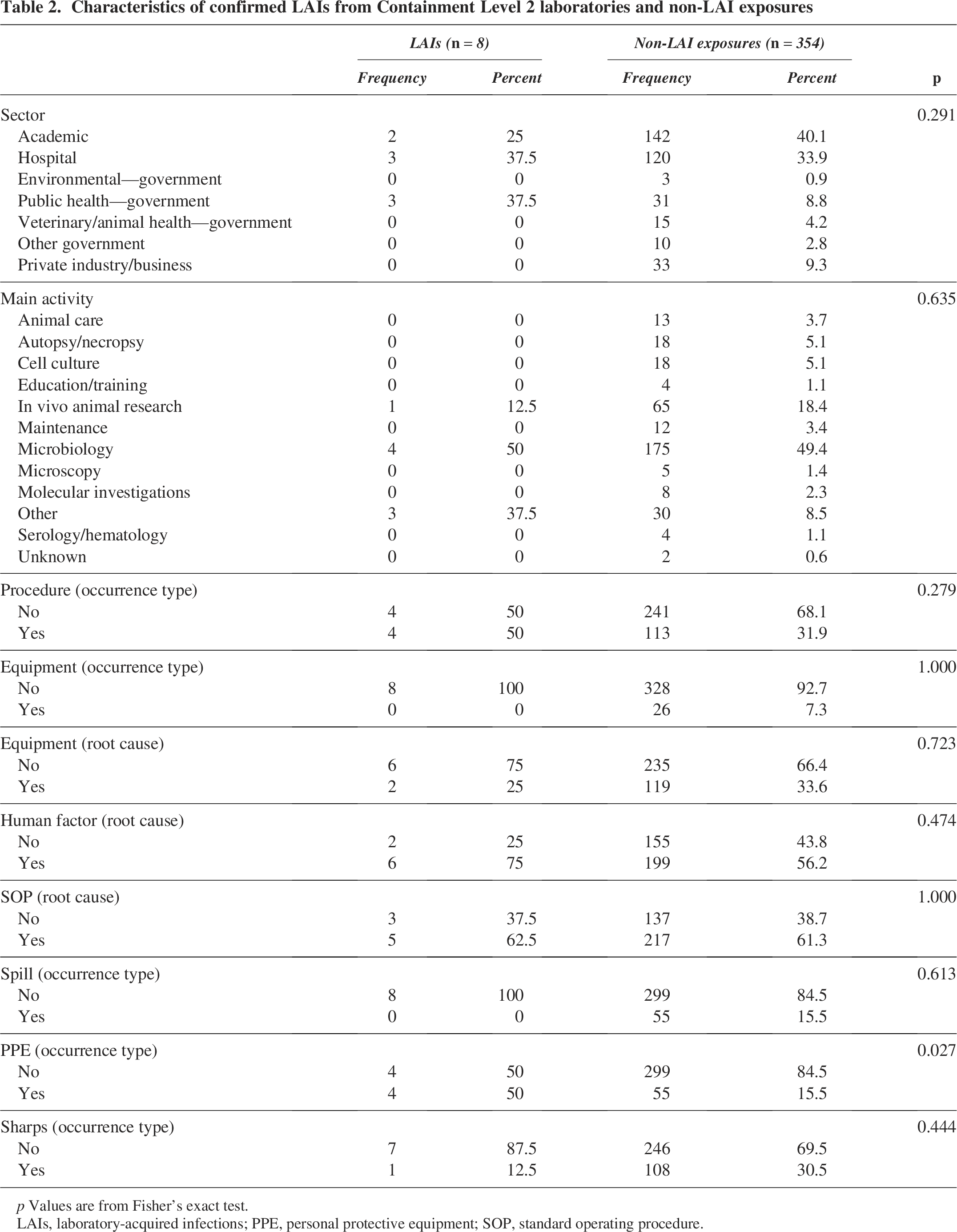
LAIs, laboratory-acquired infections; PPE, personal protective equipment; SOP, standard operating procedure.
One confirmed LAI was reported each year from 2016 to 2019, while there were two confirmed LAIs in both 2021 and 2023. One individual was affected during each of the LAI incidents in 2016, 2017, and 2019, as shown in Figure 1. In 2021, two separate incidents occurred, each affecting one person, giving a total of two affected persons in that year. Similarly, in 2023, two separate incidents took place, each one affecting one person. In 2018, there was a rise in the number of affected persons, as one incident resulted in an LAI exposure involving the possible inhalation of initially unsuspected
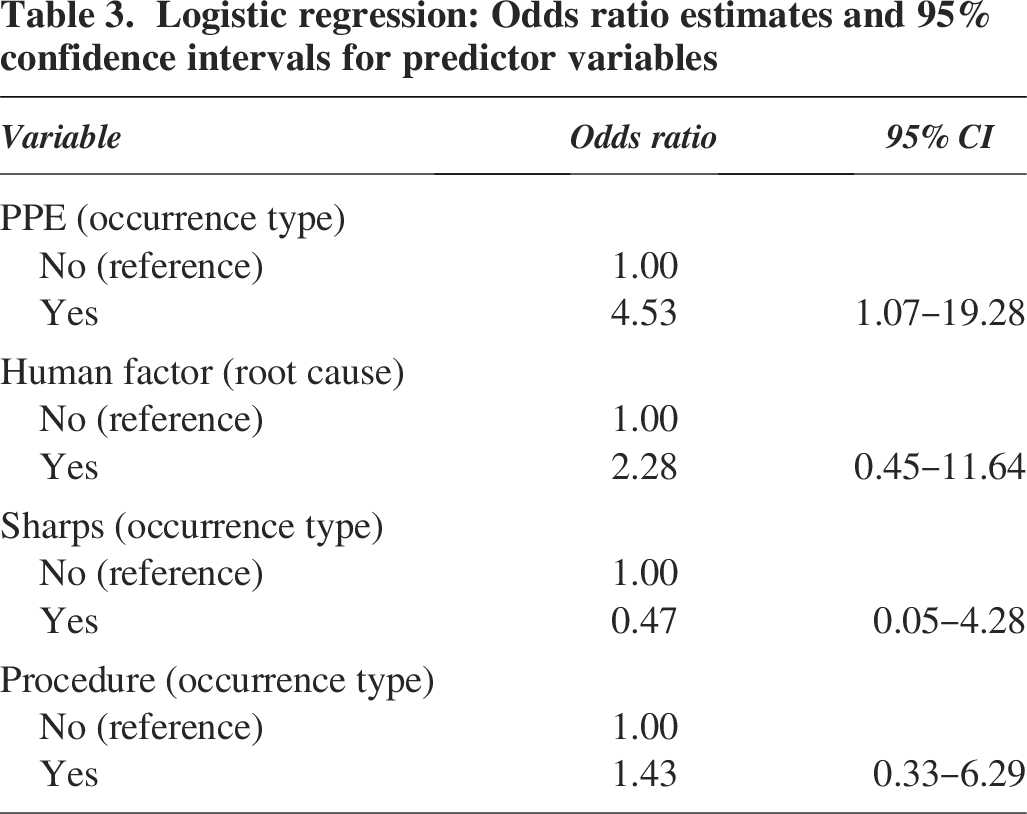
Logistic regression: Odds ratio estimates and 95% confidence intervals for predictor variables
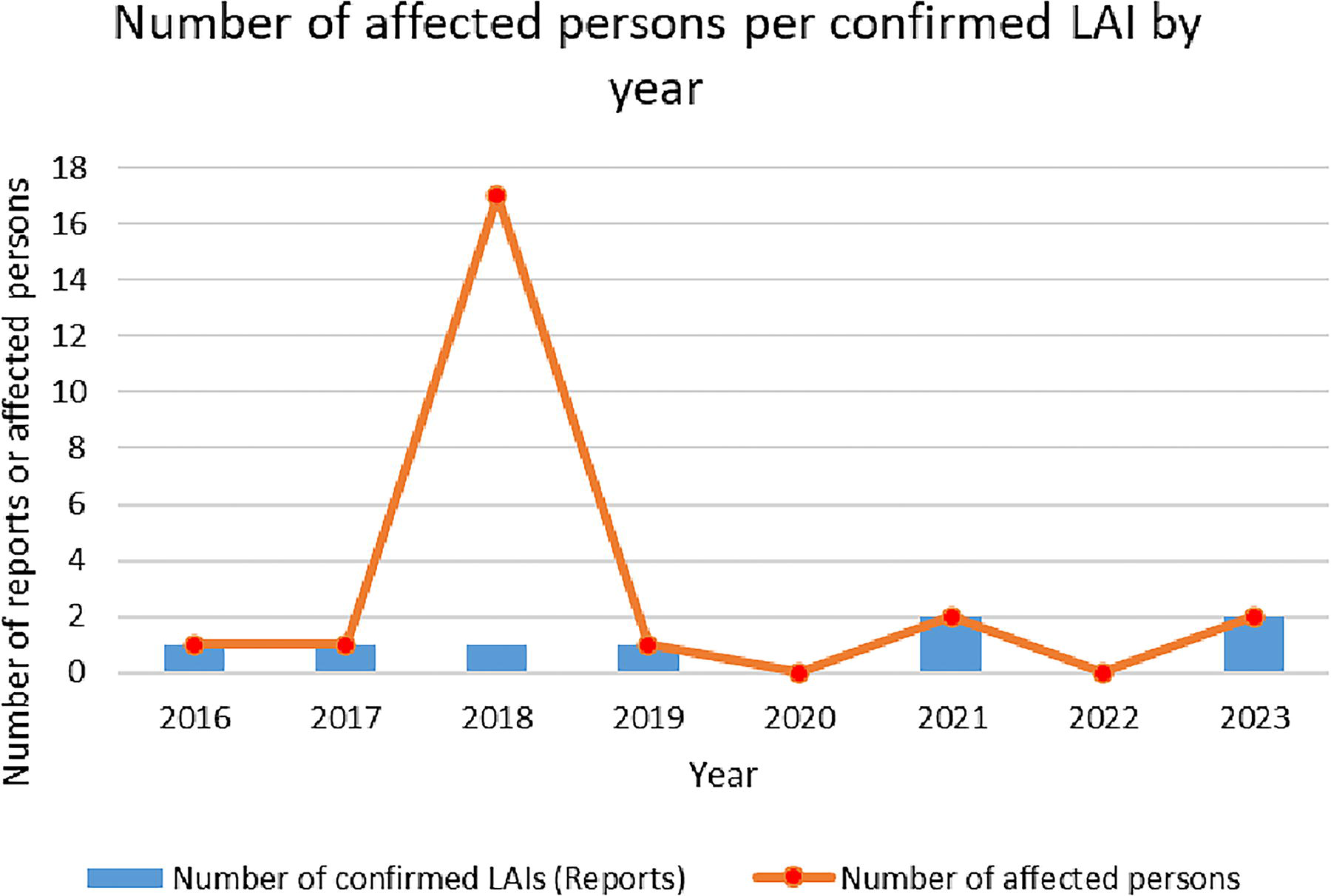
Number of affected persons per confirmed LAI by year, from 2016 to 2023. The years are represented on the
Risk Factors Identification Analysis Results
After the stepwise selection procedure and theoretical considerations, the following variables were included in the logistic regression analysis: PPE (occurrence type), human factors (root cause), sharps (occurrence type), and procedure (occurrence type). As shown in Table 3, the PPE-related occurrence type was significantly associated with the likelihood of LAIs (
Sensitivity Analyses
Although the overall sample size of exposure events was not considered small, the primary outcome (the occurrence of LAIs) was rare (2.21%). As a result, several potential risk factors could not be included in the logistic regression due to separation and convergence issues. To address this, Firth’s penalized logistic regression was used to model all potential risk factors: sector, main activity, procedure (occurrence type), equipment (occurrence type), equipment (root cause), human factors (root cause), SOP (root cause), spill (occurrence type), PPE (occurrence type), and sharps (occurrence type). Two variables were significantly associated with the likelihood of LAIs: the public health sector compared to academic (
Another sensitivity analysis was also conducted using the CATMOD procedure implemented in SAS. This procedure allows for the modeling of categorical data that can be organized into contingency tables. The logistic regression model using the CATMOD procedure initially included all the potential risk factors that were being considered. However, due to the low occurrence rate of LAIs, the maximum likelihood estimates for several potential risk factors—sector, main activity, spill, and equipment-related occurrences—were inconclusive. After removing those risk factors from the model, the results from the CATMOD procedure were similar to the main analyses and those from Firth’s penalized regression. Only PPE-related occurrences were near the statistical significance of being a risk factor (
Discussion
The purpose of this study was to identify the risk factors associated with acquiring laboratory infections while working with HPTs in the laboratory. Of the eight potential risk factors included in the stepwise selection procedure, four were included in the logistic regression analysis. Among those variables, failure of or inadequate PPE was identified as a risk factor, as it was significantly associated with an increased likelihood of contracting LAIs compared to having proper or adequate PPE. This result supports findings from other research and guidelines that emphasize the crucial role of appropriate PPE in mitigating occupational risks and reducing exposure to infectious agents in laboratories. 12 It suggests that proper or adequate PPE for the activity is fundamental to preventing infections in laboratory workers.
However, it is worth noting that LAI cases were evenly split between incidents involving proper or adequate PPE for the activity and those involving failure of or inadequate PPE for the activity, indicating potential misuse or the need for reinforced compliance with safety protocols. Human factor-related issues, which include issues related to human interaction, work demands or the work environment in relation to standards, policies and procedures, training, communication, management and oversight, or equipment and the work environment, were observed in 75% of LAI incidents, suggesting the vital role of human behavior in laboratory safety. This observation on the prevalence of human factor-related issues aligns with literature reviews that found that many LAIs could be attributed to human mistakes, which were likely a result of lack of training, competence, understanding, or a combination of such factors.19,20 Yet, the regression analysis did not identify human factor-related issues as a risk factor. While a review found that many of the reported LAIs were due to user-driven issues, such as procedural or needlestick-related issues, 19 results did not show any statistically significant association between sharps or procedure-related occurrences with the occurrence of LAIs. There was also no significant association of LAIs with procedural occurrences, which could be explained by multifactor collinearity and lower power due to the rarity of LAIs. Although these variables were not associated with LAIs, human factors, and SOP as root causes have been found to significantly affect the occurrence of exposure incidents. 21
Addressing human factor-related issues requires a comprehensive approach involving ongoing education, training, and behavioral interventions to promote a culture of compliance with safety protocols and continued vigilance among laboratory workers. Effective management of biosafety and biocontainment risks also relies on strong leadership dedication and commitment, a high level of staff proficiency, and significant investment in infrastructure. 19 PHAC promotes this culture of safety by ensuring that institutions operating regulated facilities conduct a follow-up investigation after an exposure incident. This allows the institutions to see where improvements can be made to their biosafety program so that appropriate corrective actions, which often involve additional training for staff, can be applied.
The high frequency of LAIs in microbiology-related activities, which account for 50% of reported cases, reflects a high risk associated with the handling of pathogens. Manipulations involving microorganisms present higher risks due to their infectious nature and require compliance with containment protocols and safety practices. The importance of the safe handling of HPTs is emphasized in the article by Kojima et al. which explains that local risk assessments, adequate control measures, and extra PPE may be used to mitigate risk exposure to the HPTs being handled. 22
Public health and hospital laboratories have contributed significantly to confirmed LAIs, indicating the vulnerability of these establishments despite their distinct operational structures. LAIs across different sectors show the universality of LAI risks, which justifies tailored interventions and targeted educational programs in the various laboratory environments to effectively reduce these risks.
The increase in the number of affected individuals reported in 2018, compared with other years, raises questions about the potential underlying factors contributing to such an outlier. Possible causes of this may include changes in laboratory practices, failure of or inadequate PPE for the activity, or changes in research activities. Furthermore, the total number of affected persons reported in 2018 serves to demonstrate the importance of upholding safety protocols in laboratories.
To enhance safety measures, continuous training, which has been known to contribute significantly toward cultivating a robust safety culture within laboratories, 23 should be provided with a focus on managing hazardous products, ensuring the proper use of safety equipment, and fostering compliance with safety protocols. Further exploration into decision-making dynamics related to the use of PPE could yield valuable insights. A study using a discrete choice experiment (DCE) framework to explore the decision-making processes related to safety among laboratory workers revealed that decisions about PPE use were more heavily influenced by the perception of exposure risk. 24 A deeper understanding of the interaction between personal risk assessment and the requirements of laboratory rules in the context of PPE use could provide the foundation for developing targeted interventions to improve PPE compliance.
It is important to acknowledge the limitations associated with this study. The presence of correlated variables constrained the model’s capacity to incorporate certain predictors concurrently. Utilizing stepwise regression, only one significant variable (PPE) was retained, potentially overlooking other influential factors. This limitation suggests a potential oversight of critical determinants affecting LAIs, despite their relevance. For this reason, other potential risk factors were manually evaluated for relevance based on results from univariate and bivariate analyses, and theoretical considerations. Additional sensitivity analyses were also conducted and provided similar results to the main analyses.
Although the study encompassed a wide range of factors, it may have omitted certain variables likely to have an impact such as years of experience working in laboratories and role of the person affected (technician, student, etc.). This omission could limit the overall understanding of the determinants of LAIs. Furthermore, establishing a causal link between the factors identified and LAIs is challenging due to the observational nature of the study, which limits definitive causal conclusions. Also, since the collection of data is dependent on the reporting by facility personnel, there is potential for recall bias, exposure, and outcome misclassification biases that could affect the detection of an association between risk factors and the likelihood of LAIs.
Focused solely on a specific temporal and geographical context in Canada, the results of the study may not be universally applicable to other contexts or periods. Variations between regions or periods could give rise to different trends and influencing factors, which could affect the wider relevance and applicability of the results.
Finally, when interpreting the results, it is important to remember that this study was based on a small number of LAI cases (
Conclusion
This study involved an analysis of risk factors associated with LAIs in Canada from 2016 to 2023, utilizing data collected by the LINC surveillance system. The importance of proper or adequate PPE in preventing LAIs was confirmed, establishing a relationship between the failure of or inadequate PPE for the activity and a higher risk of LAIs.
Even in cases where there was proper or adequate PPE, there were reported LAI incidents, indicating either misuse or the necessity for stricter compliance with safety protocols. Human factors and microbiology-related activities contributed to the incidence of LAIs, indicating the need for ongoing education and training, as well as a culture that prioritizes compliance with safety protocols by laboratory workers.
Key findings indicate the essential role of safety protocols, ongoing training, and targeted interventions in reducing the risk of LAIs in laboratories. Targeted interventions in specific zones of the laboratory are essential to address the common risks associated with LAIs. Although this study is focused on Canada, its findings support the need for rigorous safety protocols, constant vigilance, and further research on factors contributing to LAIs to improve laboratory safety practices worldwide.
Authors’ Contributions
C.M.G.: Conceptualization, writing—original draft, formal analysis, software, and methodology. E.F.T.: Formal analysis, validation, methodology, and writing—review and editing. A.G.: Incident monitoring and writing—review and editing. C.A.: Incident monitoring and writing—review and editing. A.N.D.: Writing—review and editing and supervision. S.B.-A.: Writing—review and editing.
Footnotes
Acknowledgments
The authors express their gratitude to the license holders for their valuable contribution to incident reporting. A special acknowledgment goes to the dedicated team at the Centre of Biosecurity for their support and knowledge.
Funding Information
This work was supported by the Public Health Agency of Canada as part of its core mandate and there are no competing financial interests.
Ethical Approval
This research was completed at the Government of Canada’s Public Health Agency of Canada in Ottawa, Ontario and did not require IRB approval.
Authors’ Disclosure Statement
No competing financial interests exist.
Appendix A. Definitions
Definition of root causes

| Root cause | Description |
|---|---|
| Communications | Includes issues with communication to guide the activity that led to the incident |
| Equipment | Includes issues with equipment used for the activity that led to the incident |
| Human factor | Includes human interaction issues related to the incident |
| Management oversight | Includes issues with the management and/or oversight to guide the activity that led to the incident |
| Additional training | Includes issues with the training that guided the activity related to the incident |
| Standards, policies, procedures | Includes issues with the standards, policies, procedures, or other expected practice documents that guided the activity that led to the incident |
