Abstract
Introduction:
A safety data sheet (SDS) is an established hazard communication tool for chemicals, for which no comparable document exists in the biotherapeutics industry. As the cell and gene therapy (CGT) field expands, industry leaders have identified a growing need to address this gap in communication of the unique occupational health and safety risks posed by CGT materials and products.
Methods:
Following the sections of a traditional chemical SDS, information was modified by industry subject matter experts, relevant to CGT biological materials. This guide was developed based on assumptions of a maximum biosafety level 2, and any chemical components present in the material were excluded from the hazard classification.
Results:
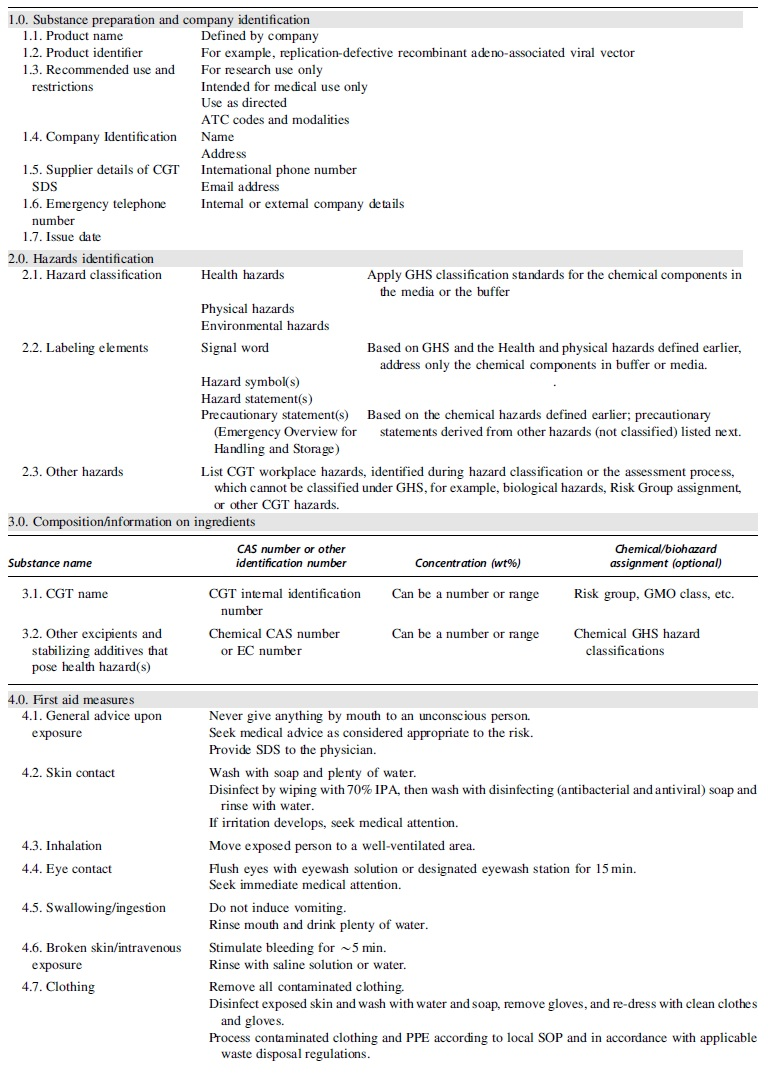
The guide contains necessary information to conduct a workplace risk assessment and communicate the unique workplace hazards posed by potential exposures to the material. The target audience is intended to be entities handling and producing these materials, plus collaborators, contractors, or operations sites receiving and handling the CGT material. An example of a CGT SDS is provided in Table 1.
Discussion:
The CGT SDS provides industry with a best practice to address an existing gap in hazard communication for CGT. We expect that, as the field evolves, so may the contents. The CGT SDS can be used as a reference for other biological modalities in the field.
Conclusions:
This initial CGT SDS communicates workplace hazards and assesses the unique risks posed by these biological materials and can assist in creating exposure control plans specific to the workplace hazards.
Introduction
The field of cell and gene therapy (CGT) is comparatively new and rapidly emerging. With the ability to use cells to carry a treatment throughout the body (cell therapy), as well as to modify or manipulate the expression of a gene (gene therapy), 1 CGT practitioners are developing technologies such as inactivating a malfunctioning gene; activating existing genes/cells ex vivo; replacing a pathogenic gene with a healthy copy; or introducing a missing, modified, or new gene into the body. 2
To effect these innovative therapies, CGT practitioners are required to synthesize and handle non-traditional, novel, or proprietary substances, and products for which Environmental Health and Safety (EHS) and biological safety (biosafety) standards and tools have yet to be firmly or universally established. For example, the gold-standard technical document for communicating chemical hazards is the Safety Data Sheet (SDS), which was developed in compliance with the Globally Harmonized System (GHS) of Classification and Labelling of Chemicals. 3
A further example is the use of a chemical SDS template for vaccines; although proprietary (company-specific) and not peer-reviewed, samples are available with an online search. Finally, there is an analogue for working with pathogenic organisms; these Pathogen Safety Data Sheets (PSDSs) describe the hazardous properties and recommendations for working with or around these agents in a laboratory setting. 4
However, there is no current analogue available for specifically CGT materials. The BioPhorum EHS and Biosafety working group have developed this analogue: a guide for SDS and biosafety experts to create SDSs for working with CGT biological materials and products.
This CGT SDS guideline also fills a gap in CGT operation risk assessment; it will not only help the biological risk assessment, but it also defines other CGT workplace hazards besides the biohazards. Therefore, it can help create a thorough CGT infection control and product hazard control/exposure control plans.
This new CGT SDS guideline largely mirrors the chemical SDS, which was standardized internationally in compliance with GHS and promulgated in the United States via the Occupational Safety & Health Administration (OSHA) Hazard Communication (HAZCOM) Standard 29. 5 The guide developed for CGT biologicals provides context and example text to assist SDS authors to make decisions on content, criticality, applicability, and other concerns that may not be immediately evident due to the novelty of the field and the EHS and biosafety niche specialty within it.
We are confident this new CGT SDS is highly versatile, comprehensive, and applicable. Although perhaps not future proof, it will retain robust utility while accommodating the dynamism inherent in a field that is unfolding. We offer it to the industry as a best practice with aspirations toward it becoming a new standard for communicating the workplace hazards of CGT biological materials and products, and can also perhaps serve as a model for other biologic products.
Methods
We present a comprehensive guide for the CGT practitioner in a pre/commercial setting, to assist with creating a document to convey hazards to downstream recipients of novel biological materials. This may include EHS personnel, biological safety officers, quality assurance personnel, chemists, or toxicologists, non-clinical, and supply chain or tech transfer staff.
A secondary audience may include downstream SDS consumers, such as shipping and receiving warehouse staff, manufacturing staff, scientists, clinical trial managers, pharmacists, transporters, and regulatory agencies. The CGT SDS should be authored by teams composed of individuals with adequate knowledge to accurately interpret relevant existing data into the workplace environment.
Using the 16 standard sections of a traditional chemical GHS SDS, the potential properties and considerations for biological materials have been assessed against corresponding chemical SDS regulations. Unlike a traditional chemical SDS, an SDS for CGT biologicals will be based on human clinical data, animal toxicity, and non-clinical data that include established biohazardous or infectious properties, pharmacology, and any additional relevant considerations that could be used to define hazards and the corresponding operational settings.
Several assumptions have been made, listed next, which potentially serve the CGT SDS author as a means to disambiguate among typical options available, as well as a platform upon which to build subsequent decisions.
The biological agents within the materials and products in question are classified as Risk Group Two (RG2) or below, operating at Biosafety Level 2 (BSL-2) or below.6,7
The SDS guide covers mainly the biological components of the biological material or product, for example, vectors, cell lines, etc. Chemical hazard classifications of ancillary chemical components such as vehicles, buffers, reagents, etc. are not discussed here and are not accommodated by this guide as they already have the GHS-compliant chemical SDS infrastructure.
Examples of novel biological materials include but are not limited to:
Somatic and T cell gene therapy drug product Viral vectors, such as Adeno-associated virus Adenovirus Herpes simplex virus Lentivirus Retrovirus Modified vaccinia virus Ankara Bacteria Bacteriophage
Examples of use cases include, but again are not limited to:
Transport of CGT bioactive product and intermediates across sites during R&D, manufacturing, or testing processes.
Technical transfer of CGT bioactive products and intermediates between client and contract manufacturing organization.
Product launch and delivery of newly marketed CGT bioactive products.
Operations in the R&D labs, manufacturing plants, packaging, clinical and health care settings.
Education and training tool for health care staff and patients utilizing CGTs.
Results
The 16 narrative sections given next correspond directly with the 16 sections of the CGT SDS in Table 1. The narrative sections provide helpful context for each category and include background, examples, and other information we believe useful to CGT SDS authors. The table itself is then presented where we have provided additional example information. Once a product has received regulatory approval, the CGT SDS should be aligned with the product label or packaging insert.
Cell and gene therapy safety data sheet guide
Substance Preparation and Company Identification
Similar to a traditional chemical SDS, this first section describes identification of the CGT products, relevant uses, the name of the supplier of the substance or product, and the supplier contact details.
Hazards Identification
This section describes the hazards of CGT products and the associated warning information by the GHS regulations, 3 and the following are usually listed:
Classification of the CGT products based on country GHS hazard classification rules
The GHS hazards listed in this section are usually only for the chemical components in the CGT products (e.g., the buffer components). Also, guidelines for ecotoxicology studies do not apply for CGT products and thus there are no data which can be used for environmental hazard classification. However, if there are existing ecotoxicology data for the buffer components, classification can be included in this section.
Labeling elements
Signal word, and hazard statements, symbols, or pictograms and precautionary statements are in accordance with country GHS hazard classification rules and only address hazards of the chemical components in the buffer or media. The precautionary statements should be listed based on the chemical hazards defined in the Classification of the CGT Products Based on Country GHS Hazard Classification Rules section.
Other hazards that cannot or are not classified under GHS, such as biological hazards and risk group assignment
This is the section where authors may list CGT workplace hazards.
Types of hazards to consider are based on
Human clinical data and preclinical safety data (including both Good Laboratory Practice [GLP] and non-GLP data): Conventional approaches to toxicity testing of pharmaceuticals may not be appropriate for CGTs due to their unique, diverse structural, and biological properties. These require species specificity, immunogenicity, and unpredicted pleiotropic activities in toxicity studies. 8
In addition, conventional toxicities, skin sensitization, genotoxicity, carcinogenicity, and reproductive toxicity are usually unavailable for biologicals. Therefore, hazard assessments must not only be based on single dose or repeat dose toxicity studies; but also a “weight of evidence” approach needs to be applied. The weight of evidence assessment should include all available data, including CGT mechanism of action (MOA), target biology, off-target effects, pharmacology, pharmacokinetics, biodistribution, germline transmission, oncogenic potential, tumorigenicity, mode of action, human clinical data, and a complete literature review. Above all, the workplace hazard assessment needs to consider administration routes of these pre-clinical and clinical safety data and to translate the data into a workplace scenario by evaluating each possible and relevant exposure route.
Allergies, immunotoxicity, and immunogenicity: Immunotoxicity refers to unanticipated effects on the immune system and may or may not be related to the pharmacology of the molecule, for example, unintended immunosuppression or enhancement. Drug-induced hypersensitivity and autoimmunity refer to the risk of allergies and should be assessed for handling a CGT product. In general, the major concerns for exposure risk associated with long-term exposures of biotherapeutics are acute reactions, cytokine release syndrome, or chronic immunosuppression leading to opportunistic infection, chronic infection, or malignancy.
Most biopharmaceuticals have been assessed for their immunotoxicity potential as a part of general single- and/or repeat-dose toxicity studies. 9 Immunogenicity, the host immune response to the drug, must also be assessed. This is not immunotoxicity per se but can become an unintended immune effect if the antidrug antibodies themselves cause toxicity either by cross-reacting with the endogenous protein and causing depletion or by forming immune complexes, causing tissue damage.
Biohazards from known pathogens: When evaluating biosafety risk from genetically transduced virus, viral vector, or bacteria, the related wild-type pathogens (e.g., replication competent viral vector or virus) should be used for reference as a conservative approach to assessing EHS and biosafety risk.
Hazards from antibiotic resistance genes: Antibiotic resistance genes are one of the most widely used selectable markers for general cloning and there are concerns they might be transferred into certain CGT products. This hazard should be considered when assessing hazard profiles for CGT products.
Composition/Information on Ingredients
This section identifies the constituent ingredients of a CGT product, including excipients and stabilizing additives. Based on the EU REACH Regulation, Annex II requirement, 10 and OSHA 11 guidance, authors should list all components (in line with disclosure limits) posing health hazards, regardless of their concentrations.
For chemical components, OSHA does not require authors to list the hazard classification. However, it is required by EU REACH Annex II, in addition to any substance that has a Community Workplace Exposure Limit assigned. Compared with a traditional chemical SDS, there is no major difference other than listing the classification of the CGT active pharmaceutical ingredients (APIs) for EU SDSs.
First Aid Measures
This section describes basic first aid in such a way that an untrained attendant would be able to respond to an exposure. If further medical attention is required, authors should state specific instructions, including the level of urgency to be applied.
High-priority exposures needing immediate attention include inhalation, needlesticks, broken skin, eye, and mucous membrane exposure. For example, eye exposure should be defined as high-risk, since the eye has a degree of immune privilege and the immune response to pathogens and foreign antigens could be suppressed. The disinfecting agent or washing solution should be named as precisely as possible to ensure appropriate use.
Firefighting Measures
This section describes appropriate fire response involving the CGT product and any specific hazards that may develop during a fire. The CGT product itself poses minimal hazards, and standard language found on chemical SDS is likely to be sufficient. However, some risks to consider in this section include CGT organism survivability and over-pressurization of heated containers, which may release biohazardous aerosols.
Accidental Release Measures
Based on EU REACH Regulation, Annex II 10 and OSHA 11 regulations, this section provides recommendations on the appropriate response to spills, leaks, or releases, including containment and clean-up practices to prevent or minimize exposure to people, property, or the environment. It also recommends distinguishing between responses for large versus small spills, where spill volume can have a significant effect on the risk to personnel, and between cell therapy and gene therapy products.
It is important to refer to appropriate country agency-approved disinfectants, 6 for example, EPA approved, for a specific CGT material, for example, 70% ethanol or isopropanol should not be the preferred method due to potential vapor exposure, flammability, rapid evaporation resulting in short contact times, and ineffectiveness against several common CGT biological agents.7,12
CGT spills should be treated as the worst-case scenario until identified otherwise. For the cells derived from donors and patients, spills should be treated at BSL-2. For spills in the clinical environment, country-specific regulations must be considered.
As the spilled materials are sanitized, their disposal does not pose any major environmental or community risk. Instead, the risk is much higher during the decontamination and cleaning process, as well as collecting the spill material. The spill response team (in-house team or external vendor) should be well trained on response to and remediation of biological versus chemical spills.
Handling and Storage
This section provides advice on safe handling and storage practices and emphasizes precautions appropriate to the unique properties of the CGT product. Specific use cases, for example, R&D or manufacturing, can also provide clarification for biosafety level controls. Referencing technical documents may be appropriate, for example, workplace hazard assessments. Advice on protection against fire and explosion: Advise on protection against fire and explosion and draw attention to operations and conditions that create new risks by altering the properties of the substance or mixture, as well as appropriate countermeasures. Precautions for safe handling: Describe safe CGT product or mixture handling, for example, containment and practices, to prevent exposure to aerosols and splashes. Provide relevant instructions, such as: Handle according to instructions on product information sheet and label, in accordance with good industrial hygiene and established laboratory procedures (per containment level). Remove gloves and wash hands when work with material is completed. Do not reuse gloves. Remove contaminated clothing and protective equipment before entering eating areas. Conditions for safe storage Specific design for storage rooms or vessels (including retention walls and ventilation). Incompatible materials. Conditions of storage (humidity limit/range, light, inert gas, etc.). Special electrical equipment and prevention of static electricity. Risk minimization for release to the environment.
This subsection also should indicate any special requirements such as the type of packaging/containers, labeling, and access or security controls for CGT materials.
Exposure Controls/Personal Protection
Based on EU REACH Regulation, Annex II 10 and OSHA 5 regulations, this section indicates the exposure limits, engineering controls, and personal protective measures used to minimize occupational exposure. The required information consists of:
National Occupational Exposure Limits (OELs), or Community Workplace Exposure Limits, Industrial Hygienists Threshold Limit Values, and any other exposure limit used or recommended by the manufacturer, importer, or employer preparing the SDS. Recommendations are aimed at API components produced by, or involving, living cells, not chemically synthesized drugs. OEL values usually apply to chemicals, as opposed to Risk Group (RG) and Biosafety Level (BSL) assignment that apply to biologicals. The RG can be obtained from the RG Database from ABSA International. 13 Usually, the RG is determined by the parental agent of the CGT and will be listed in the Hazards Identification section. The BSL assignment depends on not only the RG and CGT biohazards, but also the cell line used in the production, genetic engineering process, and scale of the process. For protein therapies, OELs and BSLs could both be applied. If production of the biotherapeutic product (e.g., protein small-molecule conjugates) contains both biological and chemical synthetic processes,14,15 it will have both BSL and OEL assignments.
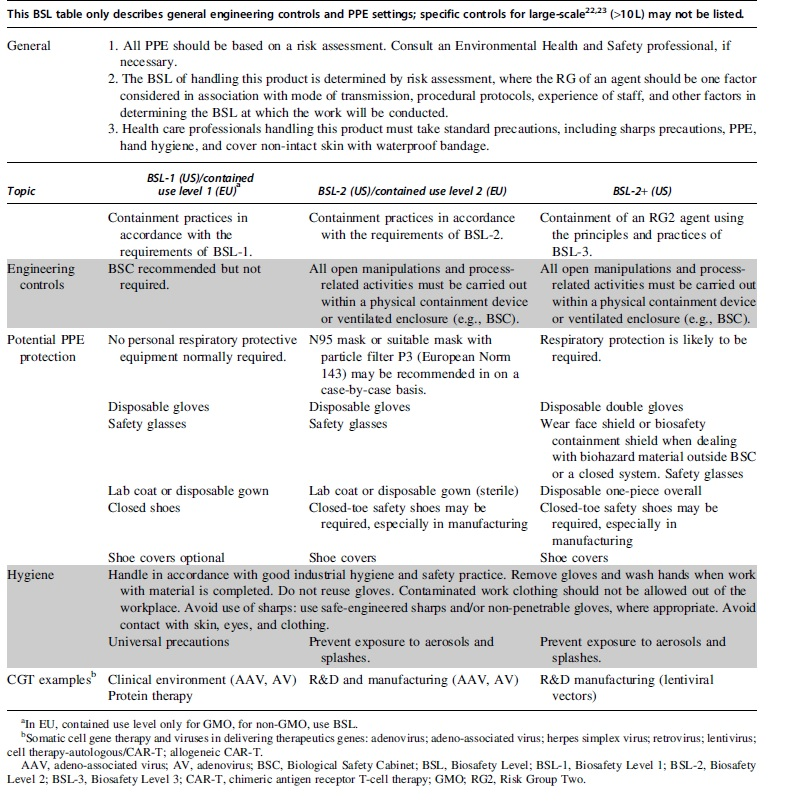
Appropriate engineering controls (e.g., using local exhaust ventilation or only in an enclosed system). For appropriate BSL controls, refer to the Biosafety in Microbiological and Biomedical Laboratories. 7 Requirements may vary depending on the BSL assignment; please refer to Table 2.
Recommendations for personal protective measures to prevent illness or injury from exposure to CGTs, such as personal protective equipment (PPE). Again, requirements may vary depending on the BSL assignment detailed in Table 2. The detailed hygiene measures used to control handling risks or to reduce the quantity of CGT material present at the workplace can also be included in this section.
Other special requirements: any health conditions affecting individual susceptibility to infection or the ability to receive medical treatment on exposure should be described here. Much in the way that, for example, vaccines made in yeast are contraindicated for patients with yeast allergies, requirements may vary and can include preventative and/or post-exposure immunization and prophylaxis, including contraindications, for example, pregnancy, the aforementioned yeast allergies, and so on.
Biosafety Levels
The RG of CGT products are usually defined or approved by authorities (i.e., National Institutes of Health or Centers for Disease Control and Prevention) and will often not change across the product development process and the product working environments (e.g., R&D, manufacturing or clinical). However, the BSL assignment will be different at different working environments, for example, the BSL assignment in clinical setting could be lower than the assignment used in R&D and/or manufacturing.
Physical, Chemical, and Biological Properties
This section identifies physical and chemical properties associated with the substance or product. Although the inclusion of a section containing physical properties may seem counterintuitive, the biological nature of CGT materials has unique properties that can vary immensely and have effects on personnel safety.
The SDS may not contain every item listed in Table 1, as information may not be relevant or available. When this occurs, a notation to that effect should be included.
Stability and Reactivity
This section communicates the tolerance of the CGT material to a variety of environments, including the laboratory and conditions by which it will be inactivated or eliminated. Environmental factors affecting stability may include chemical agents, ionizing or nonionizing radiation, temperature, humidity, time outside the host, and susceptibility to altering agents.
The section also conveys information about the by-products of material treated for inactivation or destruction. Depending on the nature of the CGT material, by-products can include but are not limited to volatile organic compounds, allergens, or halogenated compounds.
Although safety measures for handling the CGT material are emphasized, this section can also provide information to protect limited quantities of valuable material from incompatible substances and conditions.
Toxicological and Pathological Information
For a traditional chemical SDS, this section identifies information on toxicological and health effects and/or indicates whether such data are available. Though less appropriate for inclusion in a biological SDS, it is still recommended to include where applicable:
Numerical measures of toxicity, for example, acute toxicity estimates such as LD50, the median lethal dose, that is, the estimated amount of a substance expected to kill 50% of test animals in a single dose.
Indication whether the material is listed in the National Toxicology Program Report on Carcinogens 16 is a potential carcinogen in the International Agency for Research on Cancer 17 Monographs, or a potential carcinogen by OSHA.
Consider including guidance that SDS authors can implement to help SDS consumers note the risks associated with chronic, low-level occupational exposure during handling procedures.
As CGT is a new field and the breadth of the spectrum of potential downstream systemic effects of handling these biologicals is yet unknown, we also recommend SDS authors include content that can help their readers evaluate the benefit of long-term follow-up with laboratorians, manufacturing staff, and clinical staff who have occupational exposure to CGT materials. The FDA analogue recommendation for following up with research subjects can be up to 15 years after exposure; 18 for CGT materials, given concerns about, for example, increased risk for oncogenesis and off-target gene editing, etc., SDS authors should consult with their organizations' SMEs and make recommendations for follow-up that are appropriate for each product.
Information on likely routes of exposure should be considered, noting fomites which may transfer disease to a new host. Physical contact, aerosols, and splashes may contaminate objects, surfaces, and personnel, potentially exposing workers (e.g., inhalation, contact with mucous membranes) and/or making these workers capable of transporting potentially infectious agents from their facility to the environment. Employing both containment (e.g., using a biosafety cabinet [BSC], frequently changing PPE) and conventional cleaning procedures (e.g., using disinfectants, washing hands) is often enough to reduce the risk of environmentally mediated transmission.
Likely routes of potential hazardous exposure should be ascertained by conducting a white paper assessment (based on International Council for Harmonisation guideline, 8 existing toxicity, clinical, pharmacology data and literature, molecular weights, MOA, and hazard-specific information).
Considering the nature of biological materials, there is likely information beyond the scope of a chemical SDS that will be useful for authors to consider, some of which are listed in Table 1.
Ecological Information
In this section, information is provided regarding the potential environmental risks associated with the product. Documented cases demonstrate potential risk from release of biological agents and the potential environmental effects.19,20 Inadequate controls may lead to contaminated objects, surfaces, and personnel; potentially exposed workers can become unwitting vectors for the transmission of biological agents into the environment.
It is recommended a CGT SDS provides information on the results of a release: ecotoxicity, persistence and degradability, survivability, accumulation, mobility, host range, and reservoirs. Additional recommendations include examining modes of release, risks of release from an actual workstream in which the CGT material is present, theoretical workstreams of future applications, ecological and community impacts, release caused by ineffective deactivation/disinfection, and possible outcomes of a release such as infection acquired outside of control areas.
Disposal Considerations
Disposal of CGT intermediates differ by local governance and must comply with country, state, and local regulations and refer to local authorities having jurisdiction for disposal considerations. Additional consideration should be given to sustainable options and assess for solid wastes such as contaminated consumables and plastics.
Liquid biological material retaining harmful properties such as pathogenicity should be inactivated before drain disposal. Acceptable methods for inactivation may be established and validated under relevant facility conditions to ensure efficacy for large-scale. Closed industrial pre-treatment systems such as chemical or thermal effluent decontamination systems or neutralization systems are recommended for large-scale disposal.
For mixed wastes, for example, chemical and biological, handle and decontaminate the biological component first, followed by chemical inactivation.
Transport Information
This section provides guidance on shipping and transportation of the CGT product according to requirements, which will vary by mode, for example, road versus air; consult the most recent edition of UN Recommendations on the Transport of Dangerous Goods 21 for transport hazard classes, including for dry ice. Function-specific training is required by law to ensure compliance with applicable regulations for those responsible for packing and shipping hazardous materials.
Consult individuals knowledgeable on both the location of origin and destination to ensure all applicable import/export permits or regulatory requirements are obtained to accompany the shipment.
Regulatory Information
The example statement presented in Table 1 is a high-level communication currently found in many chemical SDSs. Though technically sufficient, this section provides the opportunity to present targeted regulatory requirements based on the physical location of the CGT activity. Regulations can come from diverse agencies regarding public health, commercial transportation, or agriculture.
Permit requirements may also apply for CGT products, intermediates, and/or starting material shipments.
Table 3 provides examples of supplementary resources.
Supplementary resources
APHIS, Animal and Plant Health Inspection Service; BIS, US Bureau of Industry and Security; CDC, Centers for Disease Control and Prevention; ECHA, European Chemicals Agency; NIOSH, National Institute of Occupational Safety and Health; RCRA, Resource Conservation and Recovery Act; TSCA, Toxic Substances Control Act; USDA; US Department of Agriculture.
Other Information
This section details the SDS author, dates of revision, and other useful information to the end user, including links to additional references.
Conclusions
This paper and the SDS guide in Table 1 are the original efforts in defining workplace hazards and the corresponding workplace safety setting proposals in the biotherapeutics field. Although the focus for this paper is only for CGT products, this guide may also be helpful as a reference for other biotherapeutic modalities such as protein therapies, mRNA, and plasmids.
With no established tool in the field, this guide is offered as an industry best practice with aspirations of becoming a standard communication for workplace hazards unique to CGT biological materials and products. As the field develops and more CGT products move into commercialization, regulatory oversight will likely influence documents such as this. Further, as hazards are better defined and characterized with a growing body of knowledge, the scope of certain sections is likely to evolve.
We are confident this new CGT SDS guide is versatile and comprehensive, and encourage those exchanging CGT with collaborators, colleagues, and/or contractors to utilize this guide as a means of hazard communication to provide the necessary information for conducting workplace risk assessments and establishing safe handling and operating procedures.
Footnotes
Acknowledgments
Improvements were suggested by several subject matter experts who reviewed the manuscript. Any recommendations and errors are our own and should not tarnish the reputations of these reviewers. Special thanks go to Jason Keaton (Acacia Safety Consulting, Inc.) for helpful insights and comments. The work was facilitated by BioPhorum, which since its inception in 2004 has become an open and trusted environment where senior leaders of the biopharma industry come together to openly share and discuss the emerging trends and challenges facing their industry. The BioPhorum Operations Group's mission is to create environments where the global biopharmaceutical industry can collaborate and accelerate their rate of progress, for the benefit of all. More information can be found at ![]()
Authors' Disclosure Statement
No competing financial interests exist.
Funding Information
No funding was received for this article.
Supplementary Material
The CGT Safety Data Sheet template can also be downloaded from the BioPhorum website at https://www.biophorum.com/download/cgt-safety-data-sheet-template/
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.