Abstract
Introduction:
Decontamination of farms affected by bovine tuberculosis could be very challenging during outbreaks occurring in the winter with freezing temperatures. Steam treatment has been of practical interest, but information is needed on whether such treatment is able to inactivate the causative agent, Mycobacterium bovis. This study was to evaluate the use of pressurized steam for inactivation of Mycobacterium terrae, a surrogate for M. bovis on various surfaces.
Methods:
Carrier disks made of steel, wood, or rubber were inoculated with 6.32 ± 0.38 log10 M. terrae. While being held at background temperatures of −20°C, 4°C, or 21°C, these carrier disks were treated with pressurized steam (120°C ± 5°C) for 5, 10, 15, or 20 s. Reduction in colony forming units of M. terrae and temperatures on the top and bottom surfaces of the disks were determined.
Results:
Complete inactivation of 6 log10 M. terrae on steel and wood disks was achieved by 10 s of steam treatment at all three background temperatures. In comparison, 20 s of steam treatment was needed for the complete inactivation of mycobacteria on rubber disks. Corresponding to the longer treatment time required for mycobacterial inactivation, temperatures on the bottom surface of the rubber disks rose substantially slower than those of the steel and wood disks at all three background temperatures.
Conclusion:
The results suggested that treatment with pressurized steam has potential for efficient and effective disinfection of surfaces contaminated by mycobacteria at or below freezing temperatures in winter.
Introduction
Bovine tuberculosis (TB) is a chronic contagious bacterial disease of livestock caused by infection with Mycobacterium bovis. 1 Infected animals with progressive disease shed the bacteria in respiratory secretions and aerosols, milk, feces, urine, and vaginal secretions or semen. 2 Susceptible animals usually acquire infection by the respiratory route and occasionally the oral route. Bovine TB also affects many wildlife animals such as cervids, badgers, feral pigs, possums, and buffaloes.2,3
The extensive reservoir in wildlife makes it difficult to eradicate the disease. Bovine TB is zoonotic and may be transmitted to humans through ingestion of unpasteurized dairy products, inhalation of infectious aerosols, or direct contact through breaks in the skin. 1 The disease threatens both animals and humans and may lead to significant economic damages if not controlled properly. 4
Bovine TB is a reportable disease in Canada. If the disease is confirmed on a premises, all infected and susceptible exposed animals will be subjected to humane destruction, and the premises and equipment will undergo thorough cleaning and disinfection for disease control and eradication. 5 M. bovis is an obligate aerobe bacillus with a complex thick cell wall that protects the bacterium against various environmental stresses such as pH, desiccation, humidity, freezing, and heating, and enables it to survive on affected farms for months. 6 Chemical disinfectants are commonly used for surface decontamination of equipment and tools on affected farms.
A long list of disinfectant products has been registered with claims of mycobactericidal effects within minutes in clinical and residential settings, 7 however, on farms chemical disinfection encounters significant challenges. First, a majority of chemical disinfectants are toxic to animals and may cause damage to farm equipment. 8 Heavy organic load on affected surfaces may serve as a barrier to active ingredients and reduce the effectiveness of disinfectants.9,10 More importantly, low temperatures close to or below the freezing point of water may substantially slow down the disinfection process.10,11 In addition, widespread and repeated use of disinfectants may lead to adaptation of mycobacteria to the disinfectants and increase their tolerance toward antibiotics. 12
Heat treatments, such as pasteurization, 13 direct steam injection, 14 and other thermal processes, 15 have been commonly used in the food industry for inactivation of mycobacteria in dairy and meat products. Also steam disinfection, as an alternative to chemical disinfection, has been tested for surface decontamination in food production and clinical settings where superheated steam with temperatures above the saturation point at a set pressure could inactivate Geobacillus stearothermophilus spores, 16 Salmonella enteritidis strains, 17 and multidrug-resistant nosocomial bacteria18,19 within seconds or minutes.
Encouraged by the mentioned steam disinfection trials, this study evaluated the mycobactericidal effect of treatment with pressurized steam for surface decontamination at cold weather temperatures that could occur on Canadian farms. Mycobacterium terrae was used as a surrogate for M. bovis since M. terrae has been commonly used for evaluation of microbicides against mycobacteria. 20 The evaluation was carried out using the second quantitative carrier test (QCT-2) developed by Springthorpe and Sattar 21 with carrier disks made of various materials.
Methods
Bacterial Inoculum
M. terrae (ATCC 15755) was used as a surrogate for M. bovis in this study. It was grown to log phase in 100 mL Middlebrook 7H9 broth (BD Biosciences, Mississauga, Canada) with 10% (v/v) albumin–dextrose complex (BD Biosciences) and 15% (v/v) glycerol in 75 cm2 cell culture flasks with a canted neck and a 0.2 μm vented plug cap (Sarstedt, Inc., Newton, NC) for 21 days at 37°C. The bacterial culture was centrifuged at 10,000 g for 10 min at 4°C.
The cell pellets were washed twice by resuspending in 100 mL sterile distilled water and centrifuged as before. The cell pellets were resuspended with distilled water by mixing on a Vortex mixer with 10 sterile 5-mm diameter glass beads. Colony forming units (CFU) of the cell suspension were determined by spread plating (100 μL) 10-fold serial dilutions in sterile saline on Middlebrook 7H11 agar (BD Biosciences).
Preparation of Organic Load
American Society for Testing and Materials standard method E2197-02 was followed in preparation of organic load to simulate body fluids. 22 The organic load added to the suspension of bacterial inoculum was a mixture of the following stock solutions in phosphate-buffered saline (pH 7.0; Gibco, Nepean, Canada): (1) 5% yeast extract (Oxoid, Nepean, Canada), (2) 5% bovine serum albumin (Sigma, Oakville, Canada), and (3) 0.4% bovine mucin (CalBioChem, Oakville, Canada).
The solutions were prepared separately and sterilized by filtration through a syringe filter with a pore diameter of 0.22 μm (Millipore, Bedford, MA), and stored at 4°C. To prepare 500 μL of the test inoculum with the organic load, 340 μL of the homogenized cell suspension was mixed with 25, 35, and 100 μL of the stock solutions of yeast extract, bovine serum albumin, and bovine mucin, respectively.
Preparation of Carrier Disks
Three types of carrier disks were used: brushed stainless steel disks (AISI No. 430; Muzeen & Blythe, Winnipeg, Canada), synthetic rubber disks (Viton™, product No. 27351; Sigma), and pine wood disks (custom made). Disk diameter and thickness were measured using a caliper and given in Table 1. Before use, the disks were washed three times with distilled water and dried at 60°C for 1 h. The dried disks were sterilized at 121°C for 25 min.
Carrier disk properties
Steel = stainless steel No. 430, wood = pine wood, rubber = synthetic rubber (Viton™).
Thermal conductivity is obtained from Materials Thermal Properties Database. 27
Each carrier disk made of steel, wood, or rubber was inoculated with 10 μL of a freshly vortexed mycobacterial suspension supplemented with the organic load containing ∼6 log10 CFU of M. terrae. To test the influence of the organic load on disinfection of mycobacteria, additional steel disks were inoculated with 6 log10 CFU of M. terrae in distilled water. The inoculum was deposited onto the disk center using a pipette. Inoculated disks were dried in a desiccator under vacuum for 1 h at room temperature. Each dried and inoculated disk was carefully placed with sterile forceps, with the inoculated side up, on the inside bottom surface of a 30-mL Nalgene® polypropylene straight-side vial.
Test Procedure
The steam disinfection process was evaluated using the QCT-221 with disks kept at −20°C, 4°C, and 21°C. For each type of carrier disk at each temperature, two sets of quadruplicate disks with inoculum were used, one set subjected to steam treatment as described hereunder and the other set as control for no treatment. Each carrier disk was held in a vial that was planted into a metal-bead bath in a plastic container (Figure 1). The container and its contents had been incubated at −20°C, 4°C, or 21°C for 24 h for temperature stabilization. Immediately before the steam treatment, the container was transferred into a biosafety cabinet and the lids of all vials were removed.

Pictures and diagrams showing the layout of vials containing carrier disks for steam treatment using a household steamer. Ctl, control.
A household steamer, Bissell® Steam Shot™ Hard Surface Cleaner (Bissell, Gatineau, Canada; Figure 1), was used to generate pressurized steam according to the manufacturer's instruction. In brief, the steamer was filled with 300 mL distilled water and connected to the power. When the indication light turned off, the steaming trigger was pressed to release steam until a constant stream of steam was obtained. The nozzle of the steamer was then positioned directly over the inoculated carrier disk and steam was applied for 5, 10, 15, or 20 s.
The nozzle was ∼5 mm above the carrier disk during the treatment and had no direct contact with the disk to prevent cross-contamination. Safety face shields and insulated gloves were used as personal protective equipment when performing the steaming procedures.
Immediately after the steam treatment, the carrier disk was transferred using a pair of sterile forceps into a 15-mL conical centrifuge tube that contained 10 mL phosphate buffer (0.067 M, pH 6.8) supplemented with 0.1% (v/v) Tween 80 (PBT). The contents in the conical tube were vortexed for 30 sec. The disk was then transferred into a 25-cm2 cell culture flask containing 10 mL Middlebrook 7H9 broth and incubated for 30 days to determine whether any residual M. terrae survived on the disk. The liquid content in the conical tube was subjected to 10-fold serial dilutions that covered a 6 log10 range using PBT.
Each of the six dilutions was poured onto a 47-mm membrane filter with a 0.45 μm pore diameter (Millipore). Each dilution tube was rinsed with 20 mL PBT and the wash passed through the same filter. The inside surface of the filter unit was washed with at least 40 mL PBT and passed through the same filter. The filter was removed from its holder and placed on a plate of Middlebrook 7H11 agar. The plates were incubated in an inverted position at 37°C and inspected every few days. Those plates were held for 30 days for CFU count and calculation of log10 reductions. Immediately after the removal of the carrier disk, the vial containing the steam effluent was placed on ice and then the effluent was subjected to dilution, filtration, and culture process for enumeration of M. terrae as described earlier.
Repeats and log10 Reduction in CFU
Triplicate experiments were conducted for each type of carrier disks at each background temperature for each treatment duration. In each experiment, there were four steam-treated disks and four control disks with no steam treatment. Mycobactericidal activity was determined by comparing the growth of mycobacteria from each treated disk with the mean of the growth from the control disks. The log10 reduction was calculated from log10 (mean of four controls) – log10 (CFU of each test carrier). The mean (±standard deviation) log10 reduction was calculated from the results of three experiments.
Disk and Steam Temperature
To determine disk temperature during the steam treatment, vials containing disks without mycobacterial inoculum were set up the same way as already described. A type K thermocouple connected to a thermometer (Sper Scientific 800005, Scottsdale, AZ) was in contact with the top or bottom surface of the disk to measure disk temperatures at 0, 5, 10, 15, and 20 s of steaming. To determine the temperature of steam leaving the steamer, the thermocouple was in contact with the outlet of the nozzle. Temperatures were recorded every 10 s for a period of 3 min after a constant stream of steam was obtained, and the process was repeated three times.
Statistical Analysis
The data presented are the mean values of log10 reduction in CFU of M. terrae on quadruplicate disks from three experiments. One-way analysis of variance was used to determine significant differences from different types of disks at the same background temperature for the same duration of treatment time. Data were analyzed using GraphPad Prism 8.0 software. The critical level for significance was set at p < 0.05.
Results and Discussion
Cleaning and disinfection of fences, feed troughs, and other equipment and tools could be challenging during bovine TB outbreaks on Canadian farms in winter. Freezing temperatures and heavy organic load on affected surfaces would significantly reduce the efficiency and possibly impair the effectiveness of chemical disinfection.9,10 Thus, heat treatment using steam has been of practical interest, but information is needed on whether steam treatment provides inactivation of mycobacteria at ambient temperatures close to or below the freezing point of water. Hence, in this study carrier disks were kept at background temperatures of −20°C, 4°C, and 21°C to simulate temperatures that could occur on farms.
Considering possible high titer shedding and low infectious dose of M. bovis in cattle,23,24 this study targeted a 6 log10 reduction of mycobacteria by the steam treatment, instead of a 4 log10 or 99.99% reduction required for chemical disinfectants claiming against mycobacteria. 25 As such, an inoculum containing 6.32 ± 0.38 log10 CFU of M. terrae was used to evaluate the mycobactericidal effect of pressurized steam on surfaces made of steel, wood, and rubber materials commonly found on farms.
Steel disks were used for evaluating the influence of organic load on the steam treatment. Temperature of the pressurized steam was 120°C ± 5°C at the outlet of the nozzle of the steamer. At the background temperature of 21°C, 5 and 10 s of the steam treatment were required to produce a complete inactivation of 6 log10 M. terrae in the absence and presence of organic load, respectively (Table 2). Complete inactivation was confirmed by no viable M. terrae recovered from the disk and the effluent from the steam treatment.
Reduction (log10) of Mycobacterium terrae on steel or wood disks by steam treatment
Steam treatment for inactivation of Mycobacterium terrae in the absence (−) or presence (+) of organic load on steel or wood disks was carried out at various background temperatures for various duration. All values in bold (log10 colony forming units per disk) represent complete inactivation of all inoculated bacteria on disks.
As expected, the organic load simulating body fluids reduced the efficiency of the steam treatment. Very likely, a treatment time longer than 10 s would be required for complete inactivation of 6 log10 mycobacteria in a layer of feces or soil that could hinder heat transfer. As organic matters are ubiquitous on farms, the steam treatment was evaluated in the presence of organic load for various surfaces and background temperatures. In the presence of the organic load, 10 s of the steam treatment completely inactivated 6 log10 M. terrae on steel and wood disks at all three background temperatures, 21°C, 4°C, and −20°C (Table 2).
In comparison, at the background temperature of 21°C, 10 s of the steam treatment resulted in 3.91 log10 reduction of M. terrae on the rubber disks (Table 3), which was significantly (p < 0.05) lower than those on the steel and wood disks (Table 2). Longer treatment time (20 s) was needed for complete inactivation of 6 log10 M. terrae on rubber disks (Table 3). Furthermore, no viable M. terrae was recovered from the effluent of steam treatment under any testing conditions in this study.
Reduction (log10) of Mycobacterium terrae on rubber disks by steam treatment
Steam treatment for inactivation of Mycobacterium terrae in the presence of organic load on rubber disks was carried out at various background temperatures for various duration. All values in bold (log10 colony forming units per disk) represent complete inactivation of all inoculated bacteria on disks.
Possibly due to the horizontal disk lay out, mycobacteria could remain on the disks during the entire treatment. However, surfaces on farm are of various orientations and curvatures, steam treatment may lead to early release of viable mycobacteria into the effluent. Therefore, collection and further process of the effluent may be needed for thorough disinfection.
Temperature profiles of the disk were investigated to better understand the association between disk temperature and mycobactericidal effect of the steam treatment. Since it is difficult to accurately obtain true surface temperature of objects during steam treatment, 26 temperature was measured on the top and bottom surfaces of the disks to provide a possible range containing the true value. Temperatures of control disks that received no steam treatment remained at the set background temperatures during the experiments (data not shown).
Temperature profiles on the top and bottom surfaces were similar for the steel and wood disks, reaching 73°C within 5 s and staying above that for the rest 5 s of steam treatment at all three background temperatures (Figure 2). Such combination of temperature and time should enable the complete inactivation of 6 log10 M. terrae on the steel and wood disks, as 4.86 s was expected to produce 6 log10 reduction (0.81 s per 1 log10 reduction) of mycobacteria in the high temperature short time pasteurization of milk at 72°C. 13
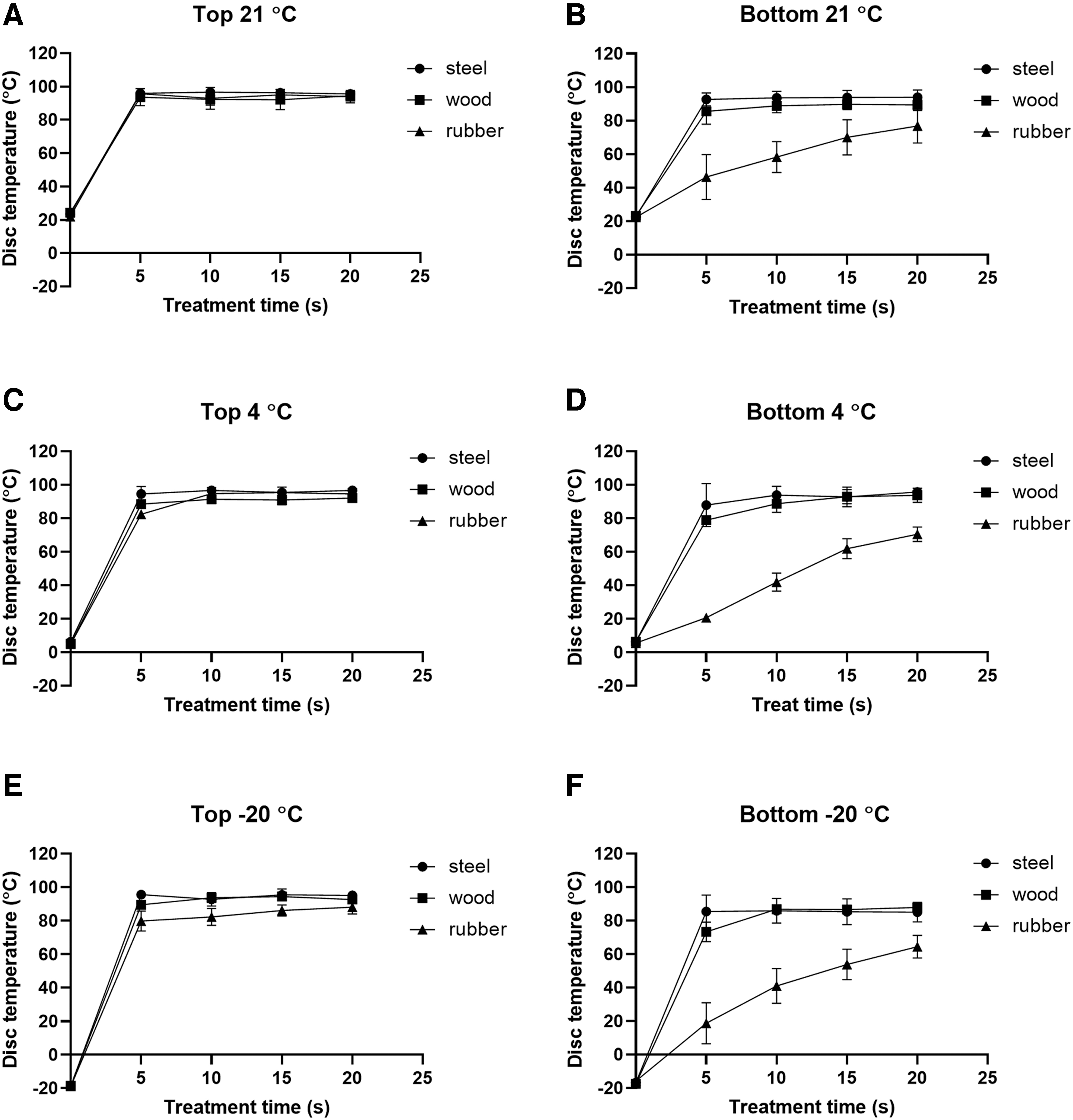
Temperature profiles on the top
For rubber disks, the temperature on the bottom surface rose significantly slower than the top surface and reached 64°C, 71°C, and 77°C by 20 s of steam treatment at the background temperatures of −20°C, 4°C, and 21°C, respectively (Figure 2). In correlation with the slower temperature rise, longer treatment time was needed for complete inactivation of 6 log10 mycobacteria on rubber disks as compared with steel and wood disks. Greater thickness and lower thermal conductivity (Table 1) might slow down the rate at which the temperature rose for rubber disks compared with steel disks.
In contrast, the thickness and thermal conductivity of wood disks were like those of rubber disks (Table 1). Likely, the porosity of wood might facilitate steam penetration and support faster temperature rise of wood disks. When steam was used for heating, a layer of condensed water would form on the surface of the object and might impede heat transfer from steam to the object and reduce the bactericidal effect by steam treatments. As an evidence, James et al. 26 reported that the static steam treatment at atmospheric pressure for 10 s reduced only 1.65 log10 CFU of total viable bacteria on 1 cm2 of chicken portions.
In comparison, pressurized steam was ejected from a proximity (∼5 mm) onto the disk surface in our study. The microdroplets of water in the steam might disturb the layer of condensed water by collisions and presumably decreased the thermal resistance of the water layer. As such, 10 or 20 s of the steam treatment was able to produce 6 log10 reduction of M. terrae on various surfaces. In support of our findings, microdroplets of hot water were able to penetrate cavities and crevices on nuts for inactivation of 5.73 log10 CFU Salmonella during spraying superheated steam and pressurized boiling water. 17
Overall, the steam treatment in this study was evaluated using small disks (∼1 cm in diameter) maintained at set background temperatures and pressurized steam generated by a small household steamer (300 mL in capacity). Results from this small-scale laboratory evaluation demonstrated that the steam treatment efficiently and effectively inactivated mycobacteria on steel, wood, and rubber surfaces at background temperatures close to and below the freezing point of water. These promising findings may serve as a basis for future large-scale on-farm evaluations of steam treatment using mobile industrial boilers following appropriate occupational health and safety rules for decontamination of surfaces affected by mycobacteria.
Footnotes
Acknowledgments
Appreciation is expressed to Dr. Olga Andrievskaia, Ottawa Laboratory (Fallowfield), Canadian Food Inspection Agency, for providing the Mycobacterium terrae (ATCC 15755) strain, and to Ms. Tamara Morrill and Mr. Gokhan Yilmaz for their assistance in determination of temperatures on disk surfaces.
Authors' Contributions
J.G. and E.R. conceptualized the study. J.G. and M.C. carried out the experiments and analyzed the data. J.G. wrote the article. All authors contributed to and approved the submitted version.
Authors' Disclosure Statement
No competing financial interests exist.
Funding Information
This study was supported by the Canadian Food Inspection Agency on evaluation of steam treatment for decontamination of surfaces affected by mycobacteria (N-000113 - OLF-A-1804).
