Abstract
Background:
Aromatherapy is widely used in women's health as a complementary therapy.
Objective:
This review aimed to critically evaluate clinical evidence of the effectiveness of aromatherapy in managing menopausal symptoms.
Design:
A systematic review and meta-analysis.
Methods:
A total of 11 electronic databases were searched up to November 5, 2020. Randomized controlled trials (RCTs) evaluating any type of aromatherapy against placebo in menopausal individuals were eligible. Two authors independently assessed the study eligibility and risk of bias as well as extracted the data for each study. Cochrane risk of bias tool was used to evaluate the methodological quality of each included studies. No ethical approval was required for this manuscript as this study did not involve human subjects or laboratory animals.
Results:
Seven RCTs met our inclusion criteria and were analyzed. Most of the included RCTs had low risk of bias in most domains except for blinding. Four studies tested the efficacy of aromatherapy for menopausal symptoms. The results showed that aromatherapy (lavender or low dose of neroli) inhalation had superior effects on reducing the total menopausal symptom score compared with the placebo. One study presented that aromatherapy massage with mixed oils reduced menopausal symptoms. Three studies investigated the effects of aromatherapy inhalation on sexual desire, and all of the studies showed that aromatherapy inhalation (lavender or low dose of neroli or mixed oils) had superior effects compared with the placebo.
Conclusion:
This review indicates limited evidence of the benefit of aromatherapy (lavender or low dose of neroli) in improving total menopausal symptoms and sexual desire. Further studies are highly warranted to validate the findings.
Introduction
Menopause often presents unpleasant symptoms such as chills, hot flashes, palpitations, sleep disturbances, fatigue, vaginal dryness, low libido, osteoporosis, emotional liability, and depression. 1 Hormone replacement therapy (HRT) of combined oestrogens and progestogens or oestrogens alone has been one of the most effective therapeutics for the management of menopausal symptoms. 2 As the risk of HRT is highly publicized, many women tend to seek alternative options out of concern. 3 –6 Aromatherapy is the therapeutic method of using aromatic essential oils, which are mainly extracted from flowering and nonflowering herbs, applied through inhalation, massage, or baths, and may improve the well-being of the body, mind, and spirit. 7
Phytoestrogens of essential oils used in aromatherapy have shown many potentials in relieving menopausal symptoms and improving postmenopausal lipid profiles in women. 7 Many clinical studies have revealed the therapeutic benefits of aromatherapy in relieving anxiety and stress, reducing pain, enhancing alertness, and inducing relaxation, by stimulating endorphin production. 7 –11
There are five systematic reviews of aromatherapy for menopausal symptoms. 12 –16 All of the reviews concluded that aromatherapy may be effective for relieving menopausal symptoms. However, all five had several limitations, which include the lack of comprehensive literature search, 12 –15 being out of date, 12 –14 assessing evidence in a mixed population, 12,14,15 analyzing duplicate studies as different trials, 16 pooling data from randomized controlled trials (RCTs) and non-RCTs together, 14,16 and inappropriate selection of RCTs. 16 Hence, their assessment of evidence exaggerates the questionable results. This systematic review aimed to summarize and critically appraise the current evidence on the effectiveness of aromatherapy for the management of menopausal symptoms.
Methods
Registration
Before perfoming the formal review, this systematic review was registered in PROSPERO 2017 (CRD42017079191: available from
Search methods for identifying studies
Electronic sources
The following 11 electronic databases were searched for studies published from database inception to November 5, 2020: MEDLINE PubMed, EBSCO Embase (EBSCO), Cochrane Central Register of Controlled Trials (CENTRAL), EBSCO Cumulative Index to Nursing and Allied Health Literature (CINAH), EBSCO Allied and Complementary Medicine Database (AMED), Korean Studies Information Services System (KISS), Research Information Service System (RISS), Oriental Medicine Advanced Search Integrated System (OASIS), Korean Medical Database (KM base), DBpia, and KoreaMed. In addition, the reference lists of eligible articles and relevant review articles were manually searched to identify additional relevant records. All the full-text articles were obtained and read in the form of hard copies. The same search strategy with modifications was used for each database.
Search strategies
The literature searches were performed using the medical subject heading (MeSH) terms “aromatherapy OR essential oil” AND “menopause OR climacteric OR menopausal.”
Eligibility criteria
Types of studies
All RCTs evaluating the therapeutic effects of aromatherapy compared with placebo were included.
Types of participants
The authors included data from studies that examined menopausal women of both perimenopausal and postmenopausal.
Types of interventions
Trials that investigated the effects of any type of aromatherapy were eligible regardless of the therapy duration, dosage, and administered routes (massage, inhaled, baths, or oral). The studies that used placebo as control treatments were included.
Outcome measures
Primary outcomes
Any menopausal symptoms that were measured with the menopause rating scale (MRS), Greene climacteric scale (GCS), Kupperman index, or other validated questionnaires.
Female sexual function, measured with validated questionnaires such as sexual functioning questionnaire, female sexual function index, and brief index of sexual functioning for women.
Secondary outcomes
Only adverse effects (AEs) were considered secondary outcomes.
Data management and analysis
Study selection
The process of screening and selection of data was performed separately by two authors (J.C. and L.A.) and was verified by a third author (H.W.L.). When disagreements on inclusion were not resolved through discussion, the arbiter (M.S.L.) made the decision after all review authors' opinions were considered. No language restrictions were imposed. The selection process is detailed in the preferred reporting items for systematic reviews and meta-analyses flow diagram.
Data extraction
After reading and obtaining all the hard copies of the eligible articles, three reviewers independently extracted the data from the articles according to the published protocol's predefined criteria. Data details of the first author's last name, publication year, trial design, sample size, interventions and control regimens, main outcome measures and findings, AEs, and authors' conclusions were extracted. The extracted data were tabulated in Microsoft Excel for further analysis.
Risk of bias assessment
The review authors independently assessed the risk of bias in the included studies according to the criteria from the Cochrane Handbook version 5.1.0, which includes “random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting and other sources of bias. The quality of each trial was categorised as having a low, unclear, or high risk of bias.” 18 Any differences in opinion were resolved through discussion with a third author.
Treatment effect measures
Continuous data are presented as mean differences (MDs) with 95% confidence intervals (CIs) to measure the treatment effect. In the case of other types of data or outcome variables with different scales, the authors converted the data into MDs or used standard MDs with 95% CIs. Dichotomous data are presented as relative risks (RRs) with 95% CIs and other binary data as RR values.
All of the statistical analyses were conducted using Cochrane Collaboration's software program, Review Manager (RevMan) version 5.4.1. for Windows (The Cochrane Collaboration, 2020). The corresponding authors of the particular study were contacted to acquire and verify the insufficient data when possible. The chi-square and I 2 tests were used for the evaluation of the heterogeneity of the included studies. For meta-analysis, the data across studies were pooled using a random-effects model, if there was no excessive statistical heterogeneity present.
The albatross plot for each outcome was generated using STATA/SE v.16.1 (StataCorp LLC, College Station, TX, USA) to understand the overall direction of results for each outcome. The albatross plot also served the purpose of complementing the available effects from the results of meta-analysis. Each outcome of an individual study was presented as a point in the plot, plotted against the sample size with p-value as effect estimates.
Analysis unit
For trials with crossover designs, the data from the first treatment period were used for the analysis of treatment effect. In the case wherein there are more than one control group evaluated, data of each control group were combined during primary analysis and the subgroup analyses were then performed for each group.
Assessment of heterogeneity
As recommended by the Cochrane Handbook for Systematic Reviews of Interventions, I 2 statistic was used to quantify the inconsistencies among the included studies with the value of 50% as the cutoff point for significant heterogeneity, if meta-analysis was possible. When heterogeneity was observed in the meta-analysis, a subgroup analysis was conducted to explore the possible causes of heterogeneity. 19 Subgroup analyses for different control types, intervention types, condition types, and study designs were conducted.
Assessment of reporting bias
If at least 10 trials or a sufficient number of studies were included, the use of funnel plots to detect reporting bias was planned. 20 If funnel plot asymmetry differed for publication bias, distinction of the possible reasons (small effects size, low methodological quality, or true heterogeneity among the included studies) for the asymmetry found was attempted.
Results
Description of the included trials
In total, 446 potentially relevant studies were identified, of which 9 articles 21 –29 from 7 studies met the inclusion criteria, as shown in Figure 1. Although one of the excluded studies claimed the study was an RCT, 30 it used random sampling, not randomization. Table 1 summarized the key data of all included studies. The authors of two trials published separate articles with different outcomes, 23,24,27,28 and the trials with the primary outcomes of interest as the representative studies were designated. 23,27 Five RCTs assessed inhalation aromatherapy, 21 –23,25,26 and two RCTs assessed massage aromatherapy. 27,29 Five of the included RCTs originated in Iran, 22,23,26,27,29 one in Taiwan, 21 and one in Korea. 25 The studies were published between 2012 and 2020. Six of the included trials used a parallel design, 21,22,25 –27,29 while one RCT employed a crossover design. 23 Two RCTs measured menopausal symptoms using the Green climacterics scale, 21,23 one used the MRS, 27 and the other used the Menopause-Specific Quality of Life (MENQOL). 25
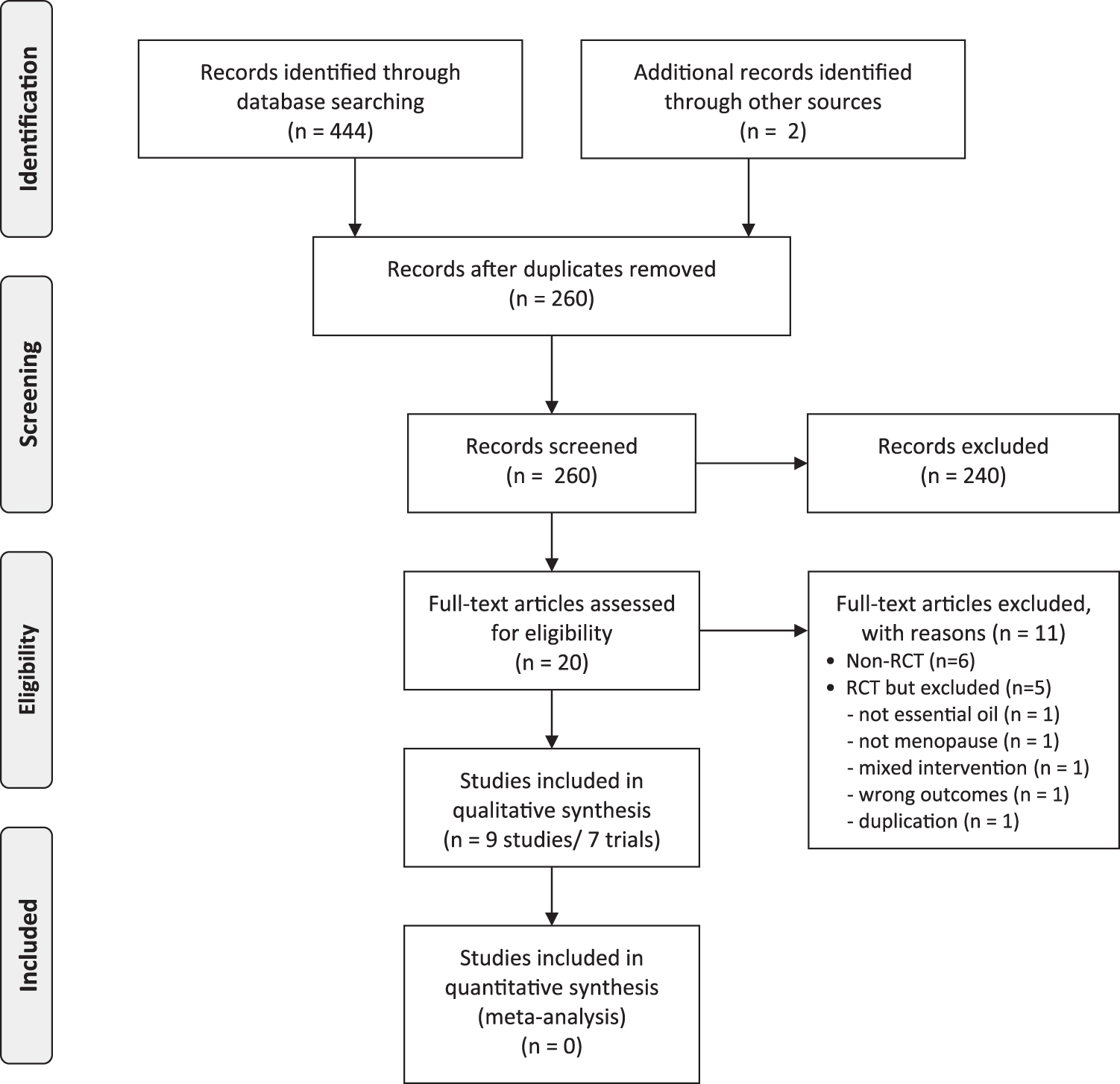
Flow chart of the trial selection process. RCT, randomized controlled trial.
Summary of Randomized Clinical Studies on Aromatherapy for Managing Menopausal Symptoms Compared with a Placebo Control
We excluded the italicized controls in the analysis because of not fit to our inclusion criteria.
Differ from other outcomes, a higher score indicates better condition.
AEs, adverse effects; BDI, Beck depression inventory; FSFI, female sexual function index; GCS, Greene climacteric scale; MENQOL, Menopause-Specific Quality of Life; MRS, menopausal rating scale; PSQI, Pittsburgh Sleep Quality Index.
Risk of bias
The risk of bias was low in most of the domains for the included RCTs (Fig. 2). All of the trials reported random sequence generation except one RCT. 29 Four studies reported allocation concealment. 21,22,25,26 Although all trials used a placebo control, the domains related to blinding were scored as unclear risk of bias because of the nature of aromatherapies. None of the included RCTs mentioned outcome assessment blinding. Two studies had a high risk of bias for selective reporting because they published other articles with different outcomes. 23,27 One study did not report the reasons for dropout and withdrawal of participants. 25
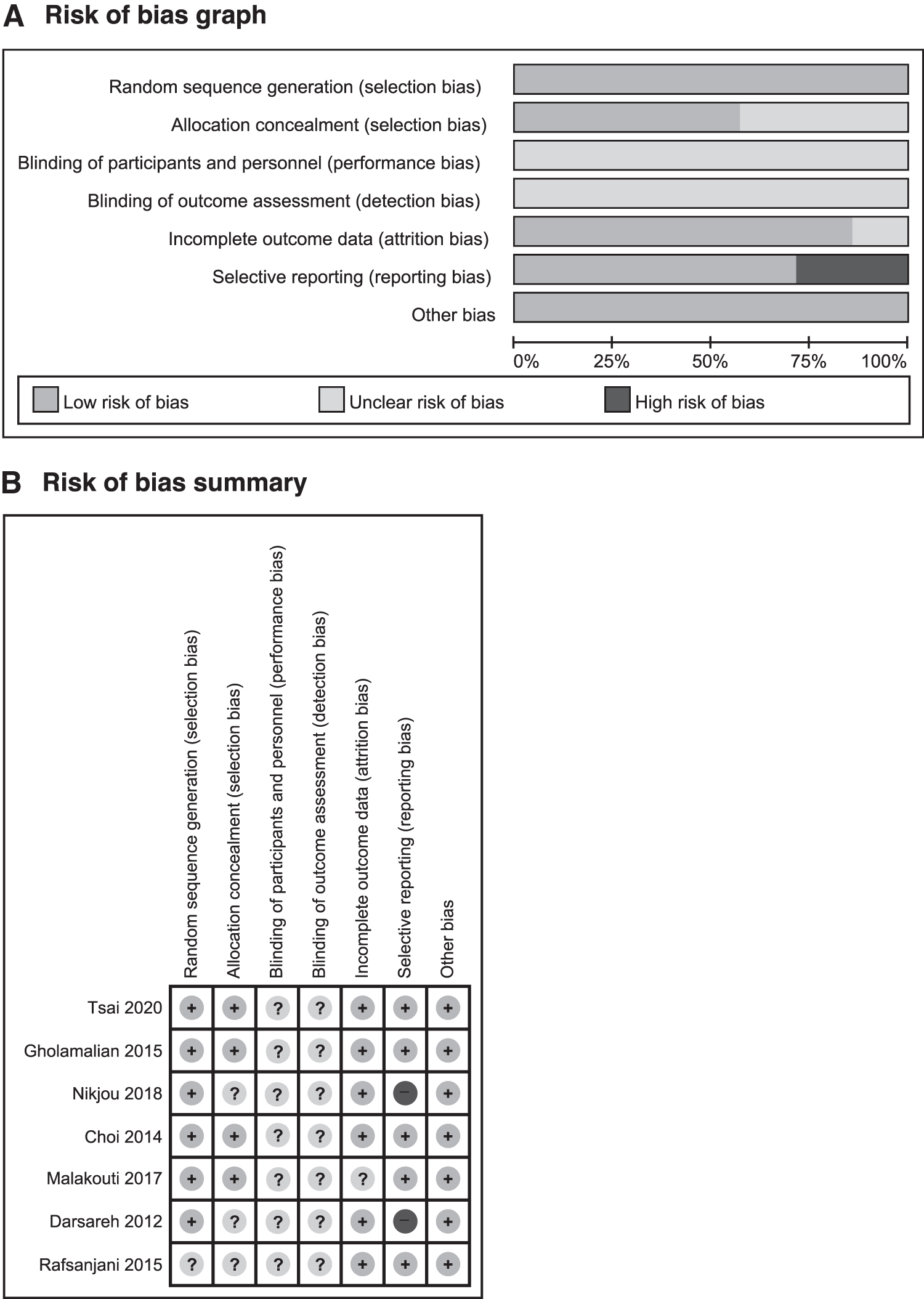
Risks of bias.
Outcome measurements
Menopausal symptoms
Four studies tested the effects of aromatherapy inhalation or aromatherapy massage on menopausal symptoms with the GCS, MRS, or MENQOL compared with placebo (Fig. 3A). 21,23,25,27 One study showed that lavender inhalation reduced menopausal symptoms compared with placebo, 23 while the other study with a combination of essential oil inhalation failed to do so. 21 A third study tested two doses of neroli oil inhalation for menopausal symptoms. A lower dose of neroli oil reduced menopausal symptoms compared with placebo, while a higher dose showed the tendency, not statistical significance. 25 One study showed that aromatherapy massage reduced menopausal symptoms. 27 The results were not pooled because of clinical heterogeneity including the type of essential oils.
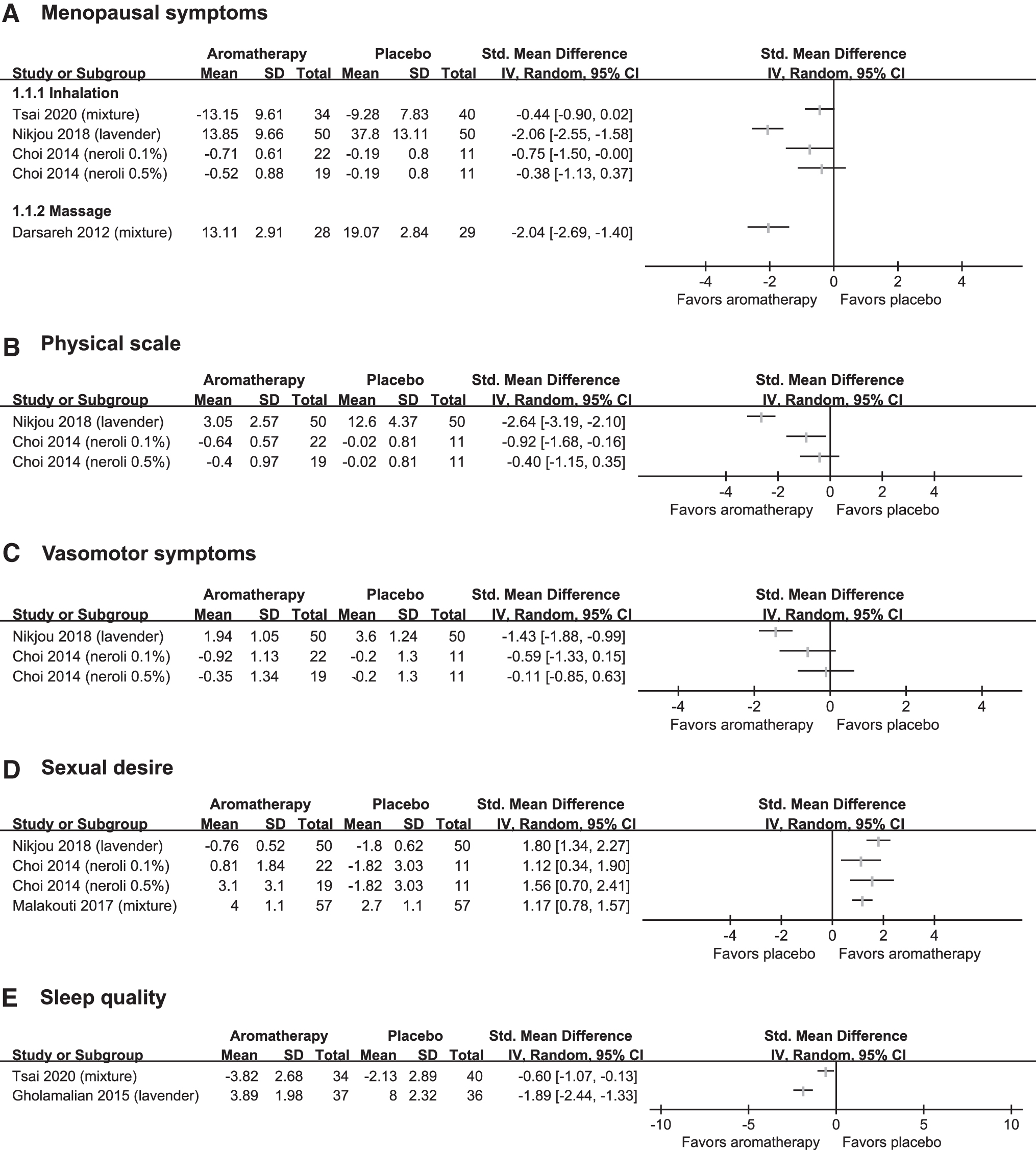
The forest plot of
Two studies reported physical subscales. 23,25 One study with high dose neroli oil inhalation failed to show that aromatherapy inhalation improved the physical score, 25 while lower dose neroli oil and another study with lavender inhalation showed beneficial effects of aromatherapy. 23
Two studies tested the effects of aromatherapy inhalation for vasomotor symptoms. 23,25 One study with lavender oil inhalation showed superior effects of aromatherapy on vasomotor symptoms compared with placebo, 23 while the other study with neroli oil inhalation failed to do so. 25
Three studies investigated the effects of aromatherapy inhalation on improving sexual desire, 23,25,26 and the results of all these studies showed superior effects of aromatherapy (lavender, neroli, and mixture) inhalation compared with the placebo (Fig. 3D).
Two studies assessed aromatherapy inhalation for improving sleep quality 21,22 and both studies showed favorable effects of aromatherapy (lavender or mixture) inhalation compared with the placebo (Fig. 3E).
Of the 22 data points (from 7 studies) included in the albatross plot, 19 data points showed positive associations of aromatherapy on menopausal symptoms (including total menopausal symptoms, sexual desire, depression, physical scores, and sleep quality), whereas three data points (psychological score from two studies and vasomotor symptom from one study) showed null association (Fig. 4).
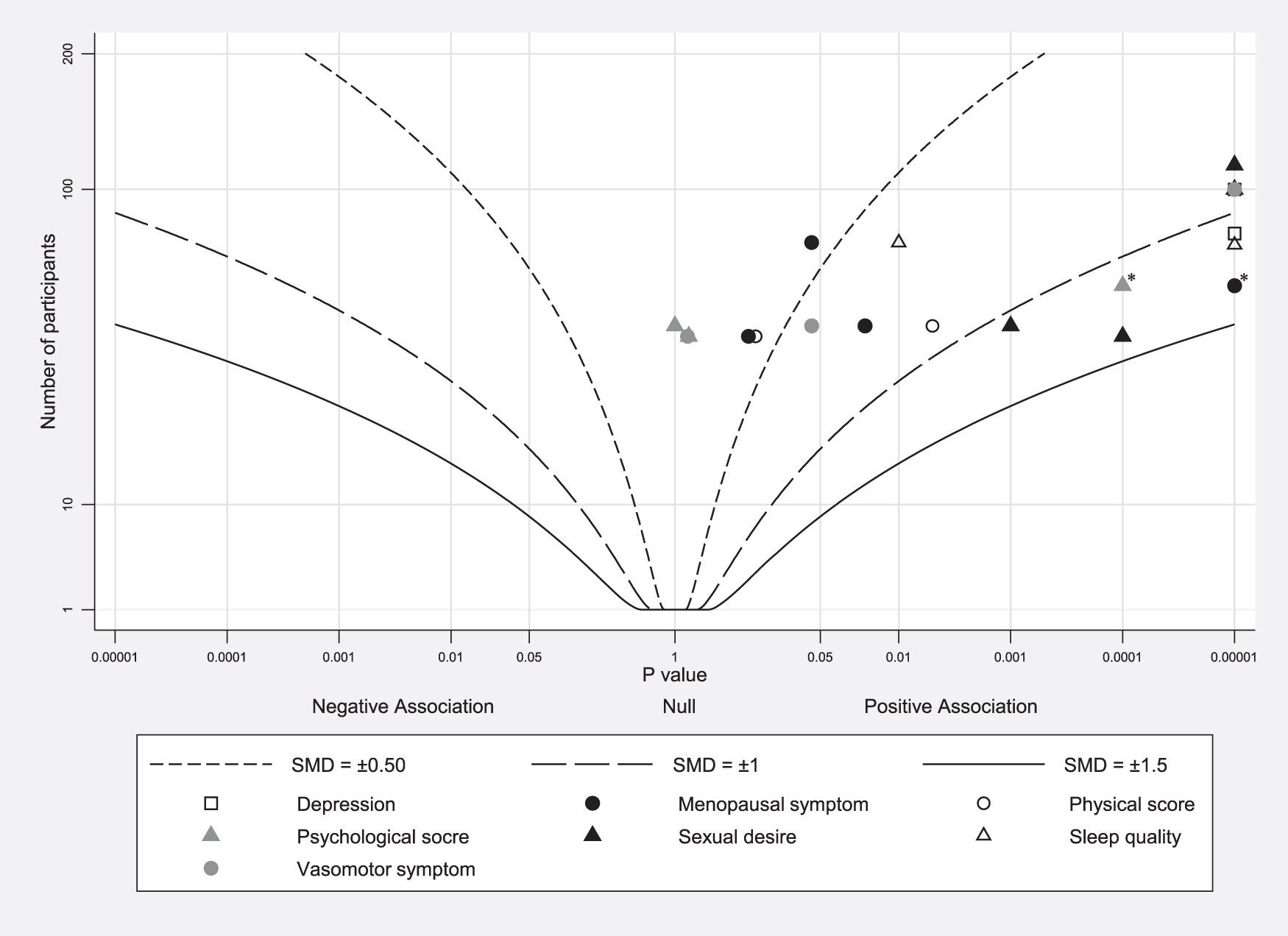
Albatross plot for each outcome.
Adverse effects
Four studies assessed AEs, 22,26,27,29 while the other three studies did not measure AEs 21,23,25 (Table 1). Three RCTs reported no AEs, 22,27,29 while one study reported several AEs in both groups, including skin redness, itching, nausea, abdominal pain, headache, and gripe. 26
Discussion
Summary of the main results
Few rigorous studies have evaluated the effect of aromatherapy on menopausal symptoms. Evidence from the included studies suggests that aromatherapy (lavender or low dose of neroli) may improve total menopausal symptoms slightly compared with a placebo. Other types of oils should be tested further. However, the small number of studies, small sample size, and unclear risk of bias prevent robust conclusions.
Overall completeness and applicability of evidence
Aromatherapy (lavender or low dose of neroli) may improve menopausal symptoms slightly than placebo. However, these results should be generalized with caution because of potential heterogeneities in the ingredients of essential oils and the countries in which trials are conducted.
Agreement or disagreement with other studies or reviews
There were five previous reviews of aromatherapy for menopausal symptoms. 12 –16 One of which included five trials of two RCTs and three controlled clinical trials on the effects of aromatherapy on menopausal symptoms. 14 Its findings suggest that aromatherapy is effective for relieving menopausal symptoms. This review was successfully updated by adding six more RCTs 21 –23,25,26,29 and formed a similar conclusion. Two recent systematic reviews from one research group that assessed sexual problems in menopausal women and psychological symptoms in postmenopausal and elderly women were published in the same year. 12,13,15 One review with three RCTs concluded that aromatherapy had beneficial effects on sexual dysfunction, 13 and the other two reviews also indicated favorable effects of aromatherapy (two RCTs: one in menopausal women and one in elderly individuals) 12 or lavender (three RCTs) on psychological symptoms. 15 However, one review combined menopausal women and elderly individuals, and all of the RCTs and non-RCTs were analyzed together without considering their limitations. Sixteen very recent studies (nine RCTs and seven non-RCTs) tested aromatherapy with lavender. 16 The authors observed beneficial effects of aromatherapy with lavender on physical and psychological symptoms in menopausal women. However, this review had several inappropriate analyses, including the analysis of duplicate studies as different trials, analysis of pooled data from RCTs and non-RCTs, and assessment of non-RCTs as RCTs. Hence, their assessment of evidence is not reliable and not convincing. Compared with these results, their review showed exaggerated values because of pooled RCT and non-RCT data. The results provide objective evidence for readers.
Implications for practice
The clinical efficacy of essential oil inhalation may be good, and it appears to be valuable for clinical use. However, due to the small number of studies and unclear minimum clinically important differences (MCIDs), the objectivity of the efficacy assessment should be confirmed with large-scale rigorous studies. The authors also relied mainly on self-reported measures to determine the effect on the main outcomes.
Potential mechanism of action of aromatherapy
If aromatherapy is assumed to be beneficial for improving menopausal symptoms, it may be interested in the possible mechanisms of action. One plausible explanation might be the involvement of the autonomic nervous system by essential oils inhalation including linalool for lavender. 7,31 It might modulate the parasympathetic nervous system through the hypothalamus related to touch and smell. 7,31 Others have postulated that essential oils inhalation or massage might stimulate the production of neurotransmitters including endorphin, serotonin, and dopamine level. 32 However, this might be different according to the type of essential oils. More basic research is needed to understand fully the mechanisms of action of aromatherapy for treating menopausal symptoms.
Potential bias in the review process
The authors judged the blinding of participants and personal according to using the measurement of success of blinding regardless of their claims. However, none of them assessed this measurement and they were scored as unclear bias. The included studies used sweet almond oil, 21,25,29 diluted milk, 23 normal saline, 22 and odorless liquid petrolatum 27 as placebo controls. Although the authors claimed that their studies were blinded in design, due to the nature of aromatherapy, many participants may have uncovered which group they were in because of the smell of essential oils. Therefore, there is a possibility for breaking of blinding and potential overestimation of the treatment effects. The results should be cautiously interpreted.
Limitations and future research
This review has several limitations. First, although considerable efforts were made to retrieve all RCTs on the subject, the authors could not be absolutely certain that they were successful. One of the main limitations of the included studies was selective outcome reporting and lack of transparency regarding the research process. Some trials were published multiple times in the authors' native language and English, with selected outcome measures. Furthermore, these studies reported only some of their results, resulting in selective reporting. It is conceivable that several negative RCTs have remained unpublished, which would distort the overall picture. Second, most of the included studies were conducted in Iran, and a degree of uncertainty regarding the application of findings remains. Finally, the reviewed studies lack full reporting of their results, and there was a paucity of published studies; therefore, the conclusions of this review might be overstated. Another limitation is not only the scarcity of data but also the heterogeneity of parameters. This prevents the drawing of a firm conclusion. Future researchers should preliminary report all of their planned outcomes and essential reporting outcomes to improve transparency in future studies and strengthen the evidence.
In conclusion, the current evidence suggests that aromatherapy inhalation appears to be effective for improving menopausal symptoms including sexual desire slightly. However, due to the small number of studies and the lack of MCIDs, the evidence is limited. Further rigorous RCTs are needed to overcome the many limitations of current studies.
Footnotes
Authors' Contributions
Conceptualization of the study was done by J.C., H.W.L., and M.S.L.; data curation was done by J.C. and L.A.; and formal analysis was carried out by H.W.L. and M.S.L. Funding acquisition was done by M.S.L.; investigation was performed by H.W.L. and J.C.; methodology was done by H.W.L. and M.S.L.; project administration was done by M.S.L.; resources were taken care of M.S.L.; software was taken care of H.W.L. and M.S.L.; supervision was done by M.S.L.; validation of the study was by M.S.L.; visualization was by H.W.L. and M.S.L.; roles/writing, original draft, was by H.W.L., J.C., and M.S.L.; writing, review and editing, was by L.A. All authors have read and agreed to the published version of the article.
Disclaimer
The authors alone are responsible for the contents of the article.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This study is supported by the Korea Institute of Oriental Medicine (K18043, KSN20134276, and KSN2013240). The funder has no role in this study.
