Abstract
Objectives:
The purpose of this study was to evaluate a traditional herbal preparation, Jobelyn,® for its effects on anemia and CD4+ T-cell counts in human immunodeficiency virus–positive (HIV+) patients in Nigeria.
Design:
An open-label pilot study involving 10 confirmed (HIV+) patients who were not receiving antiretroviral therapy (ARVT) was performed, in which the patients consumed Jobelyn for 8 weeks, at a dose of 500 mg twice daily. The pilot study was followed by a controlled trial involving 51 patients, all confirmed HIV+, where the patients with CD4+ T-cell counts below 350 cells/μL were receiving ARVT. The eight patients with baseline CD4+ T-cell counts above 350 cells/μL received Jobelyn. The remaining patients who all received ARVT were randomized to ARVT alone versus ARVT+Jobelyn for 12 weeks.
Results:
Patients receiving ARVT showed a statistically significant improvement in their CD4+ T-cell counts across the 12-week study period (p<0.01). Patients receiving ARVT+Jobelyn showed a faster improvement, reaching a high level of statistical significance compared to baseline already at 6 weeks (p<0.001), and remained highly significant at 12 weeks (p<0.001).
Conclusions:
This is the first controlled study conducted to evaluate efficacy of Jobelyn on immune status in HIV+ patients. The data suggest that consumption of Jobelyn contributed to improved hemoglobin levels and increased CD4+ T-cell counts in Nigerian HIV+ patients. Further studies are needed to examine similar effects in other populations, and to elaborate on the underlying mechanisms, specifically, whether the consumption of Jobelyn supported multiple aspects of bone marrow function.
Introduction
T
Jobelyn® is a natural product developed from tropical plants found within the Nigerian flora. Toxicological evaluation in laboratory animals has shown a satisfactory safety and side-effects profile. 2 It has been found to induce rapid and marked improvement in anemia. 3 –6 The suggested mechanism of action was an immune-boosting effect, which is interesting in light of the presence of antiviral peptides in some parts of the sorghum plant, 7 as well as unique polyphenol compounds identified in Jobelyn. 8,9 Evidence in vitro for many biological properties of Jobelyn was recently published. The data include immune modulation, activation of natural killer cells involved in antiviral immune defense actions, and up-regulating of the production of antiviral chemokines. 10
Jobelyn's effects on anemia 3 –6 lead to the speculation whether the effect on red blood cell production as well as production of other cell types may be positively affected. Limited use in uncontrolled trials in antiretroviral-naïve HIV-infected subjects as well as patients on highly active antiretroviral therapy (HAART) has suggested some improvement in several hematological parameters and quality of life. 11 HAART uses a combination of reverse transcriptase inhibitors and protease inhibitors and is the current standard drug treatment of HIV/acquired immune deficiency syndrome (AIDS). Appropriate and consistent use of HAART can help reduce viral load and allow for some immune system reconstitution, including an increase in CD4+ T-cell counts. However, even with heavy subsidy, HAART is neither accessible nor affordable to a majority of people living with HIV/AIDS in third-world countries. Use of HAART is also limited by potential toxicity and side-effects, as well as development of resistant strains of HIV. Only patients whose biologic profiles fit the eligibility criteria are typically treated with HAART. These limitations emphasize the need for continued search for viable alternatives or adjuncts to HAART.
Increasing use of Jobelyn by many HIV+ patients and people with anemia justifies further evaluation of its efficacy for support of production of lymphocytes and red blood cells in people with bone marrow suppression. This study was therefore designed to address these questions. Two clinical pilot studies were performed on patients with HIV in Lagos, Nigeria. An initial pilot study involved 10 HIV+ patients who also suffered from anemic conditions. A subsequent controlled study continued to evaluate the effects of CD4+ T-cell counts in HIV+ patients, some of whom also received antiretroviral treatment. The controlled study aimed at evaluating the role (if any) of Jobelyn in the management of HIV in the presence or absence of HAART.
Methods
Nutritional supplement
The consumable nutritional supplement Jobelyn was grown, harvested, and manufactured by Health Forever Products, Ikeja, Lagos, Nigeria, using seed stock from a recently domesticated variant of West African Sorghum bicolor. One lot was used for the open-label pilot study, and a second lot was used for the controlled trial. Both lots had similar chemistry profiles showing a very high content of polyphenols, particularly apigeninidin, which was present at approximately 4% of the dry weight of the botanical material (Table 1). This level is consistent with other lots tested for immune-modulating activities. 10
The quantification of these five polyphenol marker compounds was evaluated by the highly sensitive LC/MS-MS methodology, and their structures were confirmed by CAD-MS/MS analysis. The testing was performed by Brunswick Laboratories, Norton, MA.
Trial 1
Ten patients (4 men and 6 women, age range 23–49 years) with confirmed HIV+, a reduced number of CD4+ T lymphocytes, who did not qualify for antiretroviral therapy (ARVT), and who consumed Jobelyn, were followed through the Police Hospital in Lagos, Nigeria for 8 weeks. Potential study participants were regular patients of Dr. N. U. Eguae and were invited to participate by use of written informed consent. The number of CD4+ T lymphocytes and the hemoglobin level were evaluated at baseline, 4, and 8 weeks.
Trial 2
Based on these pilot data, a controlled trial was performed at the Clinical Research Division of the Military Hospital, Ikoyi, Lagos, Nigeria. Fifty-one patients (28 men and 23 women, age range 18–67 years) were enrolled in the study upon written informed consent, as approved by the Ikoyi Military Hospital Institutional Review Board. Potential study participants were regular patients of Dr. Ayuba, and invited to participate, by use of written informed consent. All study participants who at the time of screening had CD4+ T-cell counts below 350 cells/μL were treated with ARVT, which consisted of nevirapine (200 mg), lamivudine (150 mg), and stavudine (40 mg) twice daily. All 51 patients who participated in this clinical trial were monitored for CD4+ T-cell counts at baseline, 6, and 12 weeks. Eight patients who at the time of screening had a CD4+ T-cell count at 350 or higher were ineligible for ARVT, and received Jobelyn for the same duration of 12 weeks. The remaining 43 patients were randomized into two groups, where one group of 16 people was only treated with ARVT, and the other group of 27 people was treated with ARVT and also consumed Jobelyn daily for 12 weeks. There was no significant difference in the baseline CD4+ T-cell counts between these two groups. Data were analyzed using “within-subject” analysis using the paired two-tailed t-test, as well as “between-groups” analysis, using the unpaired two-tailed t-test.
Results
Trial 1
A statistically significant increase in CD4+ T lymphocytes (p<0.01) and hemoglobin (p<0.001) was observed within 4 weeks, using the paired two-tailed t-test. Among the 10 participants, everyone showed an improvement in both CD4+ T-cell count and in hemoglobin levels (Table 2). All 10 patients had hemoglobin levels below normal for their gender at baseline. All of them showed an improvement in hemoglobin levels. After 8 weeks of consuming Jobelyn, all 10 patients had hemoglobin levels near the lower normal range.
CD4+ T-cell counts are shown as cells/μL whole blood.
Hemoglobin levels are shown as g/dL.
Data analysis utilized the within-subject two-tailed paired t-test to compare each person's changes for Week 4 and Week 8 to baseline values.
ARVT, anti-retroviral therapy; JOB, Jobelyn,® which was consumed at a dose of 500 mg twice daily; SEM, standard error of mean.
Trial 2
Among the 51 patients participating in this controlled trial, 43 patients had baseline CD4+ T-cell counts at or below 350 cells/μL, and received ARVT.
As expected, people on ARVT showed a statistically significant improvement in their CD4+ T-cell counts across the 12-week study period (n=16, p<0.01) (Fig. 1). However, patients receiving ARVT+Jobelyn showed a more robust improvement, which reached a high level of statistical significance compared to baseline already at 6 weeks (n=27, p<0.001), and remained highly significant at 12 weeks (p<0.001) (Fig. 1). Analysis comparing the two groups showed that the CD4+ T-cell counts in the group receiving ARVT+Jobelyn was significantly higher than the group receiving ARVT alone, both at 6 and 12 weeks (p<0.001).
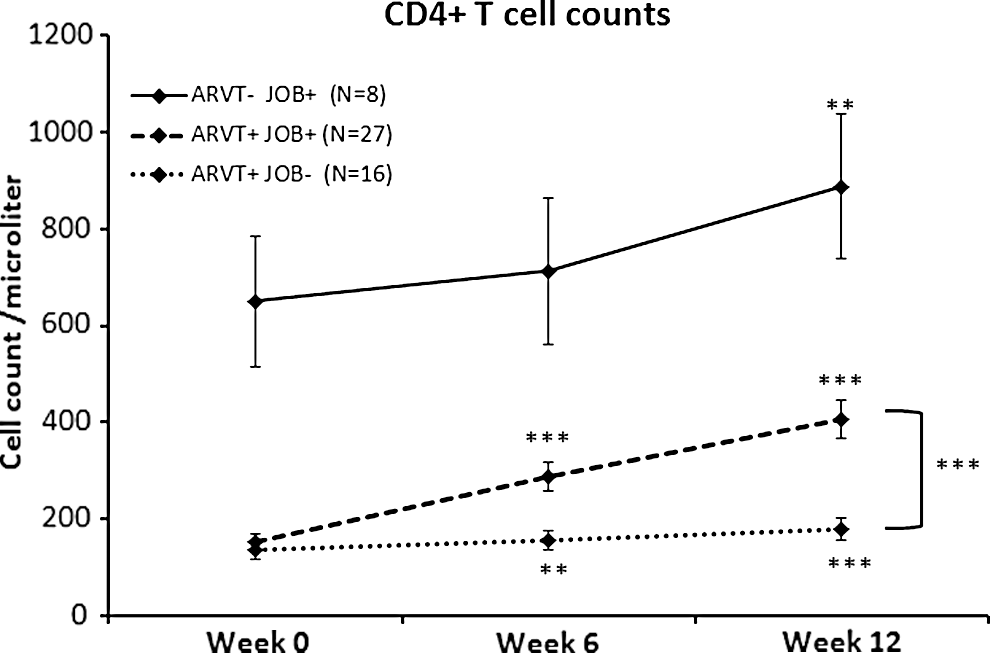
The changes in CD4+ T-cell counts from Trial 2 (a controlled study with 51 HIV+ patients, where HIV+ status was confirmed by electrophoresis) are shown as the group average±standard error of the mean. Improvements were seen for all three groups: Eight patients who started the study with CD4+ T-cell numbers higher than 350 cell/μL, and were not eligible for antiretroviral therapy (ARVT) consumed Jobelyn (JOB) for 12 weeks. Improvement reached statistical significance at 12 weeks (p<0.01). Forty-three patients with baseline CD4+ T-cell counts lower than 350 cells/μL were eligible for ARVT, and were randomized to either ARVT alone or ARVT and Jobelyn. There was no statistical significance in the CD4+ T-cell counts at baseline between these two groups. The group receiving ARVT alone showed the expected improvement in CD4+ T-cell counts, which reached statistical significance already at 6 weeks (p<0.01), and continued to improve at 12 weeks (p<0.001). The group that received ARVT and also consumed Jobelyn showed a more robust improvement, which reached a high level of significance already at 6 weeks, and remained highly significant at 12 weeks (p<0.001). Data analysis utilized the within-subject two-tailed paired t-test to compare each person's changes for week 6 and week 12 compared to baseline values, and also the between-groups unpaired t-test to compare the data for the ARVT+JOB+and the ARVT+JOB- groups at 12 weeks. Levels of statistical significance are indicated by asterisks: **for p<0.01, and ***for p<0.001.
Within the study population of 51 people, only 3 patients had lower CD4+ T-cell counts after the 12-week study than at study baseline; all 3 patients received ARVT, and 2 of them also consumed Jobelyn. These three people experienced concurrent infections (tuberculosis, pneumonia) during the study. Patients' appetite and weight increased and there was improvement in their general well-being. The maculopapular skin rash of three of the patients who had herpes zoster remitted.
The third group of patients (i.e., those eight people who received Jobelyn only, since they were not eligible for ARVT due to a higher CD4+ T-cell count at baseline) also showed an improvement during the weeks of Jobelyn consumption (Fig. 1). This improvement was statistically significant from baseline after 12 weeks (p<0.01).
Discussion
The core result presented here is the improvement in CD4+ T-cell numbers over time in the two groups that consumed Jobelyn when compared to the group treated with conventional antiretroviral treatment only. An additional observation from the initial pilot study is the improved hemoglobin levels seen in HIV+ patients consuming Jobelyn for 8 weeks.
The parallel increase in hemoglobin and CD4+ T-cell counts seen in the pilot study suggests that consumption of Jobelyn may support several aspects of bone marrow function. The data from the controlled study involving 51 people suggest support of T-cell production. The primary mechanism likely includes support of bone marrow function since anemic conditions were also reduced, possibly combined with secondary mechanisms of enhanced immune defense against viral infection. In addition, an antiviral effect may be suggested, based on the observations that Jobelyn provides several aspects of antiviral immune support in vitro, which includes activation of natural killer cells and production of chemokines MIP-1a, MIP-1b, and RANTES. 10
Future clinical studies need to address whether Jobelyn has similar effects in populations with different demographics, diet, and lifestyle, as well as different prevalence of concomitant infectious diseases. Also, the suggested mechanisms of action may be further evaluated through testing of serum cytokine/chemokine profiles, including growth factors with known effects on bone marrow function, and antiviral chemokines. These effects by Jobelyn may suggest a role for its use in broader health management, and not limited to HIV or HIV-associated anemia alone. Clinical trials are currently ongoing to examine the effects of Jobelyn on anemia in non-HIV+ study populations. Given the significant effects seen over a relatively short study period in a West African population, consideration should be given to expanding its use in other countries with different health challenges, to help augment health improvements in acute and chronic viral illnesses.
Footnotes
Acknowledgments
The two studies presented here were conducted at the Police Hospital and the Military Hospital, both in Lagos, Nigeria. The data analysis was performed at NIS Labs, Oregon, United States. The studies were sponsored by Health Forever International, Inc.
Disclosure Statement
G.S. Jensen and K.F. Benson are associated with NIS Labs, and Colonel G.I. Ayuba was employed as staff physician at the Military Hospital at the time this study was conducted. None of these people have any commercial interest in this subject matter. A. and O. Okubena are directors at Health Forever International, Inc., the sponsor of this work.
