Abstract
This study aimed to characterize alterations in select eicosanoids in experimental and human pulmonary arterial hypertension (PAH) and to assess their potential utility as predictors of outcome. Using liquid chromatography–mass spectrometry, we performed targeted lipidomic analyses of the lungs and right ventricles (RVs) of chronically hypoxic rats and plasma of consecutive PAH patients and healthy controls. In rat lungs, chronic hypoxia was associated with significantly decreased lung prostacyclin (PGI2)/thromboxane B2 (TXB2) ratio and elevated lung 8-hydroxyeicosanoid (HETE) acid concentrations. RV eicosanoids did not exhibit any changes with chronic hypoxia. PAH treatment–naïve patients had significantly increased plasma concentrations of TXB2 and 5-, 8-, 12-, and 15-HETE. The PGI2/TXB2 ratio was lower in PAH patients than in controls, especially in the treatmentnaïve cohort (median: 2.1, 0.3, and 1.3 in controls, treatmentnaïve, and treated patients, respectively,
Keywords
Pulmonary arterial hypertension (PAH) is a life-threatening disease characterized by pulmonary vascular remodeling leading to impaired pulmonary hemodynamics, right ventricular (RV) failure, and death. While biomarkers can potentially be helpful in diagnosis, follow-up, and management of PAH patients, only brain natriuretic peptide (BNP) or its precursor is currently recommended at diagnosis and for longitudinal follow-up; neither, however, is specific to PAH. 1
Alterations in lipid metabolism of the arachidonic acid (AA) derivatives (Fig. 1), such as prostaglandins (PGs), leukotrienes, and hydroxyeicosanoids (HETEs), have been implicated in the pathogenesis of pulmonary hypertension (PH).2,3 Studies suggest that metabolites of the AA derivatives mediate endothelium-dependent vasoconstriction.4–7 The most studied of these in the pulmonary vasculature are prostacyclin (PGI2), with its potent vasodilator, antiplatelet, antiproliferative, and anti-inflammatory properties, 8 and thromboxane A2 (TXA2), which opposes the actions of PGI2 by promoting vasoconstriction, platelet aggregation, and smooth muscle cell (SMC) proliferation. 9 Lung expression of PGI2 synthase is decreased 10 and urinary levels of TXB2 (the stable metabolite of TXA2) are increased 11 in patients with PAH. The lung vasculature also synthesizes various HETEs, which are implicated pathogenetically in experimental models of PH12–14 but not yet fully explored in PAH patients. Of these, 5-HETE is an enzymatic product of 5-lipoxygenase (5-LOX) and is thought to modulate the response of the lung vasculature in experimental PH; 15 12-HETE, the product of 12-LOX, is upregulated in the lungs of hypoxic rats and stimulates proliferation of pulmonary artery SMCs; 14 15-HETE, a precursor for lipoxins, promotes hypoxic pulmonary vasoconstriction, inhibits apoptosis of pulmonary artery SMCs, and promotes pulmonary vascular remodeling in some experimental PH models,16–19 whereas in other models, it has vasodilatory effects on both the systemic and pulmonary vasculature.20,21 It has also been found that 15-HETE possesses anti-inflammatory properties, 22 which may be relevant, given the growing evidence of the role of inflammation in the pathogenesis of PAH. 23 Other products of AA metabolism, such as 8-HETE, have been investigated only superficially in PAH. 2
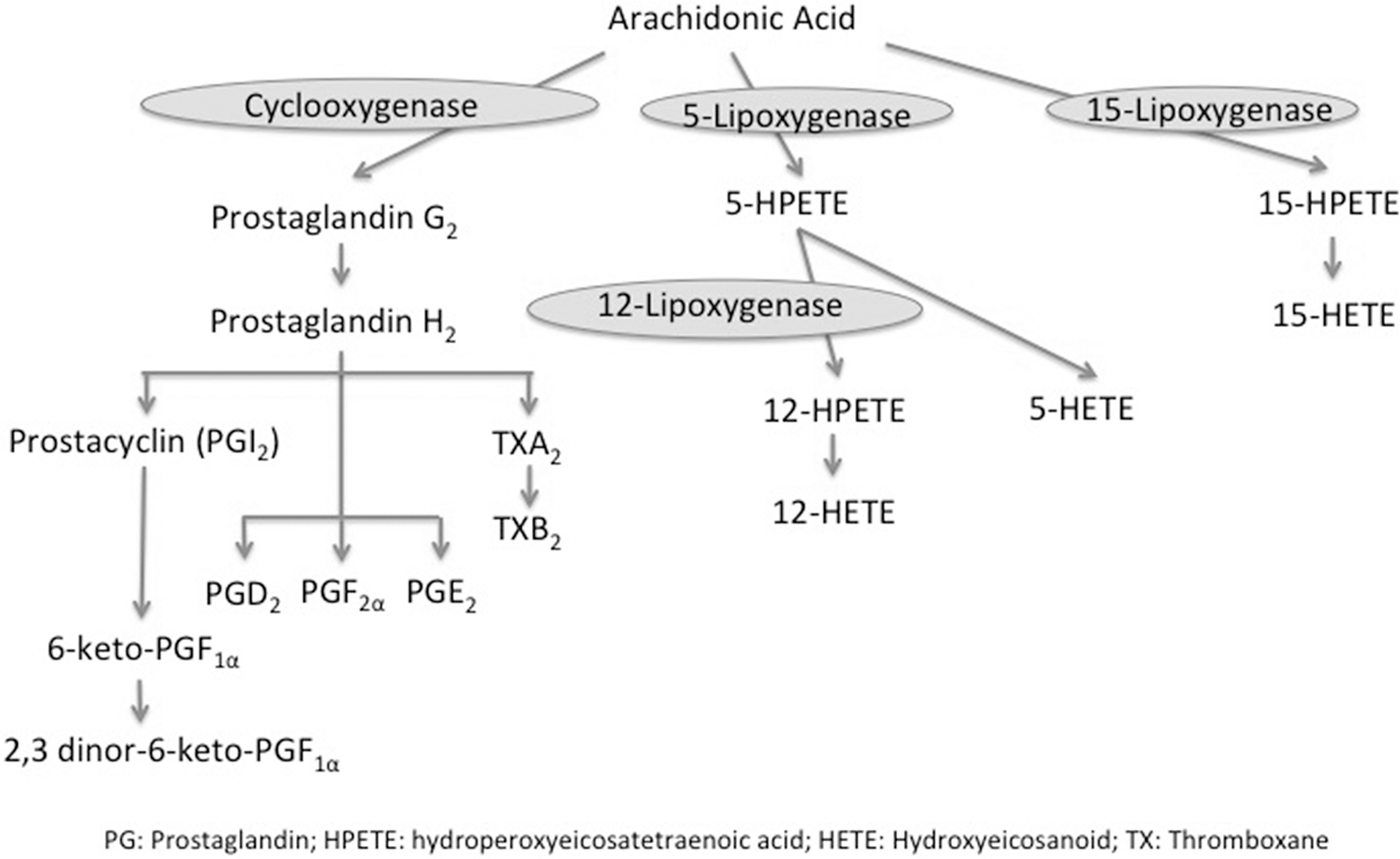
Arachidonic acid pathway.
The importance of lipid mediators derived from the AA metabolism in the pathophysiology of PAH is highlighted by the therapeutic use of PGI2 or its analogs, with resultant amelioration of the disease.24–27 However, the utility of these lipid mediators as biomarkers in PAH has not been investigated. In this study, we aimed to (1) characterize the alterations in targeted lipidomic profiles of AA derivatives of experimental and human PAH, specifically focusing on PGs and HETEs, (2) compare the lipidomic profiles of AA derivatives in experimental and human PAH, and (3) investigate the prognostic utility of these lipid mediators as potential biomarkers for PAH.
METHODS
Hypoxic exposures and tissue sampling in rats
Thirty-six male rats (Sprague-Dawley, 250–300 g), 2–4 months old, were randomized to exposure: normoxia (21% oxygen saturation for 3 weeks,
Lung and RV lipidomic analyses
Lipid extraction and analyses were carried out on homogenates of harvested lungs and RVs and analyzed by liquid chromatography–mass spectrometry (LC/MS/MS; appendix, available online). 28
Human subjects
The Tufts Medical Center Pulmonary Hypertension Database is a prospective registry of adult patients with a diagnosis of PH by right heart catheterization (RHC; appendix). The study was approved by the Tufts Medical Center Institutional Review Board, and informed consent was obtained from all subjects.
We reviewed data on consecutive patients evaluated between June 1, 2006, and March 31, 2012, and patients were classified into etiologic groups based on current guidelines. PAH was defined as mean pulmonary artery pressure (mPAP) ≥ 25 mmHg, pulmonary capillary wedge pressure (PCWP) ≤ 15 mmHg, and pulmonary vascular resistance (PVR) > 3 Wood units. Patients were included in the study if they had idiopathic PAH (IPAH) or connective-tissue disease (CTD)-associated PAH. Patients with other forms of group 1 PAH or group 2 (PH secondary to left heart disease), group 3 (PH secondary to chronic lung disease), group 4 (chronic thromboembolic PH), or group 5 (miscellaneous) PH were excluded. Patients were included in the study regardless of their treatment status (treatment naïve vs. treated).
Demographic and clinical characteristics were recorded, and blood samples were collected from fasting patients at the time of the RHC. Blood samples were also collected from healthy volunteers as controls. Death was determined from medical records and confirmed with the Social Security Death Index. All causes of death were considered in the survival analysis. Survival time was defined by the date of the RHC at which the blood samples were obtained. Survival data were censored on February 1, 2015.
Lipidomic analyses of human plasma
Plasma samples, collected at the time of the RHC in ethylene diamine tetra-acetic acid (EDTA) tubes, were centrifuged at 4°C, and plasma was isolated and stored in cryotubes at −80°C. Lipid extraction was carried out and analyzed by LC/MS/MS (appendix).
Statistical analysis
Continuous variables are presented as medians and interquartile range. Categorical variables are presented as frequencies and percentages. Wilcoxon rank-sum or Kruskal-Wallis tests were used for comparing tissue and plasma levels of eicosanoids, as appropriate. In order to evaluate the performance of different eicosanoids as predictors of outcome in PAH, the areas under the curve (AUCs) of the receiver operating characteristics (ROC) curves were calculated. The best AUCs were those of 12- and 15-HETE, and the optimal cutoff from each of their respective ROC curves was chosen. KaplanMeier survival curves were constructed, and the log-rank test was used to compared them. Cox proportional hazards were used to determine the hazard ratio of eicosanoids and survival in PAH; however, data are shown only for 12- and 15-HETE, as they were the only two biomarkers with significant associations. The results were then adjusted for the REVEAL (Registry to Evaluate Early and Long-Term PAH Disease Management) risk score.
30
Statistical analysis was performed with R, version 3.1.1, and the survival package (version survival 2.37–7). A
RESULTS
Experimental PH model
Chronic hypoxia was associated with significantly elevated RV pressures compared with normoxic controls (51 ± 6 vs. 24 ± 2 mmHg,
Hypoxia alters eicosanoid levels in rodent lungs
Levels of eicosanoids in the rat lungs were compared between the two exposures. In normoxia, several PG metabolites were below the limit of quantitation or had very low levels (Table S1; Tables S1–S5 available online).
Chronic hypoxia was associated with significant decreases in lung PGI2/TXB2 ratio (Fig. 2C). Other PG metabolites involved in bronchodilation and vasodilation, including prostaglandin E2 (PGE2), PGI2, and TBX2, were not significantly changed by hypoxia (Fig. 2A, 2B, 2D). Among different HETE molecules, chronic hypoxia was associated with high lung concentrations of 8-HETE. The 5-, 12-, and 15-HETE levels in the rat lungs were not significantly altered by hypoxia (Fig. 3A, 3C, 3D; Table S1). Levels of eicosanoids were very low in normal rodent RVs and did not exhibit any significant changes with chronic hypoxia (Table S2).
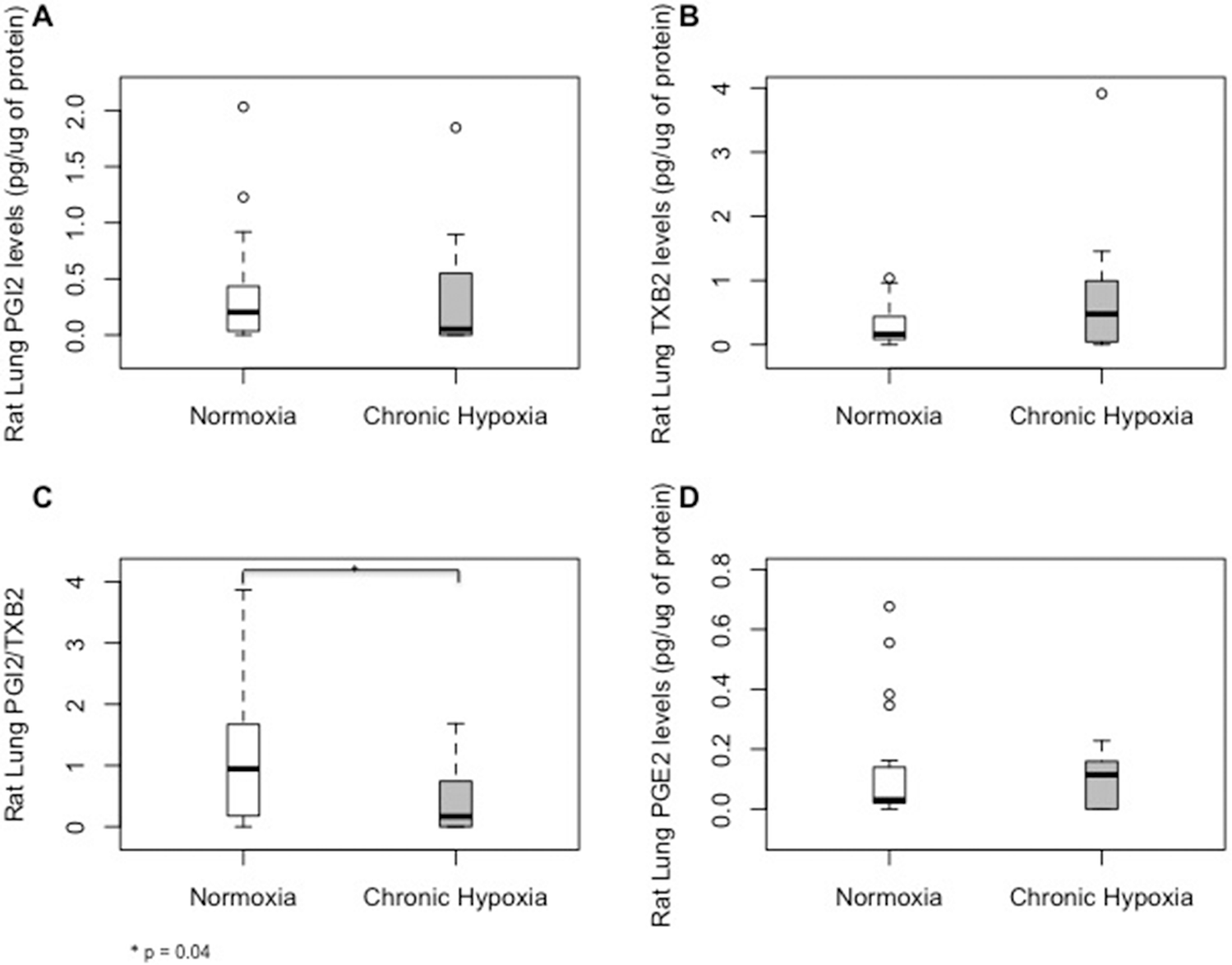
Median (interquartile range) tissue levels of prostaglandin metabolites prostacyclin (PGI2;
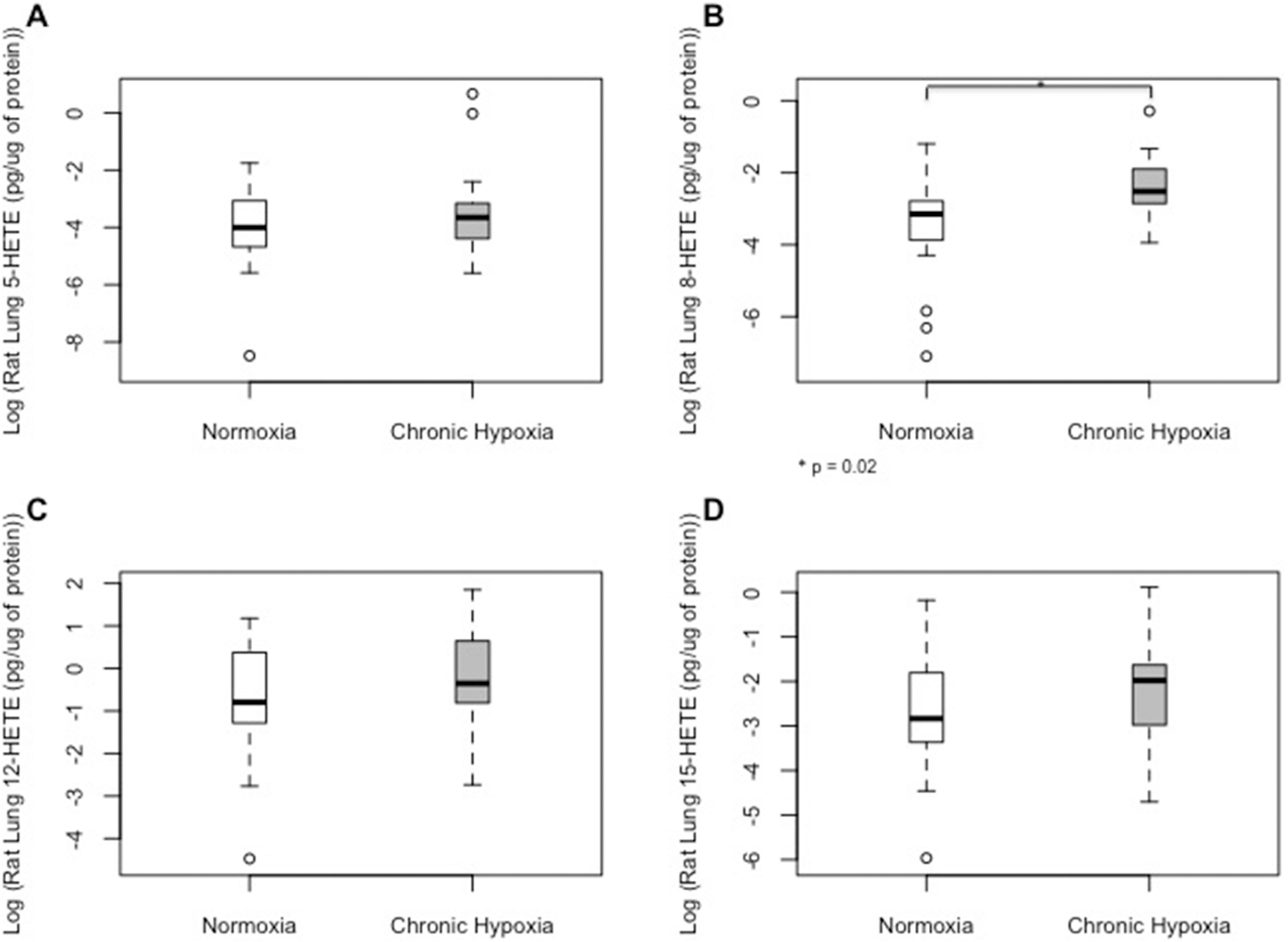
Median (interquartile range) tissue levels of 5-hydroxyeicosanoid (5-HETE;
Human plasma lipidomic analyses
Demographic and clinical characteristics of pulmonary arterial hypertension (PAH) patients and control subjects
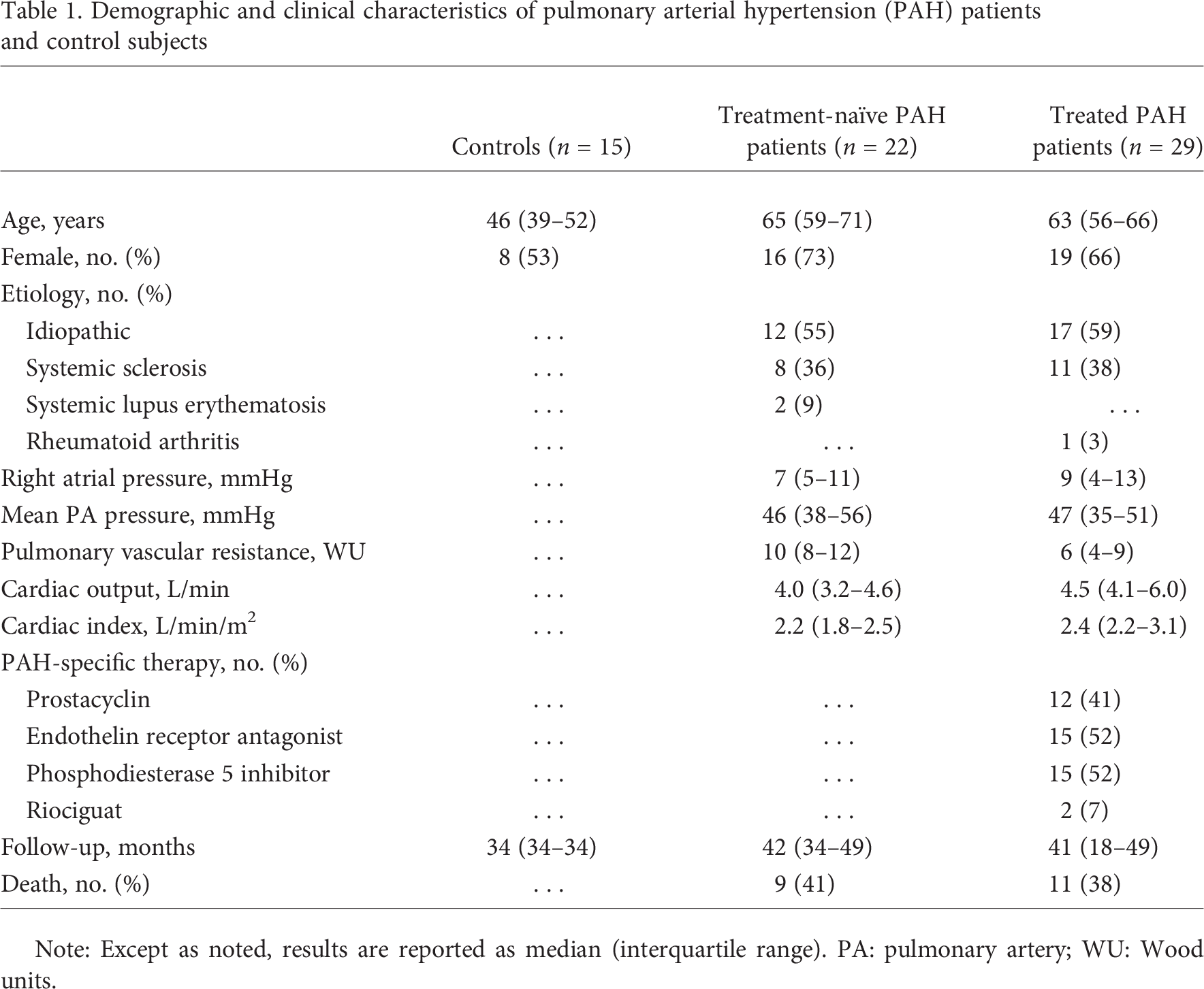
Note: Except as noted, results are reported as median (interquartile range). PA: pulmonary artery; WU: Wood units.
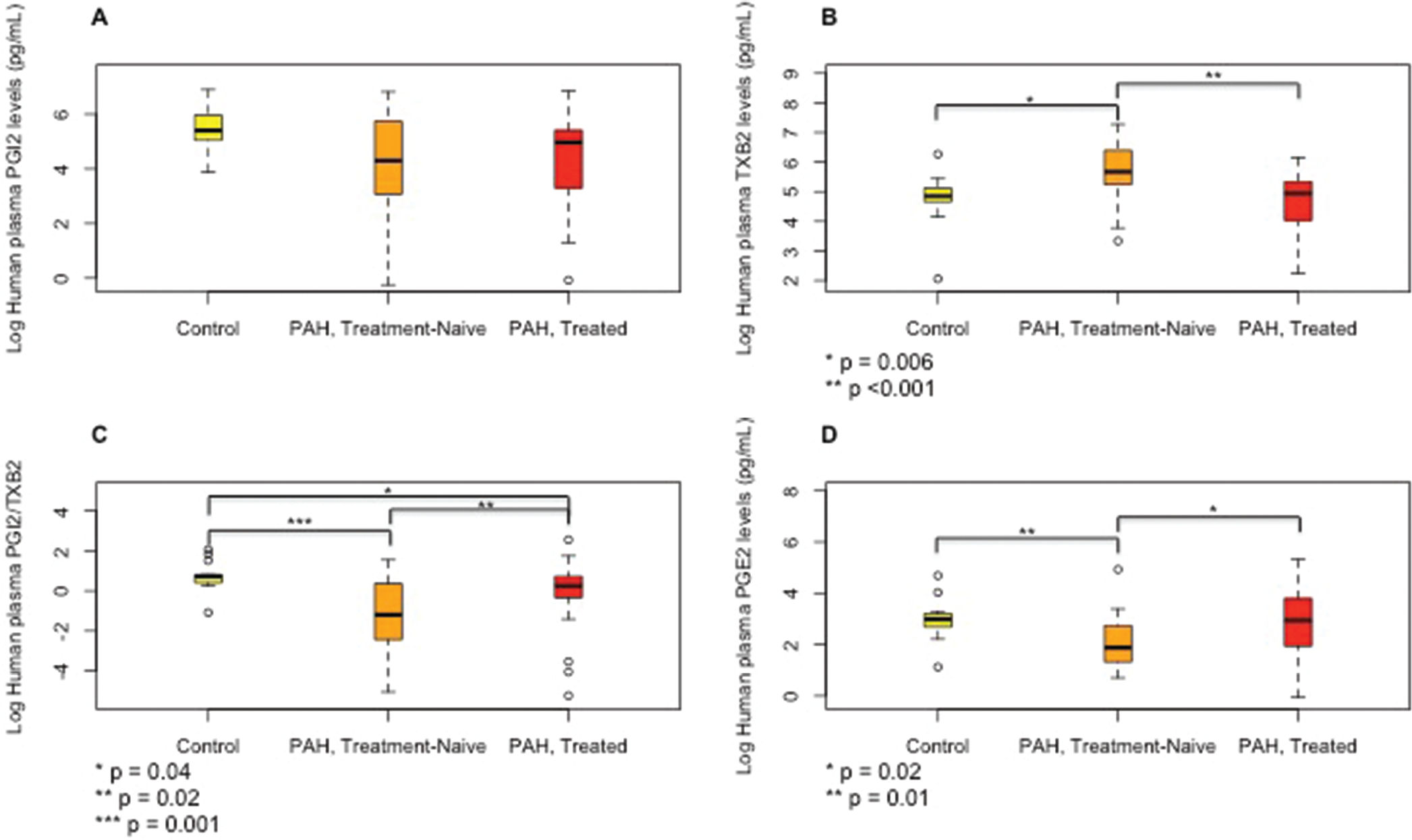
Median (interquartile range) plasma levels of prostaglandin metabolites prostacyclin (PGI2;
Treatment-naïve PAH patients had significant elevations in 5-, 8-, 12-, and 15-HETE levels (Fig. 5) compared to controls. The 5- and 8-HETE levels were considerably lower in treated PAH patients; however, 12- and 15-HETE levels, although lower than in treatment-naïve patients, still remained significantly higher in the treated patients than in controls. There was no significant difference in the levels of all HETEs measured between patients who were treated with PGI therapy and those who were not (data not shown). There were no trends observed to suggest decay over time in the levels of eicosanoids due to storage of plasma.
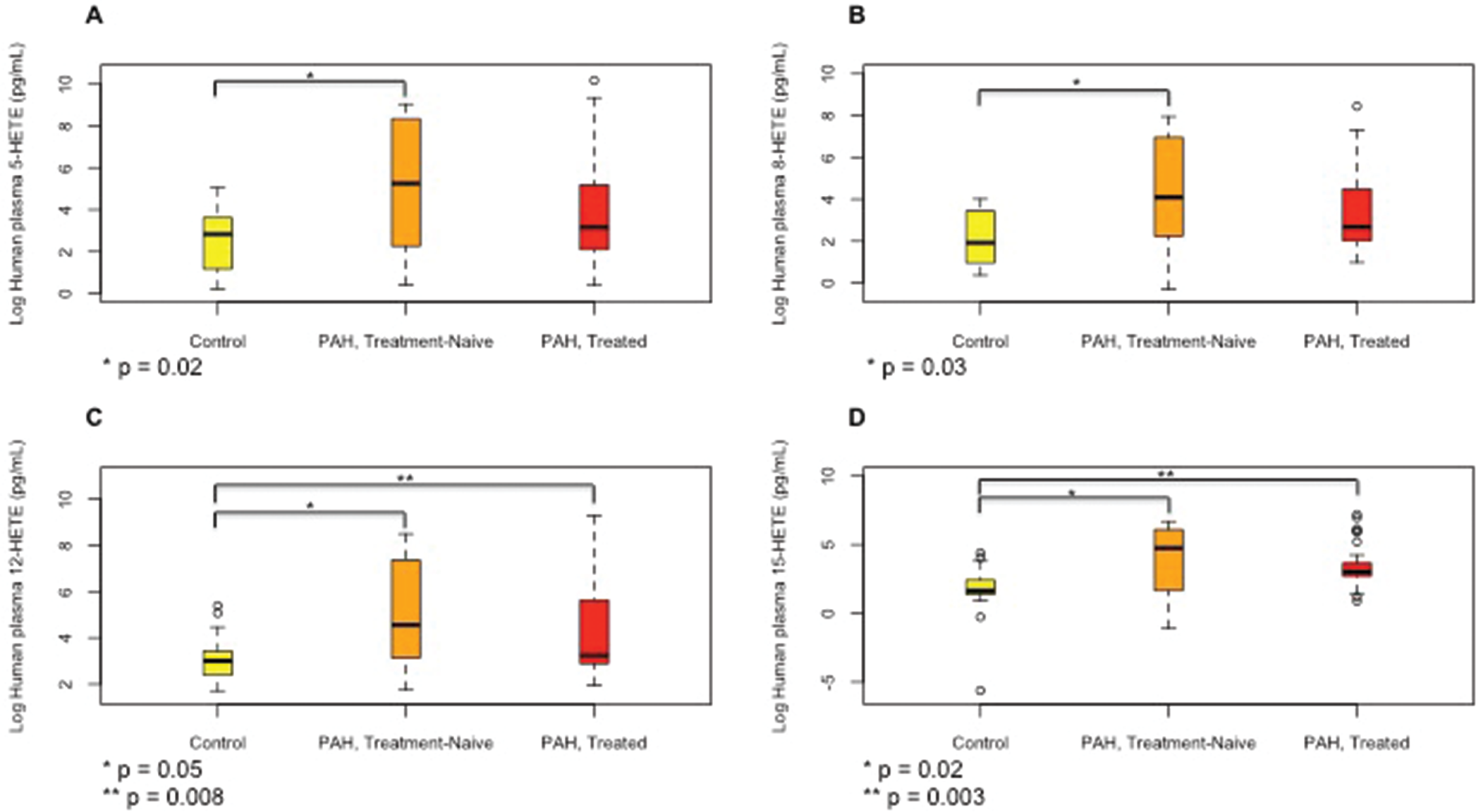
Median (interquartile range) plasma levels of 5-hydroxyeicosanoid (5-HETE;
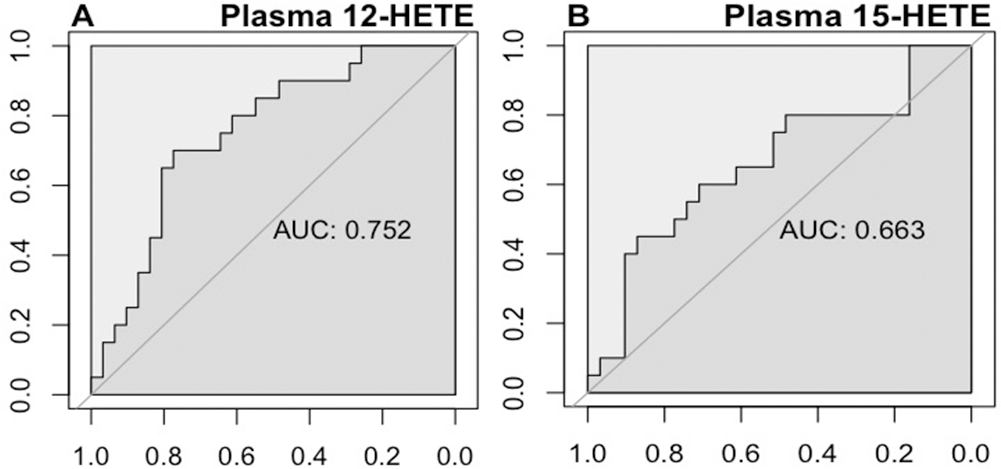
Receiver operating characteristics curves constructed to determine the optimal thresholds for human plasma 12-hydroxyeicosanoid (12-HETE;
From the ROC analyses, we established threshold plasma levels of 57 pg/mL for 12-HETE and 256 pg/mL for 15-HETE to distinguish the survivors. On the basis of these thresholds, we divided the cohort of PAH patients into either 12-HETElow (<57 pg/mL) and 12-HETEhigh (≥57 pg/mL) or 15-HETElow (<256 pg/mL) and 15-HETEhigh (≥256 pg/mL) groups. Both the 12-HETEhigh and 15-HETEhigh groups had significantly decreased survival (Fig. 7). Elevated 12- and 15-HETE levels were both associated with a significantly increased risk of death (unadjusted hazard ratio [HR]: 3.4 [95% confidence interval (CI): 1.3–8.9] and 2.5 [1.0–6.2], respectively; Table 2). After adjustment for the REVEAL risk score, both the 12-HETEhigh and 15-HETEhigh cohorts remained associated with a significantly increased risk of death (adjusted HR: 2.8 [95% CI: 1.1–7.3] and 4.3 [1.6–11.8], respectively; Table 2).
Cox proportional-hazards models for 12-HETEhigh and 15-HETEhigh
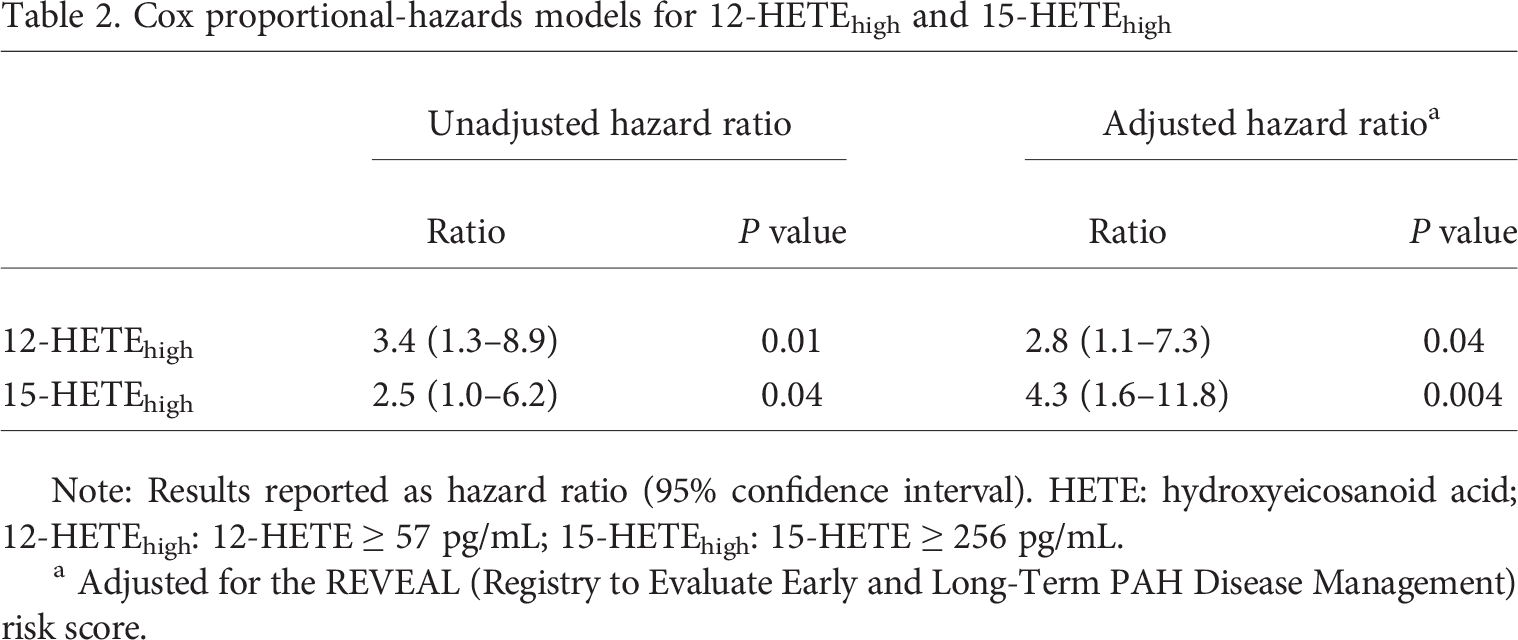
Note: Results reported as hazard ratio (95% confidence interval). HETE: hydroxyeicosanoid acid; 12-HETEhigh: 12-HETE ≥ 57 pg/mL; 15-HETEhigh: 15-HETE ≥ 256 pg/mL.
Adjusted for the REVEAL (Registry to Evaluate Early and Long-Term PAH Disease Management) risk score.
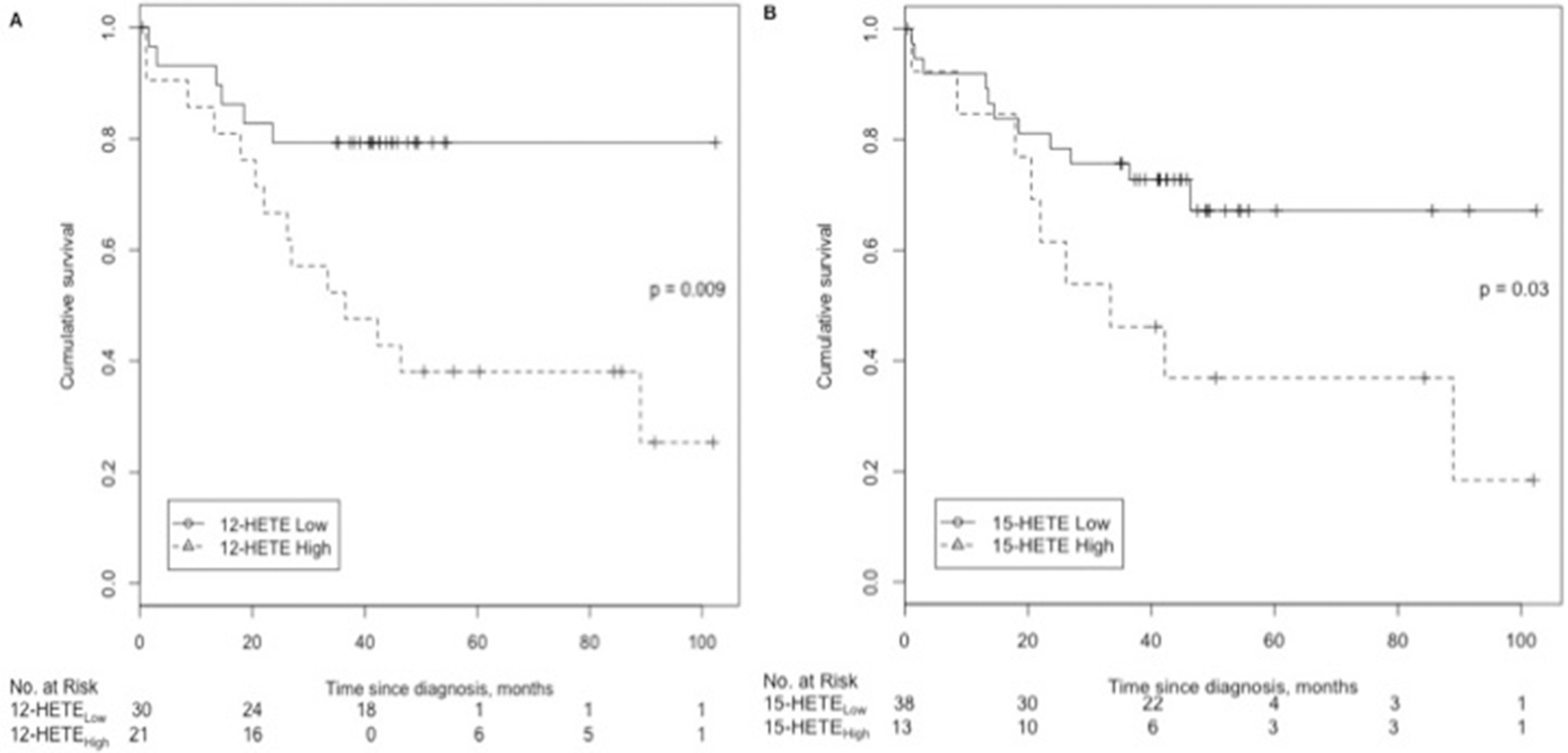
Pulmonary arterial hypertension (PAH) patient survival according to plasma hydroxyeicosanoid (HETE) levels.
DISCUSSION
This is the first study to characterize several biochemically active lipids in both experimental PH models and a well-phenotyped cohort of PAH patients. We demonstrate abnormalities in eicosanoid metabolites in the lungs of experimental rodent PH models and in the plasma of PAH patients, showing both similarities and differences. Moreover, we identified two novel potential biomarkers, 12- and 15-HETE, that were independent predictors of mortality in PAH patients, even after adjustment for known confounders.
PGs in rodent and human pulmonary vascular disease
In response to chronic hypoxia in rodents and despite lack of significant alterations in lung levels of PGI2 and TXB2, there was a decrease in PGI2/TXB2 ratio, supporting the notion of an imbalance between vasodilatory and vasoconstrictive mediators. These findings support that alterations in AA metabolism may play an active role in the pulmonary vascular response to chronic hypoxia. Our findings are in contrast to results from the Sugen 5416/hypoxia (SuHx) rat model of experimental PH, in which elevated levels of PGE2 and TXB2 were found at 3 weeks. 31 This suggests that alterations in lung eicosanoids depend on the experimental model used and/or the type and severity of PH, as the SuHx rat model has been characterized as an irreversible, more severe form of PH, compared to the reversible model of chronic hypoxia.
In PAH patients, we observed elevated plasma levels of several AA metabolites in treatment-naïve PAH patients, abnormalities that were attenuated in the treated PAH patients. The alterations in PG pathways tended to be more substantial than those in the experimental PH model. The vasodilator PGE2 changed in the direction opposite that in rodent lungs. PGE2 was lower in treatment-naïve patients than in controls and not different between the treated patients and controls, while TXB2 was elevated in treatment-naïve patients and restored in the treated PAH patients. More importantly, PGI2/TXB2 was significantly lower in our patients than in controls, and, although it increased in the treated patients, it remained abnormal. These findings are consistent with findings by Christman et al. 11 of higher urinary levels of TXB2 and TXB2/PGI2 in 8 PAH patients compared to non-PAH controls, lending further support to the notion that impaired balance between PGI2 and TXB2 contributes to the pathogenesis of PAH in humans. In addition, our results suggest that PAH treatment may modify these PGs to partially restore the balance between vasoactive metabolites.
HETEs in rodent and human pulmonary vascular disease
Among the HETEs measured in rodent lungs with experimental PH, 8-HETE was significantly higher with exposure to chronic hypoxia. This is in accordance with increased 8-HETE levels in the SuHx rat model. 31 Moreover, plasma 8-HETE levels were also higher in treatment-naïve than in treated PAH patients, suggesting that 8-HETE may play a role in pathogenesis of both experimental and human PH disease. It has been shown that 5-HETE plays a role in the monocrotaline experimental PH model; 15 however, in our study, 5-HETE levels in the rat lungs were not affected significantly by chronic hypoxia. Previously, levels of 5-HETE in the lungs from patients with primary and secondary forms of PH were reported to be increased. 2 In our study, levels of 5-HETE were also elevated in the plasma of treatment-naïve PAH patients and were not different between control subjects and treated PAH patients, suggesting that they were ameliorated by PAH treatment.
By mediating barrier function disruption, neovascularization, and angiogenesis, 12-HETE has been implicated in the pathogenesis of vasculopathies in atherosclerosis, cancer, and diabetic retinopathy.32–35 It also promotes platelet activation and aggregation, 36 and 12-HETE and the 12-LOX pathway have also been shown to play a role in the pathogenesis of the chronic hypoxia and SuHx rat models of PH.14,31 We previously showed that 12-LOX is upregulated in the lungs of chronically hypoxic rats and that 12-HETE stimulates proliferation of pulmonary artery SMCs through an extracellular signalregulated kinase–dependent mechanism. 14 In our study, although 12-HETE levels tended to increase in the lungs of rats exposed to chronic hypoxia, this increase did not achieve statistical significance. However, the levels of 12-HETE were significantly increased in the plasma of both treatment-naïve and treated PAH patients compared to those in controls.
Despite some conflicting reports, most studies have implicated 15-HETE in the pathogenesis of experimental models of PH through potentiating pulmonary artery SMC proliferation and protecting against apoptosis,17–19,37 enhancing vasoconstriction during hypoxia via the protein kinase C pathway and downregulation of potassium channels 38 and stimulating the expression of angiogenic molecules such as vascular endothelial growth factor and fibroblast growth factor 2, promoting autocrine-mediated angiogenesis.39,40 Both 15-HETE and 15-LOX have been implicated in platelet activation, pulmonary intravascular thrombosis, angiogenesis, and pulmonary vascular remodeling in experimental PH models.41–43 Contrary to most studies in rodents, 15-HETE has vasodilatory effects on both the systemic and pulmonary vasculature in rabbits.20,21 In our study, levels of 15-HETE were increased, although not significantly, in the lungs of rats exposed to chronic hypoxia but were significantly increased in the plasma of both treatment-naïve and treated PAH patients, similar to the changes in 12-HETE levels. These findings are similar to previously reported increased levels of 15-HETE in the SuHx rat model. 31
In our study, 12- and 15-HETE correlated moderately with PVR and/or mPAP, suggesting a correlation with disease severity. In a prior report, 12- and 15-HETE levels in the lungs of patients with severe PAH were found to be elevated, while patients who were treated chronically with PGI tended to have lower 12- and 15-HETE lung levels. 2 In our cohort of patients, plasma levels of 12- and 15-HETE were similar between patients who were treated with PGIs and those who were not.
Most interestingly, we showed that plasma levels of 12- and 15-HETE were independent predictors of mortality in our cohort. We elected to use the REVEAL risk score for risk adjustment because it was derived from a large United States–based registry that included IPAH and CTD-associated PAH patients and has been validated in incident and prevalent PAH patients as well as cross validated in the French registry.30,44,45 Also, the REVEAL risk score is based on the contribution of multiple variables that need not be measured concurrently. 46 Even after adjustment for the REVEAL risk score, 12- and 15-HETE remained independent predictors of mortality, implicating 12- and 15-HETE as two potential novel biomarkers for PAH. In our study, survival time was assessed from the time the blood samples were obtained in this cohort of newly diagnosed and prevalent patients with PAH. While the survival curves begin to separate after 2 years, the significantly increased risk of death associated with these markers remains an important independent prognostic factor because of its association with long-term outcomes in these patients. Taken together, the above findings suggest that 12- and 15-HETE and their pathways are involved in the pathogenesis of PAH, potentially through increased vasoconstriction, cell proliferation, and enhanced thrombosis of the pulmonary vasculature, and that these pathways may be modified by PAH treatment.
Our study has several limitations. First, we performed targeted lipidomic analyses focused on PGs and HETEs, and we did not evaluate other lipid molecules that could potentially be involved in the pathogenesis of PAH. Although we identified abnormalities in eicosanoid levels in the lungs of experimental PH rodent models and in plasma of human PAH patients, our design did not enable us to understand causal relationships between individual eicosanoids and the development of PH. Another limitation relates to our use of a convenience sample of controls who were not age and sex matched to our PAH patients. While this limits generalizability of our conclusion, our control data offer important insight into the pathobiology of eicosanoid imbalances in PAH, given the lack of data on normal values for eicosanoid levels in human plasma. Finally, our cohort included treatment-naïve and treated PAH patients; however, we did not have serial plasma samples from patients after initiation of PAH-specific therapy.
We conclude that alterations in eicosanoid metabolism are characteristic of both experimental models of PH and human PAH, suggesting a role for circulating eicosanoids in the pathogenesis of pulmonary vascular disease. While there were similarities between the rodent hypoxia model and human PAH, our study found several differences in eicosanoids, which emphasizes the lack of a representative experimental model of PAH. Furthermore, we identify two novel biomarkers that may be helpful in follow-up of PAH patients, assessment of treatment response, and prediction of outcome.
