Abstract
This study was undertaken to characterize the molecular and pathological mechanisms of pulmonary vascular remodeling in a patient who developed chronic lung allograft dysfunction and recurrent pulmonary hypertension (PH) 22 years after undergoing a right single lung transplantation for pulmonary arterial hypertension (PAH). Histopathologic examination of the explanted lungs at the time of retransplantation showed characteristics of diffuse vascular remodeling combined with features of acute and chronic thromboemboli and evidence of bronchiolitis obliterans in the right lung allograft. In contrast, the native left lung demonstrated pulmonary arterial changes in keeping with PAH associated with disseminated pulmonary ossification. Real-time polymerase chain reaction and Western blot—performed on the first lung allograft, the native lung, and the new donor lung—demonstrated increased expression of apoptotic-related gene and protein levels in the lung allograft compared with the native PAH lung and the donor lung. Localization of cell apoptosis determined by triple immunostaining for caspase 3, CD31, and smooth muscle actin was positive in the pulmonary endothelial cells but not the smooth muscle cells of the lung allograft, while no positive staining was detected for cell death in the native PAH lung. The presence of PH in the lung allograft 22 years after transplantation was associated with upregulation of apoptotic markers and evidence of apoptotic endothelial cell death compared with the native lung and donor lung.
Pulmonary arterial hypertension (PAH) is a severe vascular disease characterized by a progressive arteriopathy that leads to progressive right heart failure and premature death.1,2 The pathological mechanisms underlying this condition remain elusive. Pulmonary artery endothelial cell dysfunction and structural remodeling of the pulmonary vessels are early features of PAH. These features are characterized by hyperproliferative and antiapoptotic diathesis within the vascular wall of the resistance pulmonary arteries, leading to vascular lumen occlusion.3,4 In PAH, the vascular remodeling process includes the proliferation and migration of pulmonary artery smooth muscle cells, leading to medial hypertrophy and increased pulmonary vascular resistance.5–7 Recent evidence suggests that PAH results from the initial loss of normal endothelial cells and the replacement by endothelial cells that are hyperproliferative and antiapoptotic.8,9 The mechanisms leading to pulmonary hypertension (PH) in other conditions than PAH, such as in patients with underlying parenchymal lung disease (group 3) or chronic thromboembolic disease (group 4), are not well documented. Herein, we report a patient who underwent bilateral lung transplantation for chronic lung allograft dysfunction (CLAD) and recurrent PH 22 years after single lung transplantation for PAH. This case provided the unique opportunity to study the molecular and pathological mechanisms of pulmonary vascular remodeling in the first lung allograft and the native PAH lung.
CASE DESCRIPTION
A 50-year-old female who had undergone a single right lung transplantation 22 years ago for idiopathic PAH underwent double lung transplantation due to CLAD and recurrence of PH. Right heart catheterization before the retransplantation showed severe PH, with pulmonary artery pressures of 99/48 mmHg (mean: 74 mmHg). The pulmonary function test showed a forced expiratory volume in 1 second (FEV1) of 1.4 L (52% of predicted), forced vital capacity of 2.3 L (71% of predicted), and diffusing capacity of the lungs for carbon monoxide (DLCO) of 69% predicted. Over the past 2 years before her retransplantation, her clinical conditions rapidly deteriorated to World Health Organization functional class IV, requiring home oxygen, despite stability in her FEV1. The right ventricular systolic pressure on the echocardiogram increased to 89 mmHg during the same time period. Therefore, her clinical deterioration was felt to be related to worsening PH, and she was started on phosphodiesterase type 5 inhibitor. Her perfusion scan also demonstrated a radical reduction in the perfusion of her right lung from 82% to 21%. A CT pulmonary angiogram showed the presence of a new eccentric thrombus in the right pulmonary artery that improved after starting anticoagulation. The cause of PH at the time of retransplantation could therefore have been related to chronic thromboembolic disease (group 4), chronic lung allograft dysfunction (group 3), or progression of her idiopathic PAH (group 1).
At the time of retransplantation, lung biopsies were obtained from the new donor lung as well as both explanted lungs—namely, the right lung allograft and the native left lung—after hospital research ethics board approval. Representative samples of each were snap frozen for molecular and protein analysis or fixed in 4% formaldehyde for immunohistochemistry analysis (appendix, available online).
Transcriptomic analysis
Real-time polymerase chain reaction (PCR) was conducted to contrast the expression of apoptotic and proliferative markers in the donor lung as well as the allograft and the native lungs (Table S1, available online). Detailed procedures for transcriptomic analysis, immunoblotting, and immunohistochemistry are listed in the appendix.
Results
Morphological study.
Histopathological examination of the right lung allograft showed characteristics of diffuse vascular remodeling combined with features of acute and chronic thromboemboli and evidence of bronchiolitis obliterans. In contrast, the native left lung demonstrated pulmonary arterial changes in keeping with PAH associated with disseminated pulmonary ossification (Fig. 1). Hematoxylin-and-eosin staining was performed to characterize blood vessels in the donor lung (Fig. 1a), single-transplanted right lung, which developed PH after lung transplantation (Txd PH; Fig. 1b–1d), and left native PH lung (original PH lung; Fig. 1e, 1f). The single-transplanted lung showed diffuse vascular remodeling in the distal pulmonary artery with partial or complete intimal fibrotic formation within the thickened media layer (Fig. 1b–1d), consistent with the development of precapillary pulmonary hypertension. Severe intimal fibrotic lesions and dense calcification were seen in the native PH lung but not in the right lung.
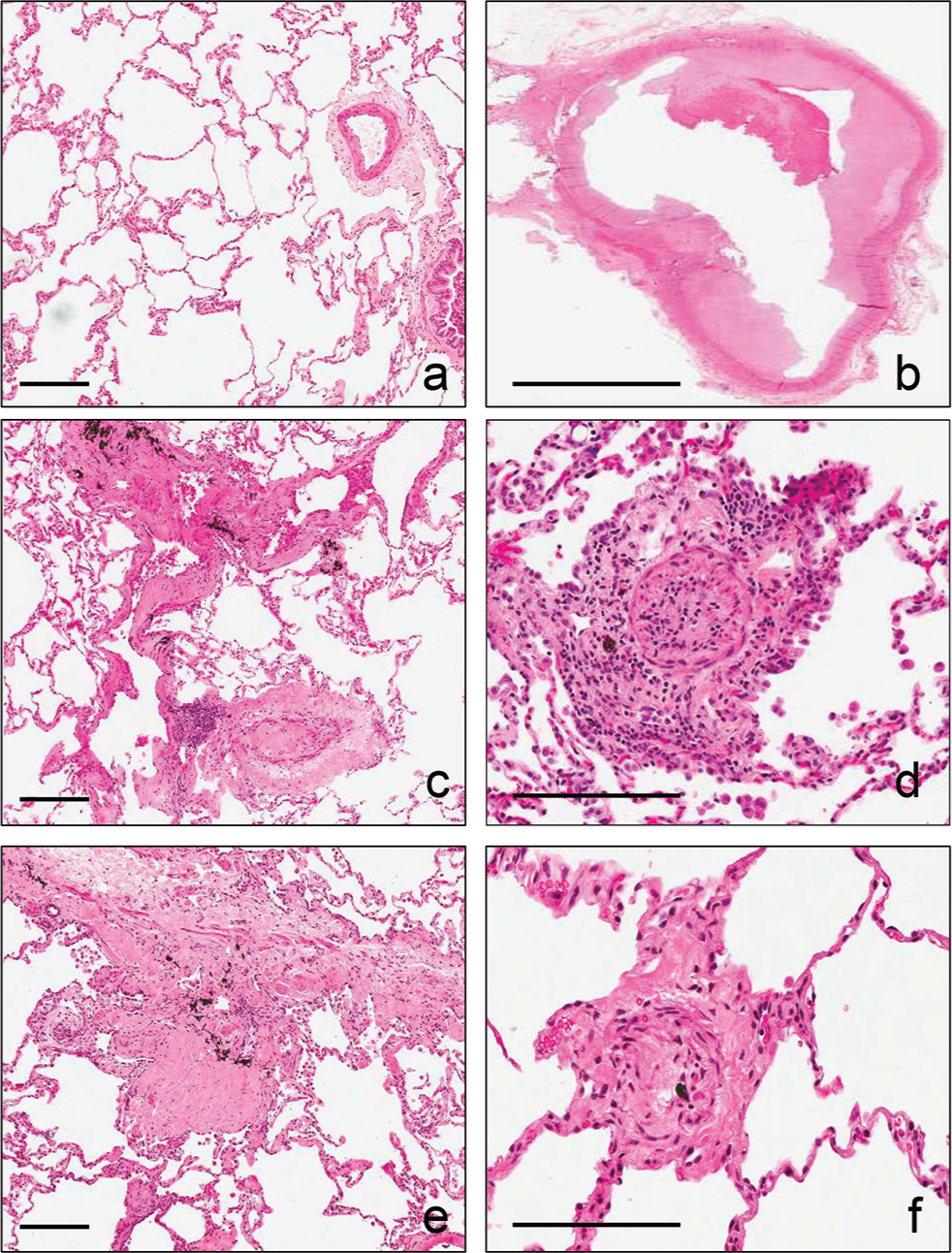
Hematoxylin and eosin staining of the donor lung (a), proximal pulmonary artery and lung from the right lung allograft (b–d), and native left pulmonary arterial hypertension lung (e, f). Evidence of bronchiolitis obliterans was observed in the right lung allograft associated with eccentric chronic thromboemboli and distal vascular remodeling (scale bars = 50 μm).
PCR and Western blot analysis.
Real-time PCR was conducted to contrast the expression of apoptotic and proliferative markers in the donor lung, the lung allograft, and the native lung (Fig. 2). Interestingly, the real-time PCR shows (Fig. 2, top) that caspase 3 was drastically increased in the lung allograft compared with the native lung and donor lung. The expression of other markers such as bone morphogenetic protein receptor 2 (BMPRII), Ras homolog gene family A (RhoA), and proliferating cell nuclear antigen (PCNA) was also increased in the lung allograft compared with the donor lung. In contrast, the cell cycle inhibitory marker p21 was decreased in the lung allograft. Western blotting (Fig. 2, bottom) showed that some of the protein expression supported the RNA data. In particular, caspase 3 and RhoA were upregulated in the lung allograft and the native PAH lung compared with the donor lung. In contrast, some protein expression, such as PCNA and cleaved poly (ADP-ribose) polymerase (cPARP), presented different trends between the PCR and Western blot data. The functional significance of these findings will require further study. RhoA activation of Rho kinases leads to phosphorylation of myosin light chains in smooth muscle cells and sustained contraction. Increased Rho expression has been previously described in cases of PAH. 10 Our data showed that RhoA expression was slightly increased in both the single Txd PH lung and the native PH lung.
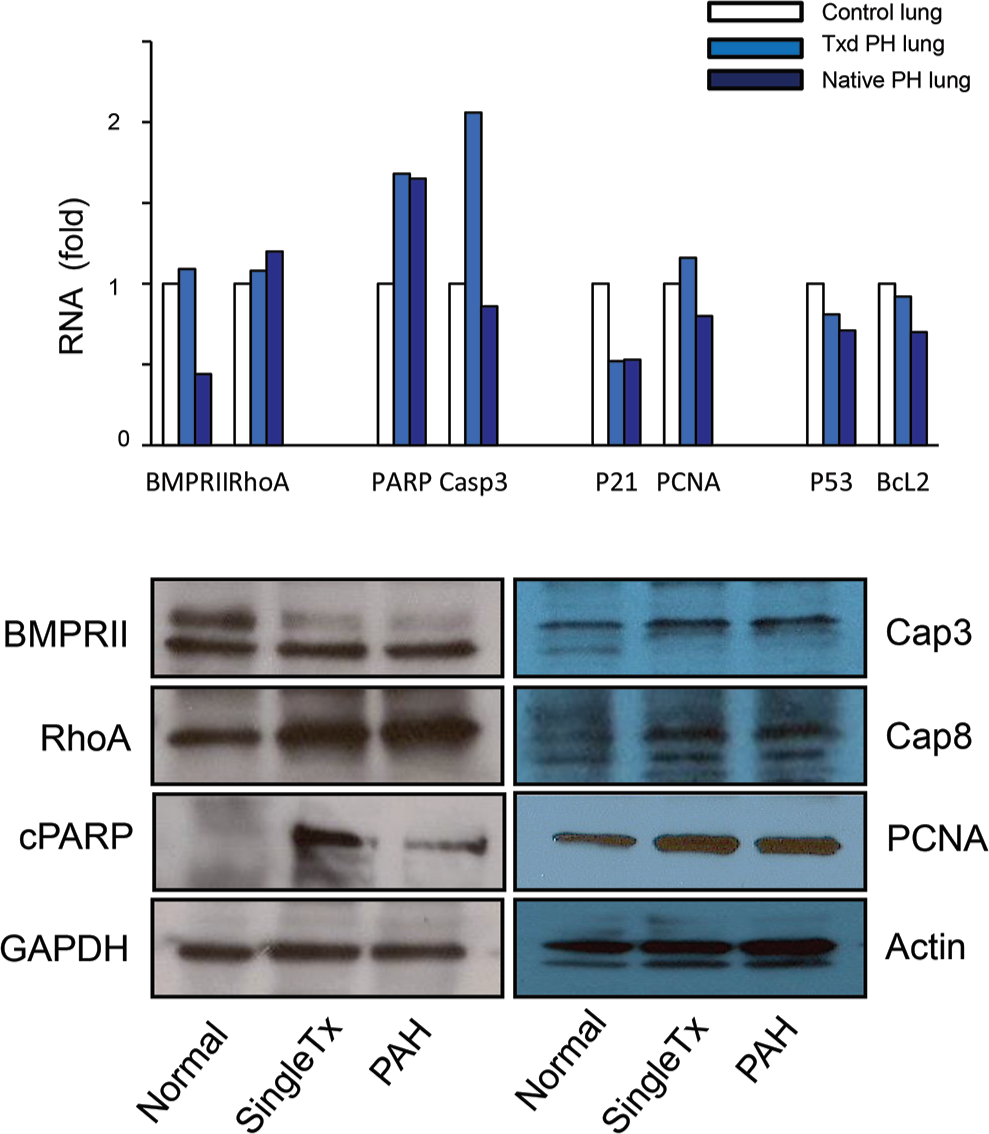
Top, real-time polymerase chain reaction was conducted to determine the expression of pulmonary hypertension–related apoptosis and proliferation genes—such as bone morphogenetic protein receptor 2 (BMPRII), Ras homolog gene family A (RhoA), cleaved poly (ADP-ribose) polymerase (cPARP), caspase 3 (Cap3), P21, proliferating cell nuclear antigen (PCNA), P53, and BCL2—in healthy donor lung (white bar), single-transplanted (Txd) lung (light blue bar), and left native pulmonary hypertension (PH) lung (dark blue bar). Bottom, Western blotting was conducted to show protein expression changes of the genetic related marker BMPRII, vasoconstrictor marker RhoA, cell proliferation marker PCNA, and apoptotic markers Cap3, 8, and cPARP against housekeeping proteins GAPDH and actin in the normal, single Txd, and pulmonary arterial hypertension (PAH) native PH lung sample.
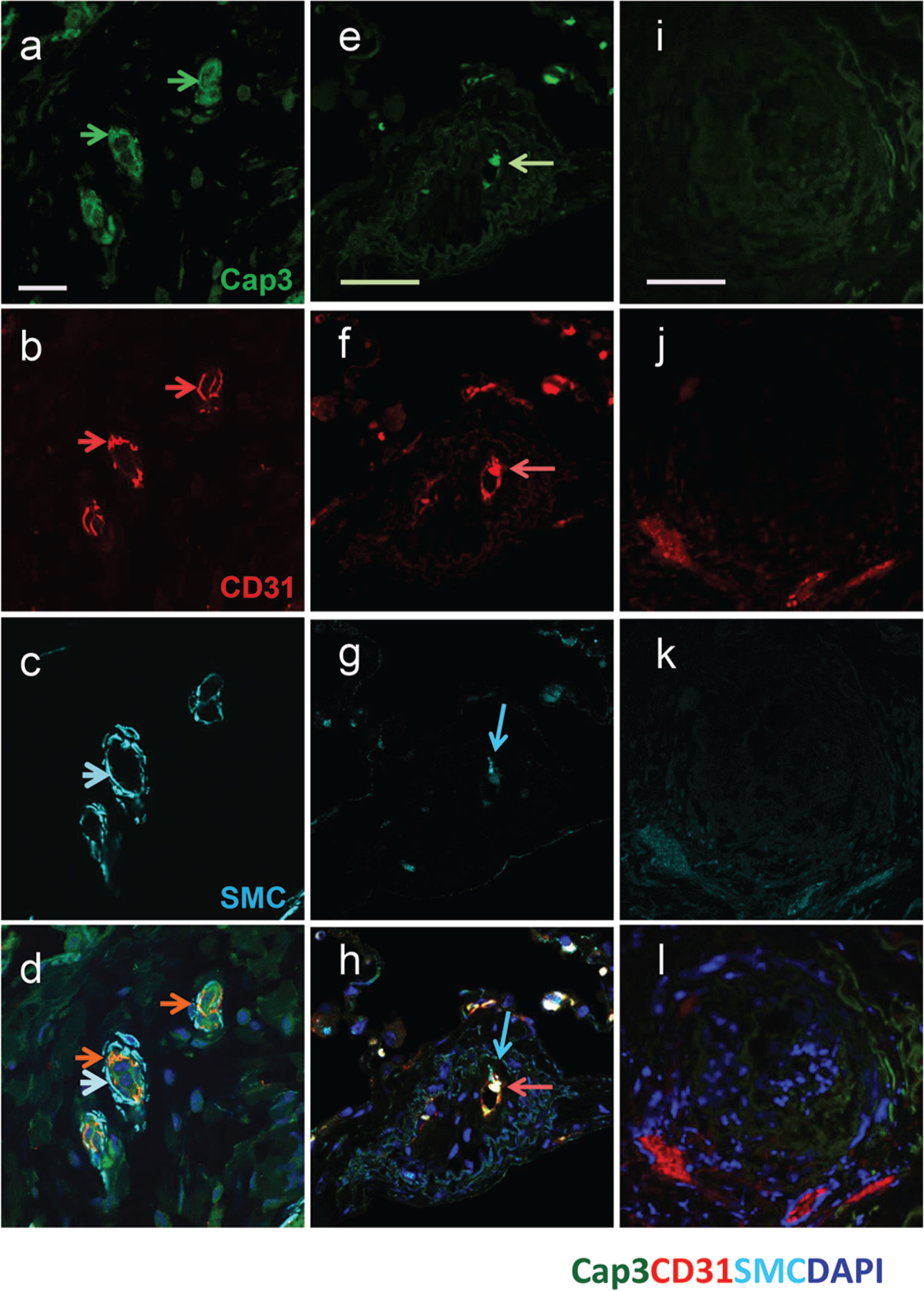
Morphological studies conducted with fluorescent confocal microscopy display images of the single-transplanted lung. Paraffin sections of the terminal pulmonary arteries were stained with apoptotic marker caspase 3 (Cap3) in green (a, e, i), endothelial cell marker CD31 in red (b, f, j), smooth muscle actin marker (SMC) in light blue (c, g, k), and DAPI in blue. Confocal microscopy showed that pulmonary endothelial cells—but not smooth muscle cells—of the single-transplanted lung were undergoing apoptosis (d, h). The newly formed small vessels were stained for apoptotic marker, caspase 3 (Cap3) in green (e), endothelial cell marker CD31 in red (f), smooth muscle actin marker (SMA) in light blue (g), and DAPI in blue. Confocal microscopy showed that newly formed endothelial cells were undergoing apoptosis. i–l, Confocal microscopy showed the plexiforms filled with mixed cells with no apoptotic cells observed (scale bars: 50 μm).
DISCUSSION
This case report is unique in several aspects. The patient is one of the longest survivors after lung transplantation for PAH. She underwent right single lung transplantation in November 1989 followed by bilateral lung transplantation in January 2012. Her retransplantation was motivated by the development of CLAD combined with PH. The mechanisms of PH before her retransplantation could have been due to either the progression of CLAD or the development of chronic thromboembolic disease, which was evident on the CT scan and on final pathology. Previous studies have shown that transplanted lungs can develop PH in association with CLAD.11,12 Although less likely, the progression of her idiopathic PAH in the native lung could also have contributed to the development of PH in the right lung allograft. It has been shown in an animal model that the presence of a nonperfused lung could induce vascular remodeling in the contralateral lung potentially through the release of factors involved in vascular smooth muscle cell proliferation 9 Her second transplant also provided us with unique material to study the mechanisms of PH. We obtained samples from her right lung allograft, her left native PAH lung, and the new donor lung. Since experimental PAH models 9 show that apoptosis may play a major role in the development of vascular remodeling, we analyzed markers of apoptosis and cell proliferation in details in all three samples. We observed that the presence of PH in the lung allograft was associated with upregulation of apoptotic markers and evidence of apoptotic endothelial cell death compared with the native lung and donor lung. Analysis of the native PH lung with confocal microscopy showed that apoptosis was mostly absent.
Footnotes
ACKNOWLEDGMENTS
The authors thank Ms. Hana Y. Z. Hong for her excellent work on molecular analysis and Drs. John Granton, David Huang, and Mingyao Liu for their scientific comments.
