Abstract
The Pulmonary Hypertension Association (PHA) is a patient advocacy organization seeking to find ways to prevent, improve treatment for, and cure pulmonary hypertension (PH) and to provide hope for the PH community through support, education, research, advocacy, and awareness. Many patients involved with PHA are also involved in various PH-specific research studies; however, the patient expectations and priorities for PH-specific research are currently unknown or not well examined. Our objective was to identify the current modes of study entry, priorities within research, and expectations over the course of study involvement for patient constituents of PHA. A 29-question online survey was designed by PHA and disseminated to the PHA patient constituency on its Facebook page through a post on November 29, 2012. Responses were collected on SurveyMonkey through December 10, 2012. Respondents were divided into parallel survey tracks, depending on whether the respondent indicated previous participation in research studies. These two cohorts of individuals were analyzed and, where appropriate, compared with tests of association. A total of 234 respondents were included in the final data analysis, with 95 (40.6%) reporting previous participation in research studies. These respondents reported an overall positive experience in their research studies (64.9% very good, 21.3% good, 12.8% neutral, 1.1% bad). Of the respondents with previous research study participation, 91.1% indicated that receipt of the study outcome after participation would positively influence their decision to participate in future research; despite this, only 41.17% reported receiving information of this sort after their participation. Research participation is a strong interest of PHA patient constituents; clear and consistent communication from the research team is an expectation of many participants. Despite this expectation, 58.83% of respondents indicated they did not receive communication from the research team after participation. This offers an opportunity not only to improve participants’ experiences but also to increase the likelihood of future study participation.
Pulmonary hypertension (PH) is a rare, complex, multisystem disorder resulting from progressively restricted pulmonary arterial circulation and increased pulmonary vascular resistance, ultimately leading to right heart failure. 1 PH can occur as an idiopathic process, be inherited through a number of genetic mutations, or arise in association with another disease, such as chronic thromboembolic disease, connective-tissue disorders, congenital heart disease, chronic liver disease, HIV infection, and illicit drug use. 2 A specific type of PH that is characterized by increased pressure directly due to remodeling of the adventitia, media, and intima in the distal pulmonary arterial bed is called pulmonary arterial hypertension (PAH). By definition, persons with PAH have a mean pulmonary artery pressure of >25 mmHg; a pulmonary artery occlusion pressure, left atrial pressure, or left ventricular end-diastolic pressure of ≤15 mmHg; and a calculated pulmonary vascular resistance of >3 Wood units. 2
There is currently no cure for PAH; however, several therapeutic options exist to reduce the disease severity. These include conventional supportive medical therapies and oral, inhaled, subcutaneous, and intravenous PAH-targeted therapies. Available therapeutic modalities continue to expand, but this requires involvement of patients who participate in research studies. Participant recruitment to the levels required for statistical power and wider external validity can be difficult, given the rarity of PAH, which has an estimated prevalence between 15 and 52 cases per million population and an incidence between 2.4 and 7.6 cases per million population. 3
The Pulmonary Hypertension Association (PHA) is a grass-roots organization that seeks to convene multiple stakeholders in PH: patients, family members, caregivers, medical professionals, and interested industry professionals. A core tenet of the PHA's mission is to facilitate patients’ active involvement throughout the course of their disease. Since 1994, thousands of patient constituents have sought this enfranchisement through participation in the PHA Research Room, a biennial opportunity to contribute data and biological specimens to the studies of 10–12 investigators at each of the PHA International PH Conference and Scientific Sessions. These biennial opportunities, however, are insufficient for the many active constituents to achieve their goal of seeking every available avenue to get ahead of their disease.
Entry into the world of scientific research is understandably complicated for the many potential participants lacking an education in the basic and clinical sciences. Patient and caregiver leaders within the PHA community, who regularly seek research study opportunities, have informally expressed confusion and frustration at the inaccessibility of study information for the health illiterate. Wishing to identify the current modes of study entry, patients’ priorities within research, and expectations over the course of the study—ultimately in an attempt to increase research study participation—the PHA completed the following survey of its patient constituency.
METHODS
Survey content
A complete version of the “Patient Expectations in Research Studies” survey is included in appendix 1 (Apps. 1–4 available online). Leadership in the PHA designed the survey, and responses were collected electronically through the online platform Survey Monkey. At the beginning of the survey, respondents were asked (1) whether they were patients with PH and (2) whether they had previously participated in a research study. Depending on the response to the historical-participation questions, respondents were placed into parallel survey tracks seeking to examine (1) how important various recruiting parties and strategies are in the study entry process, (2) whether fully understanding study benefits and risks altered one's decision to participate, (3) the importance of select study targets and (4) what information or benefits the participant expected to receive at the conclusion of the study. Finally, those respondents who reported previous participation were asked what information they had received from the research teams and their overall experience participating in research.
Survey distribution
The PHA solicited participation in this survey on its Facebook page, through a post on November 29, 2012. At the time, the PHA was connected to 6,937 individuals through this Facebook page, all of whom had the opportunity to view this notification and participate. Data were self-reported and submitted anonymously from November 29 through December 10, 2012.
Statistical analyses
Data were collected through the Survey Monkey online survey platform, and at the conclusion of the survey period, raw results were downloaded as a Microsoft Excel spreadsheet. Categorical variables were compared via the Pearson χ 2 test; continuous variables were compared via the Student t test. A 2-tailed P value of <0.05 was considered statistically significant a priori. All proportions are presented with the raw number of responses. Statistical analyses were performed with STATA 13.1 SE software (StataCorp, College Station, TX).
RESULTS
Survey population
A total of 259 anonymous survey responses were received during the open period, from 17 countries. Respondents were initially asked to confirm their status as a PH patient; 10 individuals indicated that they were caregivers to PH patients, and their responses were not included in the final analysis. An additional 15 respondents submitted a survey that was more than three-quarters incomplete, and their responses were also excluded.
Of the 234 respondents included in the final analysis, 95 (40.6%) reported previous participation in a research study (see Table 1). In this cohort of previous participants, 38 (40.0%) indicated participation in only 1 study, 31 (32.6%) in 2 or 3 studies, and 24 (25.3%) in more than 3. Nearly all (96.2%) indicated that at least one of these studies was related to PH. The remaining 3.8% were not excluded from data analysis, to potentially include participants in studies related to PH comorbidities. Although 32 (13.7%) of the survey respondents were from countries outside the United States (for these purposes, Puerto Rico was defined as a separate country), neither previous participation (P = 0.440) nor the number of studies for those reporting previous participation (P = 0.114) differed when comparing US to non-US respondents.
Survey participants

When asked to indicate the timing of their most recent study, 26 (27.7%) of respondents indicated that they were currently enrolled in a study, 25 (26.6%) had participated within the past year, 26 (27.7%) had participated within the past 3 years, and 17 (18.1%) indicated that his or her most recent research study was more than 3 years ago.
Overall, respondents reported a positive experience with the study participation: 61 (64.9%) rated his or her experience as very good, 20 (21.3%) as good, 12 (12.8%) as neutral, and 1 (1.1%) as bad. Qualitative, open-ended responses were accepted to this question as well, and examples are viewable in appendix 2.
Individuals who indicated that they had not previously participated in a research study were asked what had kept them from participating. Of those who specified, 61 (51.7%) indicated that they were unaware of any studies; 18 (15.25%) wanted to participate but ultimately did not qualify; and 39 (33.05%) indicated that they were aware of studies but were not approached.
Research study participant recruitment
Research study participants were then asked to indicate the relative importance of different factors as they decided to participate in research studies: medical team invitations, patient association encouragement, encouragement from other patients, and the individual's previous experiences. An invitation from the respondent's physicians and nurses was viewed as important to research study participation. Specifically, 46 (52.9%) rated this as extremely important, 32 (36.8%) as important, 2 (2.3%) as not important, and 7 (8.05%) as not applicable. When asked to rate the importance of a patient association encouraging the respondent to participate in a research study, 12 (14.8%) rated this as extremely important, 22 (27.2%) as important, 11 (13.6%) as not important, and 36 (44.4%) as not applicable. Similar results were noted when previous participants were asked to rate the importance of encouragement from their caregiver or family members: 14 (17.5%) rated this as extremely important, 18 (22.5%) as important, 12 (15.0%) as important, and 36 (45.0%) as not applicable. A similar distribution was also noted on the importance of encouragement from other patients in study participation: 13 (16.5%) rated this encouragement as extremely important, 15 (19.0%) as important, 13 (16.5%) as not important, and 38 (48.1%) as not applicable. The respondents’ own previous experiences were rated fairly high in importance for participation, with 25 (30.1%) rating this as extremely important, 18 (21.7%) as important, 4 (4.8%) as not important, and 36 (43.4%) as not applicable.
Individuals’ understanding of the benefits and risks of study participation was rated very high in importance for study participation. When asked to rate the importance of understanding the benefits of study participation, 73 (84.9%) rated this as extremely important, 7 (8.1%) as important, 2 (2.3%) as not important, and 4 (4.7%) as not applicable. The importance of understanding the risks was not rated quite as strongly, with 54 (66.7%) indicating that this was extremely important, 17 (21.0%) important, 0 (0.0%) not important, and 10 (12.4%) not applicable. Bivariate analysis revealed that a clear understanding of the benefits and risks positively correlated to the reported overall experience in the respondents’ research study participation (P < 0.001 and P < 0.01, respectively; see Tables 2, 3).
Understanding benefits and overall reported experience
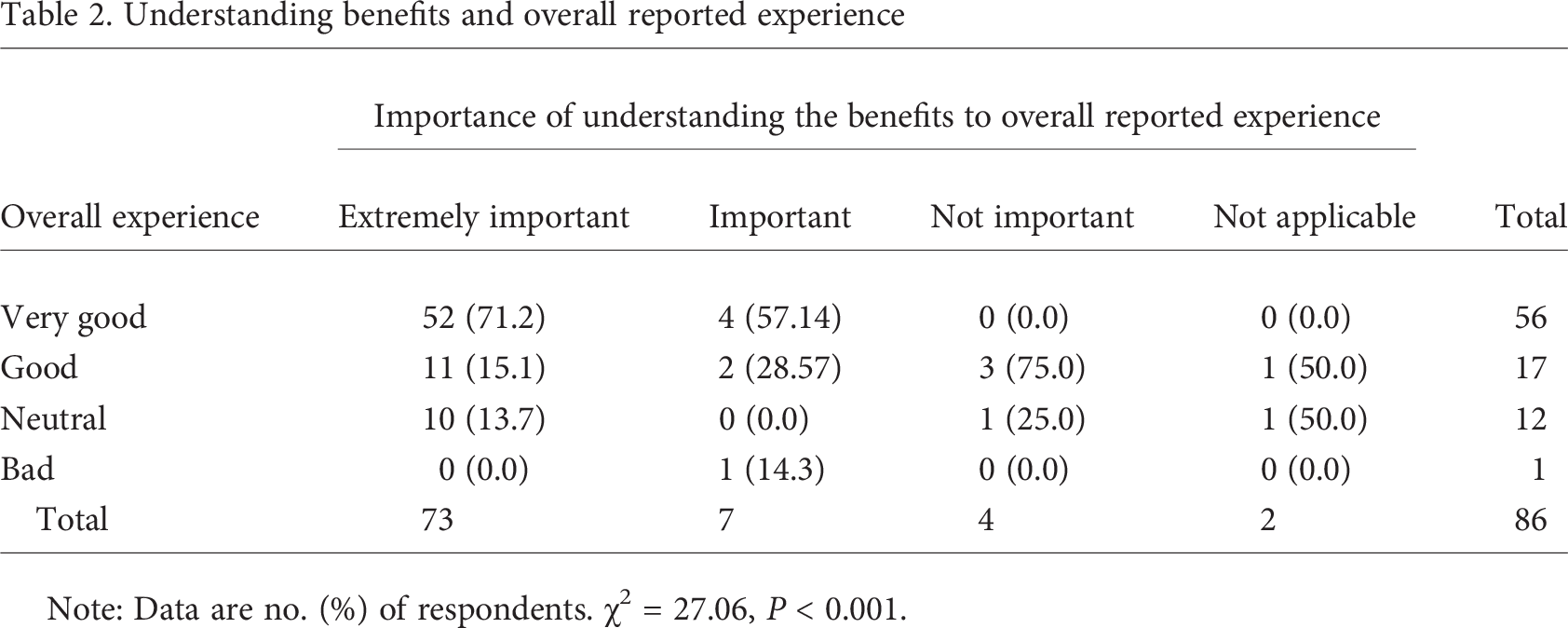
Note: Data are no. (%) of respondents. χ 2 = 27.06, P < 0.001.
Understanding risks and overall reported experience
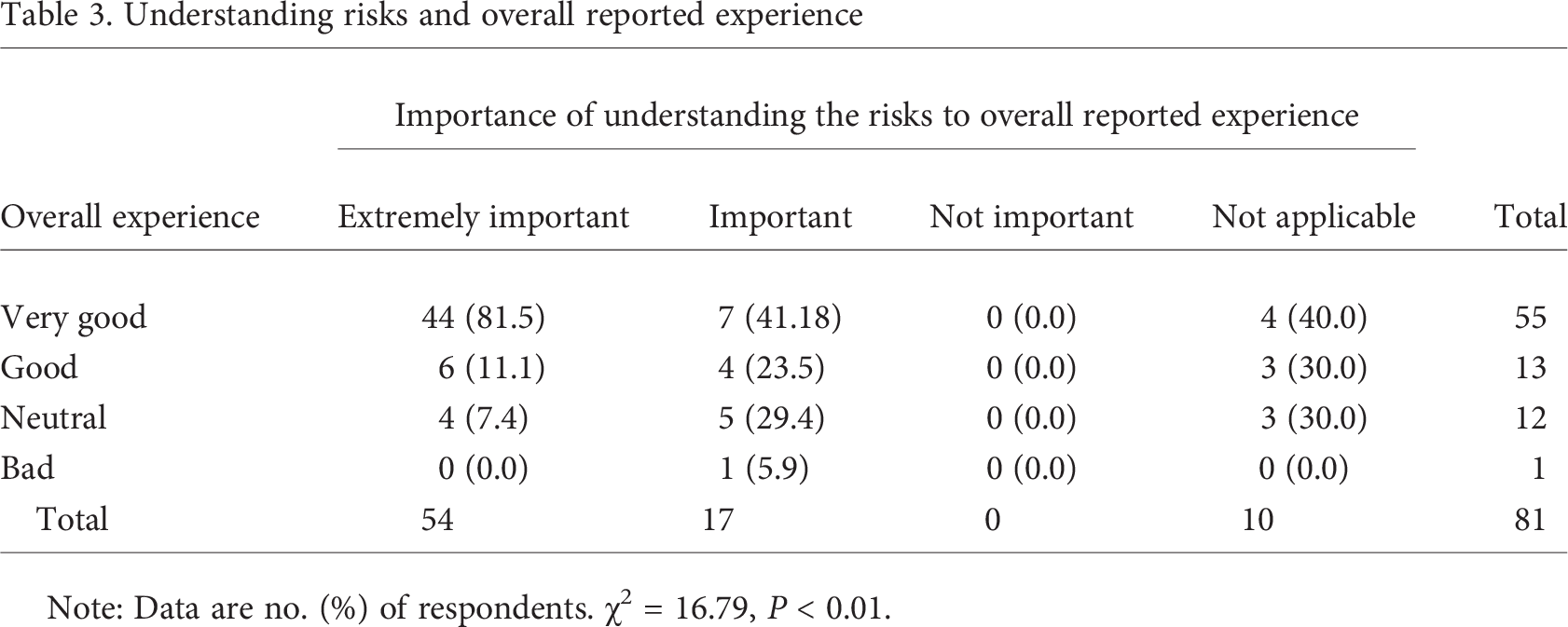
Note: Data are no. (%) of respondents. χ 2 = 16.79, P < 0.01.
Poststudy communication received
Respondents were asked to indicate whether they felt that they had received enough information, specifically on the study results, from the research team. Twenty-two (24.4%) indicated that they had not received results because the study was ongoing. Of those who reported completion of the research study, 40 (58.8%) of respondents indicated receiving no information on the study results, and 28 (41.2%) reported receiving study results. When all previous participants were asked whether receiving communication after the conclusion of the study, including the study results as they became available, would make them more likely to participate in future studies, 82 (91.1%) reported that this would positively influence their decision, 3 (3.3%) indicated this would not positively influence their decision, and 5 (5.6%) were not sure. Previous participants were more likely than nonparticipants to report that receiving communication at the conclusion of the study would positively affect their decision to participate in future research studies (P = 0.012). Ninety-eight (80.3%) of the respondents without reported research study participation indicated that receiving information would make them more likely to participate in future studies; 16 (13.1%) reported that this information would not affect their decision to participate in the future; 8 (6.6%) were not sure.
Future direction of research
Previous research study participants completed an additional section that sought to identify their priorities in future PH research. Participants were asked to rate 5 study targets on a 4-point scale (“very important,” “important,” “does not matter much,” and “does not matter at all”). When asked to rate the importance of identifying new treatments, 69 (96.6%) rated this as important or very important. Eighty-six (95.6%) rated a more robust understanding of the side effects of current pharmaceutical therapies as important or very important. Seventy-six (86.4%) indicated that further exploring the genetics of PH was important or very important. Sixty-nine (77.5%) felt that identifying less invasive diagnostic modalities (e.g., alternatives to right heart catheterization) was important or very important. Finally, 64 (71.9%) indicated that changing the clinical endpoint of studies and trials (e.g., alternatives to the 6-minute walk test) was important or very important. See Table 4 for full quantitative results and appendix 3 for responses to the open-ended portion of the section.
Research study priorities
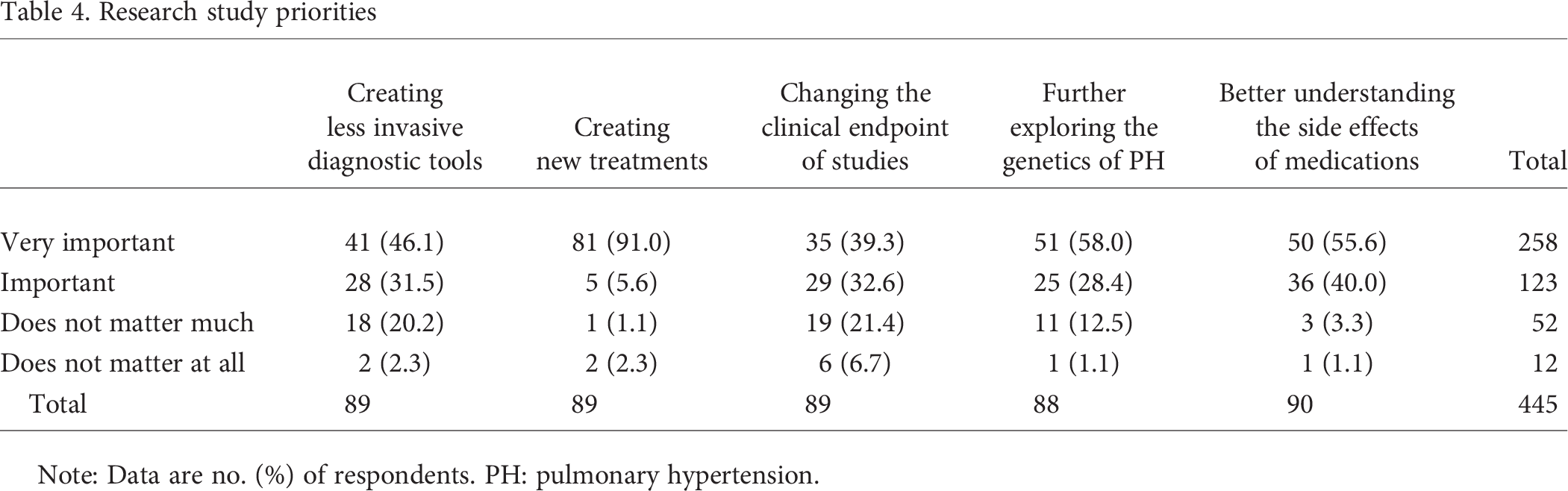
Note: Data are no. (%) of respondents. PH: pulmonary hypertension.
Nonparticipants were asked to rate the same research study foci—new treatments and a more robust understanding of side effects, genetics, diagnostic modalities, and clinical endpoints—on a similar 5-point scale (“very important,” “important,” “unsure,” unimportant,” “very unimportant”). When ranking new treatments, 121 (98.4%) of respondents without previous research experience indicated that this was important or very important. One hundred eleven (91.0%) believed that a more robust understanding of therapeutic side effects was important or very important. One hundred five (85.4%) stated that further exploring the genetics of PH was important or very important. One hundred one (82.1%) rated less invasive diagnostic modalities as important or very important. Examining alternative study endpoints was rated important or very important by 94 (76.43%) of respondents without previous research participation.
Postresearch study communications expectation
All participants, regardless of previous research study participation, were asked to indicate their poststudy communication expectations. Respondents were allowed to choose whether they expected to receive information on the study's outcome, continued treatment, information on other trials in the field, and/or nothing. Respondents without previous research study participation were significantly more likely to expect communication on each of the identified options than were those with previous research study participation (see Table 5).
Poststudy communication expectation
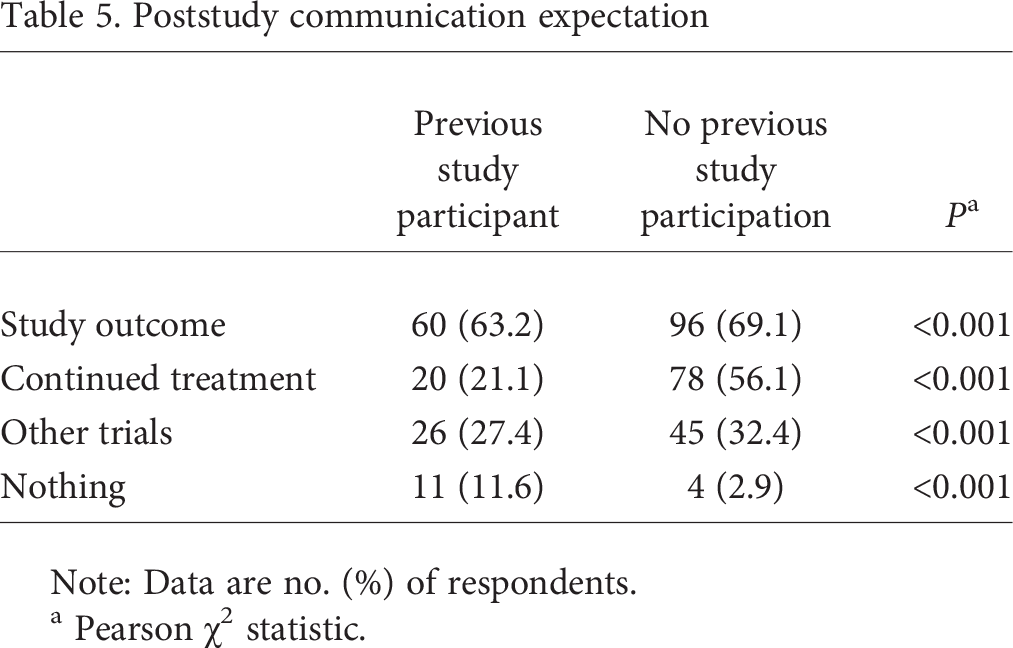
Note: Data are no. (%) of respondents.
Pearson χ 2 statistic.
Empowerment through research study participation
In order to learn what drives potential subjects into research study participation, survey respondents with no previous research study participation were asked to indicate how participating in research would make them feel. In addition to the open-ended responses in appendixes 2–4, respondents were asked to indicate whether participating made them feel empowered, that they were participating in the advancement of science, and/or that their care was being sacrificed for research purposes. Respondents could choose more than one option. Thirty-nine (28.1%) of those respondents who had no previous research experience indicated that their participation would make them feel empowered, 91 (65.5%) felt that they would be participating in the advancement of science, and only 5 (3.6%) felt that their participation would lead to their care being sacrificed.
DISCUSSION
Our survey demonstrated that PH patients connected with the PHA, regardless of previous research study involvement, are generally very interested in information and communication regarding PH-related research. Despite the limitations in survey design and the responder biases expected in survey methodology in general, to be outlined below, this survey offers important insights into the methods of entry into research studies, overall participant satisfaction, feelings due to participation, and communication expectations from the research teams.
In this survey instrument, the term “research study” was never formally defined for respondents. Principal investigators and study sponsors are able to submit clinical trials for inclusion on the PHA website at www.PHAssociation.org/ClinicalTrials. Currently, 25% of the studies listed on this page are observational prospective studies and 75% are randomized controlled trials of pharmaceutical agents and strategies. In the Research Room at the 2014 PHA International PH Conference and Scientific Sessions, 50% of the participating studies were observational and 50% were cross-sectional. Given the wide range of study types, interventions, and potential research settings of patient constituents of the PHA and the desire to include the communication expectations of all potential clinical research subjects, the survey instrument allowed respondents to interpret the term “research participation.” Unfortunately, this does not allow for comparison between different study types; however, it does present the expectations and experiences of previous participants in aggregate.
In general, respondents with previous research study participation indicated the importance of their medical teams when choosing to participate in research studies. Respondents indicated that involvement from other parties—such as encouragement from patient associations, family members, and other patients—was not as important for recruitment into the research studies in which they participated. Despite survey participation being limited to those connected to the PHA, this result is not surprising, given that the PHA as an organization does not actively encourage its patient constituents to participate in specific research studies. This is due, in part, to the inherent selection bias that is already a general concern in rare-disease case-control studies; 4 the association believes, as currently organized, that this continues to be a conversation best left to a patient and his or her medical team.
One important component of a potential subject's decision to participate in research studies is a clear and understandable explanation of the risks to the subject and the potential benefits to the subject and others. Not surprisingly, a strong correlation exists with the stated importance of benefit and risk of the research study and the subjects’ overall positive experience in previous research studies.
Our survey highlighted several important expectations of research study participants regarding communication with the research team. More than half (40 [58.8%]) of respondents who reported full completion of their participation did not receive any information on the outcome of the research study in which they participated. This was overwhelmingly the largest expectation of all previous participants; 60 (63.2%) indicated that they expected to receive this information, and a total of 82 (91.1%) reported that receipt of this information would positively affect their decision to participate in future studies.
Continuous communication to patient participants places an understandably large administrative burden on research teams. For multicenter studies, which are often required for rare-disease research and wider external results validation, the party responsible for this consistent, understandable, regular, unbiased communication may not be clear. This is an important issue to address, however, as the expectation of poststudy communication on the outcome was rated much higher than other expectations—such as continued treatment or information on other trials in the field.
Messaging is extremely important when working with potential research subjects, and this survey asked those who had not previously participated in a research study how participating would make them feel. More than half (91 [65.5%]) felt that they would be participating in the advancement of science, nearly one-third (39 [28.06%]) indicated that they would feel empowered, and only 5 (3.6%) felt that their care would potentially be sacrificed by participating. Despite the very low level of health literacy in the United States, 5 we demonstrate that patients are generally very interested in actively participating both in the advancement of science and throughout the course of their progressive disease state. Messages should therefore be communicated to patient subjects explaining, to the extent possible, the immediate and larger scientific implications of their involvement.
Limitations
While the results from this survey provide an important insight into the thoughts and expectations of research study participants, there are a number of limitations, in both design and methodology, to the scope and external validation of the results previously described. First, this was an online survey that was publicized through a single e-communication channel and is therefore subject to voluntary-response bias. Moreover, the survey was available only in English, despite the global reach of the PHA Facebook page. In addition, the population from which responses were collected is directly connected to the PHA and is likely an active subset of the general PH community, furthering the selection bias inherent in survey methodology. Second, basic demographic data were not collected to provide an insight into the responses ultimately included in, or excluded from, the final data analysis. Third, the term “research study” was never formally defined and therefore could reasonably be interpreted in a number of ways, ranging from observational registry inclusion to randomized clinical trials. Finally, although respondents were placed into parallel surveys on the basis of their previous participation history in research studies, response options were not always parallel and limited the tests of association between these two cohorts.
Conclusion
The PHA Patient Expectations in Research Studies survey, despite its limitations, provides useful insight into the expectations and priorities of patients participating in PH-specific research studies. Research teams have a clear incentive to improve communication practices with patient participants, specifically in how benefits and risks are communicated to the participant before participation and how results are communicated after participation. Further study and focus groups are clearly needed to maximize participants’ experiences, to ensure that expectations are met and that patient research priorities are scientifically examined.
Footnotes
ACKNOWLEDGMENTS
We thank those who participated in the Patient Expectations in Research Studies online survey. In addition, we acknowledge and thank B. Rivas-Morello (Pulmonary Hypertension Association) for her assistance in data preparation for the survey analysis.
