Abstract
Decreased synthesis of nitric oxide (NO) by NO synthases (NOS) is believed to play an important role in the pathogenesis of pulmonary arterial hypertension (PAH). Multiple factors may contribute to decreased NO bioavailability, including increased activity of arginase, the enzyme that converts arginine to ornithine and urea, which may compete with NOS for arginine; inadequate de novo arginine production from citrulline; and increased concentration of asymmetric dimethylarginine (ADMA), an endogenous inhibitor of NOS. We hypothesized that PAH patients with the lowest arginine availability secondary to increased arginase activity and/or inadequate de novo arginine synthesis might have a slower rate of NO synthesis and greater pulmonary vascular resistance. Nine patients with group 1 PAH and 10 healthy controls were given primed, constant intravenous infusions of 15N2-arginine, 13C,2H4-citrulline, 15N2-ornithine, and 13C-urea in the postabsorptive state. The results showed that, compared with healthy controls, PAH patients had a tendency toward increased arginine clearance and ornithine flux but no difference in arginine and citrulline flux, de novo arginine synthesis, or NO synthesis. Arginine-to-ADMA ratio was increased in PAH patients. Two endotypes of patients with low and high arginase activity were identified; compared with the low-arginase group, the patients with high arginase had increased arginine flux, slower NO synthesis, and lower plasma concentrations of ADMA. These results demonstrate that increased breakdown of arginine by arginase occurs in PAH and affects NO synthesis. Furthermore, there is no compensatory increase in de novo arginine synthesis to overcome this increased utilization of arginine by arginase.
INTRODUCTION
Pulmonary arterial hypertension (PAH) is a disease of unknown etiology characterized by an increase in pulmonary vascular resistance (PVR) secondary to pulmonary arterial vasoconstriction, vascular remodeling, and thrombosis. Decreased bioavailability of the vasodilatory molecule nitric oxide (NO) is believed to play an important role in the pathogenesis of PAH.1–3 In support of this, intrapulmonary NO, biochemical reaction products of NO in bronchoalveolar fluid, and exhaled NO have been found to be decreased in PAH patients compared with controls.3,4 NO is produced in the body by a family of enzymes known as NO synthases (NOS). In a reaction that consumes molecular oxygen and cofactors such as nicotinamide adenine dinucleotide phosphate and tetrahydrobiopterin (BH4), NOS converts
The synthesis of NO via NOS depends on the availability of substrate and cofactors as well as the presence of endogenous inhibitors of the enzyme. In humans, the major source of arginine is whole-body protein breakdown. De novo arginine synthesis occurs in the kidney via a two-step reaction starting with citrulline and with argininosuccinate as an intermediate 7 and constitutes 5%–15% of endogenous arginine flux in the fasting state. 8 Under conditions of stress or disease states, rates of endogenous arginine synthesis may be insufficient to meet the body's demands.9,10 Utilization of arginine by other enzymes, particularly arginase, may also decrease its availability for reaction with NOS. Arginases, which exist in two isoforms, convert arginine to ornithine and urea and compete with NOS for arginine.8,11 Arginase I is expressed in the liver and contributes to the majority of the body's total arginase activity, whereas arginase II is present in most tissues, including the lungs. 12 Increased arginase I expression has been implicated in the development of PAH in sickle cell disease, 13 while increased arginase II expression has been found in pulmonary endothelial cells of patients with World Health Organization (WHO) class I pulmonary hypertension. 12 Endogenous inhibitors of NOS may also affect NO production. The dimethylarginines, including asymmetric dimethylarginine (ADMA) and symmetric dimethylarginine (SDMA), are synthesized by the methylation of arginine residues in protein, which are released by proteolysis. 14 ADMA is the most potent endogenous inhibitor of NOS and competes with L-arginine for active sites on endothelial NOS (eNOS). Both ADMA and SDMA interfere with y+-mediated transport of arginine into cells. 14
The relationship among arginases, dimethylarginases, and eNOS is complex and provides a potential explanation for the “arginine paradox.” The arginine paradox describes the phenomenon that increased extracellular arginine is needed to produce maximal NO production despite the fact that both extracellular and intracellular concentrations of arginine far exceed the theoretical Km of the enzyme. 15 The ratios of arginine to its catabolic products and to the dimethylarginines may better reflect systemic arginine availability, and thus arginine supplementation under conditions where these ratios are low could improve arginine availability for NO production (Fig. 1).
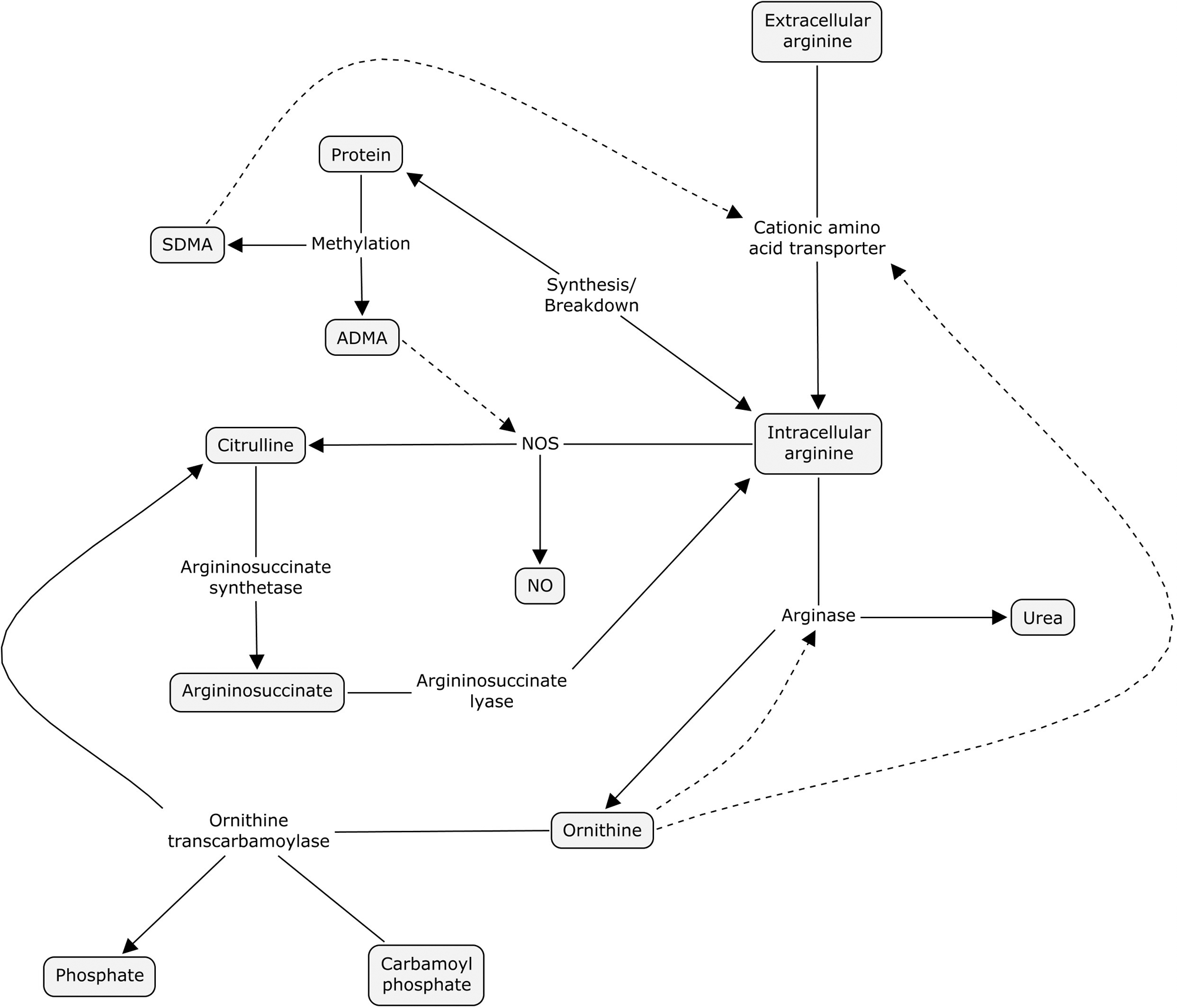
Arginine metabolic pathways. Extracellular arginine is transported intracellularly by y+ cationic amino acid transporters. Intracellular arginine is derived from protein breakdown and de novo synthesis from citrulline via argininosuccinate. Nitric oxide synthases (NOS) converts arginine to NO and citrulline. Arginase hydrolyzes arginine to ornithine and urea. Endogenous methyl-arginines (asymmetric dimethylarginine [ADMA], symmetric dimethylarginine [SDMA]) are produced after proteolysis from methylated arginine residues in protein and inhibit NOS as well as arginine transport. Dotted lines indicate inhibition.
Despite the evidence supporting derangement of arginine and NO metabolism as an important mechanism underlying the development of PAH, in vivo studies comprehensively dissecting the relevant metabolic pathways are lacking. One study found decreased whole-body NO production, as measured by conversion of 15N2-arginine to urinary 15N-nitrite and 15N-nitrate, in 4 patients with primary pulmonary hypertension. 16 We hypothesized that PAH patients with the lowest arginine availability, secondary to increased arginase activity and/or inadequate de novo arginine synthesis, might have a slower rate of NO synthesis and greater PVR. We tested this hypothesis by using stable-isotope tracer techniques and biochemical methods to determine the metabolic fates of arginine in PAH patients in comparison with healthy controls and relative to clinical parameters of pulmonary hypertension.
METHODS
Subjects
Nine patients with group I pulmonary hypertension as defined by the WHO 17 were enrolled from the pulmonary clinics of Baylor College of Medicine and the Cleveland Clinic. For comparison, 10 healthy volunteers without chronic illnesses matched as closely as possible for age and sex were also enrolled. All participants provided written informed consent, and the study protocol and consents were approved by the institutional review boards at both institutions. All PAH patients were on stable medications and had undergone right heart catheterization as part of their standard care. On the day of isotope infusion, a subgroup of participants also had fractional exhaled NO (FeNO) measured by an online method at a constant flow rate of 50 mL s−1 according to the standards published by the American Thoracic Society. 18
Isotope infusions
Tracer infusions were performed in all subjects at the Clinical Research Unit of the Cleveland Clinic, the General Clinical Research Center of Baylor College of Medicine, and the Metabolic Research Unit of the Children's Nutrition Research Center. Sterile solutions of guanidino-15N2-arginine, 13C,2H4-citrulline, 15N-citrulline, 13C-urea, and 15N2-ornithine (Cambridge Isotope Laboratories, Tewks-bury, MA) were prepared using strict aseptic techniques and were tested for sterility and lack of pyrogens prior to infusion.
After an 8-hour overnight fast, all participants had an intravenous catheter placed in the antecubital vein for isotope infusions and in a hand vein of the contralateral arm for blood sampling. The hand was heated to arterialize blood samples.19,20 After a baseline blood sample was obtained, primed, continuous, intravenous infusions of 15N2-arginine (prime = 8 μmol kg−1, infusion = 8 μmol kg−1 h−1), 13C,2H4-citrulline (prime = 1.5 μmol kg−1, infusion = 1 μmol kg−1 h−1), 13C-urea (prime = 80 μmol kg−1, infusion = 8 μmol kg−1 h−1), and 15N2-ornithine (prime = 2 μmol kg−1, infusion = 2 μmol kg−1 h−1) were started and maintained for 6 hours. In addition, a prime of 15N-citrulline (0.16 μmol kg−1) was administered to prime the secondary 15N-citrulline pool. Blood samples were obtained every 30 minutes between 4.5 and 6 hours (4 samples) of the isotope infusions.
Sample analyses
Blood samples were drawn into prechilled tubes containing sodium heparin or EDTA. Tubes were centrifuged immediately at 4°C, and the plasma was separated and stored immediately at −70°C for later analysis.
The plasma isotope enrichments of arginine, citrulline, and ornithine were measured by tandem liquid chromatography mass spectrometry. The amino acids were converted into their 5-dimethylamino-1-napthalene sulfonamide derivatives, and ions were analyzed by selected reaction monitoring on a triple-quadrupole mass spectrometer. The transitions observed were precursor ion
Plasma amino acid concentrations were measured by ultraperformance liquid chromatography using precolumn derivitization with 6-amino-quinolyl-N-hydroxysuccinimidyl carbamate. Plasma concentrations of ADMA and NO metabolites were determined by stable isotope dilution with 2H7-ADMA and Na15NO3 as internal standards, as described elsewhere.21,22
Arginase activity
Plasma arginase activity was determined with aliquots of 50 μL of plasma using a modification of the radioisotope method. 23 Arginase activity was measured by the conversion of N2-arginine to N2-urea. Concentrations of 15N2-urea at baseline and after reaction were measured using isotope dilution with 13C, 15N2-arginine as an internal standard. The unit of arginase activity is defined as microliters of urea per minute.
Calculations
The rate of appearance or total flux (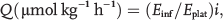
Under steady state conditions, the rate of appearance of arginine equals the rate of disappearance. Therefore, arginine clearance is
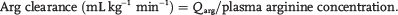
Because arginine is converted to citrulline and NO at a 1:1 ratio, the rate of conversion of N2-arginine to 15N-citrulline is an index of NO synthesis. Similarly, the conversion of 13C, 2H4-citrulline to 13C, 2H 4-argimne is a measure of de novo arginine synthesis, and the conversion of N2-arginine to N2-urea is an index of arginase activity. The conversion of precursor to product is calculated by
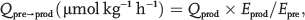
The fractional synthesis rate (FSR) of NO was calculated according to the precursor-product equation as described elsewhere,
21
using the following equation:
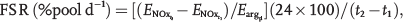
Additional cohort
A second cohort of 9 healthy individuals and 9 patients diagnosed with class I PAH between the ages of 18 and 60 years was used to explore the relationship between plasma arginase activity and cardiac function. Plasma arginase activity and plasma amino acid concentrations were measured as described above. Echocardiograms were performed on the PAH patients by a single experienced sonographer, and multiple parameters were measured as described elsewhere. 24
Statistical analysis
Continuous variables were summarized by group as means ± SE, unless otherwise indicated. Normality of data was confirmed using the Kolmogorov-Smirnov test. Differences between groups of subjects were assessed by the unpaired Student's
RESULTS
Subject characteristics
PAH patients and controls were similar in age, sex, ethnicity, weight, and body mass index (all
Study population
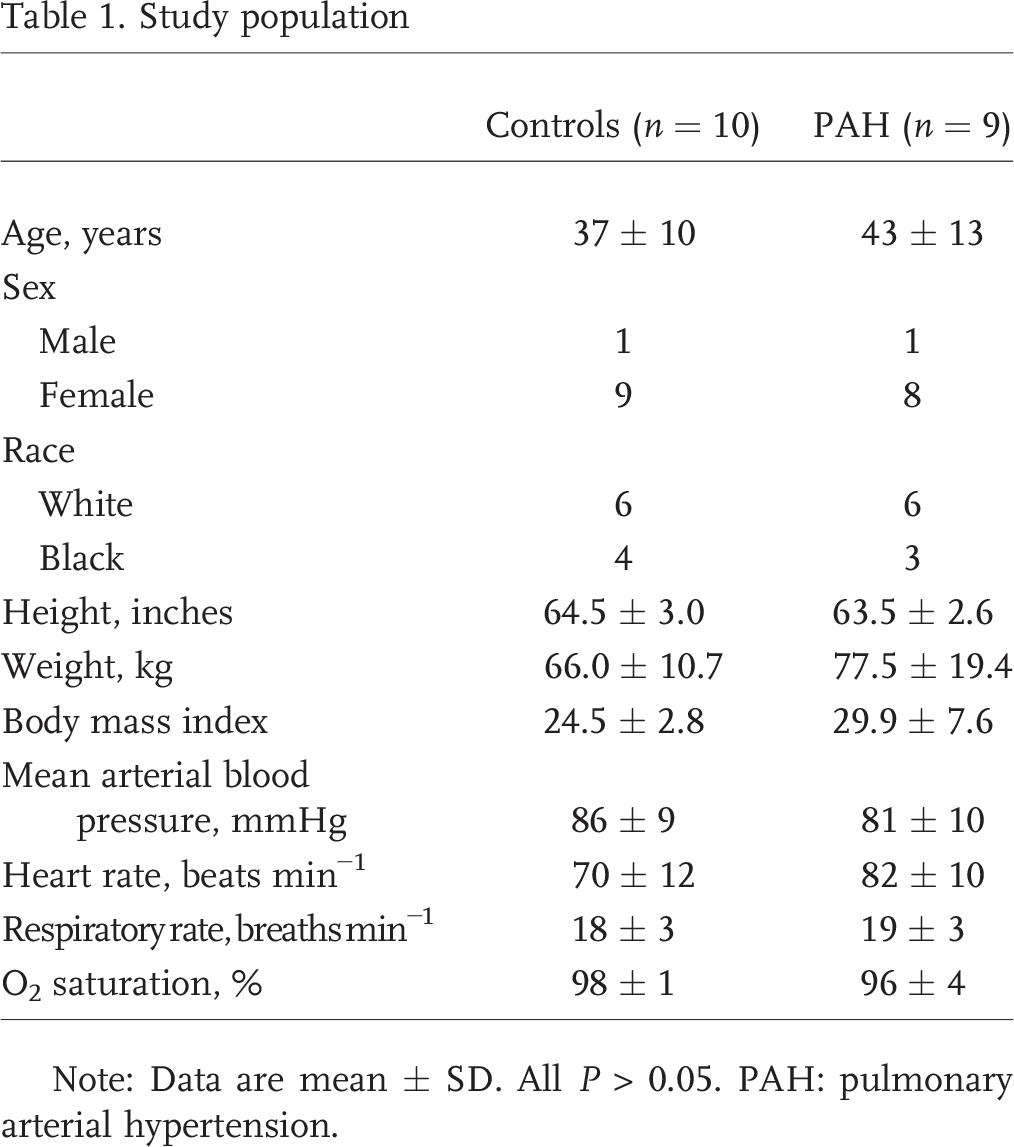
Note: Data are mean ± SD. All
Clinical characteristics of pulmonary arterial hypertension (PAH) patients
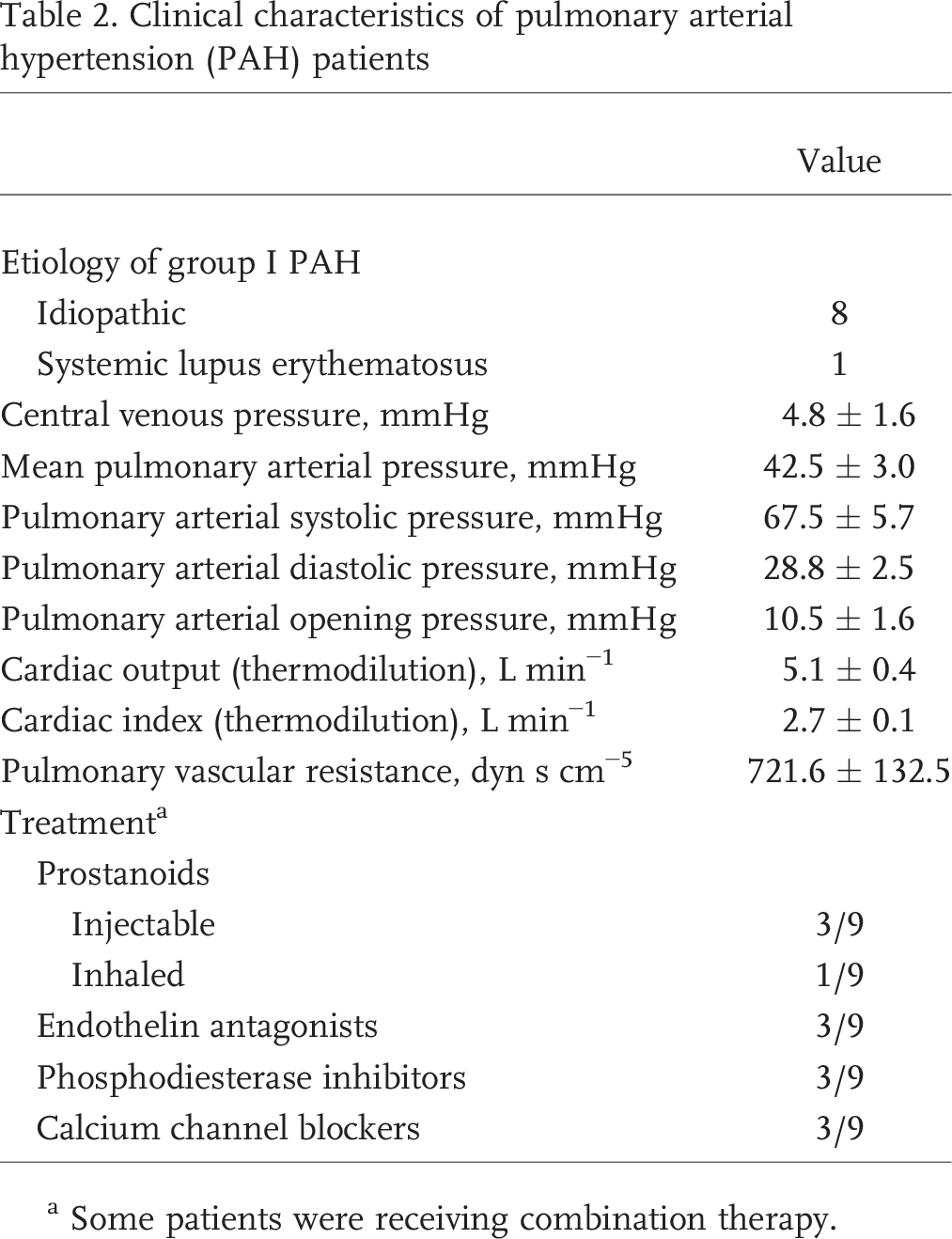
Some patients were receiving combination therapy.
Arginine, citrulline, and NO kinetics
Arginine and citrulline kinetics and plasma concentrations were similar among PAH patients and controls (Table 3). There was no difference in arginine or citrulline flux between controls and PAH patients. De novo arginine production and conversion of 15N2-arginine to 15N-citrulline, an index of NO synthesis (Fig. 2), were also similar between PAH patients and controls. Citrulline flux was negatively correlated with mean PAP (mPAP; Table 4), although this did not meet statistical significance. There was also a significant negative correlation between de novo arginine production and cardiac output (Table 4).
Parameters of arginine production
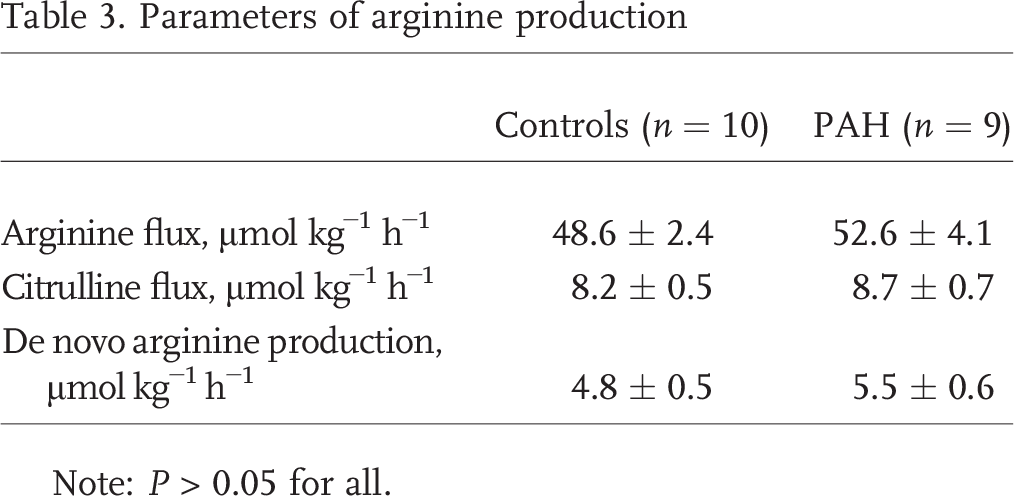
Note:
Significant correlations between metabolic/kinetic and clinical parameters
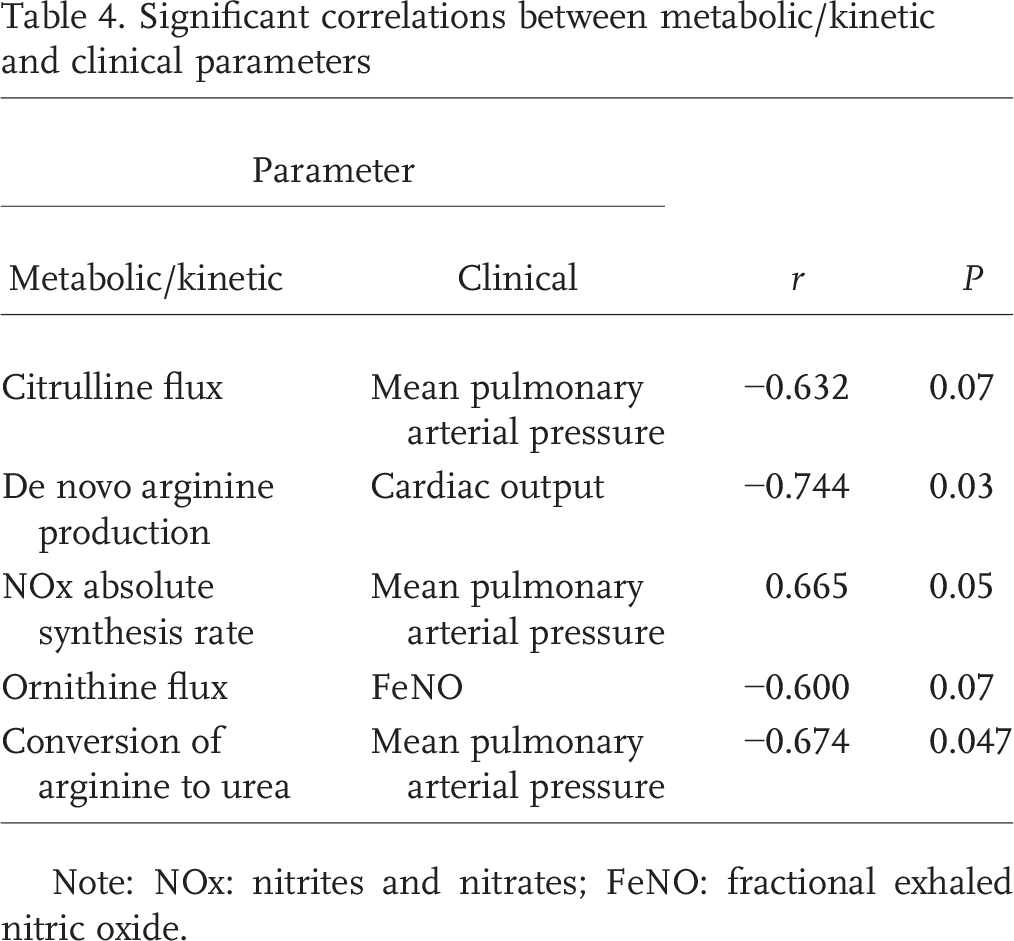
Note: NOx: nitrites and nitrates; FeNO: fractional exhaled nitric oxide.
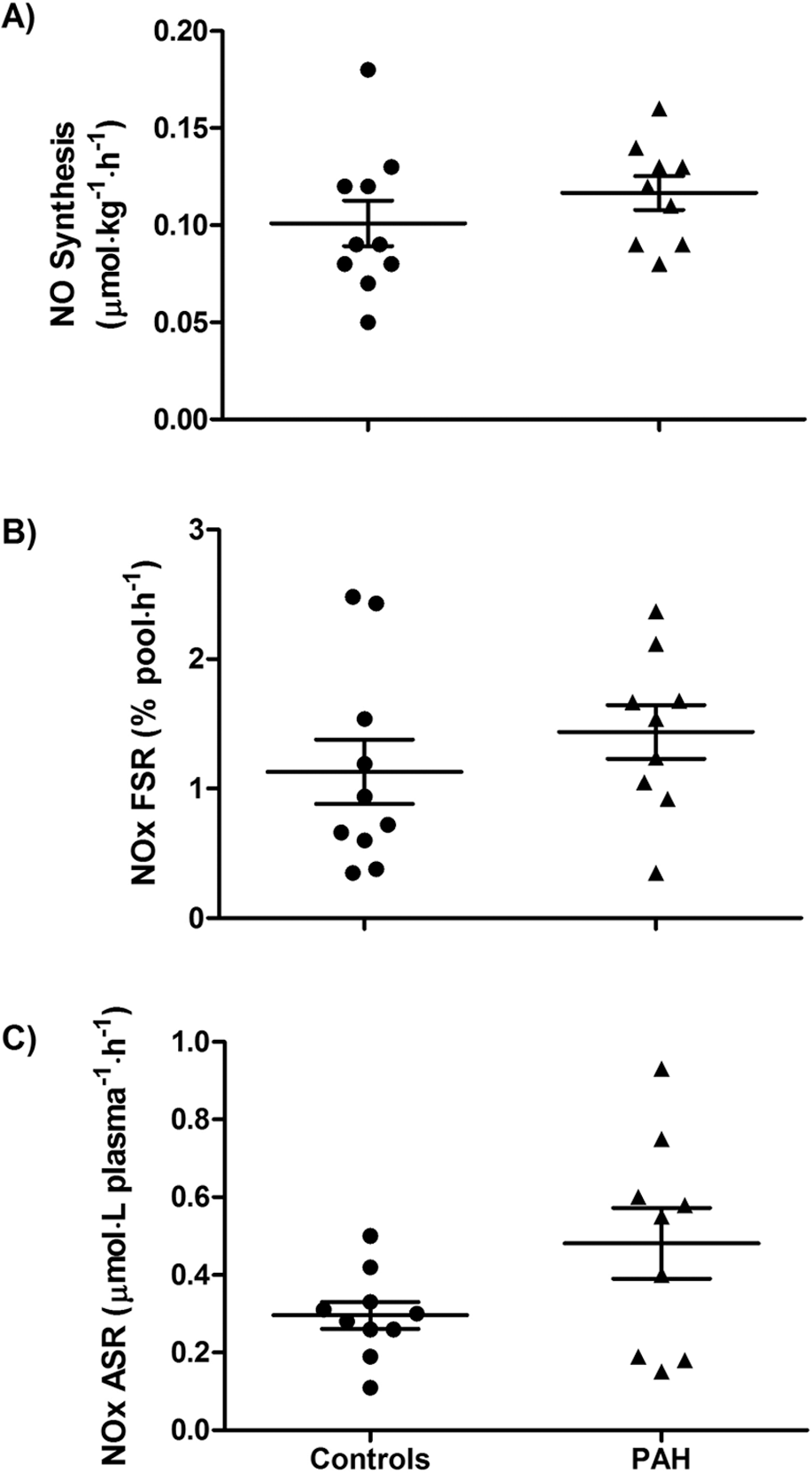
Parameters of nitric oxide (NO) synthesis. The conversion of 15N2-arginine to 15N-citrulline, an index of NO synthesis, was similar in pulmonary arterial hypertension (PAH) patients and controls (
Plasma NOx concentration was not different between PAH patients and controls (PAH = 32.0 ± 4.6 μM, controls = 30.8 ± 4.0 μM;
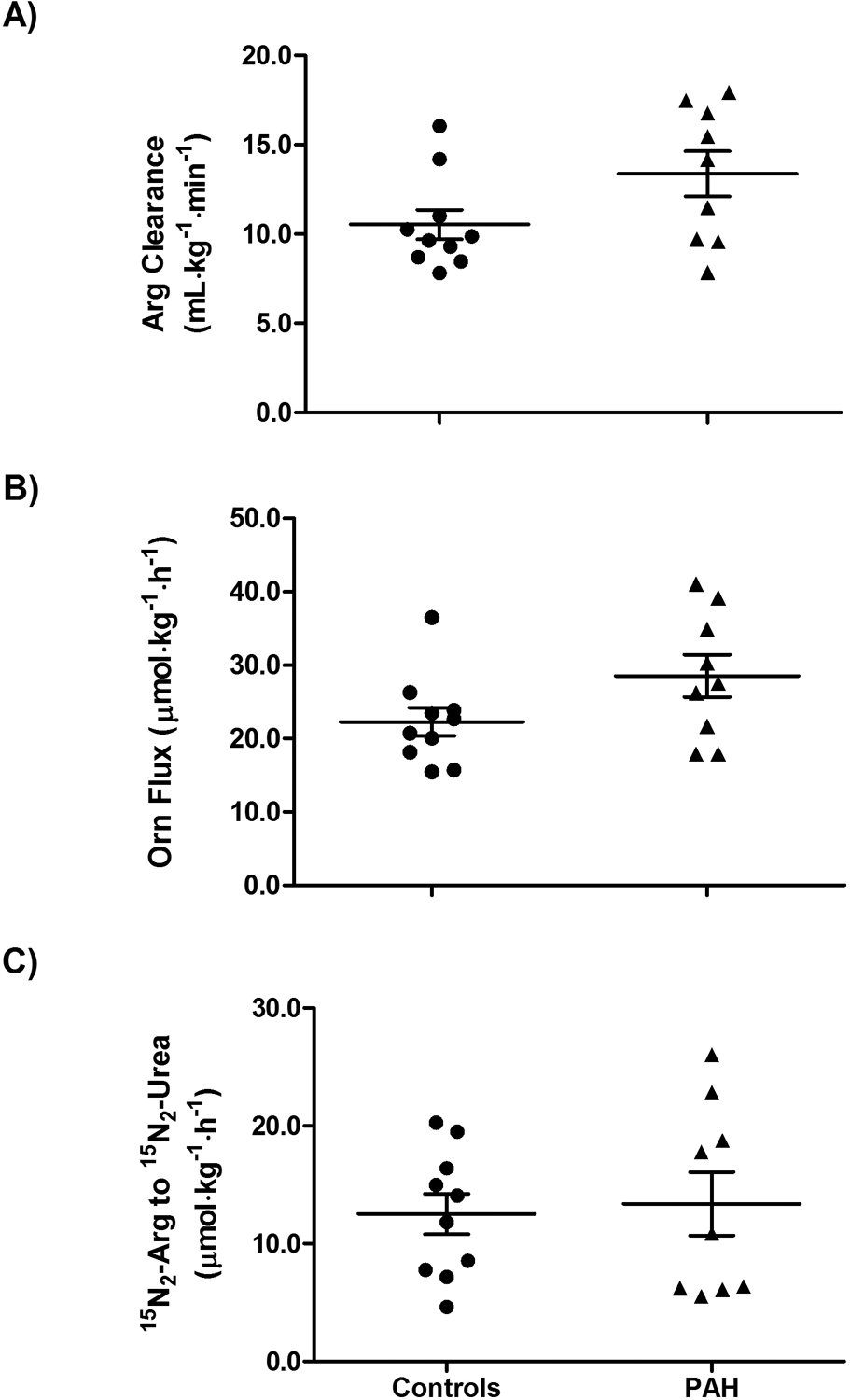
Arginine clearance and catabolism. Compared with controls, pulmonary arterial hypertension (PAH) patients had a tendency toward greater arginine clearance (
FeNO
In the subgroup of participants who underwent FeNO testing, there was no difference in FeNO between PAH patients and controls (PAH = 14.4 ± 2.8 ppb, controls = 17.7 ± 4.7 ppb;
The arginine metabolome: arginine, ornithine, citrulline, and ADMA
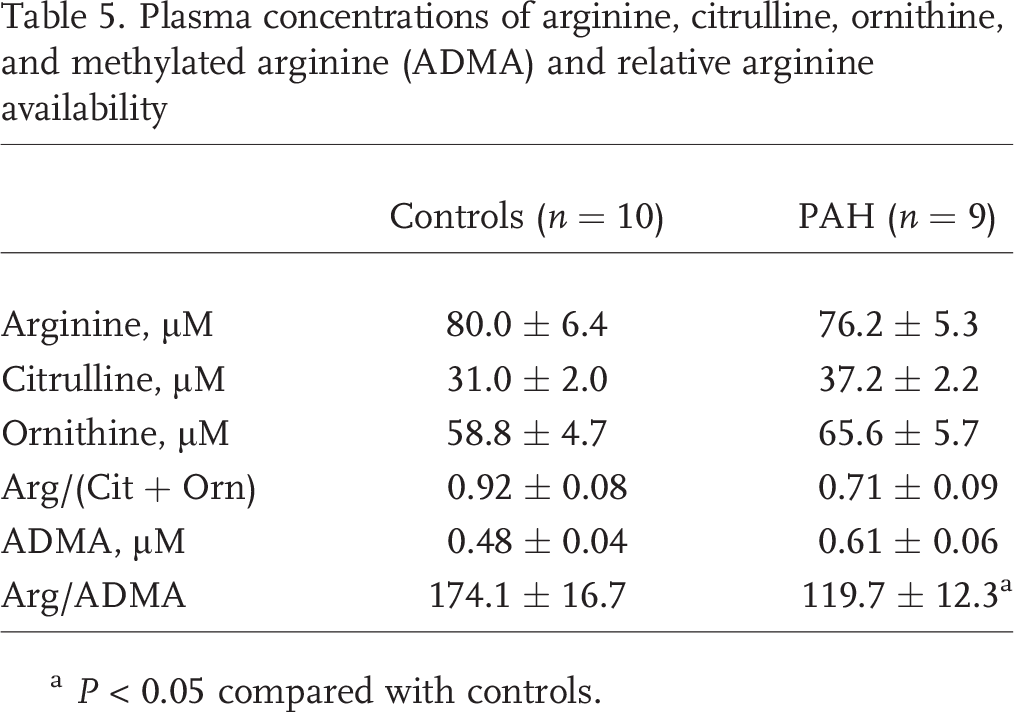
Plasma concentrations of arginine, citrulline, and ornithine were similar among healthy controls and PAH patients (Table 5). When arginine availability was evaluated using the ratio of arginine to products generated from catabolism (Arg/[Cit + Orn]), arginine availability was 30% lower in PAH patients, although the difference was not statistically significant (
Plasma concentrations of arginine, citrulline, ornithine, and methylated arginine (ADMA) and relative arginine availability
Arginase activity
Because ornithine and urea are products of arginine hydrolysis by arginase, their rates of production reflect arginase activity. The rate of conversion of 15N2-arginine to 15N2-urea is also an index of arginase activity. There was a trend toward increased ornithine flux in PAH patients (
There was a strong correlation between the different measurements of arginase pathway activity (Fig. 4). There was a significant negative correlation between conversion of arginine to urea and mPAP (Table 4). On further inspection of the data, the conversion of arginine to urea was clearly clustered into two groups among PAH patients: 4 patients had low conversion (less than mean −1 SD of controls), and 5 patients had high conversion, that is, low and high arginase activity.
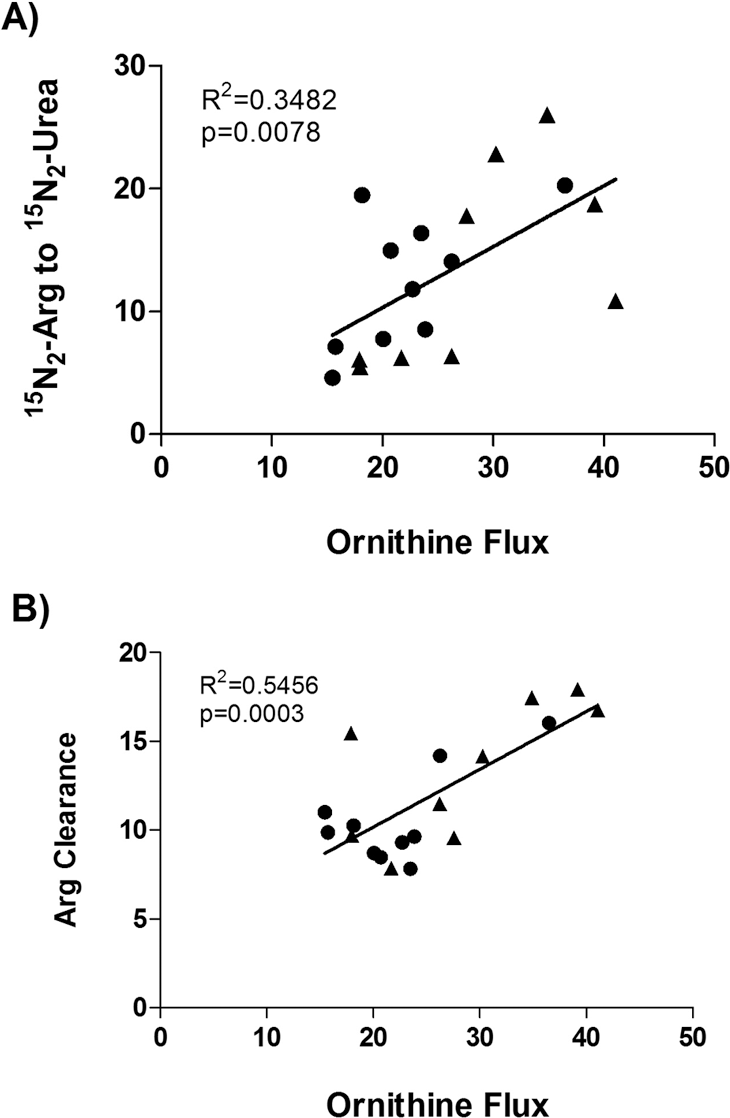
Correlations among measures of arginine catabolism. Arginine clearance and catabolism are strongly related to measures of ornithine flux. Circles indicate controls, and triangles indicate pulmonary arterial hypertension patients.
Hemodynamic and kinetic parameters by arginase activity
Because PAH patients appeared to be divided into those with low and those with high arginase activity, hemodynamic and kinetic parameters were analyzed in these two endotypes. PAH patients with high arginase activity had higher arginine flux, lower NO synthesis, lower ASR of NOx, and lower plasma concentration of ADMA (Fig. 5). There was no difference in PAPs (mean [
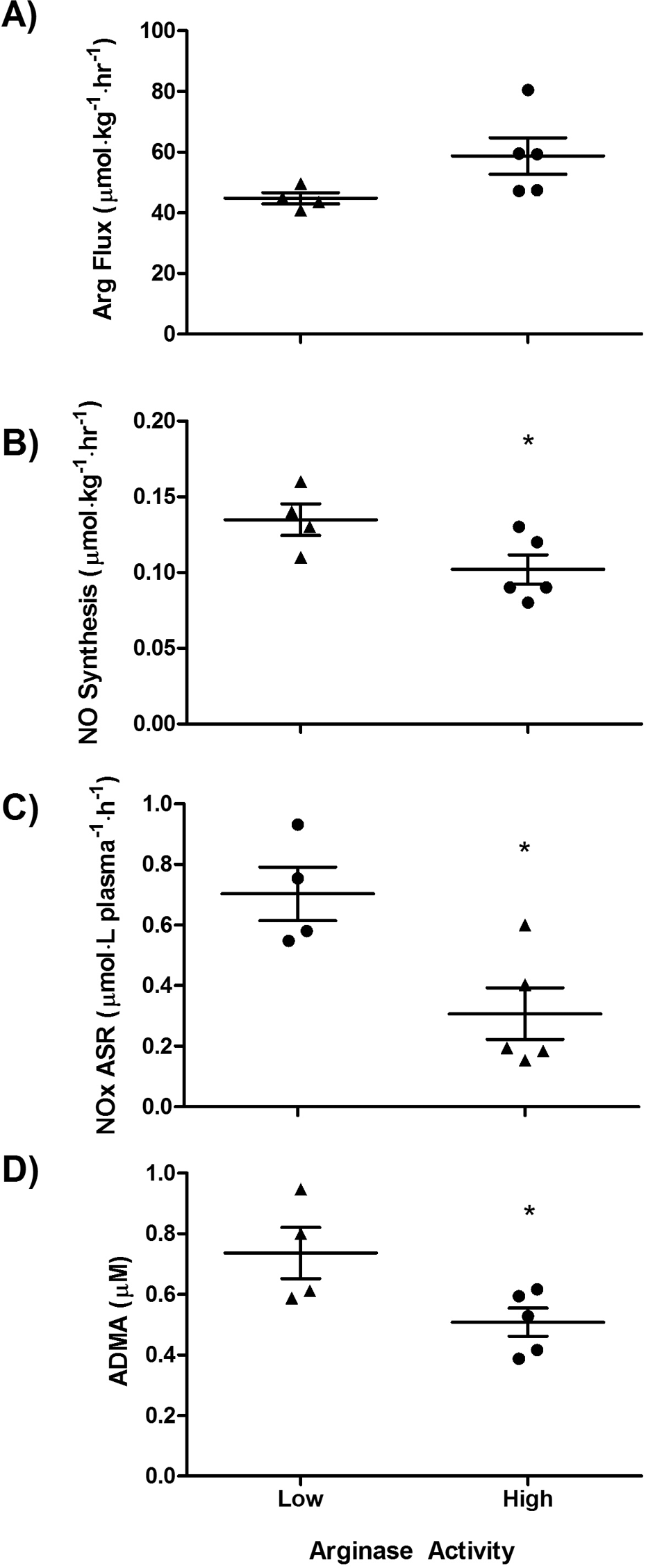
Arginine metabolic endotypes of pulmonary arterial hypertension (PAH). A subgroup of PAH patients with high arginase activity had greater arginine flux (
Additional cohort
In the additional cohort of 9 PAH patients and 9 controls, there was a trend toward higher plasma arginase activity in PAH patients than in controls (PAH = 0.78 ± 0.05 μmol urea min−1, controls = 0.65 ± 0.05 μmol urea min−1;
DISCUSSION
There is substantial evidence that NO deficiency is an important mechanism underlying the development of PAH, although the specific metabolic derangements contributing to decreased NO bioavailability are not completely understood. 25 This is the first study, to our knowledge, to use stable isotope tracer techniques to characterize the production and catabolism of arginine and its relationship to NO synthesis in pulmonary hypertension, thereby increasing our knowledge of the role played by arginine metabolism in the disease process. The results demonstrate that there is decreased arginine bioavailability in patients with pulmonary hypertension as a result of increased catabolism by arginase and higher concentrations of ADMA. The global arginine bioavailability ratio (the ratio of arginine to ornithine plus citrulline) has been shown to be associated with mortality in patients with sickle cell disease and pulmonary hypertension. 13 This study similarly found decreased arginine bioavailability measured by low global arginine availability ratio in two separate cohorts of PAH patients compared with controls but also substantiates this finding with kinetic measurements showing faster arginine clearance and ornithine production in PAH patients. Furthermore, there is decreased NO synthesis in the subgroup of patients with high compared with low arginase activity, demonstrating that arginase activity regulates arginine availability for NOS.
Alterations in arginase (specifically arginase II) expression and activity have been implicated in the pathophysiology of pulmonary hypertension. Rats with pulmonary hypertension induced by monocrotaline have significantly accelerated arginase activity in their pulmonary arterial endothelial cells, along with decreased NOS activity and expression. 26 Similarly, human endothelial cells isolated from PAH patients have increased expression of arginase II and lower NO production than endothelial cells from controls. 12 On a systemic level, these PAH patients also had higher serum arginase activity and a lower arginine-to-ornithine ratio. This is the first study to confirm these findings in vivo. A study by Castillo et al. 27 first established the use of stable isotope techniques to characterize in vivo arginine catabolism and demonstrated the importance of arginine degradation in the regulation of arginine availability. When healthy adult men were given an arginine-free diet, there was a significant decline in ornithine flux as well as arginine conversion to ornithine in an effort to conserve body arginine. A study by the same authors in burn patients subsequently found that plasma arginine to ornithine conversion constitutes only 7% of total urea production, 28 indicating that the majority of urea flux is derived from turnover of arginine via the urea cycle in the liver rather than plasma conversion of arginine to urea. Ornithine flux, on the other hand, is a measure of non-urea-cycle arginine metabolism and serves as a surrogate for arginine turnover and catabolism, since changes in arginine turnover and oxidation were paralleled by changes in ornithine flux. In this study, we found that PAH patients and controls had similar urea flux, and, in line with the prior tracer studies, plasma conversion of arginine to urea constituted 7.8% of urea production in the healthy controls. However, the PAH patients had a tendency toward increased ornithine flux and arginine clearance, two parameters that were highly correlated. Furthermore, ornithine flux was also significantly correlated with in vitro plasma arginase activity, and findings of increased in vitro arginase activity were confirmed in a second cohort. Taken together, these results indicate that there is increased catabolism of arginine by arginase in PAH. The finding that ornithine but not urea flux differs between PAH patients and controls suggests that this is specifically due to accelerated activity of arginase II.
Increased arginase catabolism is believed to be important in PAH due to decreased arginine availability for reaction with NOS and thus slower NO synthesis. However, contrary to our hypothesis, despite increased arginase activity PAH patients as a whole in this study did not have slower NO production; in fact, the results suggest that NO production may be increased in PAH patients. One potential explanation is that arginase contributes to the development of PAH via NO-independent pathways. For example, in a hypoxia model of pulmonary hypertension, arginase II was responsible for proliferation of pulmonary arterial smooth muscle cells, possibly via ornithine metabolism to polyamines, which are a key component needed for cellular growth. 29 In addition, upregulation of arginase expression increases production of proline, 30 an important precursor for synthesis of collagen. Thus, increased ornithine production in PAH patients could help sustain vascular remodeling via increased metabolism to polyamine and proline, although this was not directly measured in this study. We also found that there was a subgroup of patients with high arginase activity as measured by arginine to urea conversion, and, as predicted, this group had slower NO synthesis and faster arginine production compared with the group with low arginase activity. At the same time, there was no difference in de novo arginine production between the two groups. These findings suggest that, in PAH patients with high arginine catabolism by arginase, increased arginine production results from increased arginine release from protein breakdown and that there is an inadequate response in de novo arginine synthesis to compensate for increased arginine catabolism. This may be because de novo arginine synthesis from citrulline does not readily adapt to changes in arginine availability; in support of this, healthy men fed arginine-free diets also did not have compensatory increase in de novo arginine production. 31 In this study, citrulline plasma concentration and flux were negatively correlated with PAPs and de novo arginine production was negatively correlated with cardiac output, suggesting that citrulline supply—and subsequently its conversion to arginine—are important for improving hemodynamic parameters, likely via NO production. Therefore, citrulline may be a potential treatment for PAH. Citrulline supplementation has been shown to be superior to arginine supplementation for increasing NO synthesis in patients with mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes syndrome as a result of marked increase in de novo arginine synthesis. 32 Oral citrulline supplementation raised arginine plasma concentrations in patients with sickle cell disease 33 and increased de novo arginine production and NO synthesis in piglets with hypoxia-induced PAH, thereby improving pulmonary arterial pressure, pulmonary vascular resistance, and NO production. 34 Further studies are needed to determine the efficacy of citrulline supplementation in PAH.
PAH patients with high arginase activity also had lower plasma ADMA concentrations than patients with low arginase activity. The lungs are known to be a major source of ADMA, 35 and increased ADMA has been previously reported in patients with pulmonary hypertension12,36,37 and is an independent predictor of mortality. 37 Elevated ADMA concentrations are thought to result from reduced activity of dimethylaminohydrolase, the enzyme responsible for ADMA clearance. 38 Although PAH patients had increased ADMA and a significant reduction in the arginine/ADMA ratio in the current study, this was not accompanied by the expected decrease in NO synthesis. In fact, whole-body NO synthesis was increased, and the rate of NO production as measured by conversion of 15N2-arginine to 15N-citrulline was correlated with exhaled NO, demonstrating an association between whole-body and pulmonary NO production. However, although the rate of NO production may be accelerated, not all the NO formed may participate in physiological processes. In some cases where eNOS expression is unexpectedly increased, it is likely uncoupled, leading to superoxide production instead of NO, and the NO produced may then be inactivated by scavenging reactions. 25 In addition, the majority of PAH patients in this study were receiving treatment for PAH, and although none of the medications act directly on NO synthesis, they may have indirectly affected NO production. In fact, administration of prostacyclin to PAH patients has been shown to increase exhaled NO, and successful treatment of PAH has led to increases in FeNO. 39 Finally, NO production was measured in the whole body by stable isotopes, which may not account for compartmentalization intracellularly. 40 Nevertheless, the important finding that ADMA was significantly higher in the patients with low arginase activity suggests that different mechanisms may be responsible for alterations in arginine metabolism in different patients.
This study has several limitations. It was conducted in a small number of patients with group I PAH and therefore may not be applicable to all types of pulmonary hypertension. Patients were studied while receiving PAH treatment, as it would not have been feasible to stop the medications for the study, and therefore potential effects of therapy on arginine metabolism and NO synthesis could not be excluded. In addition, lung-specific in vivo measurements could not be obtained, as this would have required invasive procedures and sampling of the pulmonary circulation. However, this study provides a rationale for future potential therapies for PAH, including citrulline supplementation, inhibition of arginase II, and enhancing degradation of ADMA.
Footnotes
ACKNOWLEDGMENTS
We are grateful to the nursing staff of the Clinical Research Unit, the General Clinical Research Center, and the Metabolic Research Unit for care of the participants and to Grace Tang for assistance in laboratory analyses.
