Abstract
The pathogenesis of pulmonary arterial hypertension (PAH) exhibits many neoplastic-like features. Cowden syndrome is a difficult-to-recognize heritable cancer syndrome caused by a germline mutation in the phosphatase-and-tensin homolog deleted on the chromosome 10 (PTEN) gene. PTEN regulation has been implicated in cancer development and, more recently, PAH pathogenesis. Here we report a case of PAH in a patient with Cowden syndrome and the response to pulmonary vasodilators.
CASE DESCRIPTION
A 54-year-old woman was referred to the pulmonary clinic for evaluation of progressive dyspnea over the last 6months. She reported dyspnea after walking two blocks or going up a flight of stairs. She denied cough, wheezing, hemoptysis, chest pain, orthopnea, paroxysmal nocturnal dyspnea, palpitations, and leg swelling.
She was diagnosed with Cowden syndrome (CS) 4 years ago, molecularly confirmed by identifying a germline heterozygous mutation in the PTEN gene. Her CS was characterized by acral keratosis of the torso; abdominal and pelvic arteriovenous malformations (AVMs) treated with coil embolization and surgery; breast cancer treated with lumpectomy, radiotherapy, and tamoxifen; and colon cancer treated with partial colectomy and adjuvant chemotherapy. She was a retired social worker and a never-smoker. She denied using anorexigens. There was no family history of CS or pulmonary hypertension (PH).
Her blood pressure was 111/61 mmHg; heart rate, 66 beats/min; oxygen saturation, 96% on ambient air; and body mass index (BMI), 28. She had no crackles, wheezing, clubbing, cyanosis, or skin thickening. Her abdomen was without organomegaly. There was a 3/4 systolic murmur over the left sternal border. Her hemoglobin was normal. Pulmonary function testing showed forced expiratory volume in first second of expiration (FEV1) of 54%, forced vital capacity (FVC) of 58%, FEV1/FVC of 72%, total lung capacity of 88%, and carbon monoxide diffusing capacity of 92% predicted. Of note, there was early termination of expiration and no expiratory plateau. A recent computed tomography chest angiogram showed no evidence of pulmonary embolism, parenchymal lung disease, or lymphadenopathy. Transthoracic echocardiogram showed normal left ventricular function, a severely dilated right ventricle (RV) with moderately to severely decreased function, pulmonic valve regurgitation, and an estimated right ventricular systolic pressure (RVSP) of 115 mmHg. Compared with an echocardiogram performed ?6 months ago, there was worsening RV dilation and function and increased RVSP (previously at 55 mmHg).
Right heart catheterization (RHC) showed a mean right atrial pressure of 10 mmHg, RV pressure of 84/3 mmHg, mean pulmonary arterial pressure (mPAP) of 47 mmHg, mean pulmonary capillary wedge pressure (PCWP) of 10 mmHg, cardiac output (CO) of 7.72 L/min, and pulmonary vascular resistance (PVR) of ?5 Wood units. Interestingly, an RHC performed 7 years ago showed mPAP of 45 mmHg, PCWP of 18 mmHg, CO of 13.25 L/min, and PVR of ?2 Wood units, consistent with PH secondary to a high-output state. Since that time, she had undergone multiple successful coil embolizations to treat her AVMs.
She was diagnosed with PAH and started on tadalafil. On follow-up, she reported improvement in both dyspnea and exercise capacity. Echocardiogram showed decreased RVSP and improvement in RV function.
DISCUSSION
PH is defined by mPAP above 25 mmHg at rest and is classified into five groups according to pathophysiologic and therapeutic characteristics. 1 PAH is a distinct subgroup of PH that comprises idiopathic PAH, heritable forms, PAH associated with connective tissue disease, congenital heart disease, portal hypertension, human immunodeficiency virus infection, and some other conditions. 1 It affects the pulmonary microcirculation and is characterized by arterial obstruction due to vascular proliferation and remodeling. Plexiform lesions are the hallmark of PAH and exhibit neoplastic-like features. 2 In fact, the pulmonary vascular cells in PAH exhibit a switch in metabolic phenotype characterized by low numbers of mitochondria and decreased oxygen consumption, significantly higher glycolytic rate, apoptosis resistance, and increased cell proliferation. These metabolic changes are associated with pathologic expression of hypoxia-inducible factor 1α (HIF-1α). Studies have shown higher levels of circulating HIFinducible factors, including erythropoietin, stem cell factor, and hepatocyte growth factor (HGF) in PAH patients compared with controls. 3 In addition, pulmonary arterial endothelial cells (PAECs) from human PAH lungs produce greater HGF and stromal-derived factor 1α (SDF-1α) than control PAECs. 3
Moreover, heterozygous mutations in bone morphogenetic protein (BMP) receptor type 2 (BMPR2), a growth factor belonging to the transforming growth factor β superfamily, have been reported in ?70% of patients with hereditary PAH and in ?25% of patients with sporadic idiopathic PAH. 2 BMPs and their receptors play a role in cell growth control, even in cancers. 2 Downstream signaling of BMPR2 includes the PI3K-AKT-mTOR pathway, which, when activated, leads to cellular proliferation. This pathway is negatively regulated by PTEN, a major tumor-suppressor gene. 4 PTEN mutations are responsible for many of the sporadic cancer syndromes, including CS. 4 They lead to uncontrolled cell growth and are associated with endothelial cell hyperproliferation and impaired vascular remodeling. 5 Recently, data from animal studies show a relationship between PTEN and cardiopulmonary vasculature remodeling, as PTEN activation was shown to regulate cardiomyocyte hypertrophy and survival as well as pulmonary smooth muscle cell proliferation and survival. 6 Inactivation of PTEN, on the other hand, was associated with AKT-dependent upregulation of HIF-1α. It promoted medial smooth muscle cell hyperplasia, vascular remodeling, and histopathology consistent with PH through increased HIF-1α-mediated production of SDF-1α and recruitment of hematopoietic progenitor cells to the vasculature.5,6
CS is a difficult-to-recognize heritable disorder, of which 25%–85% have been shown to be caused by germline PTEN mutations.7,8 Clinically, CS is characterized bymucocutaneous lesions (oral papillomas, trichilemmonas, acral keratoses, and papules; Fig. 1) and a predisposition to the development ofmalignancy in many tissues, notably involving the breast, thyroid, and endometrium(Table 1). 4 Vascular anomalies can be present in ?50% of CS patients, the majority of which are fast flowing and include AVMs. 7 To the best of our knowledge, the only other case of PAH associated with CS was reported in a patient with exposure to the anorexigen dexfenfluramine, a potential trigger of PAH. 10 Our case report is the first to describe PAH in a patient with CS without other predisposing factors. Our patient had no underlying left heart disease, intrinsic lung diseases, or chronic thromboembolism. Her spirometry showed diminished FEV1 and FVC, although in the setting of a poor expiratory effort and elevated BMI. Her initial RHC showed elevated mPAP with high CO and normal PVR consistent with a high-output state. She since underwent multiple embolizations for AVM treatment; however, follow-up echocardiograms did not show any improvement in RV function and pressures after embolization. Her repeat RHC showed elevated mPAP, normal CO, and an elevated PVR consistent with a diagnosis of PAH. While it is possible that a long-standing high-output state secondary to AVMs could have led to PAH, we propose that germline PTEN mutations may be a predisposing factor for the development of PAH, plausibly mediated via HIF-1α, a shared proangiogenic pathway.
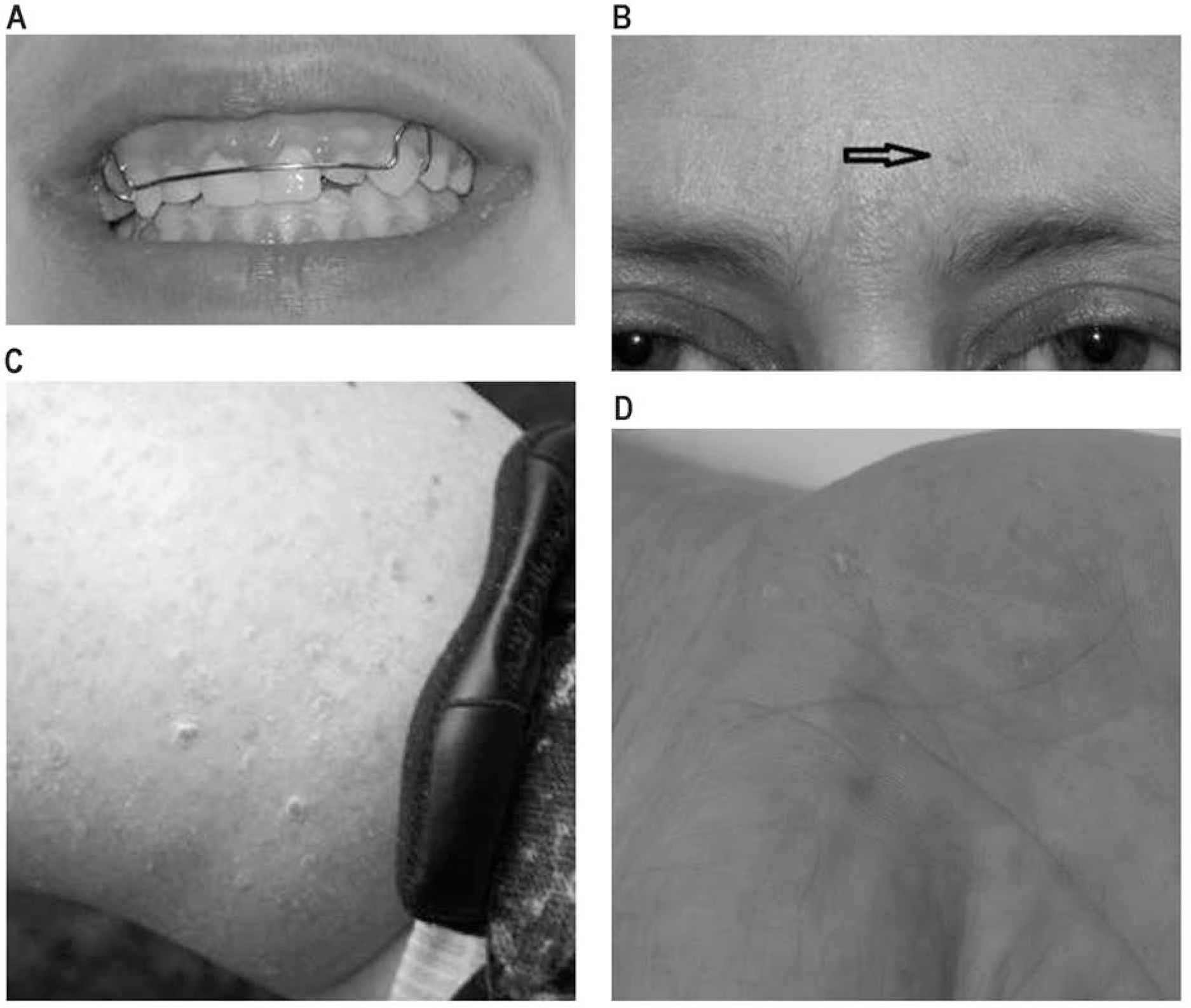
Mucocutaneous features of Cowden syndrome. A, Gum papillomas. B, Biopsy-proven trichilemmoma. C, Acral keratosis. D, Palmar pits/papules. A color version of this figure is available online.
Features of three syndromes associated with pulmonary arterial hypertension (PAH)

Note: HHT: hereditary hemorrhagic telangiectasia; ENG: endoglin; ACVRL1: activin receptor-like kinase 1; SMAD4: SMAD family member 4; PTEN: phosphatase-and-tensin homolog deleted on chromosome 10; BMPR1A: bone morphogenetic protein receptor 1A; SDHB/D: succinate dehydrogenase B/D; AKT1: v-akt murine thymoma viral oncogene homolog 1; PIK3CA: phosphatidylinositol 3-kinase catalytic subunit A; KLLN: killin; BMPR2: bone morphogenetic protein receptor 2; AVM: ateriovenous malformation; AVF: arteriovenous fistula; PH: pulmonary hypertension; GI: gastrointestinal.
