Abstract
The functional and prognostic significance of pulmonary hypertension (PH) is well established, yet our understanding of circulating peptides and metabolites that might mark or contribute to various forms of PH remains limited. Metabolites are the end result of all the regulatory complexity present in a cell, tissue, or organism and therefore serve as the most proximal reporters of the body's response to a disease process or drug therapy. This review presents the rationale, methodology, and preliminary findings from studies that apply comprehensive metabolite profiling to gain knowledge of new circulating markers of PH.
The development of elevated pulmonary arterial pressure (PAP), either in unselected populations or in individuals with a variety of cardiopulmonary diseases, is associated with poor prognosis.1–5 Pulmonary hypertension (PH, i.e., mean PAP > 25 mmHg) leads to right ventricular (RV) dysfunction that is closely associated with impaired exercise capacity 6 and renal and hepatic dysfunction 7 as well as morbidity 8 and mortality.3,9 Despite the functional and prognostic significance of right ventricular–pulmonary vascular (RV-PV) dysfunction, peptides and metabolites that might mark or contribute to RV-PV dysfunction remain incompletely defined. Knowledge of new circulating markers of RV-PV dysfunction or susceptibility to RV-PV dysfunction may provide more precise estimates of risk while also defining the pathways perturbed in individual patients, revealing new targets for intervention and ultimately enabling an individualized approach to care.10,11
To address unmet needs in disease biomarkers, investigators have turned to comprehensive profiling of large numbers of endogenous metabolites and proteins in biofluids, which has been termed “metabolomics” and “proteomics,” respectively (Figure 1). Metabolites are the end result of all the regulatory complexity present in a cell, tissue, or organism, including transcriptional and translational regulation as well as posttranslational modifications. Therefore, metabolites are the most proximal reporters of the body's response to a disease process or drug therapy (Figure 2). As described below, emerging metabolomics techniques will allow us to “overlay” new biomarkers and multimarker scores on existing cardiovascular disease diagnostic tests. 12 It is anticipated that some new markers will be uncorrelated with or “orthogonal” to existing markers, thus providing additional pathobiological insights and information for cardiovascular disease management. 13 In addition to serving as disease biomarkers, circulating metabolites may themselves participate in previously unanticipated roles as regulatory signals with hormone-like functions. 14
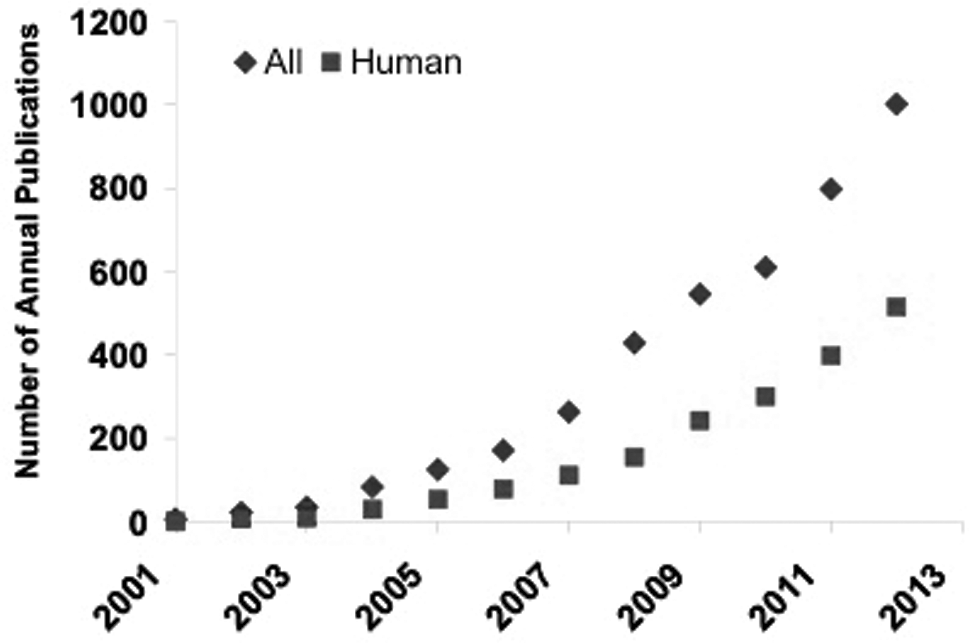
Rapid expansion of proteomics and metabolomics in biomedical research: the number of annual publications in metabolomics from 2001 to 2012. Criteria for inclusion of publications included the terms “metabolome” and “metabolomics.” A color version of this figure is available online.
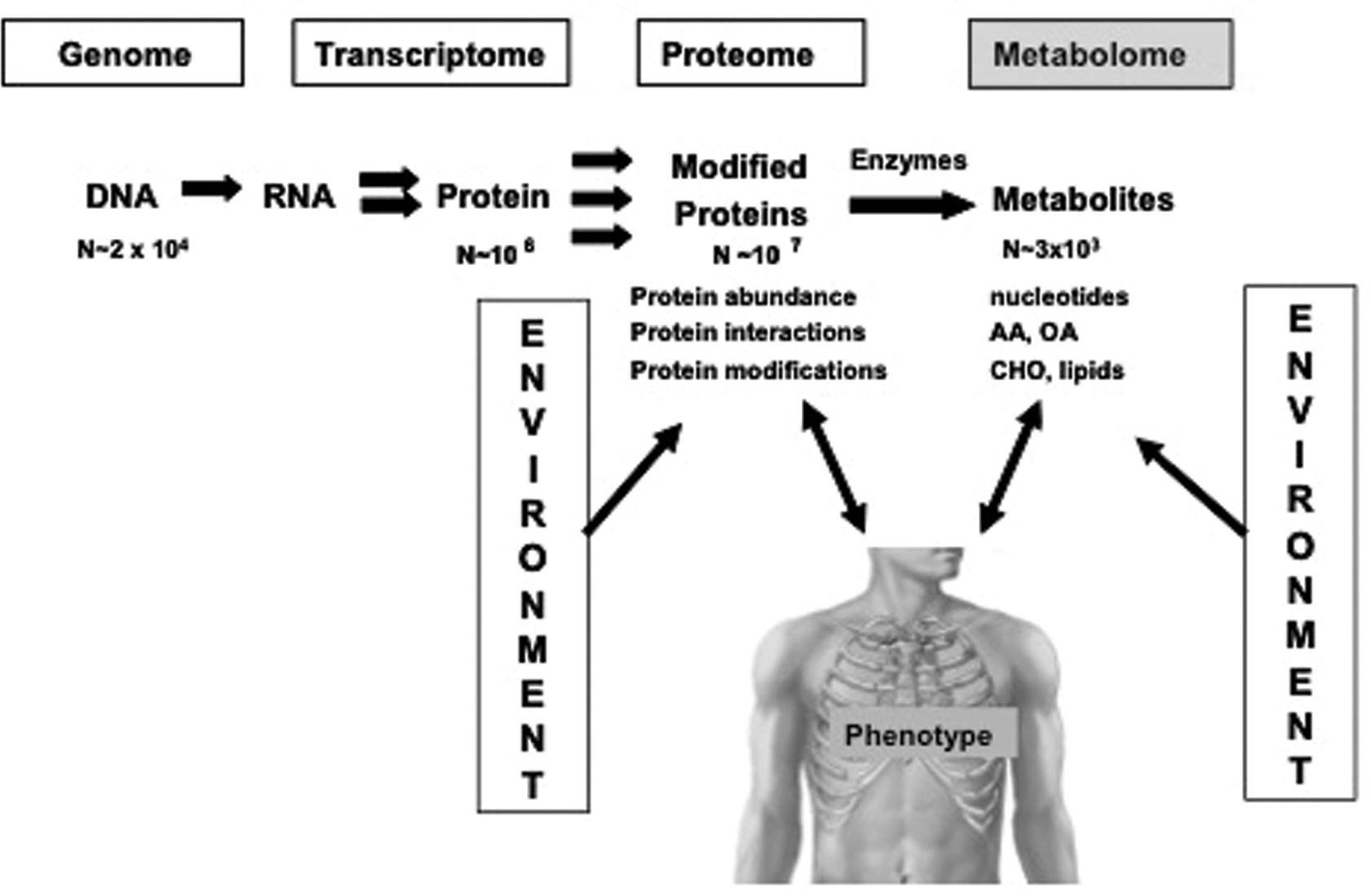
Integration of “omics” approaches and their relationship to phenotype. AA: amino acids; OA: organic acids; CHO: carbohydrates. A color version of this figure is available online.
METHODS FOR METABOLIC PROFILING
Present estimates suggest that the human metabolome consists of approximately 3,000–5,000 endogenous small molecules (i.e., <1 kDa in size), as described in the Human Metabolome Project (http://www.hmdb.ca), and ∼40,000 total metabolites, including exogenous compounds originating from nutrients, microbiota, drugs, and other sources.15,16 The metabolome spans a variety of chemical compound classes, including those that are anionic versus cationic or lipophilic versus hydrophilic. Therefore, no single analytical method is capable of analyzing all metabolites.
Two core technologies are utilized to perform metabolic profiling: nuclear magnetic resonance (NMR) and tandem mass spectrometry (MS/MS). For recent reviews of metabolomics technologies, please see articles by Dunn, 17 Kuehnbaum, 18 and Ramautar. 19 NMR requires relatively little sample preparation and is nondestructive, allowing for subsequent structural analyses. However, the method tends to have low sensitivity and can detect only highly abundant analytes. MS/MS coupled with liquid chromatography (LC), on the other hand, has higher sensitivity for small molecules and is also applicable to a wide range of biological fluids (including serum, plasma, and urine). Recent advances in MS technology now enable researchers to determine analyte masses with such high precision and accuracy that metabolites can be identified unambiguously even in complex fluids. 19
These technologies can be used to characterize biological samples either in a targeted manner or in a pattern discovery manner. In the former, the investigator targets a predefined set of metabolites for analysis. In the latter, the investigator is faced with a complex pattern of peaks, many of which are anonymous: the molecular identities of the species giving rise to the peaks are generally not known. While the targeted approach is more limited, the analysis is more straightforward, as the biochemicals giving rise to the signals have already been identified. An example of metabolite coverage by a targeted platform that is being actively applied to metabolic profiling of cardiovascular diseases20–23 is shown in Table 1. This platform can acquire the data for 290 metabolites in ∼60 minutes from less than 200 μL of plasma. As can be seen in Table 1, the platform provides nearly uniform coverage of all pathways in the KEGG database. Selection of metabolites for incorporation into our platform is based on (1) their biological diversity (i.e., “sentinels” in as many of the known metabolic pathways as possible), (2) their biological relevance (i.e., arginine-nitric oxide [NO] signaling pathway intermediates), and (3) their ability to be detected by LC-MS methods. The pattern discovery, or fingerprint, approach is inherently less biased, but the need for subsequent unambiguous identification of the peaks can be difficult.
Approximate coverage of KEGG metabolism areas
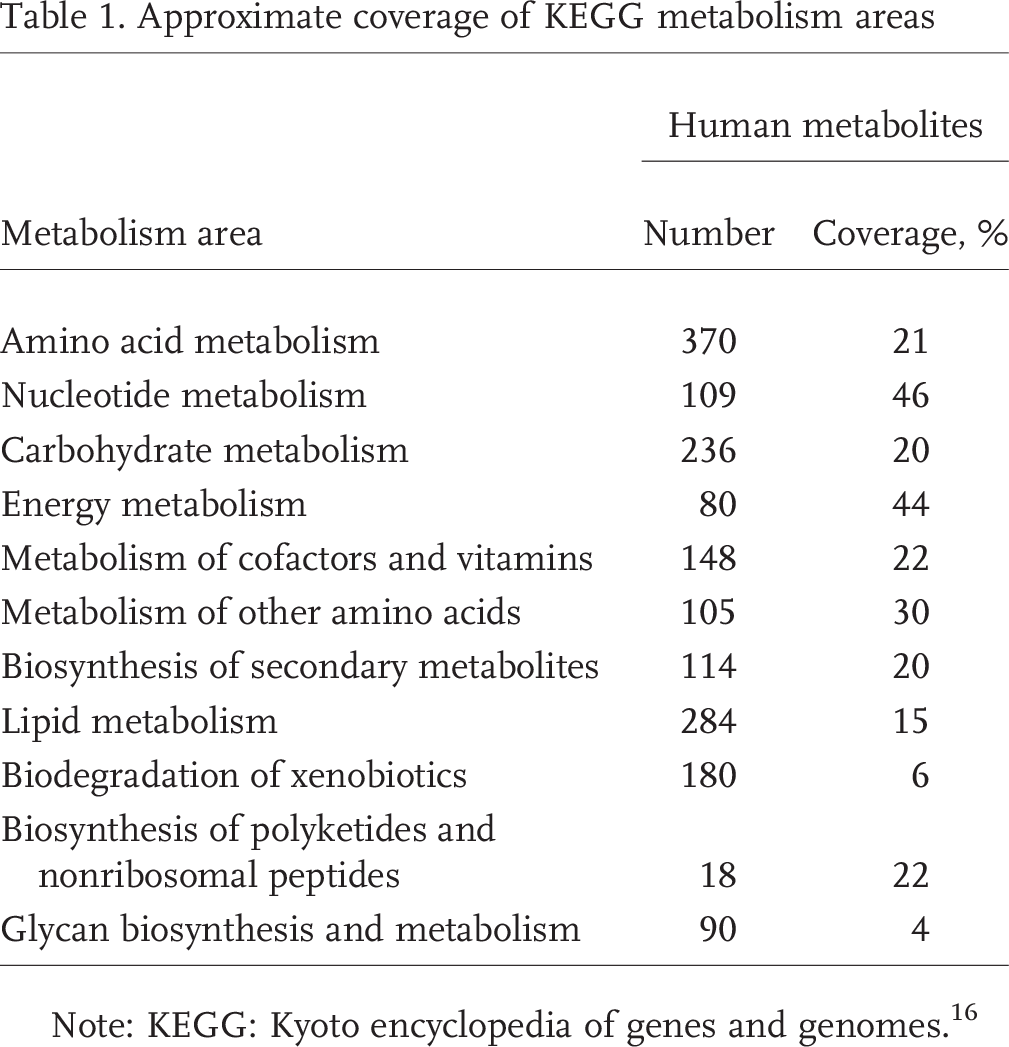
Note: KEGG: Kyoto encyclopedia of genes and genomes. 16
STATISTICAL APPROACHES AND DATA REDUCTION FOR “OMICS” APPROACHES
Although high-throughput metabolomics approaches to biomarker discovery bring many advantages, they also bring the danger of generating false-positive associations as a result of multiple testing and overfitting of data. Replication is imperative to minimize overfitting of data. 24 Application of traditional statistical approaches (e.g., Bonferroni correction) in this setting tends to levy an insurmountable statistical penalty that can obscure biologically relevant associations. Newer statistical techniques, 25 such as advanced resampling methods or control of the false-discovery rate, 26 coupled with functional trend analysis in which changes in constituents of common pathways are analyzed together, can aid in detecting subtle but important changes in multiple variables identified in “omics” approaches.27–29 Several data reduction strategies can be used to construct multivariate metabolite biomarker profiles, 30 including discriminant analysis, 31 partial least squares, principal components analysis, 32 and artificial neural networks. 33
APPLICATION OF METABOLOMICS FOR CARDIOVASCULAR BIOMARKER DISCOVERY
A very limited number of metabolites, such as glucose, lipids, creatinine, urea, and uric acid, have been used for decades to assess an individual's (pre-)disease condition. In 1971, Arthur Robinson and Linus Pauling 34 conceived the core idea that information-rich data reflecting the functional status of a complex biological system resides in the quantitative and qualitative pattern of metabolites in body fluids. Currently, in the field of inborn errors of metabolism, an extensive repertoire of metabolites is used as biomarkers for diagnosis, progression, and response to treatment. 35 As might be expected, the application of metabolomics to complex cardiovascular diseases with multiple potential clinical confounders (i.e., medication exposure, age, gender, comorbidities) has been more difficult than that to Mendelian disorders. 36 Interindividual variability and adequacy of signal-to-noise intensity for certain metabolites represent additional hurdles in metabolomics studies.
To help circumvent these problems, investigators have applied these emerging technologies to unique clinical scenarios where serial sampling can be performed in patients both before and after a controlled perturbation, thereby allowing each patient to serve as his or her own biological control.20,21,29 As proof of principle, our group has applied targeted MS-based metabolomics to patients undergoing “planned myocardial ischemia,”21,22,29 elicited by exercise, and “planned myocardial infarction,” 20 induced by alcohol septal ablation in patients with hypertrophic obstructive cardiomyopathy. In both cases, robust metabolic changes were evident within minutes of the physiologic perturbation, when traditional markers of myocardial injury such as troponin remained unchanged.
A global metabolomics analysis of plasma revealed a pathway in both humans and mice linking microbiota metabolism of dietary choline and phosphatidylcholine to cardiovascular disease pathogenesis. 37 Metabolic profiling has also demonstrated distinct patterns of myocardial substrate utilization in individuals undergoing surgical coronary revascularization. Turer et al. 38 found that patients with left ventricular dysfunction demonstrated global suppression of metabolic fuel uptake and limited myocardial metabolic reserve and flexibility after global ischemia/reperfusion stress in the setting of cardiac surgery. Shah and colleagues 39 used a targeted MS/MS-based platform to profile 69 metabolites so as to evaluate suspected ischemic heart disease in more than 600 subjects from the Duke CATHGEN biorepository who underwent cardiac catheterization. They demonstrated that peripheral blood metabolite profiles, one enriched for branched-chain amino acid metabolites and another comprising urea cycle metabolites, add to the discriminative capability for coronary artery disease, compared with models containing only clinical variables.
METABOLOMICS IN PH
The profiling of metabolites, including lipids, sugars, amino acids, and amines, is particularly relevant to RV-PV dysfunction. Some circulating metabolites (i.e., cyclic guanosine monophosphate [cGMP] and other NO pathway intermediates such as arginine, asymmetric dimethylarginine [ADMA], ornithine, citrulline, tryptophan hydroxylase metabolites, and catecholamines) have already been implicated in mediating pulmonary vascular tone, as described below. Altered metabolic substrate utilization, in the form of a mitochondrial metabolic switch from glucose oxidation to glycolysis, has been observed in the setting of PV remodeling,40,41 RV hypertrophy, 42 and RV dysfunction, 43 offering further incentives to apply metabolite profiling to PH in left ventricular dysfunction. Through metabolomic profiling studies in PH, it will be important to ascertain the extent to which plasma metabolite levels reflect alterations in RV-PV metabolism while also evaluating small molecules with known vasoactive properties, as well as metabolites not previously associated with PH, as potential biomarkers of RV-PV dysfunction.
There is an emerging paradigm of PH as a systemic, multiorgan disease characterized by mitochondrial dysfunction. 44 Mitochondria integrate inputs regarding fuel supply (oxygen, glucose, lipids) and have recently been recognized to secrete mitokines that act on distant organs. This exciting work suggests that when mitochondria from one organ become dysfunctional, they can signal mitochondria from other tissues by releasing diffusible factors termed mitokines. 45 PH patients demonstrate insulin resistance in skeletal muscle analogous to observations in type 2 diabetes. This is particularly relevant because circulating metabolite clusters consisting of elevated levels of branched-chain and aromatic amino acids have been shown to independently predict future onset of insulin resistance and diabetes more than a decade before overt disease onset. 23
Recently, Fessel and colleagues 46 reported findings from metabolomic analyses of human pulmonary microvascular endothelial cells expressing two different disease-causing mutations in the bone morphogenetic protein receptor type 2 gene (BMPR2). This study confirmed previously described increases in glycolysis but also found significant upregulation of the pentose phosphate pathway, increases in nucleotide salvage and polyamine biosynthesis pathways, decreases in fatty acid oxidation pathways, and impairment of tricarboxylic acid (TCA) cycle anaplerosis. The study went on to confirm increased isocitrate dehydrogenase activity in the setting of elevated isocitrate levels in BMPR2-mutant cells. This proof-of-principle study provides insight into potential metabolic pathways altered in RV-PV dysfunction beyond the primary focus to date on altered glucose homeostasis in PH.
Plasma-based metabolomics studies in PH are just beginning to emerge. One recent study in a porcine model of pulmonary embolism examined pre- versus postembolism plasma metabolite levels and found alterations in metabolites related to energy balance in hypoxic conditions, including glycolysis-derived metabolites, ketone bodies, and TCA cycle intermediates. 47 The extent to which easily measured circulating metabolite levels will reflect metabolic changes in experimental models requires further investigation. For example, physiologic perturbations such as hypoxia and exercise can result in increases in circulating TCA cycle intermediates, 21 which may or may not reflect intracellular TCA cycle activity.
METABOLOMICS IN WHO GROUP 2 PH
There may be a particularly important role for application of metabolite profiling in furthering understanding of RV-PV dysfunction in the setting of left-heart disease (i.e., WHO group 2 PH). Heart failure (HF) due to left ventricular systolic dysfunction (LVSD) is commonly associated with the development of PH. 6 However, the ability of the RV to respond to increased PAP at rest and during exercise is highly variable in HF and remains poorly understood. 48 Studies in both experimental models and patients have suggested a maladaptive imbalance between small-molecule pulmonary vasodilators and vasoconstrictors in HF. Altered NO signaling, including decreased NO synthase (NOS) bioavailability and increased production of endogenous small-molecule NOS inhibitors (i.e., ADMA), may contribute to the pathophysiology of PH in HF. 49 Like impaired NO responsiveness, decreased production of prostacyclin (PGI2) and dysregulation of PGI2 metabolism may also contribute to PH in LVSD. 50 Furthermore, insulin resistance with decreased peroxisome proliferatoractivated receptor g activation can accelerate adverse remodeling of the pulmonary vasculature in HF. 51
Shao et al. 52 recently reported that arginine-NO pathway intermediates can distinguish patients with differential burdens of WHO group 2 PH. Melenovsky et al. 53 found that impaired transpulmonary cGMP release relative to transpulmonary B-type natriuretic peptide uptake was present in HF patients with high pulmonary vascular resistance and that phosphodiesterase-5 inhibition restored cGMP responsiveness. That study is an example of careful physiologic phenotyping with multisite sampling that can serve as a basis for potentially inferring local metabolism in the pulmonary circulation via measurement of circulating small molecules such as cGMP. While these studies provide promise that circulating markers of arginine-NO pathway intermediates associate with hemodynamic profiles, further investigations are needed to determine whether circulating metabolite levels from metabolic profiling will predict responses to therapies and prognosis in RV-PV dysfunction.
CONCLUSION
Although metabolomics technologies are still under development, they complement other functional genomic approaches, such as high-throughput genome sequencing and RNA expression analysis. 54 Together, they hold great promise to transform our ability to profile samples and discover multiple new biomarkers for RV-PV dysfunction. Novel multimarker approaches may also permit identification of alterations in specific pathways to inform the best preventative and therapeutic interventions for each individual, by restoring the specific perturbed pathways to their normal function.
