Abstract
Bovine pulmonary hypertension, brisket disease, causes significant morbidity and mortality at elevations above 2,000 m. Mean pulmonary arterial pressure (mPAP) is moderately heritable, with inheritance estimated to lie within a few major genes. Invasive mPAP measurement is currently the only tool available to identify cattle at risk of hypoxia-induced pulmonary hypertension. A genetic test could allow selection of cattle suitable for high altitude without the need for invasive testing. In this study we evaluated three candidate genes (myosin heavy chain 15 [MYH15], NADH dehydrogenase flavoprotein 2, and FK binding protein 1A) for association with mPAP in 166 yearling Angus bulls grazing at 2,182 m. The T allele (rs29016420) of MYH15 was linked to lower mPAP in a dominant manner (CC 47.2 ± 1.6 mmHg [mean ± standard error of the mean]; CT/TT 42.8 ± 0.7 mmHg; P = 0.02). The proportions of cattle with MYH15 CC, CT, and TT genotypes were 55%, 41%, and 4%, respectively. Given the high frequency of the deleterious allele, it is likely that the relative contribution of MYH15 polymorphisms to pulmonary hypertension is small, supporting previous predictions that the disease is polygenic. We evaluated allelic frequency of MYH15 in the Himalayan yak (Bos grunniens), a closely related species adapted to high altitude, and found 100% prevalence of T allele homozygosity. In summary, we identified a polymorphism in MYH15 significantly associated with mPAP. This finding may aid selection of cattle suitable for high altitude and contribute to understanding human hypoxia-induced pulmonary hypertension.
INTRODUCTION
Bovine pulmonary hypertension (BPH) was first identified 100 years ago. 1 The disease became commonly known as high-altitude disease because historically it was observed to occur only at altitudes over 2,440 m and as brisket disease because the clinical signs of right-sided congestive heart failure typically include edema of the brisket region. The average herd incidence has been estimated to be 2% but as high as 10%. 2 The majority of BPH cases have been reported to occur in calves, 3 but, more recently, it has been reported to occur in both beef and dairy cattle up to 2 years of age and at more moderate altitudes.4,5
Currently, the only management tool available to mitigate the risk of pulmonary hypertension is to screen cattle for hyperresponders to hypoxia through invasive measurement of mean pulmonary arterial pressure (mPAP). 6 This method of screening is costly and time-consuming, and the sensitivity of the test is dependent on the altitude at which it is performed: the higher the altitude, the higher the sensitivity. This creates logistical problems for seedstock producers located at low altitude that may want to sell bulls to producers at higher altitudes. A bull susceptible to hypoxia-induced pulmonary hypertension is at great risk of congestive heart failure when taken to high altitude for testing purposes.
The mPAP in cattle has been estimated to be moderately heritable. 7 Inheritance related to a few major genes in an autosomal-dominant manner has been suggested.8–10 The identity of these genes is unknown. The high incidence of BPH at high altitude suggests that causative genetic polymorphisms are common in the general population. Affected and resistant cattle appear phenotypically indistinguishable at low altitude; differential expression of the polymorphic genes may occur only in response to the hypobaric hypoxia associated with increasing altitude. A genetic test would allow producers to select cattle suitable for use at high altitude without the need for invasive pressure testing at high altitude.
Until recently, there were no known major genetic variants associated with the hypoxic vasoconstrictor response of cattle. In 2011, six single nucleotide polymorphisms (SNPs) were identified to be associated with increased mPAP in cattle, but statistical significance was not found because of a small sample size. 11 Several genetic associations with pulmonary responses to altitude have been reported in humans.12–14 A major advantage of genetic studies in cattle relative to humans is the ability to control and/or account for environmental effects on phenotype.
In this study, we measured mean, diastolic, and systolic PAPs in a cohort of 166 Black Angus bulls residing in Wyoming at an elevation of 2,182 m. These hemodynamic data were then correlated with three genes previously identified as being plausibly associated with BPH: 11 myosin heavy chain 15 (MYH15; rs29016420), NADH dehydrogenase flavoprotein 2 (NDUFV2; rs29024944), and FK binding protein 1A (FKBP1A; rs29013210). We also examined the allelic frequency at the same three loci in the Himalayan yak (Bos grunniens), a closely related species acclimatized to life at high altitude. We hypothesized that alleles associated with a lower PAP in our cattle cohort would be overrepresented in the yak.
METHODS
Ethical approval
This study was approved by Colorado State University Animal Care and Use Committee. DNA and RNA were exported from the United States to the United Kingdom under article 4 of the Importation of Animal Products and Poultry Products Order 1980 (license IMP/GEN/2008/03). All other animal samples were transported under license TARP/2011/327 from the United Kingdom's Department for Environment, Food, and Rural Affairs issued by the Animal Health and Veterinary Laboratories Agency.
Cattle cohort
DNA was collected from a herd of black Aberdeen Angus bulls raised and PAP tested at an altitude of 2,182 m in southern Wyoming, on September 13, 2011. The bulls had access to grazing at elevations ranging from 2,000 to 3,000 m. Selection of bulls for low PAP has been routinely performed for more than 20 years. The bulls in this study were progeny of low-PAP sires (bulls with mPAP <42 mmHg). DNA from 166 bulls was collected using Performagene nasal swabs (DNA Genotek, Ottawa, Canada). The kit contains a sponge applicator and a tube containing Performagene solution, which acts as a preservative and a cell lysis agent. The sponge is rubbed gently in the animals' nostril for 5 seconds, after which the sponge is replaced into the collection tube. Animals required restraint in a chute and with a head harness. Samples were then stored at room temperature and posted from the United States to the United Kingdom before DNA purification.
Yak cohort
Yak DNA samples were obtained from one herd in Elbert, Colorado, at an elevation of 1,500 m on August 30, 2011. The yaks either were imported from Tibet or had been raised in Colorado for no more than two generations. In total 24 yaks were sampled using Performagene nasal swabs as described for the cattle cohort.
Assessment of PAP in cattle
The methods used for measurement of PAP are described in detail elsewhere. 6 In brief, the jugular furrow was scrubbed with disinfectant and the vein occluded at the thoracic inlet, causing jugular vein distension. The jugular vein was then punctured with a 13-gauge 8.9-cm (3.5-inch) needle until blood flowed freely from the needle. A polyethylene catheter (Fisher Health Care, Waltham, MA) containing sterile isotonic saline (0.9 % NaCl) was then passed through the needle and into the jugular vein. The catheter was flushed while being gently advanced down the jugular vein, aiding its passage. When approximately 30–45 cm of the catheter had been passed into the jugular vein, the proximal end was then connected to the pressure transducer by way of a three-way stopcock. The transducer, supported on an adjustable stand, was positioned so that it was level with the base of the heart. The transducer and catheter were again flushed to ensure that no air had entered the system.
The catheter was then advanced through the right side of the heart and into the pulmonary artery. The catheter was held still until the pressure remained constant (approximately 10 seconds) and mean, diastolic, and systolic PAPs were recorded. Correct placement of the catheter in the pulmonary artery was verified by pulling the catheter back into the right ventricle and observing a change in pressure waveform.
DNA extraction from nasal swabs
DNA was extracted 3 weeks after collection using PG-L2P solution, purchased from DNA Genotek with the nasal swabs. Briefly, the nasal swabs containing Performagene solution were put in a 50°C oven for 2 hours to ensure the inactivation of nucleases; 500 mL of the nasal swab solution was then mixed with 20 mL PG-L2P purifier reagent and incubated on ice for 10 minutes in order to precipitate impurities. The tubes were spun for 15 minutes at 13,000 rpm and the supernatant was then transferred to a fresh microcentrifuge tube; 25 mL of 5 M NaCl was added to precipitate the DNA and the solution was mixed before 600 mL of 100% ethanol was added. The solution was centrifuged at 13,000 rpm for 2 minutes. The supernatant was discarded and the pellet washed with 70% ethanol. The pellet was re-dissolved in 100 mL of TE buffer.
Genotyping
SNP genotyping of the three candidate genes was performed by LGC Genomics, formerly KBioscience (Teddington, UK), using a competitive allele-specific polymerase chain reaction genotyping system. This was a homogeneous fluorescent resonance energy transfer–based system. Probe sequences can be found in the appendix.
Statistical methods
The data are presented as mean ± standard error of the mean, unless otherwise indicated. Statistical associations between mPAP and the predictor alleles were evaluated by unpaired t tests. Tukey whisker plots were used to show the median and quartile ranges. Individuals were classed as outliers if they had mPAP greater than 1.5 times the interquartile range (the difference between the seventyfifth and twenty-fifth percentiles) away from the median. Outliers are represented as dots on all plots. Probability values <0.05 were reported as statistically significant. Graphical presentation was performed using GraphPad Prism 6.0d (GraphPad Software, La Jolla, CA).
RESULTS
PAP scores among Black Angus herd at 2,182 m
The mean age of bulls tested was 512 ± 13.2 days (mean ± standard deviation [SD]). The mPAP was 45.2 ± 12.2 mmHg (mean ± SD) and ranged from 34 to 119 mmHg.
Variation in MYH15, NDUFV2, and FKBP1A alleles among Black Angus bulls
The proportions of cattle with MYH15 CC, CT, and TT genotypes were 55%, 41%, and 4%, respectively (n = 163; genotyping was undetermined in 3 samples; Table 1). The T allele of the MYH15 SNP was associated with reduced mPAP in a dominant manner (CC 47.2 ± 1.6 mmHg; CT 42.8 ± 0.8 mmHg; TT 43.4 ± 2.0 mmHg; CT/TT 42.8 ± 0.7 mmHg; CC vs. CT P = 0.02; CC vs. CT/TT P = 0.02; Table 1; Fig. 1). The CC versus TT comparison did not reach statistical significance despite the sustained reduction in mPAP from CC to CT and TT, most likely because of the small number of T homozygotes (n = 7) compared to CT heterozygotes (n = 67). Although there was a similar proportion of CC and CT/TT genotypes, 7 C homozygotes had sufficiently high mPAPs to be classed as outliers, compared to only 1 CT heterozygote and 0 T homozygotes. There was a trend toward a lower systolic pressure associated with the T allele (CC vs. CT/TT P = 0.1).
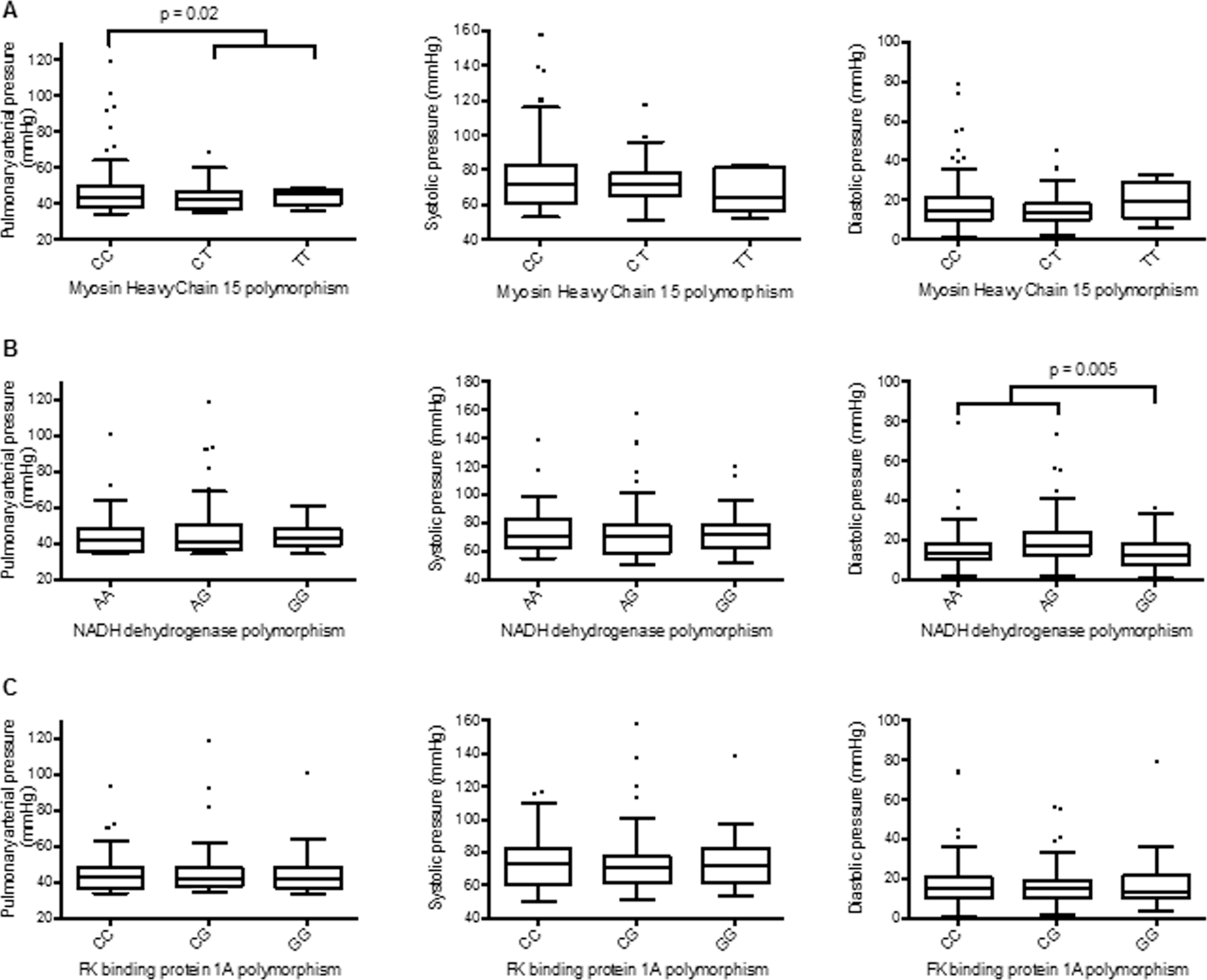
Association of single nucleotide polymorphisms with mean pulmonary arterial pressure and pulmonary systolic and diastolic pressure in cattle at high altitude. The Tukey whisker plots demonstrate the affect of myosin heavy chain 15 (A; n = 163), NADH dehydrogenase flavoprotein 2 (B; n = 161), and FK binding protein 1A (C; n = 161) alleles on mean pulmonary arterial, systolic, and diastolic pressures in Black Angus cattle at high altitude. P values represent a 2-tailed unpaired t test.
Pulmonary arterial pressures in a cohort of bulls at an elevation of 2,182 m according to genotype
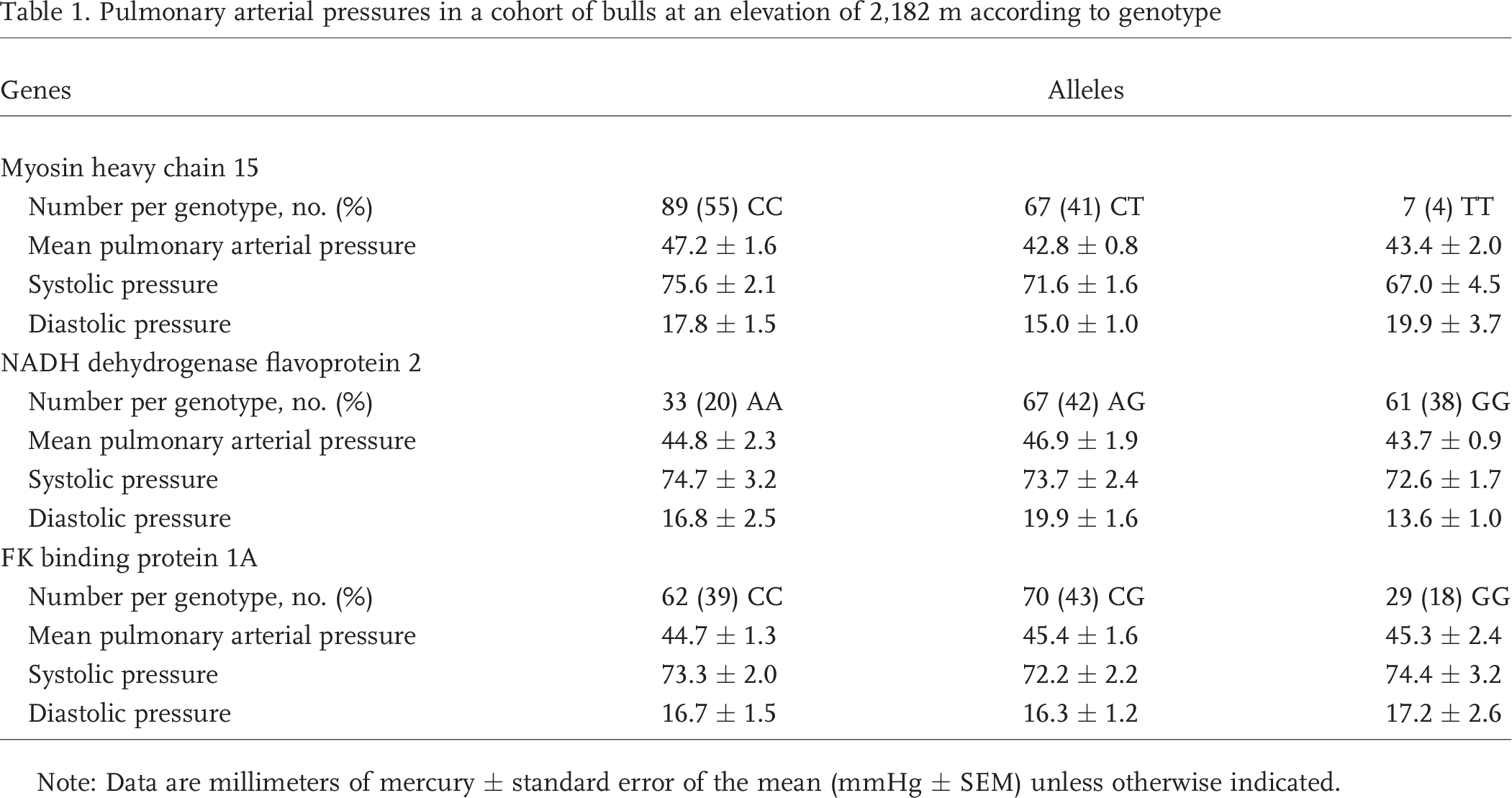
Note: Data are millimeters of mercury ± standard error of the mean (mmHg ± SEM) unless otherwise indicated.
The proportions of cattle with the NDUFV2 AA, AG, and GG genotypes were 20%, 42%, and 38%, respectively (n = 161; genotyping was undetermined in 5 samples; Table 1). G homozygotes of NDUFV2 had significantly lower diastolic PAPs than the cattle with the AG allele (GG 13.6 ± 1.0 mmHg; AG 19.9 ± 1.6 mmHg; P = 0.001; Table 1; Fig. 1). However, A homozygotes did not have a diastolic PAP (16.8 ± 2.5 mmHg) that was statistically greater than that of G homozygotes (P = 0.16). This may have been due to small number of A homozygotes; the AA/AG versus GG comparison was significant (P = 0.005). Seven cattle with either the AA genotype or the AG genotype had sufficiently high diastolic PAP to be considered outliers compared to only 1 G homozygote, despite a 3 : 2 ratio of these genotypes.
The proportions of cattle with the FKBP1A genotypes CC, CG, and GG were 39%, 43%, and 18%, respectively (n = 161; genotyping was undetermined in 5 samples; Table 1). There was no association between mPAP or diastolic or systolic pulmonary pressures and genetic polymorphisms of FKBP1A (Table 1; Fig. 1).
The adaptation of yaks to a high-altitude environment
All yaks were homozygous for the T allele of MYH15, which corresponded to a significantly lower mPAP in cattle than cattle homozygous for the C allele. Similarly, all yaks were homozygous for the G allele of NDUFV2, which was associated with lower diastolic PAP in cattle.
DISCUSSION
BPH: candidate genes
Elucidating the genetic signature of BPH remains one of the largest enigmas in high-altitude livestock management. Identifying the genes and pathways that predict susceptibility to hypoxia-induced pulmonary vasoconstriction and remodeling would direct the genetic selection of cattle suited for life at high altitude without the need for invasive PAP testing.
One recent study used 10,000 SNP chips to compare a cohort of 10 hypertensive cattle with 10 normotensive cattle. 11 They identified six possible allelic associations corresponding to six genes, but because of small sample size, the associations were not statistically significant. Three of these had potential biological relevance to pulmonary hypertension: MYH15 (rs29016420), NDUFV2 (rs29024944), and FKBP1A (rs29013210). We evaluated their association with PAPs in a larger sample of Black Angus cattle.
MYH15
The T allele of the rs29016420 SNP corresponding to the MYH15 gene was significantly associated with a lower pulmonary pressure in a dominant fashion: CC versus CT and CC versus CT/TT comparisons were both statistically significant (Fig. 1). There was a trend toward a lower systolic pressure associated with the T allele (Table 1), which suggests that systolic pressure was the main contributor to the difference in mPAP. In humans, the rs3900940 polymorphism in MYH15 causes an amino acid substitution: Thr1125Ala, located in the coiled-coil rod domain. Thr1125 has shown to be phosphorylated,15,16 whereas Alan1125 cannot be phosphorylated. This substitution may alter protein structure and function. Phenotypic effects may depend on environmental cues such as hypoxia.
MYH15 was first described in 2002. 17 The gene has evolutionary lineage distinct from cardiac and skeletal myosins 18 and is a member of the nonmuscle myosin II (NMII) subgroup of the myosin superfamily. Structurally, the protein resembles a cardiac beta or slow skeletal type 1 myosin heavy chain. 17 Until recently it was thought that the NMII proteins were limited to nonmuscle cells, where contractile activity is important for a diverse range of cellular processes such as cell division, cell migration, and cellcell and cell-matrix adhesion. Despite its name, however, NMII has been shown to be important in the tonic contraction of smooth muscle. 19 MYH15 has also been observed in extraocular muscles 20 and the heart, as well as lung, liver, platelets, brain, and plasma. 21
Studies in humans have associated MYH15 polymorphisms with coronary heart disease risk. 22 The hazard ratios of coronary heart disease in humans with the CC and CT MYH15 alleles were 1.47 (P < 0.001) and 1.09 (P = 0.18), respectively, relative to TT homozygous individuals. 23 The T allele was also associated with a decreased risk of myocardial infarction, 23 hypertrophic cardiomyopathy, 23 noncardioembolic stroke, 24 and ischemic stroke. 24 A link between other cardiac myosin heavy chain polymorphisms and hypertrophic cardiomyopathy has also been implicated in increased risk of sudden infant death syndrome in humans, 25 the prevalence of which increases in association with environmental hypoxia.26–28
Myosin heavy chain expression and hypoxia have been linked. The mouse embryo develops in hypoxic conditions and does not express MYH15 until after birth. 20 Furthermore, cardiac expression of myosin heavy chain isoforms is directed by hypoxic signaling, 29 an adaptation that alters metabolic substrate preference and improves efficiency of oxygen utilization.
The associations between MYH15 polymorphisms and hypoxic disease states might suggest that the alleles are differentially regulated by hypoxia. We found large variation of mPAP values and a disproportionate number of outliers among C homozygotes compared to CT heterozygotes and T homozygotes (Fig. 1). This may indicate that differential regulation of the MYH15 gene was occurring under the hypoxic conditions associated with the moderately high altitude at which these measurements were taken. This is consistent with BPH-susceptible and BPHresistant cattle having no phenotypic disparity at low altitude. Epigenetic regulation of MYH15 expression has been shown previously: MYH15 expression in chickens varied with growth rate and methylation status. 30 The role of epigenetics in maintaining and enhancing the hypoxic response has only recently become apparent. 31 Xia et al. 32 have described genes that are in an active configuration and more likely to bind hypoxia inducible factor. MYH15 gene polymorphisms could affect the predisposition to epigenetic modification and thus the influence of hypoxiaregulated expression at high altitude. Genomic sequencing of the MYH15 locus, along with prospective studies on the effect of MYH15 genotype on PAP will definitively prove the importance of this locus on PAP in cattle. It is unknown whether this gene has influence on PAP in humans.
NDUFV2
An association between the rs29024944 SNP corresponding to NDUFV2 and mPAP was not found. However, there was a statistically significant decrease in diastolic pressure in GG genotypes compared to AG/AA genotypes. There were a disproportionate number of bulls with A alleles that had a sufficiently high diastolic PAP to be classed as outliers in the Tukey whisker plot (Fig. 1). As discussed above, this may be suggestive of an epigenetic mechanism in action at high altitude. The NADH dehydrogenase complex has been highlighted as a genetic player in the adaption to high altitude in previous studies.33–36 NADH6 was found to be under positive selection in high-altitude-living Tibetan horses.34,35 NADH dehydrogenase (ubiquinone) enzymes constitute complex I in the electron transport chain. The complex receives electrons from the oxidation of NADH and provides electrons for the reduction of quinone to quinol, which are then passed to complex II of the electron transport chain. It is not clear how the rs29024944 SNP affects bovine NADH dehydrogenase activity. It has been suggested that polymorphisms in the NADH complex could interfere with proton-pumping efficiency, 33 which in turn influences metabolic performance. 37 How this translates into diastolic pressure is not obvious, although an effect on smooth muscle tone in the arterial wall could be one possibility.
FKBP1A
The SNP linked to FKBP1A was not associated with mean, systolic, or diastolic PAP. The protein encoded by the FKBP1A is a cis-trans prolyl isomerase. The protein interacts with many intracellular signal transduction proteins, including type I TGF-b receptor, and binds several intracellular calcium release channels, thus coordinating complex formation of the ryanodine receptor. 38 The TGF-b signaling pathway is a strong candidate in susceptibility to pulmonary hypertension39–42 especially in the context of high altitude since hypoxic signaling increases TGF-b signaling and the two pathways often work in tandem.43,44 The FKBP1A gene is also important in heart development, with deletion of FKBP1A in the mouse resulting in congenital heart defects including noncompaction of left ventricular myocardium. 45 This evidence makes FKBP1A a candidate for high-altitude adaptation, although this was not evident in this study.
The adaptation of yaks to a high-altitude environment
The role of genetics in BPH is supported not only by high familial incidence within cattle but also by marked variation in susceptibility between species. The closely related and cross-fertile yak (B. grunniens) is an epitome of successful high-altitude adaptation. Many anatomical and physiological traits equip yaks for life at high altitude, including large lungs and hearts and a greatly diminished pulmonary vasopressor response to alveolar hypoxia. 46 By comparing yak genes and cattle genes, we can understand which genes and alleles were positively selected to aid survival in a high-altitude environment. We predicted that any alleles that were associated with a lower PAP would be overrepresented in the yak.
As hypothesized, the yaks were homozygous for the T allele in MYH15, which was associated with a lower PAP in the cattle. Cross-breeding experiments between yak and cattle show that an attenuated hypoxic vasoconstrictor response is inherited in an autosomal-dominant manner since dzos (a cow-yak cross) exhibit PAPs similar to those of the yak despite the stature of the dzo resembling that of cattle. 46 Interestingly, in our study, the lower mPAP exhibited by the cattle homozygous for the T allele is also present in the heterozygous cattle, further supporting an autosomal-dominant mode of inheritance. However, since it was necessary to verify the SNP genotyping assay against the bovine genome (Bos taurus) rather than the yak genome (B. grunniens), these comparisons should be treated with some caution.
Summary
In summary, we have provided evidence for a significant allelic association with PAP in cattle (MYH15) and given evidence of positive natural selection for alleles predisposing to lower PAP in the yak. Cattle are an excellent model of human pulmonary hypertension. 47 Therefore, our findings are likely of high interest to the human medical community in addition to their relevance to the cattle industry.
Footnotes
APPENDIX
Myosin heavy chain 15 (MYH15; rs29016420):
NADH dehydrogenase flavoprotein 2 (NDUFV2; rs29024944):
FK binding protein 1A (FKBP1A; rs29013210):
ACKNOWLEDGMENTS
The authors acknowledge Jim Jensen (Lucky 7 Ranch) and Eddy Sanders (Grunniens Ranch) for making this research possible and the Clinical Pathology Laboratory at Colorado State Veterinary Hospital for providing equipment.
