Abstract
Pulmonary blood vessel structure and tone are maintained by a complex interplay between endogenous vasoactive factors and oxygen-sensing intermediaries. Under physiological conditions, these signaling networks function as an adaptive interface between the pulmonary circulation and environmental or acquired perturbations to preserve oxygenation and maintain systemic delivery of oxygen-rich hemoglobin. Chronic exposure to hypoxia, however, triggers a range of pathogenetic mechanisms that include hypoxia-inducible factor 1α (HIF-1α)–dependent upregulation of the vasoconstrictor peptide endothelin 1 in pulmonary endothelial cells. In pulmonary arterial smooth muscle cells, chronic hypoxia induces HIF-1α-mediated upregulation of canonical transient receptor potential proteins, as well as increased Rho kinase-Ca2+ signaling and pulmonary arteriole synthesis of the profibrotic hormone aldosterone. Collectively, these mechanisms contribute to a contractile or hypertrophic pulmonary vascular phenotype. Genetically inherited disorders in hemoglobin structure are also an important etiology of abnormal pulmonary vasoreactivity. In sickle cell anemia, for example, consumption of the vasodilator and antimitogenic molecule nitric oxide by cell-free hemoglobin is an important mechanism underpinning pulmonary hypertension. Contemporary genomic and transcriptomic analytic methods have also allowed for the discovery of novel risk factors relevant to sickle cell disease, including GALNT13 gene variants. In this report, we review cutting-edge observations characterizing these and other pathobiological mechanisms that contribute to pulmonary vascular and right ventricular vulnerability.
In the autumn of 2015 at the Lost Valley Ranch in Sedalia, Colorado, the 34th Grover Conference, on pulmonary circulation in the “omics” era, convened for a 4-day symposium to discuss contemporary scientific concepts in the genetics, genomics, proteomics, and epigenetics of pulmonary vascular diseases. This biannual meeting, sponsored by the American Thoracic Society and co-led this year by Drs. C. Gregory Elliot, Wendy K. Chung, D. Hunter Best, and Eric D. Austin, commemorates the perpetual and transformative scientific contributions of Dr. Robert Grover 1 to the fields of high-altitude medicine and pulmonary vascular disease. This review is part of an ongoing Pulmonary Circulation thematic series that aims to provide a synopsis of key concepts presented at this year's Grover Conference.
Here, we summarize the discussions that constituted a section of the conference emphasizing novel genetic and molecular mechanisms underpinning “pulmonary vascular and ventricular dysfunction in the susceptible patient.” To accomplish this, three concepts are reviewed in detail: the functional relevance of chronic hypoxia (CH), discovered through genomic analyses; novel molecular mechanisms and phenotypic signatures of pulmonary vascular disease in sickle cell disease; and hormonal regulation of vascular fibrosis. Although distinct in many regards, findings presented in these works intersect by virtue of their common intent to clarify the relationship between classical risk factors for vascular injury and the development of pulmonary vascular disease. In particular, these efforts capitalize on unbiased scientific methodologies, big data sets, and/or cell-specific properties unique to the pulmonary circulation in order to elucidate the pathobiological mechanisms that link hypoxia, hemoglobinopathies, and vasoactive hormone exposure to pulmonary hypertension (PH).
HYPOXIC PH
It has long been recognized that protracted exposure to alveolar hypoxia has a negative impact on the pulmonary vasculature. In the mid-twentieth century, the migration of Han Chinese from mainland China into the highlands of Tibet uncovered the deleterious effects of residence at high altitude. Han infants born at low altitude and subsequently moved to altitudes of 3,000–4,000 m suffered symptoms consistent with congestive heart failure, including dyspnea, cyanosis, enlarged right heart, and pulmonary vascular remodeling (reviewed by Anand and Wu 2 ). Similar symptoms were described with military troop movements into the Himalayas3,4 and later in studies performed in lowland adults who were stationed at high altitude.5,6 These results were consistent with studies being performed on the other side of the world in native high-altitude populations in the Peruvian Andes, which revealed elevations in pulmonary arterial pressure (PPA) and pulmonary vascular resistance (PVR), right heart enlargement, and pulmonary vascular remodeling, including medial hypertrophy, pulmonary arterial smooth muscle cell (PASMC) hyperplasia, and muscle extension into distal arterioles.7–13
Starting in the 1960s, experiments conducted by Robert Grover and his colleagues in the high mountains of Colorado (>8,000 ft [2,428.4 m]) provided insights into the cardiac problems that develop as a result of PH. 14 Studying cattle living at altitude, Will et al. 14 described animals with edema surrounding the brisket, or lower chest, and enlarged, dilated, and “flabby” hearts. In subsequent studies, extensive vascular remodeling was also observed in this model, characterized by collagen deposition, thickened adventitia, increased medial thickness, extension of muscle down to precapillary arterioles, and intimal narrowing in small pulmonary arteries (PAs).15–17
It is now clear that for those not adapted to living at altitude (usually >2,500 m), ascent to and residence at high altitude results in the development of PH,8,18,19 defined as a resting PPA of ≥25 mmHg, pulmonary vascular remodeling,7,12 right ventricular hypertrophy, 20 and, in some cases, right ventricular failure.21,22 What is also clear is that certain populations have genetically adapted to life at altitude, and genomic studies in these groups, coupled with laboratory studies in animal models, have provided insight into some of the mechanisms involved in the pathogenesis of hypoxic PH (HPH).
Mechanisms of HPH: lessons from animal models
The exact mechanisms underlying the development of HPH are still being unraveled. To aid in this endeavor, a number of animal models have been developed, one of the most reliable being the rodent CH model. Rats or mice placed in normobaric or hypobaric hypoxia (fraction of inspired oxygen [FIO2] ∼ 10%) develop significant increases in PPA, right ventricular mass, and PVR resulting from both active contraction and remodeling (reviewed by Stenmark et al. 15 ). In rats, substantial vascular remodeling has been described,23–28 findings consistent with observations reported in humans residing at high attitude.12,29 Similar results have been reported in mice, pigs, sheep, and cows, although the exact amount of remodeling and levels of PPA vary with species (reviewed by Rhodes 16 ).
Given that initial experiments detailing the hypoxia-induced structural changes in the pulmonary circulation revealed thickening of the medial layer due to increased smooth muscle and fibroblast proliferation and that acute exposure to O2, calcium channel blockers, or other vasodilators had little effect on PPA,30–33 it was widely regarded that contraction played a minimal role in sustained HPH and that “fixed” remodeling was the underlying cause of the increased PVR. This assertion was challenged, however, by striking studies demonstrating that inhibitors of Rho kinase (ROCK), an important mediator of smooth muscle cell contractility, acutely normalized PPA in chronically hypoxic rats when administered systemically34,35 or via inhalation, 36 indicating that active contraction may, in fact, be the most important factor determining PPA. This hypothesis was supported by morphological studies demonstrating that with CH, the increase in smooth muscle cells extended outward and did not, in fact, cause luminal narrowing. 37 Thus, while hypoxic exposure might increase muscularity of the small PAs, this remodeling likely contributes to the increase in PVR primarily by facilitating vasoconstriction.
The mechanisms underlying smooth muscle contraction and remodeling include factors that are both intrinsic and extrinsic to smooth muscle. With respect to contraction, under normal conditions, excess production of vasodilators, such as nitric oxide (NO) and prostacyclin, results in low pulmonary vasomotor tone. During hypoxia, reduced synthesis of vasodilators, coupled with increased production of vasoconstrictors, such as endothelin 1 (ET-1) and serotonin, shifts the balance to enhanced contraction (reviewed by Stenmark et al. 38 ). In addition, increased ROCK activation enhances the Ca2+ sensitivity of the contractile apparatus,39,40 rendering the PAs more sensitive to vasoconstrictor stimuli.
Abnormal mechanisms intrinsic to PASMCs that contribute to the increased contractile nature of PASMCs include alterations in ion channels and transporters, leading to changes in membrane potential, elevated intracellular calcium concentration ([Ca2+]i), and increased responsiveness to vasoconstrictors. PASMCs derived from chronically hypoxic rats were reported to be depolarized, 41 a finding later confirmed in other species (reviewed by Shimoda and Polak 42 ). Reduced K+ channel expression and activity were subsequently identified as factors contributing to depolarization in HPH.43–46 Depolarization was initially hypothesized to drive activation of voltage-gated calcium channels (VGCCs) and calcium influx leading to elevated basal [Ca2+]i; however, later work suggested that VGCC inhibitors have little effect on basal [Ca2+]i in PASMCs from hypoxic animals,47–49 although increased expression and Ca2+ influx through VGCCs in response to stimulation has been noted in hypoxic PASMCs.50–52 Rather, elevated basal [Ca2+]i in PASMCs derived from chronically hypoxic animals appears to occur primarily via upregulation of canonical transient receptor potential (TRPC) proteins,48,49 which form nonselective cation channels (NSCCs) that are permeable to Ca2+. 42 Decreasing the activity of NSCCs, either pharmacologically or by RNA silencing, reduced [Ca2+]i48,49 in PASMCs from animals with HPH.
Direct effects of hypoxia on PASMCs are also likely to play a role in vascular remodeling. For example, cells exposed to hypoxia ex vivo or derived from chronically hypoxic animals exhibit increases in both migration and proliferation,53–56 and both K+ channel activity and [Ca2+]i are likely to play a role. Agents that activate and/or increase expression of K+ channels, including the steroid hormone dehydroepiandrosterone and the metabolic modulator dichloroacetate, reduced remodeling in chronically hypoxic rats,46,57,58 likely because increased intracellular K+ confers resistance to apoptosis. 59 Increased [Ca2+]i is required for PASMC growth60,61 and migration 55 and has been documented in cells from hypoxic animals. 42 VGCCs contribute to stimulated proliferation 62 and may participate in the remodeling process in HPH, particularly in the presence of excessive growth factors. Increased [Ca2+]i activates a number of downstream signal transduction pathways and transcription factors that could be involved in PASMC proliferation (reviewed by Kuhr et al. 63 ). In particular, Ca2+ activates nuclear factor of activated T cells (NFAT), which reduces K+ channel expression, 64 providing a link between alterations in [Ca2+]i, dysregulated K+ channel expression/activity, and PASMC growth. Moreover, hypoxia causes Ca2+-dependent increases in the expression of aquaporin 1 (AQP1), a water channel, which is required for proliferation and migration of PASMCs. 55 In endothelial and tumor cells, increased AQP1 levels may result in localized control of water flux across the cell membrane, possibly allowing for directed cell movement. 65 However, in PASMCs, the actions of AQP1 appear to be independent of water transport 56 and instead require the C-terminal tail portion of the protein, which may act to regulate the levels of β-catenin, 66 a dual-function protein that controls both migration and proliferation.
The Na+/H+ exchanger (NHE) is another membrane transporter that contributes to hypoxia-induced remodeling. NHE is a major contributor to maintenance of PASMC pH homeostasis,67,68 and NHE activation mediates growth factor–induced proliferation. 69 Expression of NHE isoform 1 (NHE1) is upregulated in PASMCs from chronically hypoxic animals, leading to increased NHE activity and alkaline pH.70,71 Pharmacological inhibition 72 or genetic deletion73,74 of NHE1 decreased hypoxia-induced vascular remodeling and PASMC proliferation and migration. 74 In PASMCs, loss of NHE1 increased p27, a cyclin-dependent kinase inhibitor, and decreased E2F1, a nuclear transcription factor that controls proliferation, 74 suggesting that NHE1 activation represses a growth inhibitor pathway while stimulating proliferation. NHE1 may also facilitate cell growth and migration via regulation of cytoskeletal arrangement. 75
ROCK may represent a node of convergence with channels/transporters involved in remodeling, as ROCK activation opens Ca2+ channels51,76 and stimulates the activity of 77 and is activated by 73 NHE1. Chronic treatment with ROCK inhibitors reduced neovascularization and remodeling in HPH models;34,78,79 ROCK activation is necessary for migration and proliferation in a variety of vascular cell types, including PASMCs;80–83 and the upstream activators of ROCK, RhoA and RhoB, mediate cytoskeletal rearrangement in PASMCs, 84 a process necessary for cell movement, a key component of both migratory and proliferative processes.
All of the pathways outlined above are linked, in that they are regulated by the oxygen-sensitive transcription factor hypoxia-inducible factor (HIF). Hypoxia-inducible factor 1 (HIF-1) was originally identified as a heterodimeric transcription factor consisting of a β subunit (HIF-1β) that is constitutively expressed and an α subunit (HIF-1α) that is typically not detectable under normoxic conditions. 85 Subsequent studies identified HIF-2α, which is structurally similar to HIF-1α and also binds HIF-1β, resulting in the HIF-2 transcription factor. The mystery of how HIFs are regulated by changes in oxygen was solved when it was reported that HIF-1α is hydroxylated on two proline residues (via prolyl hydroxyalse domain proteins), using molecular oxygen as a substrate, allowing binding to the von Hippel–Lindau protein, ubiquitination, and targeting for proteasomal degradation (reviewed by Semenza 86 and Prabhakar and Semenza 87 ). Thus, falling oxygen levels limit hydroxylation, allowing HIF-1α/HIF-2α accumulation. Unlike HIF-1α, which is found in all cells, HIF-2α exhibits a more restricted expression pattern.
Animal studies revealed a role for HIFs in the development of HPH. Generation of mice homozygous for a null Hif1α allele resulted in embryonic lethality, 88 whereas mice heterozygous for the null allele (Hif1a+/−) were viable, with phenotypically normal appearance under normoxic conditions. When exposed to CH, Hif1a+/− mice exhibited impaired development of HPH and reduced vascular remodeling. 89 Similar results were observed in mice with partial deficiency in Hif2a. 90 When studied ex vivo, PASMCs from Hif1a+/− mice also exhibited reduced hypoxia-induced proliferation. 91 Surprisingly, mice with a selective Hif1a homozygous deletion targeted to vascular smooth muscle cells 92 or Hif2a homozygous deletion targeted to endothelial cells 93 resulted in enhanced HPH, although inducible smooth muscle–specific deletion of Hif1a exhibited protection from HPH similar to global Hif1a heterozygosity. 94 The differences observed between targeted homozygous and inducible targeted or global heterozygous genetic modifications clearly require further investigation.
Recent studies using pharmacologic inhibitors of HIF activity demonstrated that targeting HIF reduced CH-induced vascular remodeling in rodents. 95 The exact mechanisms by which HIF mediates contraction and/or remodeling during CH are still being investigated (Fig. 1) but involve both Ca2+ and pH homeostasis.71,91,96 HIF also regulates K+ channel expression as well as other factors involved in the pathogenesis of HPH, including ET-1 and vascular endothelial derived growth factor (VEGF). 87 While HIF can induce ET-1 production, ET-1 also upregulates HIF-1,97,98 creating feed-forward enhancement of HIF-1 expression. PASMCs treated with exogenous ET-1 accumulated HIF-1α protein and exhibited downregulation of prolyl hydroxylase domain 2 (PHD2) expression, 97 Moreover, blockade of endothelin type A (ETA) receptors prevented upregulation of HIF-1α in PASMCs exposed to moderate hypoxia. In contrast, ET-1 failed to augment HIF-1α in aortic smooth muscle cells, suggesting a feature unique to PASMCs. 97 Since the primary source of ET-1 in vivo appears to be endothelial cells, these data, taken as a whole, suggest a possible model whereby enhanced HIF-2α expression in endothelial cells during CH produces ET-1, which then augments HIF-1α in PASMCs, initiating a feed-forward mechanism for upregulation of HIF-1 and, consequently, HIF target genes to promote the development of HPH.
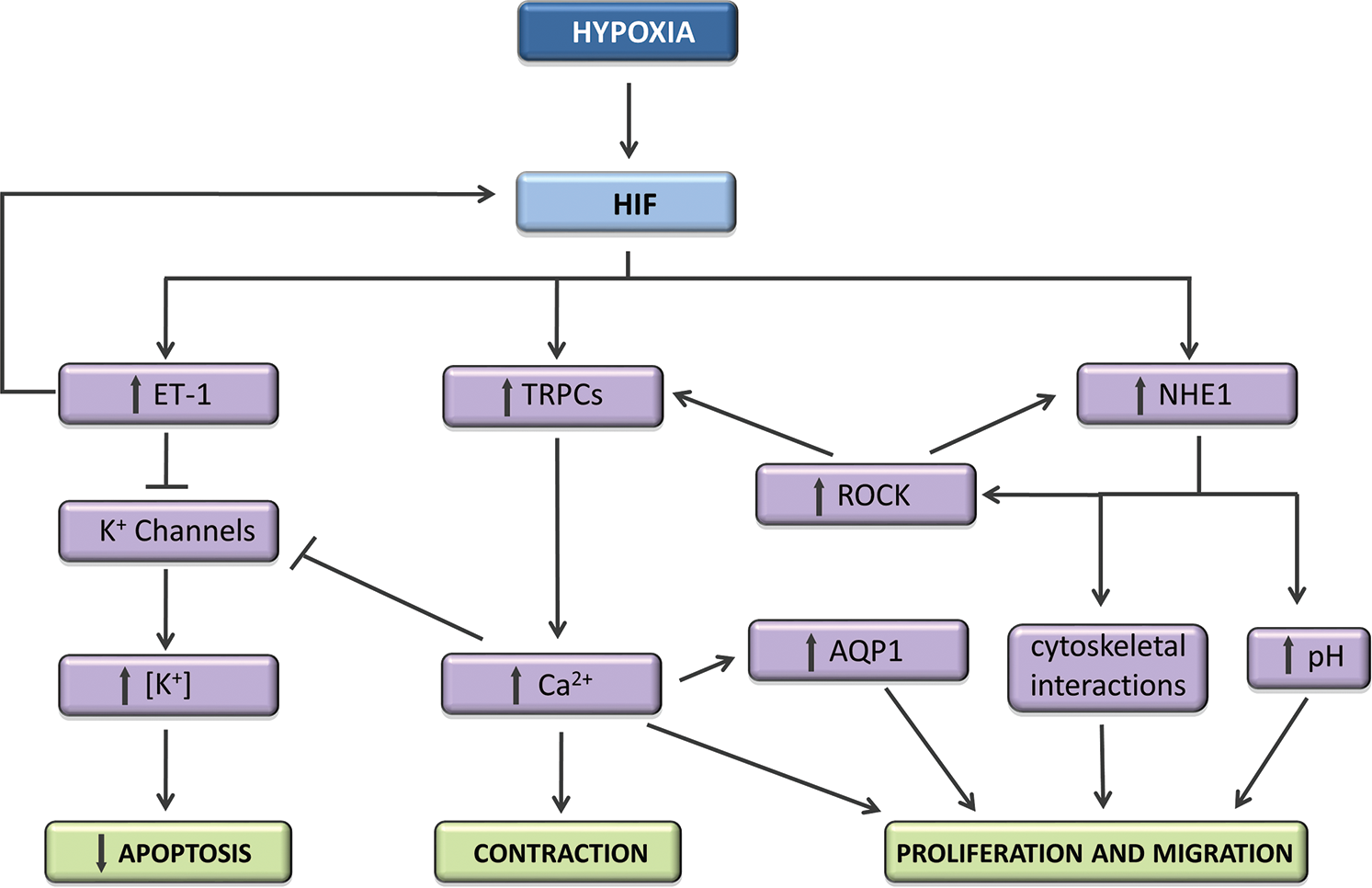
Schematic showing some of the pathways by which hypoxia-inducible factor (HIF) mediates pulmonary arterial smooth muscle changes during chronic hypoxia. ET-1: endothelin 1; TRPCs: transient potential receptors (canonical); ROCK: Rho kinase; NHE1: Na+/H+ exchanger 1; AQP1: aquaporin 1.
Mechanisms of HPH: lessons from high-altitude genomics
While experiments conducted in laboratories using animal models revealed significant effects of hypoxia on the pulmonary circulation and began to unmask some of the mechanisms involved, the molecular-biology revolution and development of techniques for genetic screening allowed for “experiments in nature” to be conducted in populations indigenous to, and therefore adapted to residence at, high altitude. Currently, it is estimated that 140 million people reside in high-altitude regions throughout the world. Three major native high-altitude populations have been studied in depth, and, not surprisingly, those indigenous populations who have adapted to survive in the harsh environments of high-altitude regions have done so differently. In Tibetans, who have been documented to live at high altitude for 30,000 years, resting PPA and PVR were normal or borderline in residents at high altitude. 99 Native Tibetans do not exhibit polycythemia but do exhibit higher hypoxic ventilatory drive and higher exhaled NO levels and rarely develop chronic mountain sickness. Separate genomic studies detailed variants in Tibetan highlanders in the genes EPAS1 (encoding HIF-2α) and EGLN1 (one of the prolyl hydroxylases that regulate HIF1α/2α).100–104 Furthermore, a genetic mutation resulting in HIF-2α overexpression was associated with development of PH. 105
Andeans have lived at high altitude for approximately 11,000 years. Although PPA was normal in the Aymara and Quenchua Indians of the Peruvian Andes, 99 at elevations of 4,540 there was a mild increase in PPA.8,106 Development of PH and polycythemia is fairly common in residents of the Andean plateau,18,107 unlike Tibetans. Variants in EPAS1 have not been identified in adapted Andeans, although recent studies have identified other genes that may be associated with adaptation, including VEGFB, 108 EDNRB (encoding the endothelin receptor subtype B), 109 and the prolyl hydroxylase PHD3.110,111
The Amarah have been documented to reside on the Ethiopian plateau for at least 5,000 years, but evidence suggests that they have lived at 2,300–2,400 ft (∼700–730 m) for ∼70,000 years. This last finding may explain why this population appears the best adapted to living at high altitude, with a lack of polycythemia or increased PPA, hemoglobin with higher O2 affinity, and higher O2 saturations. Gene variants associated with adaptation in this population include ARNT (encoding HIF-1β) and EDNRB. 112
Finally, in addition to studies conducted in humans residing at high altitude, recent work has also examined the genetics associated with development of HPH in cattle. Comparison between cattle that developed PH at altitude and those that did not revealed variants in EPAS1 that likely confer gain of function and were associated with HPH susceptibility. 113 Taken together, the genomic studies in both humans and cattle at high altitude independently identified variants in the HIF signaling pathway that confer protection or susceptibility to high-altitude PH.
Thus, while many “-omics” studies result in discovery science that is then tested in animal models to establish relevance, the field of HPH is one case in which the opposite occurred, with studies in animal models initially paving the way in identifying a prime role for the HIF pathway that was subsequently confirmed by data from large genomic studies in high-altitude populations. While additional downstream and parallel pathways continue to be identified, the central role of HIFs in the development of HPH appears to have been firmly established.
PH IN SICKLE CELL DISEASE AND THE HYPOXIC RESPONSE
Sickle cell disease
Sickle cell disease (SCD) is an autosomal recessive disorder affecting millions of individuals worldwide, making it one of the most common monogenetic diseases. Sickle cell anemia, the most common and severe form of SCD, occurs in patients with a single amino acid substitution of glutamic acid for valine in the β-globin gene, resulting in an abnormal hemoglobin form called hemoglobin S (HbS) within the erythrocytes. Mutant HbS has reduced solubility when deoxygenated, compared to normal hemoglobin A (HbA), leading to polymerization and aggregation of sickle erythrocytes in the microvasculature. Structural abnormalities and cumulative damage to the cellular membrane of sickled erythrocytes results in hemolysis and the development of chronic hemolytic anemia, as well as vaso-occlusion and multiorgan pathology. Compound heterozygosity of HbS with other β-globin gene mutations results in additional types of SCD, such as hemoglobin SC or hemoglobin S-β-thalassemia. 114 The pathobiology of SCD is multifactorial and results from the structural and functional changes of sickled erythrocytes. Deoxygenated HbS polymerizes and aggregates inside erythrocytes, forming rigid, sickled cells. The abundance of polymerized HbS is strongly influenced by both the level of deoxygenation and the concentration of intracellular HbS 115 and determines the severity of SCD. 116 Sickled erythrocytes navigating the microcirculation are continually exposed to mechanical and oxidant injury, damaging the cell membrane and leading to a shortened erythrocyte life span, increased hemolysis, and the development of chronic hemolytic anemia in patients. Importantly, intravascular hemolysis depletes the bioavailability of NO levels through the abundant release of both cell-free Hb, an NO scavenger, and arginase, which catabolizes arginine, a substrate necessary for NO synthesis. 117 Reduction in NO disrupts vascular homeostasis, leading to endothelial dysfunction, proliferation of cells within the vascular wall, thrombosis, and inflammatory stress.118–121 In addition, pathologic sickled erythrocytes have increased adherence to the vascular endothelium, leading to microvasculature occlusion and subsequent multiorgan damage. Ischemia-reperfusion injury at sites of vessel occlusion can also promote inflammation and oxidative stress.122–125 Finally, the low pressure and oxygen tension within the pulmonary arterial bed can promote HbS polymerization and erythrocyte sickling. 126
PH in SCD
In spite of significant improvements in the life expectancy of patients with SCD, estimates of the median age at death range from 42 to 53 years for men and from 48 to 58.5 years for women.127,128 Cardiopulmonary disorders are a major cause of death in patients with SCD.118,127,129–131 Among these complications, PH occurs in 6%–10% of adults with SCD, and the presence of PH is associated with significant increase in the risk of death in these patients.132–135 Although the etiology of PH in patients with SCD is multifactorial, including pulmonary arterial hypertension (PAH), pulmonary venous hypertension, and chronic thromboembolic disease, data suggest that mortality in adults with SCD and PH is proportional to the severity of precapillary PH. 136
Transcriptome analysis, PH, and the hypoxic response in SCD
Despite SCD's Mendelian inheritance, the clinical course of SCD patients is highly variable. There is significant phenotypic heterogeneity in the rate of development of acute and chronic complications, including PH, suggesting that other genetic and genomic modifiers could play a role in these complications. Therefore, we explored the usefulness of peripheral blood mononuclear cell (PBMC)–derived gene signatures as biomarkers for PH in SCD. 137 Genome-wide gene and micro-RNA (miRNA) expression profiles were correlated against estimated right ventricular systolic pressure (RVSP), yielding 631 transcripts and 12 miRNAs. Biological-pathway analysis of these 631 genes revealed Wnt signaling, calcium signaling, vascular smooth muscle contraction, and cancer pathways as significantly represented in patients with elevated RVSP. These pathways have been associated with pulmonary vascular remodeling in other studies and suggest biological relevance to these findings. In order to identify a more succinct gene list that could be capable of identifying patients with an elevated RVSP, we conducted support vector machine analysis and identified a 10-gene signature including GALNT13 (encoding polypeptide N-acetylgalactosaminyltransferase 13, a glycosyltransferase enzyme responsible for the synthesis of O-glycan 138 ) that distinguishes patients with and without increased RVSP with 100% accuracy. This finding was then validated in a separate cohort of patients who had SCD without (n = 10) or with (n = 10) PH, with an overall accuracy of 90%. Increased RVSP-related miRNAs revealed strong in-silico binding predictions of miR-301a to GALNT13, corroborated by microarray analyses demonstrating an inverse correlation between their expression. A genetic-association study comparing patients with an elevated (n = 49) and those with a normal (n = 63) RVSP revealed five significant single-nucleotide polymorphisms within GALNT13 associated with an elevated RVSP. These studies suggest the potential clinical usefulness of genomic signatures as biomarkers of PH in SCD (Fig. 2). Given that aberrant glycosylation patterns are a hallmark of the tumor phenotype influencing proliferation, invasion, angiogenesis, and metastasis potentially involved in pulmonary vascular remodeling, GALNT13 may be speculated as a potential candidate gene in SCD-associated PH.
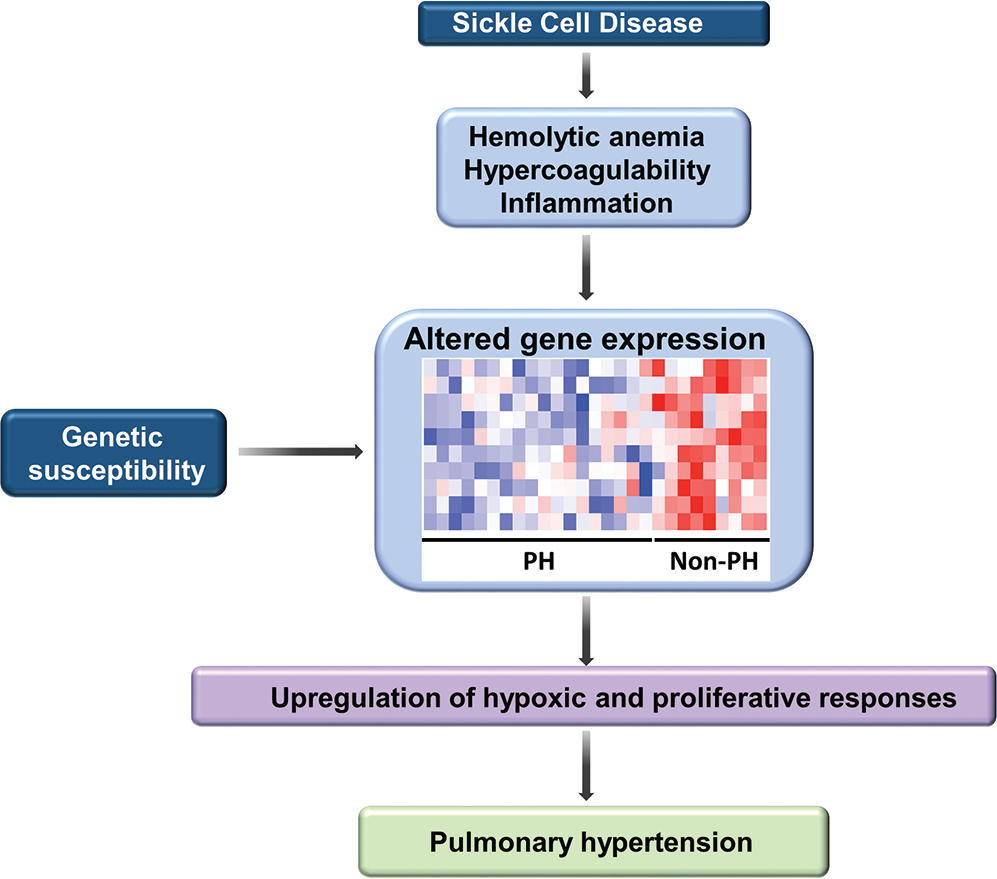
Schematic diagramming mechanisms involved in the development of pulmonary hypertension in sickle cell disease. Abnormalities characteristically observed in sickle cell disease, including hemolytic anemia, hypercoagulability, and inflammation, coupled with individual genetic susceptibility, result in altered expression of a wide panel of genes. Pathways associated with vascular remodeling, including Wnt signaling, calcium signaling, vascular smooth muscle contraction, and cancer pathways, were significantly represented in patients with elevated right systolic pressures, suggesting that these pathways are likely to play a contributory role in the development of pulmonary hypertension (PH).
As discussed above, CH is recognized to be an important cause of PH. In addition, normoxic activation of HIF-1α is involved in the pathobiology of PH and pulmonary vascular remodeling. 139 SCD is characterized by steady-state high circulating erythropoietin concentrations, 140 suggesting that this chronic hemolytic anemia is accompanied by chronic upregulation of the hypoxic response. In addition to the anemic state, increases in placental growth factor, a mediator that activates HIF-1α in normoxia, have been associated with elevated systolic PA pressures in SCD. 141 On the basis of these observations, we hypothesized that the upregulated hypoxic response in SCD might contribute to altered gene expression and the development of PH. To test this hypothesis, we compared clinical data and PBMC genomic profiles of subjects with SCD to those of subjects with Chuvash polycythemia, a monogenic hematologic disorder characterized by an upregulated hypoxic response and elevated systolic PA pressures.142,143 In Chuvash polycythemia, homozygosity for VHLR200W leads to posttranslational stabilization of the alpha subunits of HIF at normoxia via decreased binding of these subunits to the mutant VHL (von Hippel–Lindau) protein, leading to increased levels of HIF-1 and HIF-2 and altered transcription of several genes.142,144 We observed a strong correlation between gene expression profiles in hemoglobin SS subjects and VHLR200W homozygote polycythemic subjects, whereas a control analysis did not show correlations between differential gene expression in hemoglobin SS and Down syndrome patients. 145 Taken together, these data suggest that a substantial proportion of PBMC gene expression variation in hemoglobin SS patients may be related to the hypoxic response. Specifically, at a 5% false-discovery rate, 1,040 genes exhibited a more than 1.15-fold change in both conditions; 297 were upregulated and 743 downregulated, including MAPK8, which encodes a mitogen-activated protein kinase important for apoptosis, T-cell differentiation, and inflammatory responses. 146 Expression of MAPK8 was downregulated in SCD and Chuvash polycythemia, where the hypoxic response is upregulated. Association mapping with a focus on local regulatory polymorphisms in 61 patients with SCD identified expression quantitative trait loci (eQTLs) for 103 of the hypoxia-response genes. Among these findings, an eQTL of MAPK8 (rs10857560) was associated with precapillary PH in SCD (odds ratio: 13.8, P = 0.00036). The A allele of rs10857560 was associated with a further decrease in expression in SCD. Homozygosity for the A allele was present in all 14 of the patients with precapillary PH that we examined, suggesting that the dosage effect of the A allele on MAPK8 gene expression might contribute to the pathogenic mechanism of precapillary PH in SCD. 145
HYPOXIA, ENDOTHELIAL ALDOSTERONE SYNTHESIS, AND PULMONARY VASCULAR FIBROSIS
The functional consequences of CH or cell-free Hb on circulatory performance differ between the pulmonary and systemic vascular circuits. This observation is in agreement with a wide body of literature indicating cell-specific properties that delineate pulmonary endothelial and vascular smooth muscle cells from corresponding cell types in resistance blood vessels. Along these lines, a unique relationship between hypoxia, the vasoactive hormone aldosterone, and remodeling proteins in pulmonary endothelial cells was explored recently.
Vascular fibrosis is a cornerstone histopathologic feature of remodeled pulmonary arterioles that is observed across the spectrum of pulmonary vascular disease, including hypoxic lung disease. 147 Overactivation of hypoxia-dependent signaling in pulmonary vascular cells results in vascular and perivascular collagen deposition that decreases pulmonary circulatory compliance to promote PH and a predilection for right heart failure. 148 However, effective treatments to abrogate vascular fibrosis in patients with PH due to chronic hypoxic lung disease are not currently available. Thus, further understanding of the basic mechanisms by which hypoxia regulates vascular remodeling, particularly fibrosis, is required to identify novel treatments that improve on elevated rates of morbidity and mortality in at-risk patients.149–151
Endothelial-mesenchymal transition (EndMT) is a principal mechanism implicated in the development of perivascular myofibroblast proliferation that evinces collagen deposition in PAs. 152 Under hypoxic conditions or in the setting of hypoxia-independent upregulation of HIF signaling, as is the case in PAH, 153 EndMT induces a complex sequence of molecular events characterized by an acquired loss of cell-cell contact membrane proteins, such as vascular endothelial cadherin and PECAM-1 (platelet endothelial cell adhesion molecule), which permits dissociation of pulmonary artery endothelial cells (PAECs) and their migration inward toward the blood vessel lumen. In PAH, endothelial cell phenotype switching to myofibroblasts occurs in parallel to the histological rearrangement of PAECs in remodeled pulmonary arterioles and is linked to dysregulated bone morphogenetic protein receptor type 2 (BMPR-2) signaling, cell proliferation, and, presumably, fibrosis. 154 However, diethylaminoethylcellulose chromatography and other sensitive methods have confirmed detectable expression levels of (fibrillar) collagen III and the profibrotic protein connective tissue growth factor (CTGF) in PAECs under basal conditions.155,156 Importantly, however, CTGF–collagen III signaling is increased in PAECs exposed to hypoxia for 24 hours, 157 which is a time point well in advance of the 3–7-day duration necessary to complete EndMT. 152 Observations from coculture systems in vitro that demonstrate upregulation of fibrillar collagen synthesis in PASMCs and resident fibroblasts induced by PAECs overexpressing CTGF lend additional evidence indicating that EndMT is not required for vascular or perivascular fibrosis.
There are accumulating data to suggest that the mineralocorticoid hormone aldosterone contributes to the pathobiology of pulmonary vascular fibrosis that occurs concomitantly with chronic hypoxic lung injury. For example, in one study of 25 patients referred for right heart catheterization, pulmonary arterial plasma levels of aldosterone were increased in PAH patients compared to controls (595 ± 281 [mean ± SD] vs. 120 ± 42.3 pg/dL, P < 0.02) and correlated positively with hemodynamic measurements of pulmonary vascular remodeling, such as PVR (r = 0.72, P < 0.01) and transpulmonary gradient (r = 0.69, P < 0.02). 158 Findings from other groups support these observations and suggest further that elevated circulating levels of aldosterone correlate with disease burden in PAH patients. 159 In the rat monocrotaline (inflammatory), rat SU-5416/hypoxia (angioproliferative), and porcine vein banding (hypertrophic remodeling) experimental models of PAH, circulating aldosterone levels are increased, compared to those in control animals, by 4.2-fold (P < 0.05), 4.7-fold (P < 0.05), 160 and 2.5-fold (P < 0.05), 161 respectively.
Overactivation of the renin-angiotensin axis is a likely contributor to hyperaldosteronism in PAH, because pulmonary aldosterone levels associate inversely with cardiac output (r = −0.79, P < 0.005). 158 However, extra-adrenal steroidogenesis is also suggested by the finding that, aldosterone levels are increased in plasma-free saline-perfused lung tissue homogenates harvested from PAH rats, compared to those in controls in vivo (median: 100.0 [interquartile range: 87.3–113.4] vs. 183.1 [126.1–197.4] pg/μg protein, P < 0.04). 160 The possibility that local aldosterone synthesis in lung is relevant to patients is supported by data from a recent study in which hormone levels were measured across the pulmonary circulation in a cohort of controls (N = 18) and patients with mild PH and heart failure due to reduced left ventricular ejection fraction (HFrEF; N = 42) referred for invasive cardiopulmonary exercise testing. In that study, a transpulmonary (i.e., PA levels – radial artery levels) increase in aldosterone was also observed between controls and HFrEF patients at rest (7.5 [–54 to 40] vs. 61.6 [–13.6 to 165] ng/dL, P = 0.01) and during peak exercise (–20.7 [–39.6 to 79.1] vs. 25.8 [–29.2 to 109.3] ng/dL, P = 0.02), while peak volume of oxygen consumption (pVO2), which is a clinical measure of exercise reserve capacity, correlated inversely with PA (r = −0.31, P = 0.01) and radial (r = −0.32, P = 0.01) aldosterone levels. 162 Taken together, these observations are in agreement with previous findings suggesting paracrine functionality of lung tissue, as a transpulmonary difference has been reported for ET-1 and the vasodilator second-messenger molecule cyclic guanosine monophosphate (cGMP), as well as other vasoactive intermediates relevant to pulmonary vascular disease.163,164
The possibility of extra-adrenal aldosterone synthesis in the lung is supported further by data indicating that lung tissue homogenates, PASMCs, and/or PAECs express key intermediaries required for aldosterone biosynthesis from cholesterol, including steroidogenic acute regulatory protein (StAR),165,166 which promotes transport of cholesterol across the mitochondrial membrane to the site of cholesterol metabolism and thus catalyzes the first and rate-limiting step of aldosterone biosynthesis, as well as CYP11β2 (aldosterone synthase),160,167 which catalyzes the final step of aldosterone biosynthesis from 18-hydroxy-corticosterone. Hypoxia as a regulator of aldosterone bioactivity in the pulmonary circulation has been suggested by findings from population studies indicating that genetic variants coding for the angiotensin II receptor 1 (T174M polymorphism; 1166C/C) and CYP11β2 (T-344C promoter region) cluster at a greater frequency in patients with PAH 168 or those with a diagnosis of acute high-altitude pulmonary edema, compared to individuals tolerant of acute high-altitude exposure or high-altitude dwellers. 169
There is indirect evidence that hypoxia may affect aldosterone synthesis in PAECs through ET-1 signaling. For example, PAEC exposure to FIO2 of 1% for 24 hours was associated with a 2.8-fold increase in messenger RNA transcription of the ET-1 precursor pre-proendothelin 1, which corresponded to an increase in ET-1 protein expression at 24 and 48 hours. Although a number of mechanisms have been proposed to account for this effect, hypoxia-induced HIF-1 binding to the ET-1 hypoxia response element has been shown to correlate with increased ET-1 activity in various vascular cell lines.170,171 In turn, ET-1, which is an established aldosterone secretogogue in adrenocortical cells, has also been shown in PAECs to induce binding of the steroidogenic transcription factors PGC-1α and steroidogenic factor 1 (SF-1) to the promoter region of CYP11β2 that increases synthesis of aldosterone. 160 On the basis of observations indicating that StAR expression is increased in fibrotic pulmonary arterioles from mice exposed to CH, SU-5416/hypoxia-PAH rats, and patients with PAH, the direct relationship between hypoxia and aldosterone through StAR signaling has been assessed. Indeed, compared to normoxia, PAEC exposure to hypoxia (2% FIO2) for 24 hours increases aldosterone levels in the cell culture media significantly (187.2 ± 86.0 [mean ± SEM] vs. 376.0 ± 94.0 pg/μg protein, P < 0.02), as assessed by enzyme immunoassay and subsequently confirmed by liquid chromatography–mass spectrometry. 157 Hypoxia increased expression of the steroidogenic transcription factors c-Jun and c-Fos and induced their binding to the activator protein 1 (AP-1) site of the StAR promoter, whereas pharmacological antagonism of the AP-1 site or molecular inhibition of StAR prevented endothelial synthesis of aldosterone by hypoxia, providing one possible mechanism to account for regulation of steroid biogenesis by oxygen tension in PAECs. Interestingly, stimulation of c-Fos/c-Jun-StAR signaling by hypoxia was selective to PAECs, and in that study 157 it was not evident in systemic vascular cells, lung fibroblasts, or other cell types involved in pulmonary arterial fibrosis (Fig. 3). The mechanistic basis for this observation remains incompletely characterized but overall appears to be in line with expanding evidence demonstrating critical differences in the response pattern to hypoxia distinguishing pulmonary vascular (and right ventricular) cell types from their systemic (and left ventricular) cell type correlates. 172
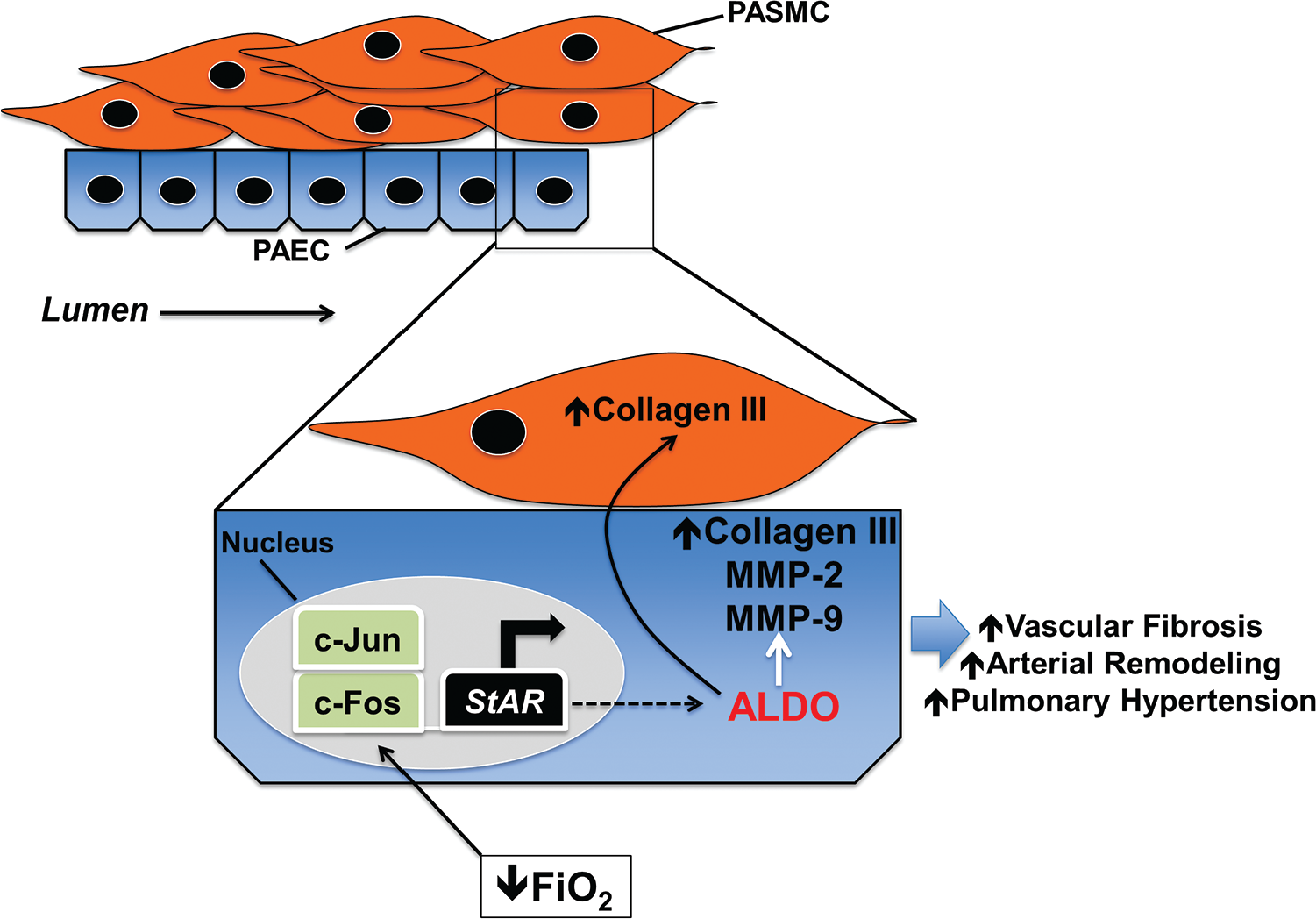
Hypoxia increases aldosterone (ALDO) synthesis in pulmonary endothelial cells to promote vascular fibrosis and pulmonary hypertension. In pulmonary artery endothelial cells (PAECs), chronic hypoxia induces binding of the transcription factors c-Fos and c-Jun to the activator protein 1 site upstream of the StAR promoter. Increased StAR expression and activity induced by hypoxia increase pulmonary endothelial ALDO production, which, in turn, upregulates expression of (fibrillar) collagen III and the remodeling proteins matrix metalloproteinase 2 (MMP-2) and MMP-9. Hypoxia-aldosterone signaling in PAECs is also associated with induced collagen III synthesis in pulmonary artery smooth muscle cells (PASMCs) in vitro and fibrotic vascular remodeling of distal pulmonary arterioles and pulmonary hypertension in vivo. FiO2: fraction of inspired oxygen; StAR: steroidogenic acute regulatory protein.
The relevance of aldosterone to the vasculopathy of hypoxia appears to involve activation of remodeling and fibrotic proteins. For example, pharmacological inhibition of the mineralocorticoid receptor with spironolactone or eplerenone prevented hypoxia-induced upregulation of matrix metalloproteinase 2 (MMP-2), MMP-9, CTGF, and collagen III in PAECs in vitro. Furthermore, administration of these pharmacotherapies, using disease prevention or reversal protocols, decreased distal pulmonary arteriole collagen burden significantly to improve PPA, PVR, right ventricular mass, and cardiac index in vivo in experimental models of hypoxic pulmonary vascular disease, including PH.160,173 Preston and colleagues 173 noted further that the inhibitory effects of mineralocorticoid receptor antagonism on fibrosis were extended beyond the pulmonary circulation and abrogated fully in the right ventricular interstitial compartment in HPH in vivo. These converging lines of evidence provide the rationale for several ongoing randomized clinical trials assessing the effect of mineralocorticoid receptor inhibition on clinical outcome in PAH patients. 174
CONCLUSIONS
The principal determinates of pulmonary vascular and right ventricular susceptibility include inherited predispositions, such as specific variants in GALNT13 or VHLR200W homozygosity that promote pathogenic interactions between red blood cells and the pulmonary arterial blood vessel wall, as well as HIF-dependent upregulation of ROCK and intracellular Ca2+ flux due to chronic exposure to hypoxia that induces a contractile phenotype in pulmonary arterioles. CH is also linked to upregulation of StAR through activation of the oxygen-sensitive transcription factors c-Fos and c-Jun that induces pulmonary endothelial aldosterone synthesis and vascular fibrosis. Taken together, these findings illustrate complex interplay between genetic, environmental, and hormonal factors that mediate pulmonary vascular injury, but they also identify numerous potential therapeutic targets to improve on increased rates of morbidity reported in patients with PH due to hypoxia, hemoglobinopathies, and other etiologies sharing related pathobiology.
Footnotes
Acknowledgments
We acknowledge the American Thoracic Society and Drs. Eric Austin and Gregory Eliot for the opportunity to contribute to the 2015 Grover Conference.
