Abstract
Patients present with a wide range of hypoxemia after acute pulmonary thromboembolism (APTE). Recent studies using fluorescent microspheres demonstrated that the scattering of regional blood flows after APTE, created by the embolic obstruction unique in each patient, significantly worsened regional ventilation/perfusion (V/Q) heterogeneity and explained the variability in gas exchange. Furthermore, earlier investigators suggested the roles of released vasoactive mediators in affecting pulmonary hypertension after APTE, but their quantification remained challenging. The latest study reported that mechanical obstruction by clots accounted for most of the increase in pulmonary vascular resistance, but that endothelin-mediated vasoconstriction also persisted at significant level during the early phase.
Keywords
More than 30 years ago, Eugene Robin commented on the problems of diagnosing acute pulmonary thromboembolism (APTE) by referring the physicians' attitudes to the emperor's vanity in Hans Christian Andersen's fable.1,2 Although significant advances have occurred over the years,3–5 some aspects of its pathophysiology remain puzzling. 6
This report provides an updated review of the literature, published mostly from 1980 onward, specifically addressing the mechanisms of hypoxemia and pulmonary hypertension (PH) after APTE. The emphasis is on the elucidation of the underlying pathophysiological principles and bringing them to the bedside, rather than on presenting meta-analysis or clinical trials. It also allows for occasional personal perspectives but has excluded recent works at the molecular level.
GAS EXCHANGE IN APTE
Hypoxemia is a common and nonspecific feature of APTE.7,8 It remains mysterious because the patient's arterial oxygen tension (PaO2) is often unpredictable and correlates poorly with embolic load.9–11 It is also associated with very different V/Q (ventilation/perfusion ratio) patterns measured by the multiple inert gas elimination technique (MIGET) among similar subjects 12 or in the same subject over time.12,13 The roles of anatomical shunts may be important among a minority of patients but are questionable in the rest. These variations in gas exchange are clinically relevant in a negative way, because blood gas results do not contribute to defining pretest probabilities in the interpretation of nuclear scans.1,14,15
The major purpose of this communication is to discuss the key factors that determine the degree of hypoxemia after APTE and their relevance to 5 commonly asked questions:
Why do patients with APTE present with such a wide range of hypoxemia?
Why do many patients with APTE have relatively clear chest X-rays while severely hypoxic?
Can patients with significant APTE present with relatively normal PaO2?
Why does hypoxemia worsen when cardiac output is increased?
Why do some patients with APTE have pulmonary infarcts and the others do not?
MECHANISMS OF HYPOXEMIA FOLLOWING APTE
Ventilation-perfusion mismatch as a key factor
Previous investigators have attempted to map V/Q abnormalities in the embolized lung. Burton et al. 16 showed the redistribution of regional blood flow (Q) versus regional ventilation (V), using nuclear scans, but their data lacked the required resolution. Others used radioactive macroaggregates to confirm that regional blood flow was significantly diverted from the embolized area but reported no data on ventilation.17,18 Results obtained from MIGET studies on APTE showed only abstract V/Q distributions based on a mathematical model. 12 In recent years, the fine images from computed tomography angiograms (CTAs) provided excellent information on the pulmonary vasculature but none on ventilation, even though there are now encouraging developments in single-photon-emission computed tomography (SPECT V/Q) that can facilitate more research in this field. 19
The simultaneous measurement of regional V and Q in small regions of the embolized lung was first accomplished more than a decade ago, by investigators who used a technique based on aerosolized and injected fluorescent microspheres (FMSs).20–22 Their data made it possible to reconstruct all regions of the embolized lung post mortem, with each region designated for its own V/Q ratio 23 and mapped in three-dimensional (3D) coordinates. All emboli locations were also marked. 24 Using appropriate computer software, their reports provided more reliable and comprehensive results than previous publications25,26 because of the direct measurements of V and Q in situ and the additional information that could be visualized on an anatomical model, e.g., dorsal versus ventral and hilar versus subpleural areas.
In an established porcine model of APTE and at different time points during the experiment, regional V and Q of the lungs were marked before and after APTE by FMSs of various colors and sizes, i.e., using 1-μm FMSs for aerosolization into the airway for labeling V and 15-μm FMSs for injection into the femoral vein for labeling Q. 24 Pigs were chosen for these experiments because of their strong pulmonary vasoconstrictive response to various physiological stimuli, such as hypoxemia. 27 They were studied in the prone position because it is their normal physiological posture. 24
At the end of the experiment, the lungs were removed from the thorax, the pulmonary vasculature was flushed with normal saline, and the inflated lungs were air-dried at room temperature for 3 days. The lungs were then sliced into about 1,000 pieces, with each sample (2 cm3) recorded for its weight, 3D location, presence of emboli, large airway, etc. (Fig. 1). The fluorescent intensities of all FMSs embedded in each sample provided the simultaneous V and Q data at the time of marking (Fig. 2). After the regional V/Q and measured gas tensions in the venous blood were obtained, the estimated pulmonary capillary O2 and CO2 tension in each sample could be calculated (see below),23,24 along with the information on its anatomical location in relation to the clots. Furthermore, the absolute amount of V and Q in each sample, in milliliters per minute, could be calculated when the cardiac output, minute ventilation, and dead space were known.
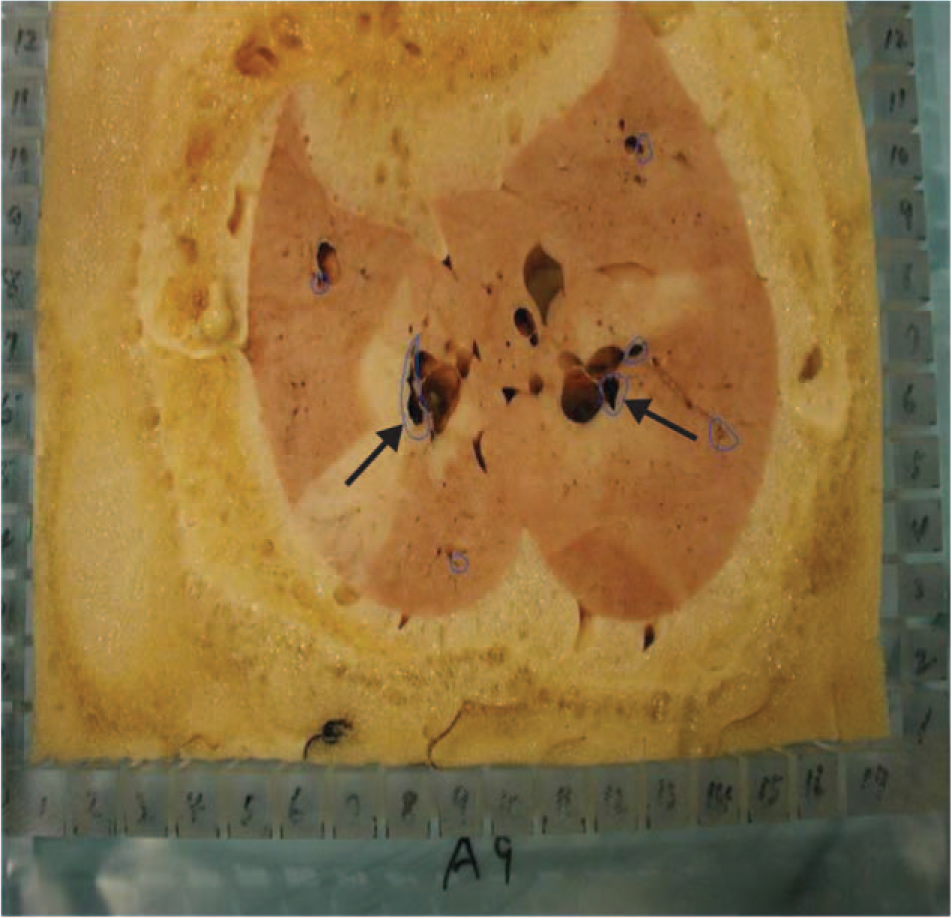
Cross section of the embolized lung embedded in a foam case. Each lung was then sliced vertically and horizontally into approximately 1,000 pieces of 2 cm3 each. The arrows point to the locations of clots.
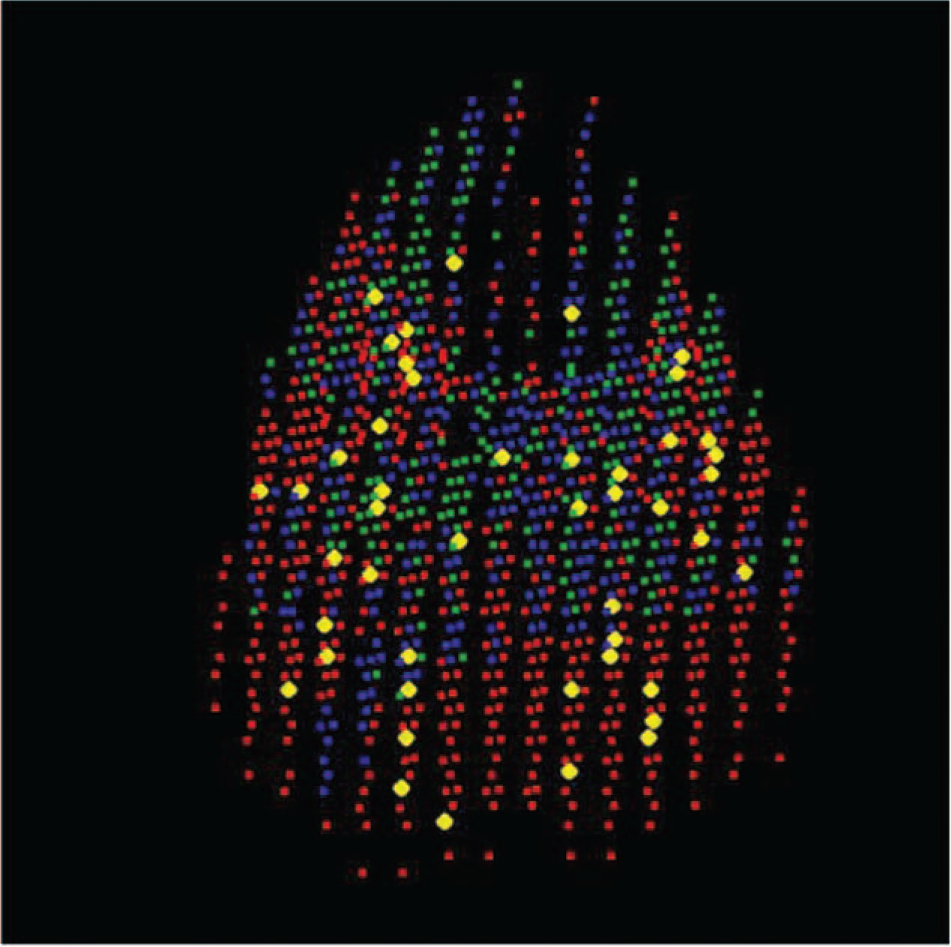
Red dots represent regions where the blood flow was reduced after acute pulmonary thromboembolism, blue dots regions where blood flow was maintained, and green dots regions where blood flow was increased. Yellow dots mark the locations of major emboli.
Because of the high affinity of oxygen to hemoglobin and the complete diffusion of oxygen across the alveolar membrane, the oxygen tension at the alveolar (PAO2), pulmonary capillary (PcO2), and arterial (PaO2) levels were considered equivalent for practical purposes, i.e., PAO2 = PcO2 = PaO2. Similarly, for CO2, because of its high diffusibility and solubility, PACO2 = PcCO2 = PaCO2, using the same notation.
The data showed that a considerable amount of the regional blood flow in the embolized lung was mechanically diverted to the less embolized regions, resulting in many low-V/Q units there. This was mainly due to the increased denominator (Q) in the V/Q of these regions, where V generally remained stable. 24 Small decreases in V/Q between good matching, or V/Q = 0.86, and V/Q = 0 could dramatically reduce PcO2, as the curvilinear line moved leftward along the flat portion of the O2-CO2 diagram (Fig. 3).28–30 (For more details, see the appendix.)
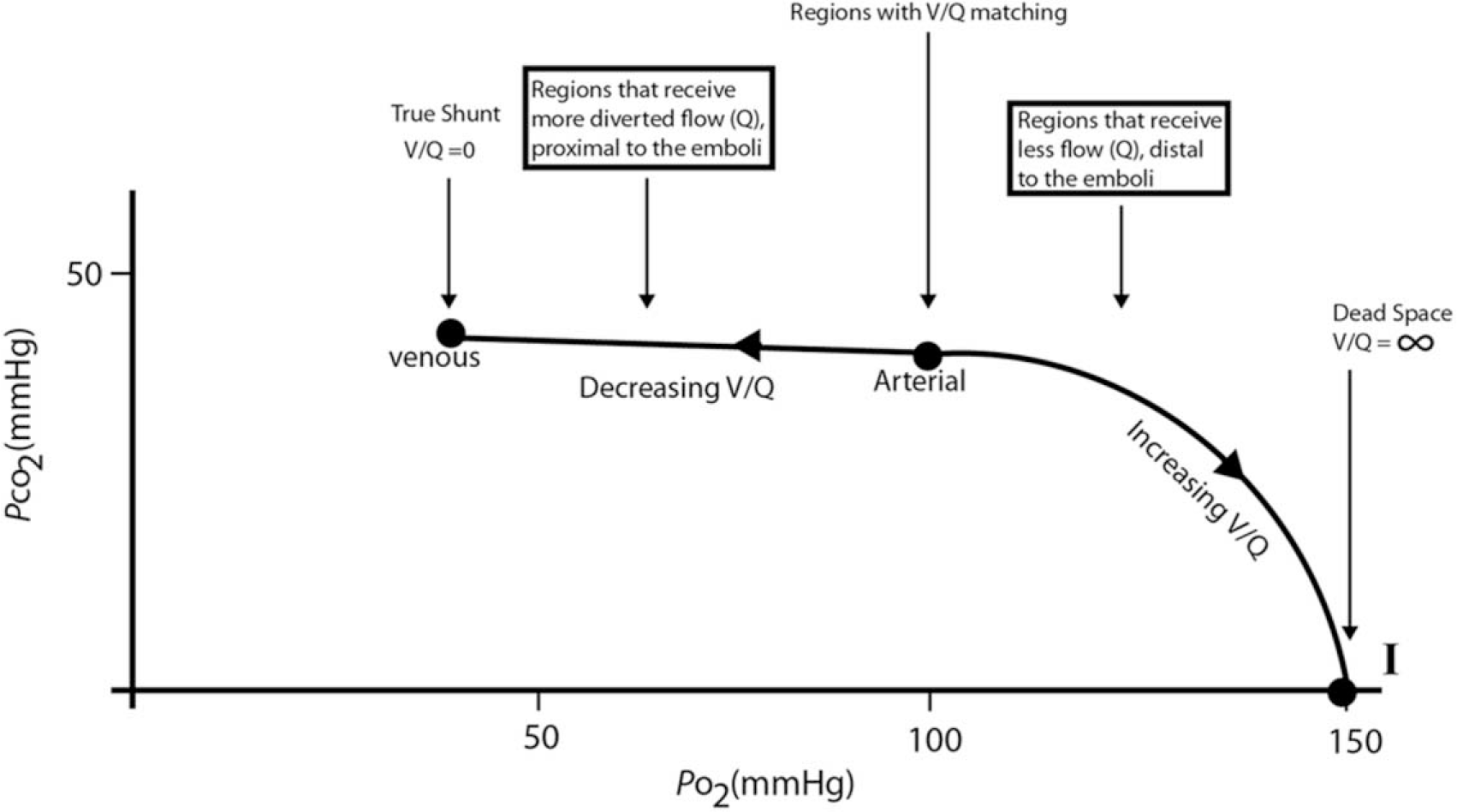
O2-CO2 diagram after acute pulmonary thromboembolism in room air. Normal arterial PO2 and PCO2 are marked at 100 and 40 Torr, respectively, with good ventilation-perfusion (V-Q) matching, representing V/Q at about 0.86. For practical purposes, PAO2 = PcO2 = PaO2, and PACO2 = PcCO2 = PaCO2 (i.e., alveolar, capillary, and arterial values are the same; see text). I: inspired gas point.
Thus, the most important mechanism of hypoxemia following APTE is the creation of these low-V/Q units as a result of the mechanical obstruction by the clots in situ.24,31 Simultaneously, high-V/Q units and physiological dead space are also created in areas where the denominator Q is much reduced, usually in regions distal to the emboli, i.e., the peripheral subpleural regions. These high-V/Q units, created in the way opposite that for the low-V/Q units, are described in the bent portion on the right side of the curve (Fig. 3) and have a greater affect on PACO2 than on PAO2. In short, there is a remarkable V/Q heterogeneity due to the scattering of Q in the presence of obstructing clots within the pulmonary vasculature. Even though V changes modestly in the same direction as Q, it does so to a much lesser extent. 24 APTE is a vascular disease and not so much a ventilation problem. 32 The observation that V remains comparatively stable compared to Q following APTE probably explains why many MIGET studies showed increases in low-V/Q units but little true shunt (V/Q = 0), because V is rarely 0 even at higher Q.33,34
Cardiac output as a confounding factor
Previous studies have shown that gas exchange often worsened with increasing cardiac output (Qt) after APTE. 34 However, this is opposite to what one may expect from Fick's equation, in that higher Qt in a steady state leads to improvement in venous oxygen tension (PvO2) and subsequently better PaO2 at a given venous admixture.
On the other hand, this contradiction is explained by an important and opposite mechanism, in which any increase in total pulmonary blood flow (or Qt) after APTE also results in a greater diversion of flow to the less embolized regions (with an already low V/Q), since the extra flow cannot not go beyond the obstructing clots. Consequently, the even lower-V/Q units in these open regions worsen the hypoxemia.
Manier and Castaing 35 reported that oxygen transport in patients with APTE was improved by dobutamine, mostly because of improved Qt, even though their concomitant PaO2 fell because of worsening V-Q mismatch shown in the simultaneous MIGET data. Others showed similar results using fluid and other vasopressors. 18 A recent study also confirmed that when Qt was increased intermittently by dobutamine after APTE, PaO2 did not improve and sometimes worsened. 36 Upon further analysis, this variation of hypoxemia at higher Qt was mostly accounted for by the interplay between the beneficial effect of higher PvO2 on PaO2 and the deleterious effect of worsening V-Q mismatch, caused by the maldistribution of regional Q, as demonstrated by the FMS data. 36
Earlier reports have shown that during severe shock following acute pulmonary embolism, the degree of hypoxemia did not reflect the severity of embolic obstruction. 37 To interpret these data in another way, when pulmonary blood flow becomes dangerously low, there is simply insufficient Q to create more low-V/Q units, and patient's blood gases may look less life-threatening than expected.
Thus, if a patient is being resuscitated during severe shock when PvO2 is extremely low, significant increases in Qt may result in dramatic improvement in PvO2 and subsequently PaO2. On the other hand, if the patient is hemodynamically more stable, higher Qt will result in even more Q to the less embolized regions, resulting in worsening hypoxemia by outweighing the positive effect of a slightly higher PvO2; i.e., the improvement in PvO2 in a stable patient is less useful, as it sits on the flatter portion of the hemoglobin-oxygen dissociation curve. Thus, Qt serves as a confounding variable on hypoxemia because it can affect PaO2 in either direction after APTE, depending on PvO2, which reflects on the adequacy of the systemic circulation.
Preexisting cardiopulmonary status as a contributing factor
Patients with preexisting cardiopulmonary diseases and disabilities are susceptible to APTE38,39 and readily suffer more hypoxemia beyond their compromised baseline conditions. Given their already lower PaO2 or PvO2 prior to this aggravation, any increase in V-Q mismatch or compromise in hemodynamics will result in a sharper decrease in PaO2, 40 as the oxygen saturation drops quickly along the steep slope of the hemoglobin-oxygen dissociation curve. 41
Because of its stealthy nature, APTE sometimes coexists with and is masked by the underlying conditions, such as exacerbation of chronic obstructive pulmonary disease (COPD),42–44 idiopathic pulmonary fibrosis, 45 congestive heart failure, 46 adult respiratory distress syndrome,47,48 and sepsis, 49 to name just a few.
Time as a forgotten factor
APTE is a dynamic disease. Hypoxemia occurs acutely at the onset but generally resolves toward the preembolized level over time, with or without treatment with heparin.12,24,50 This is partly due to the movement or breakdown of some nonfibrinized clots and partly due to the accommodation of the more recruited pulmonary vasculature, as regional blood flow becomes more evenly distributed. 24
Previous data obtained from MIGET were reported as histograms, showing V/Q distribution from 0 to infinity. They consistently showed that under normal conditions, the vast majority of V and Q occurred when V/Q was about 1, depicted as a narrow central peak on the graph. However, after APTE, the V/Q pattern changed immediately, with many V/Q units in the low (V/Q < 0.5) and high (V/Q > 10) ranges created where there were none before. However, over time these remarkable changes gradually became more homogeneous, with ratios in the extreme ranges disappearing and merging toward the middle, where a much broadened central peak now appeared, at V/Q ≥ 1. 50
Pulmonary edema, anatomical shunts, and atelectasis as minor factors
Data obtained from animal studies have shown only mild pulmonary edema following embolic injury.51,52 The extravascular lung water was 4.8 ± 0.84 g/g of dry lung, which was higher than the control value of 4.1 ± 0.5 53 but not in the range in which alveolar flooding resulted in significant hypoxemia.
Except in patients with significant heart failure, pulmonary edema after APTE is generally not a primary determinant of hypoxemia. Indeed, chest X-ray and CTA studies often fail to demonstrate pulmonary vascular congestion in most cases of APTE5,54 and sometimes show oligemia instead. 55 In fact, when clinicians encounter patients with severe hypoxemia and their unremarkable chest X-rays, they should suspect APTE as the cause of the hypoxemia.
Patent foramen ovale has been observed as an occasional cause of hypoxemia after APTE, and it has independent prognostic implications. 56 However, it is not common in the general population, and its relevance depends on the size of the foramen and concomitant right atrial pressure. Recent advances in ultrasound imaging have also revealed previously less well-known intrapulmonary shunts,57–59 but their roles in APTE specifically remain to be ascertained.
Atelectasis and pleural effusion are seen on chest X-ray in patients with isolated APTE, even without other comorbidities,54,60 such as trauma or malignancies. In many cases, the involved areas are too small to account for life-threatening hypoxemia. They generally play a minor role in the overall venous admixture and are unlikely to be physiologically significant. In fact, most of these patients hyperventilate with high minute ventilation. On presentation they have low PaCO2, 61 except in massive cases, 62 where the dead space is huge.
Baroreceptors, surfactant, and red blood cell transit time as speculative factors
Several ideas have been propagated through the years but not backed up by more thorough research. These speculations include changes in baroreceptor control,63,64 loss of surfactant,65,66 reduction in carbon monoxide transfer,67,68 and shortened red blood cell transit time (TRBC). 69 While the latter may be shortened because of high flow in some regions, it is always sufficient for diffusion of oxygen to hemoglobin across the alveolar epithelium.70,71 The calculation of this transit time, TRBC (s) = regional blood volume (mL)/regional blood flow (mL/s), is to be differentiated from the V/Q with no unit, which describes the relative intake of oxygen from V in relation to the removal of oxygen by red blood cells delivered by Q.
ALVEOLAR CO2 TENSION (PACO2) AND REGIONAL VENTILATION SHIFTS FOLLOWING APTE
Because of the increased V/Q heterogeneity after APTE, PACO2 also varies significantly. The arterial to end-expiratory CO2 gradient is often seen clinically. 72 Some investigators even suggested using this CO2 gradient to confirm the diagnosis. 73 At any rate, the alterations of PACO2 have their own pathophysiological effects in the embolized lung.
Previous studies reported that temporary unilateral pulmonary artery occlusion by inflation of a balloon-tipped catheter resulted in shifts of ventilation away from the obstructed side, where the overall V/Q was high.74,75 Furthermore, such changes could be mitigated by inhalation of CO2. 76 The proposed mechanism was presumably one of hypocapnic bronchoconstriction and reduction in parenchymal compliance (pneumoconstriction) in the affected lung (high V/Q), where local airway narrowing and increased tissue resistance were mediated by lowered PACO2.77,78 On the other hand, the contralateral lung, which received more diverted flow (Q), now had a lower overall V/Q and thus higher PACO2. This resulted in more bronchodilation and increased parenchymal compliance (pneumodilation), rendering this lung able to receive more ventilation. Thus, V/Q or PACO2 affected bronchial tone in a homeostatic way.
In the hypocapnic regions, the rise in tissue pH was mainly due to the decrease in blood flow and CO2 delivery. When acetazolamide, a carbonic anhydrase inhibitor, was given to slow the change in tissue alkalosis during vascular occlusion, it reduced the rate of ventilation shift. This observation further supported the concept of local regulation of ventilation by regional CO2. 79
These experimental findings were later extended to studies of APTE. Nadel et al. 80 reported that after barium sulfate microembolism, there was a shift in ventilation to the unaffected areas, resulting in more atelectasis of the embolized regions due to alveolar duct constriction. Levy and Simmons 81 also showed that there were again shifts in ventilation between different regions of the embolized lung, where blood clots created hypoperfused areas beyond the vascular obstruction, not unlike those created by the balloon-tipped catheters. They reported that regions of higher V/Q, and thus lower PACO2, redistributed ventilation away to the lower-V/Q regions and that these changes were again attenuated by inhalation of CO2. Using FMSs, Tsang et al. 82 showed that the magnitude of these ventilation shifts was related to the changes in PACO2, particularly when it went below 20 Torr. Note that these changes in regional ventilation were not caused by any mechanical factors, such as airway edema or secretions, as seen in other forms of acute lung injury.
Hypoxic pulmonary vasoconstriction and hypocapnic bronchoconstriction are both homeostatic mechanisms to improve V-Q matching in a diseased lung, with the former occurring in the vasculature, responding to regional O2 tension,83,84 and the latter occurring in the airway, responding to regional CO2 tension. The roles of collateral ventilation and bronchial circulation in the regulation of regional ventilation in APTE are not well studied but are arguably mitigated by their random distribution between the embolized and nonembolized regions.
Clinically, patients often hyperventilate, and yet regional atelectasis is still seen. 60 One may postulate whether these reductions in regional ventilation are caused by ventilation shifts near the embolized regions, rather than from loss of surfactant65,66 or splinting due to pleuritic pain. Whether these changes are potential sources of error in the interpretation of nuclear scan should be debated, as finding low V and Q at the same embolized area diminishes the diagnostic certainty reported by the radiologists.85,86
ANSWERING THE 5 COMMONLY ASKED QUESTIONS
Why do patients with APTE present with such a wide range of hypoxemia?
Clinically, blood gas results are nondiagnostic.87,88 Earlier studies reported that patients with APTE present with a wide range of PaO2 and very different V/Q patterns. 89 Some authors have proposed odd mechanisms, such as partial inhibition of hypoxic pulmonary vasoconstriction. 90 The actual reasons, with better hindsight, include (1) the unpredictable 3D locations of the clots, leading to infinite possibilities of regional blood flow diversion, thus creating a very different V/Q pattern each time;91,92 (2) the concomitant Qt acting as a confounding factor, which can range from hyperdynamic state to profound shock; (3) the time delay from the onset of disease; and (4) the simultaneous minute ventilation in compensation, which can also be highly variable. Furthermore, the preexisting conditions of the patients, such as cardiopulmonary diseases, obesity, postoperative pain, neuromuscular disorders, and presence of patent foramen ovale, introduce even more complexities.
Can patients with significant APTE present with relatively normal PaO2?
It is rare. Table 1 shows the favorable and unfavorable factors that can determine PaO2 after APTE. Unless all the favorable factors are lined up simultaneously, increases in the alveolar arterial oxygen gradient can be expected. Note that both saddle and subsegmental emboli can result in less mechanical scattering of pulmonary blood flow, even though their hemodynamic impacts are very different. To the extent that PvO2 becomes very low in central emboli, causing obstructive shock, PaO2 will still be depressed despite reduced diversion of blood flow. The explanation of normal PaO2 in massive APTE based on blockade of the prostaglandin pathway is unfounded. 10
Favorable and unfavorable conditions that determine the degree of hypoxemia after acute pulmonary thromboembolism
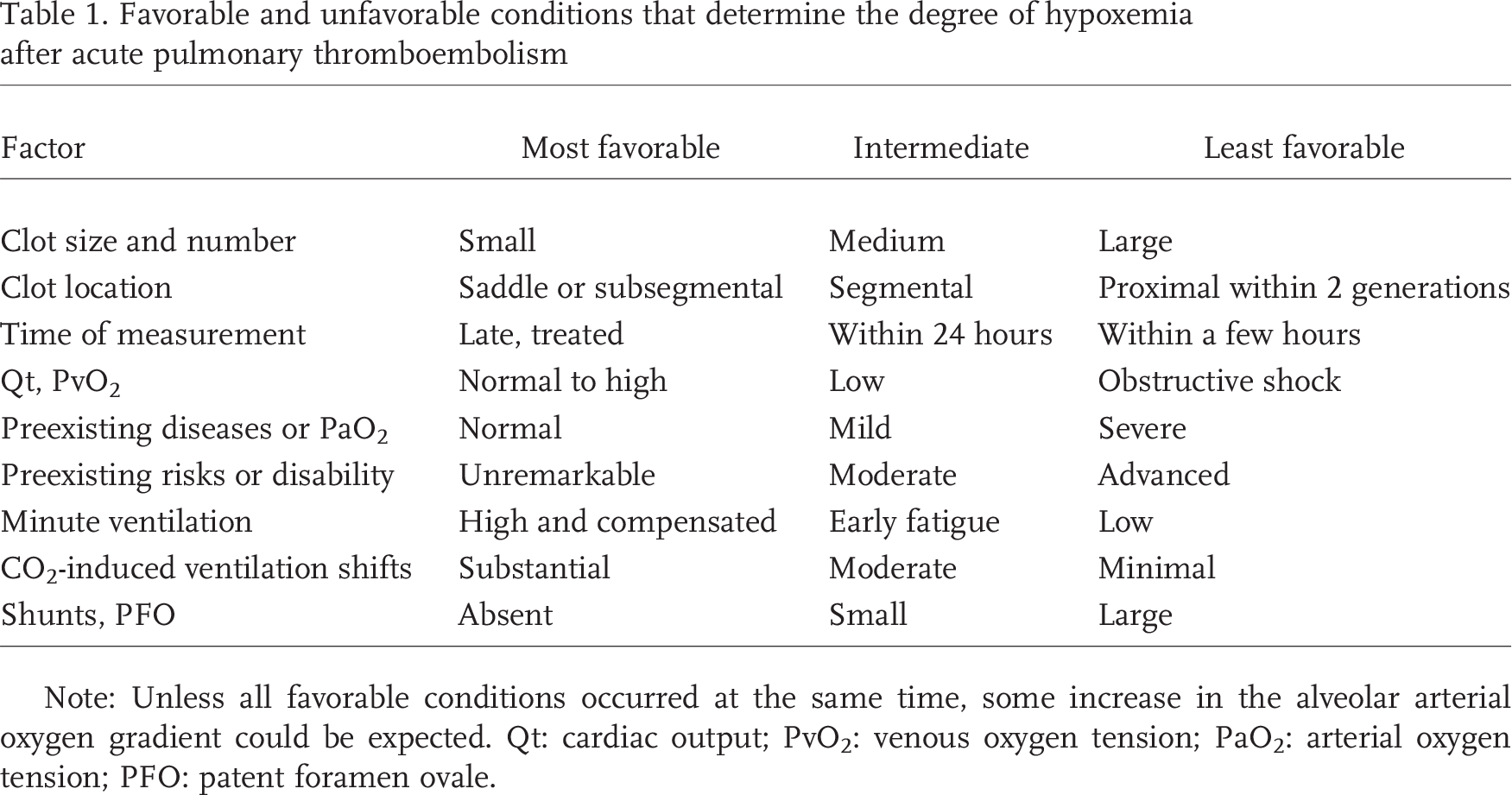
Note: Unless all favorable conditions occurred at the same time, some increase in the alveolar arterial oxygen gradient could be expected. Qt: cardiac output; PvO2: venous oxygen tension; PaO2: arterial oxygen tension; PFO: patent foramen ovale.
Why do many patients with APTE have clear chest X-rays while severely hypoxic?
Mathematically, V/Q is determined by both the numerator and the denominator. Lowering of V/Q can be achieved by reducing V, increasing Q, or both. Severity of hypoxemia is often intuitively interpreted with the amount of chest X-ray infiltrates, because the extent of pus, fluid, blood, or proteinaceous materials in the airspace correlates roughly to the reduction in alveolar ventilation by these materials (Fig. 4). 93 However, in the case of APTE, the opposite is true. The low V/Q is due to high Q rather than low V (Fig. 5). Thus, when V is maintained, the chest X-ray will be surprisingly unremarkable,54,55 despite significant hypoxemia. A V/Q scan 15 or CTA 94 will further confirm the diagnosis.
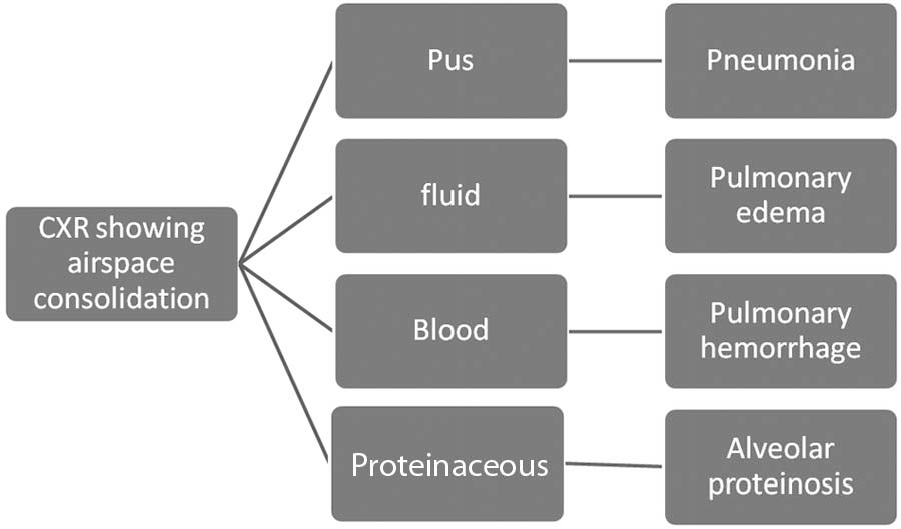
How hypoxemia occurs from low V/Q (ventilation/perfusion ratio) in the lung, mainly because of its low numerator (V), and how it can be intuitively correlated with infiltrates on chest X-ray (CXR), e.g., from pus, water, blood, or proteinaceous materials.
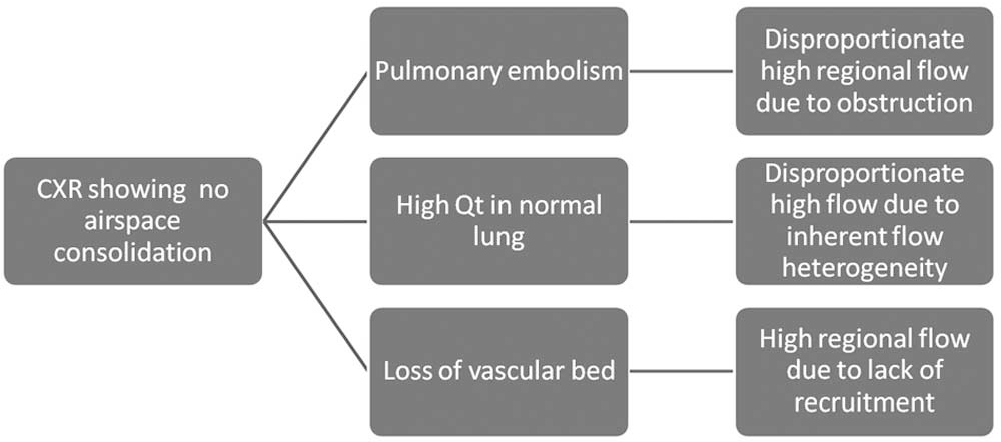
How hypoxemia occurs from low V/Q (ventilation/perfusion ratio) in the lung, mainly because of its high denominator (Q), and how it could be counterintuitive in an unremarkable chest X-ray (CXR), if V is maintained.
Why does hypoxemia worsen when Qt is increased?
Previous investigators reported that PaO2 correlated inversely with Qt in many different settings, i.e., under normal conditions 95 and in pulmonary edema, 96 COPD, 97 and APTE.25,35,98 Some suggested that this inverse correlation was due to the abolition of hypoxic pulmonary vasoconstriction due to the concomitant higher PvO2. 99 As it turns out, the answer lies mostly in the heterogeneity of regional blood flow.17,18
Despite the neat depiction of regional blood flow into different zones, as defined by pulmonary arteriolar pressure (Pa), alveolar pressure (PA), and pulmonary venous pressure (Pv),100–102 there is inherently a good deal of heterogeneity of Q even within the same zone, as defined by the Starling forces. Besides gravity, other determinants include radial gradient from the hilum, 103 metabolic factors such as PaO2 and PaCO2, 104 posture,105,106 temporal heterogeneity, 107 and uneven distribution of vascular resistance in arterial and venous beds.108–110
Given any preexisting perfusion heterogeneity, even in normal condition, it worsens with increased total flow. In other words, when Qt increases, the mean of all Qs within the lung increases. So does the standard deviation of Q; 69 i.e., some regions receive disproportionately higher flow at the expense of others. Thus, unless V can very precisely match Q in the millions of alveoli, an unlikely scenario, V/Q heterogeneity will increase at higher Qt. The culminating result is a higher alveolar arterial oxygen gradient.
Specifically in APTE, when total pulmonary blood flow or Qt increases, it will cause even more exacerbation of V/Q heterogeneity by diverting more Q to the already lower-V/Q units, because the remaining pulmonary vasculature is mechanically obstructed by the clots.17,18,24 Thus, PaO2 sometimes drops when Qt increases in APTE.
Why do some patients with APTE have pulmonary infarcts and others do not?
Pulmonary infarcts 111 after APTE are usually found in the subpleural regions because they are located beyond the embolic obstruction, where the regional blood flow is low. 24 They predispose to tissue ischemia, leading to necrosis.
In addition, the increased inflow of blood from the bronchial circulation under systemic pressure to these underperfused locations does not always allow good drainage into the pulmonary veins, especially when left atrial pressure is concomitantly high. Consequently, there is also venous infarct of congestion, stasis, and hemorrhage. 112 Reported risk factors for this complication include (1) lung cancer, in which Virchow's triad operates in situ, (2) left heart failure, in which the pulmonary venous congestion occurs, and (3) shock, in which the pulmonary and collateral flows are poor.113,114 Patients who have normal heart function are less likely to progress into the final stage of venous infarction. 112 The healthier patients with APTE who developed only pulmonary hemorrhage but stopped short of infarction, according to symptoms and follow-up chest X-rays, also recovered more quickly, usually within 7 days.
Interestingly, the use of vasodilators, such as nitroglycerin, hydralazine, nitroprusside, and prazosin, increased the risk of pulmonary infarct after APTE. 115 Some attribute this observation to the severity of the associated refractory heart failure that necessitated such treatments. However, it is possible that these drugs can cause vasodilatation in the pulmonary circulation, where regional blood flow becomes even more diverted to the unobstructed areas, thus causing more ischemia in the subpleural areas beyond the emboli and increasing the likelihood of pulmonary infarcts there. 115
PULMONARY HYPERTENSION (PH) IN APTE
PH occurs after APTE, as a result of the increased pulmonary vascular resistance (PVR), which is in turn due to the clot obstruction in the pulmonary vasculature and possibly the release of vasoactive mediators. Substances such as histamine, 116 serotonin,117,118 thrombin, 119 prostaglandins, 120 and the endothelins 121 have all been implicated. The observation that PH following APTE correlates poorly with embolic load 122 supports their roles. It is reasonable to suggest that the differing patterns of their release, in both timing and intensity,123,124 may partly account for the variations in the resulting pulmonary hemodynamics. However, others have asserted that the change in PVR was essentially the result of mechanical obstruction and have ruled out changes in pulmonary vascular tone, as suggested in a Starling model. 125 Research in this field became even more complicated because many concomitant variables that could confound the conclusions were not regularly reported, e.g., the degree of hypoxemia, 126 hematocrit, 127 Qt, 128 lung volume,129,130 fluid status, and time from onset.26,50 In this article, different views will be addressed, along with answering 5 additional questions to illustrate the controversies:
How should pulmonary vasoconstriction be estimated after APTE?
What is the temporal pattern of PH after APTE?
Does vasoconstriction also affect gas exchange after APTE?
Are vasodilator drugs harmful in the treatment of APTE?
Why does pulmonary arterial pressure (Ppa) usually reach a maximum of 40 Torr in healthy patients with APTE?
Pulmonary circulation is a low-pressure, high-flow system, 131 with a vast cross-sectional area and a huge capacity for recruitment at higher flow.132–134 In order to estimate the contribution of pulmonary vasoconstriction following APTE, we presume that the increase in PVR can be roughly divided into two major components, i.e., the obstructive component due to presence of clots and the vasoconstrictive component due the release of mediators.
Endothelins, 135 which affect both bronchial and vascular tone in the lung, have been the focus of considerable interest in the pathogenesis in PH since their discovery. They are omnipresent in the lung, along with their receptors,136,137 and are thought to be the key mediators in many forms of vascular lung injury, such as adult respiratory distress syndrome,138,139 primary 140 or secondary PH,141–143 scleroderma, 144 sepsis, 145 air embolism,146,147 and APTE.121,148,149 Because of their potency and universal nature in regulating vascular smooth muscle tone, some have asserted that other mediators implicated in APTE, including thromboxane 150 and serotonin, 151 manifest their vasoconstriction via the endothelin pathway.152–154 Lee et al. 121 showed that endothelin-1 was expressed in situ in the embolized lung and that its antagonist mitigated the subsequent PH.
In a porcine model of APTE, two groups of animals were induced with APTE, and their hemodynamics were measured in parallel over time, with one group receiving endothelin antagonist infusion and the other serving as controls. Other potentially confounding physiological parameters, including Qt, PaO2, PaCO2, lung volume, fluid status, and hematocrit or blood viscosity, were all kept relatively constant during the experiment in the same group and between groups. More importantly, consistent embolic loads and embolic patterns were achieved by injecting uniform-sized preformed clots into the lung until Ppa was about twice baseline value within 10 minutes. Their similar embolic patterns were subsequently documented by regional blood flow data using FMSs. The results showed that in the first 2 hours after APTE, the obstructive component accounted for 75% of the increase in PVR, while the vasoconstrictive component steadily accounted for 25%, when the Ppa approximately doubled after embolic injury (Fig. 6). 155
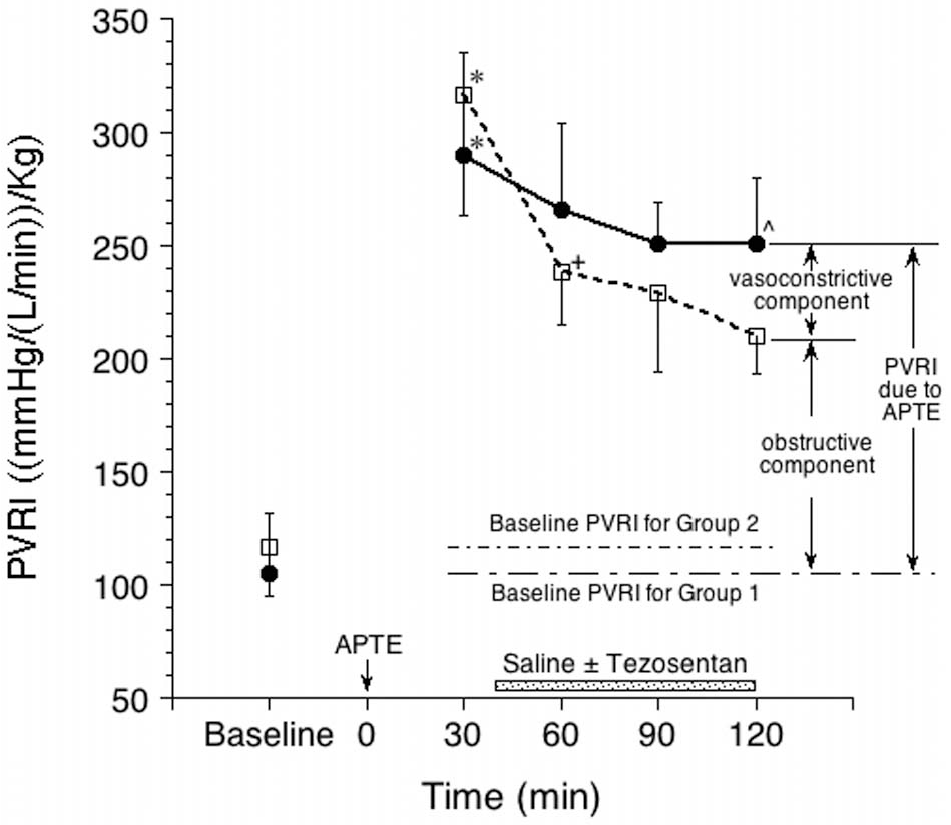
Endothelin-mediated vasoconstrictive component after acute pulmonary thromboembolism (APTE) accounted for about 25% of the total increase in the pulmonary vascular resistance index (PVRI). Group 1 is the control group; group 2 received tezosentan (see text).
ANSWERING THE 5 ADDITIONAL QUESTIONS
How should pulmonary vasoconstriction be estimated after APTE?
The animal models should mimic the clinical setting to be more relevant. Preferably, they would be set up in vivo with a closed chest, using minimal surgery and avoiding excessive vascular cannulation. 156 The premises of the models should not be based on heavy assumptions, nor should the conclusions be drawn from an arbitrary dichotomy between preset choices.125,157,158 Pigs should usually be chosen because of their stronger vascular reactivity to physiological stimuli such as hypoxemia. 27
Thromboemboli of standardized size and load,159,160 rather than glass or inert beads, should be used. The clots containing coagulation factors, e.g., thrombin, have inherent vasoactive properties, whereas the use of inert emboli compromises the purpose. 125 The pattern of embolization between groups should be documented by FMSs and post mortem markings 155 because it affects the obstructive component of PVR directly.
Animals of different sizes and species have different Qts and recruitable vascular beds in the lung. Since body weight (BW) is generally proportional to Qt, the normalization of Qt into cardiac index (CI, or Qt/BW) is preferred in the calculation of the PVR index: PVRI = 80 × (Ppa – Pw)/CI, where Pw is pulmonary capillary wedge pressure.
Because APTE is a dynamic disease, time is an unavoidable factor in affecting pulmonary hemodynamics because of the recovery nature of regional blood flow toward the preembolized state. Therefore, the hemodynamics in both control and experimental (using pharmacological antagonist) groups must be measured at the same time points, and the FMS data at each time point must be recorded to document how the regional flow pattern changes over time. If the obstructive pattern by the emboli is significantly different between groups immediately after APTE, then the subsequent data become moot.
In order to estimate PVR after APTE, all relevant parameters must be held steady as much as possible to avoid their confounding effects. They include Qt, PaO2, PaCO2, pH, hematocrit, and fluid status. Lung volume should be maintained throughout by mechanical ventilation and positive end-expiratory pressure. No vasopressor should be used. Thus, if one uses Ppa as the key parameter in drawing conclusions about PVR 161 without taking all the rest into considerations, the conclusion will be on questionable ground.
What is the temporal pattern of PH after APTE?
In our experiments, Ppa acutely increased as embolization began, and then it leveled off to a plateau level. 155 It did so after each additional load of clots, with the peak Ppa often exceeding 40 Torr temporarily, followed by a subsequent plateau below 40 Torr. These patterns were compatible with those of Smulders 124 The temporal profile of Ppa correlated well with the release timing of vasoactive mediators. Tsang et al. 148 reported a biphasic release of immunoreactive endothelins. These intense but short-lived increases in Ppa within minutes of APTE were likely due to a combination of severe initial vasoconstriction and the maximal impact of embolic obstruction prior to any accommodation of the pulmonary vasculature to the clots or their movement. Acute right ventricular failure occurred as a consequence of such a sudden increase in afterload, which resulted in paradoxical septal motion, leading to an abrupt compromise in left ventricular function. These chain reactions could have fatal consequences, often in the first hour if left untreated. 162 Gradual accumulation of multiple pulmonary emboli in the lung would likely be better tolerated than the sudden arrival of a large one with the same total mass.
Does vascular tone affect gas exchange after APTE?
Previous investigation using a nonspecific endothelin antagonist to attenuate vasoconstriction did not show a beneficial effect on V-Q mismatch after APTE. 163 Actually, the data showed a negative trend toward a slower recovery of V/Q heterogeneity in the tezosentan treatment group, compared to controls. This was presumably due to the persistently higher flow to the unobstructed and vasodilated areas, where V/Q was already low. Thus, the relative vasodilatation under the influence of tezosentan compounded the V/Q heterogeneity and worsened gas exchange. In contrast, vasodilatation in locations beyond embolic obstruction could not allow more flow there as long as the existing clots were in place.
It has also been reported that potent vasodilators had an adverse effect on oxygenation in other forms of acute lung injury, e.g., adult respiratory distress syndrome 164 and obliterative PH. 165 Some researchers have suggested that maintaining pulmonary vascular tone could improve V-Q matching in these conditions and have advocated against mitigating the existing vascular tone by vasodilators. In order to decide whether potent vasodilators are useful in a given setting, the systemic oxygen delivery must be obtained. First, do no harm.
Are vasodilator drugs harmful in the treatment of heart failure after APTE?
Yes. First, there are very few truly specific vasodilators for the pulmonary circulation. Many cause systemic hypotension and have minimal flow-independent effect, e.g., hydralazine, nitroprusside, and even dobutamine.166,167 Second, if patients are either underfilled or have ventricular septal shift, using this class of drugs causes more harm than good and complicates fluid therapy, not to mention the decreased coronary perfusion in a hypotensive state. The more relevant resuscitation end points are optimal Qt and systemic blood pressure, rather than Ppa per se. Third, some vasodilators have been shown to increase the risk of pulmonary infarcts. 115
Recent study has suggested that epoprostenol did not change right ventricular diameter in patients with APTE. However, these measurements were somewhat indirect in relation to the primary end points, such as Qt and systemic blood pressure. Transthoracic echocardiogram estimation of right ventricular size and flow were not as precise as those for the left ventricle. 168 Nevertheless, there was no benefit.
Why does pulmonary arterial pressure (Ppa) generally reach a maximum of 40 Torr in healthy patients with APTE?
It has often been observed that among otherwise healthy patients suffering from APTE, Ppa rarely exceeds 40 Torr.169,170 This phenomenon is usually explained by the fact that the thin-walled right ventricle (RV) cannot suddenly generate sufficient pressure on demand to overcome the increased afterload. On the other hand, patients with previous cor pulmonale can do so because their muscular RV is hypertrophied over time.
However, there is another consideration. Pulmonary circulation, with its normally large cross-sectional area, can readily accommodate increased flow in many regions. Thus, this large reserve capacity can attenuate the rise in Ppa by progressively recruiting more and more vasculature. In healthy subjects suffering from APTE, this recruitment will be limited to the nonembolized regions, but the reserve is usually still sufficient. Consequently, Ppa reaches a certain limit, say 40 Torr, only because any further increase in Ppa is mitigated by further increases in recruitment, either by distension or by opening of the previously closed vessels as the critical opening pressure is reached. On the other hand, if Qt is so low during shock from a large embolic load, Ppa will not be high, in accordance with the Poiseuille Law.
In addition, if the patient has a significant reduction of recruitment potential due to the loss of pulmonary vasculature, e.g., emphysema, 171 the Ppa can reach a higher level after APTE. Similarly, in patients with mitral stenosis or congestive heart failure, their fully recruited pulmonary vasculature becomes stiffer, and their Ppa also can go even higher than 40 Torr when some regional flow is increased after APTE. 34
CONCLUDING REMARKS
The remarkable variability of hypoxemia in APTE is explained by the differing embolic sizes, loads, and 3D locations that result in an infinite pattern of regional blood flow diversion and V/Q heterogeneity. Two key physiological principles account for the subsequent hypoxemia, i.e., (1) low-V/Q units are created by the increase in the denominator of that ratio, or Q, in the less embolized areas, and (2) PaO2 is extraordinarily sensitive to any decrease in V/Q between 1 and 0, to the extent that this same change exceeds that when V/Q decreases from infinity to 1. It is further confounded by the concomitant Qt and a patient's preexisting cardiopulmonary status. Generally speaking, pulmonary edema and occasional atelectasis are minor contributing factors, while incidental anatomical shunts are relevant only in some cases. Ideas involving baroreceptor, surfactant, ventilator control, and red blood cell transit time are speculative. Ventilation shift within the embolized lung mediated via hypocapnic bronchoconstriction is a homeostatic mechanism but generally does not affect oxygenation much.
In terms of pulmonary hemodynamics after APTE, the lack of vascular accommodation and severe pulmonary vasoconstriction immediately after APTE explains the disproportionately high PVR and shock among some patients during the early phase, leading to their higher mortality in the first hour. It subsides to a steadier but still significant level afterward. Endothelin-mediated vasoconstriction accounts for about 25% of the total increase in PVR. Unfortunately, the literature on this topic is confusing, because of the variable experimental designs, use of inert embolic materials, invasive animal preparations, and neglect of controlling for time in this dynamic disease. Any use of vasodilators in treating PH must be discouraged, as they can do more harm than good. Patients have died of obstructive shock, and definitive relief is achieved by timely anticoagulation or judicious thrombolysis. The value of Ppa by itself is not prognostic, unless it is interpreted with other key data, such as Qt.
This update is made possible by the tremendous contributions by many pioneers in the field. Reflecting on Dr. Robin's comment at the beginning of the article, readers can draw their own conclusions as to whether the emperor finally has got some new clothes. Much remains to be learned.
Footnotes
APPENDIX
ACKNOWLEDGMENTS
We thank Mr. Wayne Lamm, at the University of Washington, for his insights in the preparation of the manuscript.
