Abstract
Pulmonary hypertension (PH) is a disorder that develops as a result of remodeling of the pulmonary vasculature and is characterized by narrowing/obliteration of small pulmonary arteries, leading to increased mean pulmonary artery pressure and pulmonary vascular resistance. Subsequently, PH increases the right ventricular afterload, which leads to right ventricular hypertrophy and eventually right ventricular failure. The pathophysiology of PH is not fully elucidated, and current treatments have only a modest impact on patient survival and quality of life. Thus, there is an urgent need for improved treatments or a cure. The use of animal models has contributed extensively to the current understanding of PH pathophysiology and the investigation of experimental treatments. However, PH in current animal models may not fully represent current clinical observations. For example, PH in animal models appears to be curable with many therapeutic interventions, and the severity of PH in animal models is also believed to correlate poorly with that observed in humans. In this review, we discuss a variety of animal models in PH research, some of their contributions to the field, their shortcomings, and how these have been addressed. We highlight the fact that the constant development and evolution of animal models will help us to more closely model the severity and heterogeneity of PH observed in humans.
INTRODUCTION
Though often referred to as a disease, pulmonary hypertension (PH) is probably most accurately described as a pathophysiological parameter defined by certain hemodynamic measurements, including pulmonary arterial systolic pressure greater than 35 mmHg, mean pulmonary arterial pressure (mPAP) greater than 25 mmHg at rest or 30 mmHg with exercise, pulmonary capillary wedge pressure less than 15 mmHg, and a pulmonary vascular resistance greater than 3 Wood units.1,2 PH leads to an increase in right ventricular (RV) afterload and hypertrophic remodeling that eventually causes RV failure.3,4
PH is associated with infectious diseases, such as human immunodeficiency virus/acquired immune deficiency syndrome (HIV/AIDS), schistosomiasis, and viral hepatitis, as well as chronic noncommunicable conditions such as sickle cell disease, systemic sclerosis, congenital heart defects, and chronic obstructive pulmonary disease. 2 It is heterogeneous because of its etiology, gender differences (in response to treatment), epidemiology, and variations in mortality/survival rates.5–7 PH remains a deadly disease with a 3-year mortality rate between 20% (in PH associated with congenital heart disease) and 80% (in PH associated with HIV or connective-tissue disease). 8
Despite significant advancement in the management of PH in the past 12 years, it remains without cure, which is a major concern because it affects a significant part of the population.9–16 Unfortunately, current treatments improve PH only to a certain extent and do not afford permanent reversal of pulmonary vascular remodeling or reduction of mPAP.9–16 The absence of a cure and the limited success of current PH treatment are mainly due to the fact that the pathophysiology of PH is poorly understood. Animal models aid in the understanding of the disease pathophysiology because they provide the ideal platform to investigate the pathophysiological processes that underlie the disease progression. Animal models are also useful for testing novel experimental treatments.
There are several animal models available to study PH (see Table 1). We describe these animal models as single-pathological-insult (SPI) models, multiple-pathological-insult (MPI) models, knockout models, and overexpression models. In SPI models, PH is induced by a single pathological insult, such as monocrotaline or chronic hypoxia. In MPI models, multiple insults are combined, such as chronic hypoxia and SU5416, in order to induce severe PH. Concerns were raised that SPI models do not completely display the severity of PH observed in humans with respect to histological and/or hemodynamic parameters. However, we would like to suggest the possibility that PH in humans may already be late in the disease progression and that to compare this to the severity of PH observed in animal models is incorrect. Although animal models do not fully recapitulate the severity of full-blown human PH, they may very well correlate with milder forms of human PH, a stage that is often missed at the time of diagnosis. 17 Nevertheless, animal models have undergone major developments and improvements over the years, and MPI models appear to correlate better with PH in humans. In this review, we discuss the roles of various animal models in PH research, describing how they have contributed to the current understanding of the pathogenesis of PH and how they evolved over time. We also highlight the importance of MPI models as an improvement on SPI models of PH, and we comment on the future of translational research in the field of pulmonary vascular disease.
Overview of various animal models in pulmonary hypertension (PH)
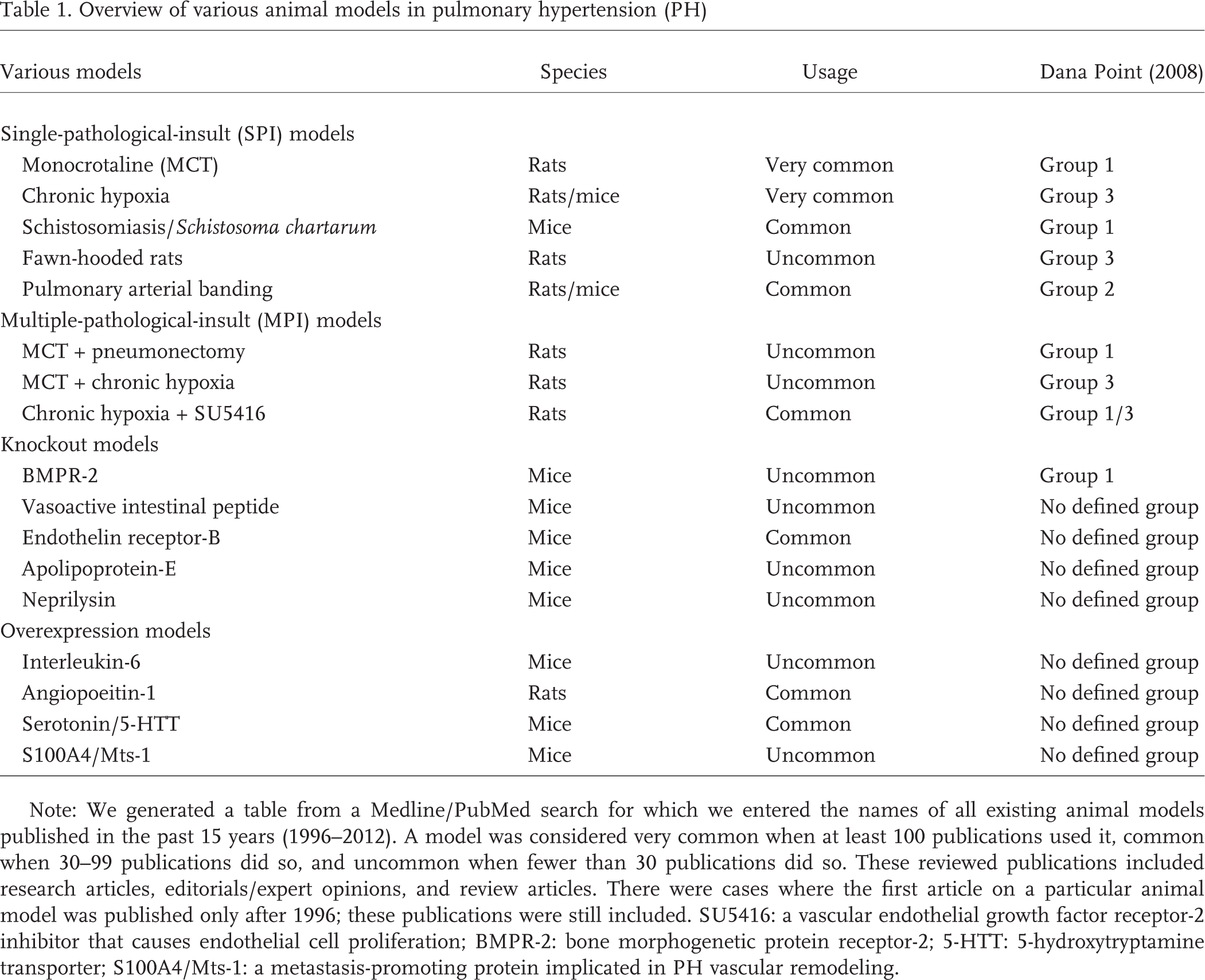
Note: We generated a table from a Medline/PubMed search for which we entered the names of all existing animal models published in the past 15 years (1996–2012). A model was considered very common when at least 100 publications used it, common when 30–99 publications did so, and uncommon when fewer than 30 publications did so. These reviewed publications included research articles, editorials/expert opinions, and review articles. There were cases where the first article on a particular animal model was published only after 1996; these publications were still included. SU5416: a vascular endothelial growth factor receptor-2 inhibitor that causes endothelial cell proliferation; BMPR-2: bone morphogenetic protein receptor-2; 5-HTT: 5-hydroxytryptamine transporter; S100A4/Mts-1: a metastasis-promoting protein implicated in PH vascular remodeling.
THE ANIMAL MODELS USED IN PH RESEARCH
Monocrotaline model
Monocrotaline (MCT) is an alkaloid from the plant Crotalaria spectabilis and a constituent of an herbal tea used by natives in the West Indies many years ago, and it is known for its ability to cause hepatotoxicity and PH.18,19 In 1967, a model of PH was established by feeding rats the seeds of C. spectabilis or by injecting nonhuman primates with a suspension of MCT.20–23 It is now general practice to induce PH in rats with a single subcutaneous/intra-peritoneal injection of MCT (60–80 mg/kg). 24 The mechanism whereby MCT causes PH includes metabolism of MCT in the liver by the enzyme cytochrome-P450 into pyrrolic derivatives that initiate endothelial injury in the pulmonary vasculature. 25 The endothelial injury is the initial trigger for pulmonary vasculitis and obstructive pulmonary vascular remodeling, characterized by narrowing/obliteration of the vascular lumen. Histological investigation of the pulmonary vasculature in PH shows features such as intimal hyperplasia, medial hypertrophy, and adventitial thickening (see Table 2).26,27 PH in the MCT model is also characterized by increased apoptosis of endothelial cells, proliferation of pulmonary arterial smooth muscle cells (PASMCs), and resistance of PASMCs to apoptosis.21–25
Broad overview of the main histological features of pulmonary hypertension (PH) within the different groups of human PH

The MCT model is commonly used by researchers, is reproducible and inexpensive, and does not require meticulous technical skills. 24 Therefore, one of the greatest benefits of this model is that it has helped us to understand the pulmonary vascular remodeling process and its pathophysiology. This was mostly shown by studies that explored the role of a genetic mutation in genes that are responsible for the regulation of endothelial cell and PASMC apoptosis and proliferation. Researchers discovered a mutation in the gene that encodes for bone morphogenetic protein receptor-2 (BMPR-2) and forms part of the transforming growth factor superfamily of proteins.28–31 Normally, transforming growth factor-β (TGF-β) signaling is initiated by signals transmitted from smad (small mothers against decapentaplegia) molecules and binding of TGF-β ligands to the TGF-β receptor.32–34 Subsequently, smad 2,3 or smad 1, 5, and 8 are phosphorylated and collocalize with smad 4, which translocates to the nucleus, where it modulates transcription of target genes (see Fig. 1). Furthermore, with the use of the MCT model, the TGF-β receptor, activin-A receptor-like kinase-1, and expression of smad 3,4 were shown to be reduced in the lungs of MCT-treated rats. This was associated with a decrease in the expression of the full BMPR-2 protein in the lungs of MCT rats, suggesting that TGF-smad and BMPR-2 signaling is impaired in MCT-induced PH, a characteristic shared with human PH.32–34
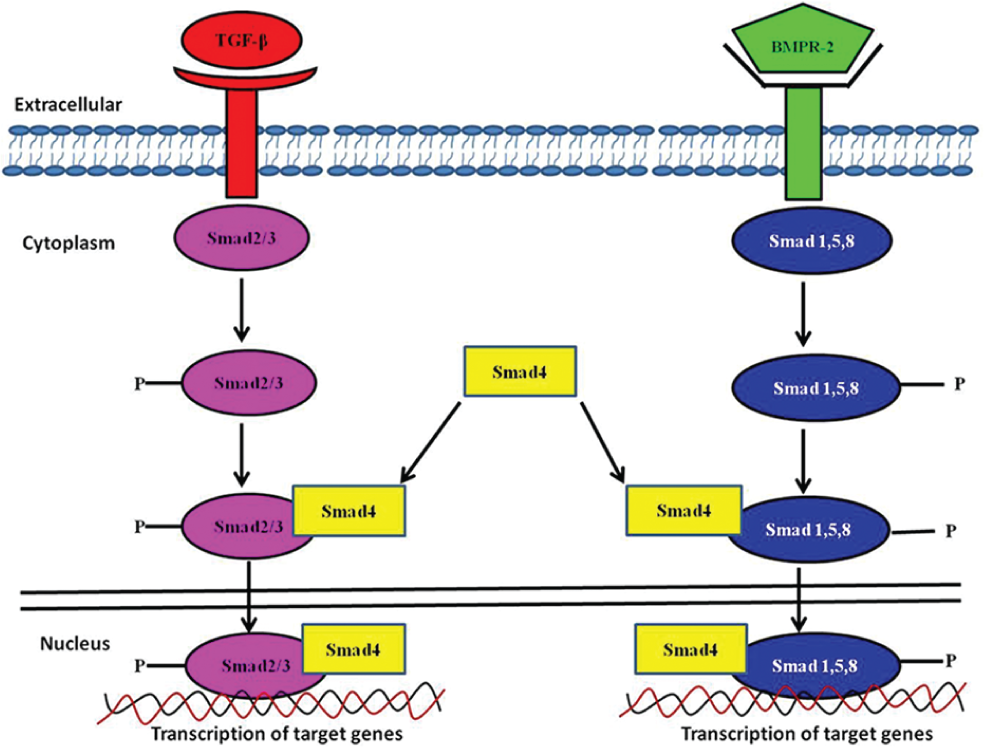
TGF-β/BMPR-2 signaling axis in pulmonary hypertension (PH): a simplified schematic diagram of the TGF-β and BMPR-2 signaling pathways that underlie the pathogenesis of PH. TGF-β and BMPR-2 bind to their receptors, where TGF-β activates smad 2,3 and BMPR-2 activates smad 1,5,8. Upon phosphorylation of these smad molecules, the smad 4 molecule is activated by both pathways and translocates to the nucleus, where it promotes transcription of target genes involved in cell growth, proliferation, and apoptosis. BMPR-2: bone morphogenetic protein receptor-2; TGF-β: transforming growth factor-β; smad: small mothers against decapentaplegia. Adapted from Santibañez et al. 186
Other studies done in the MCT model also highlighted the pivotal role of inflammatory cells (macrophages, dendritic cells, and mast cells) and cytokines (interleukin-6, interleukin-1) in the early stages of pulmonary vascular remodeling observed in PH.35–37 Macrophages and dendritic cells, both antigen-presenting cells, are believed to facilitate the inflammatory response in PH, thereby contributing to pulmonary vascular remodeling.38–40 Mast cells are specialized myeloid hematopoietic cells that are proposed to have direct vasoactive effects and to stimulate remodeling by increased production of matrix metalloproteinases.38–40 The exact role of these inflammatory cells in PH is not fully elucidated.38–40 A very attractive concept in the development of therapeutic treatments is the use of gene and cell therapy, which is now also considered in PH.41,42 The principle of gene therapy is that genes that are either not expressed or overexpressed are therapeutically stimulated or inhibited.41,42 This has been tested in the MCT model and is of particular interest because many genes are differentially expressed in PH.43–46 Hamidi et al. 47 showed that administration of the vasoactive intestinal peptide gene significantly reversed and completely prevented MCT-induced PH. Similar success has been achieved with therapy that modulates the expression of the angiopoietin-1, superoxide dismutase, and interleukin-1 genes.37,48,49 Cell therapy, in particular stem cell therapy, is also useful in PH because this may restore or replace damaged cells in the pulmonary vasculature.50–52 Various methods of administering exogenous cells have been investigated, including intravenous, intratracheal, and direct implantation into the lung.50–52 Cell therapy using mesenchymal stem cells is considered as a treatment for PH because of the potential of these cells to differentiate into other cell types and to secrete paracrine factors that may initiate tissue repair. 52 The MCT model was also used to test mesenchymal stem cell therapy; the result was significant improvement of RV function. 52
One major shortcoming of the MCT model is that most experimental treatments seem to improve, reverse, and prevent pulmonary vascular damage and PH. Experimental treatments that have been tested in this model include serine elastase inhibition (M249314 or ZD0892), platelet-derived growth factor inhibition (imatinib), Rho-kinase inhibition (fasudil), endothelin receptor antagonists (endothelin-1), serotonin transporter inhibition (fluoxetine), phosphodiesterase-5 inhibition (sildenafil and phosphodiesterase-3/4 inhibitors such as pumafentrine), statins (simvastatin, pravastatin, and rosuvastatin), and many others.53–64 The excessive degree of improvement with these experimental treatments has been criticized in the literature because it is believed to reflect imperfect models, inadequate duration of the studies, and endpoints that do not correlate with the progression of PH observed in humans.26,27 Gomez-Arroyo et al. 24 suggest that the MCT model harbors an MCT syndrome consisting of pulmonary interstitial edema, myocarditis, and hepatic veno-oclusive disease that is uncharacteristic of severe human PH. 24 Furthermore, in the MCT model, vasoconstriction seems to be an important mechanism, whereas this is found only in a subset of PH patients. 24 It is clear that the MCT model does not truly mimic severe PH as observed in humans. 24 This issue is partially addressed by MPI animal models in which PH is induced by combining pathological insults. 24 One such example is when PH is induced by combining MCT with contralateral pneumonectomy. 64 As opposed to SPI models, MPI models display features of severe PH, such as excessive RV hypertrophy (RVH) and increased hemodynamic measurements (such as mPAP and RV diastolic and systolic pressures). 64 Furthermore, in the MCT model, the disease progression toward death might be too short for compensatory mechanisms to develop, and this has been addressed by reducing the dose of MCT (30 mg/kg) that causes compensated RVH. 46 Studies like these show that the MCT model has undergone important evolutionary developments that has definitely allowed it to better model PH observed in humans.
Chronic hypoxia model
In the early 1970s, researchers initiated experiments that showed that chronic hypoxia (CHP) leads to PH in several animals. 65 CHP is observed in chronic obstructive pulmonary disease, interstitial lung disease, sleep apnea, and exposure to high altitudes and is thought to play a crucial role in the development of PH.65,66 The CHP model is assumed to be representative of group 3 (PH secondary to lung disease and/or hypoxia) in the Dana Point classification.26,27 In a typical model of CHP, rats are placed in a hypobaric chamber (10% Fio2 for 3–4 weeks or hypobaric pressure of 380 mmHg, which is equivalent to half that at sea level).66–70 A number of experimental treatments have been shown to attenuate PH in this model, including digoxin, A-17 (an inhibitor of microRNA-17), hypercapnia at a CO2 saturation of 6.5% combined with CHP (10% Fio2), bosentan, dichloroacetate, and targeted gene delivery of BMPR-2.43,55,67,70–72
The CHP model has greatly contributed to our current understanding of the molecular processes involved in the vascular remodeling induced by chronic pulmonary disease and oxygen deprivation.65–72 In the CHP model, it was recently shown that there is an impairment of the signaling pathway that induces vasodilatation, irrespective of an increase in inducible nitric oxide synthase expression.71–74 This is believed to contribute to CHP-induced PH and may have major implications for PH associated with chronic obstructive pulmonary disease. This model remains relevant but, as is the case with most animal models, it is questioned whether CHP really models human PH.75–78
A shortcoming of the CHP model is that it does not fully recapitulate the pulmonary vascular damage observed in humans with PH.76–78 Furthermore, the CHP model does not display obstructive intimal lesions in the peripheral pulmonary arteries.76–78 CHP is used to induce PH, but it rarely results in PH of the same severity observed in humans, and the model should, therefore, be further developed.76–78 In an attempt to improve this model, Morimatsu et al. 79 combined CHP with MCT. Rats were first intraperitoneally injected with MCT (60 mg/kg) and then exposed to hypobaric hypoxia at 380 mmHg for 3 weeks. Rats developed PH and RVH with concentric neointimal thickening, abnormal endothelial cell proliferation, plexiform lesions, and vascular occlusion with fibrin thrombi. 79 These histological features are similar to those seen in the lungs of patients with PH. So this improvement of the model seems to be successful in displaying severe PH with associated histological features. The reproducibility of PH by CHP is inconsistent across animal strains.80–82 When neonatal calves are exposed to CHP, they develop severe PH, with mPAPs that approach systemic level and vascular remodeling that is much more severe than that in rats or mice.83–89 Furthermore, in hamsters, CHP causes less muscularization of the precapillary arteries than in rats. 89 The extent of vascular remodeling in response to CHP seems to be genetically determined, and genes such as HIF-1α, eNOS, complement-3 (a component of innate immunity), and BMPR-2 appear to play an instrumental role.81,90–97 The mouse model of CHP has helped to achieve great insights into the vascular remodeling in PH with regard to the role of growth factors, reactive oxygen species, and the nitric oxide pathway.98–104 Mice exposed to CHP develop increased mPAP but minimal pulmonary vascular remodeling, compared to rats.80–84 This limits the use of wild-type mouse models that exhibit severe vascular lesions similar to human PH. However, this limitation was addressed by creating a mouse model of severe PH by combining CHP and SU-5416, an inhibitor of the vascular endothelial growth factor (VEGF) receptor-2.102–106 SU-5416 inhibits VEGFR-2, which plays a prosurvival role in endothelial cells and leads to endothelial apoptosis that contributes to the development of PH.102–106 Constant improvement of the model appears to bring us one step closer to better modeling and understanding of the full complexity of PH.
CHP combined with SU-5416
In 2011, Ciuclan et al. 105 developed this mouse model of severe PH by injecting mice weekly with SU-5416 (20 mg/kg) and then exposing them to CHP (10% Fio2) for 7, 14, or 21 days. This is different from the protocol for rats, during which they receive a single injection of SU-5416 (20 mg/kg) and are then exposed to CHP (10% Fio2) for 3 weeks. 105 This fairly new development of the CHP model allows researchers to study the angioproliferative features of PH, in addition to hemodynamic changes. In rats, CHP+SU-5416 causes PH with pulmonary arterial changes resembling plexiform lesions that are virtually unresponsive to treatments such as iloprost and C-type natriuretic peptide.107,108 This model displays increased RVH and RV systolic pressure.105–108 Although some may view this as negative results (i.e., failing to attenuate PH), it does indicate that the experimental PH correlates well with the unresponsiveness of PH to some treatments in humans. The unresponsiveness to treatment and irreversibility of PH are important features of this model and may mean that the model relates more closely to human PH. This improvement of the CHP model will add greater clinical relevance to data generated from MPI models.
Fawn-hooded rat model
The fawn-hooded (FH) rat is an outbred strain developed from German brown, albino, and long Evan's rats.26,27 In 1988, these rats were shown to be hypoxia sensitive, developing mild PH at sea level and severe PH when exposed to mild hypoxia.26,27 At Denver altitude, the FH rat develops PH within a month after birth, and at half this altitude it develops PH at approximately 20–40 weeks of life.109,110 On closer investigation, it becomes evident that the FH rat model is crucial in elucidating certain aspects of the development of PH with regard to increased PASMC proliferation, resistance to apoptosis, and a preference for glycolytic metabolism (known as the Warburg hypothesis). 111 Over the years, many studies done in this SPI model of PH have allowed us to investigate the mechanisms underlying these aspects. It is now known that FH rats have (1) immature developed lungs with a reduced number of alveoli, (2) an inherited platelet disorder characterized by deficient serotonin uptake into platelets, and (3) a chromosome-1 abnormality that disrupts the mitochondrial reactive oxygen species–hypoxia inducible factor alpha (HIF-1α)–potassium channel pathway that underlies PH development.26,27,111 Other mechanisms include downregulation of superoxide dismutase-2 (SOD-2) and activation of HIF-1α and pyruvate dehydrogenase kinase (PDK). 111 Therefore, SOD-2, HIF-1α, and PDK manipulation may be therapeutic targets, as shown by studies done in the FH rat model. 111 Available data are limited on therapeutic treatments that have been tested in this model, with the exception of a few. Fasudil, a Rho-kinase inhibitor (known to cause vasodilation), has been shown to reduce severity of PH in the FH rat and to improve lung alveolarization and vascularization. 112 Another study showed that vasoactive intestinal peptide (known to cause vasorelaxation) attenuated PH in the FH rat by suppression of endothelin and interaction with its receptors. 113
Pulmonary artery banding model
Patients with congenital cardiac malformations involving large left-to-right shunts have a poor prognosis if early surgery is not performed. 114 In 1952, Muller and Dammann 115 described a surgical technique called pulmonary artery banding (PAB) as a form of surgical palliation for patients with such cardiac malformations.116–118 Later, it was noticed that placing the band too distally on the main pulmonary artery trunk can lead to pulmonary artery stenosis. 118 In 2002, a slight variation of this technique was used to create the PAB rat model for PH, which is characterized by progressive pulmonary artery stenosis and RVH.119,120
During the PAB procedure, a left thoracotomy is performed and the pulmonary artery is dissected away from the aorta.119,120 An 18-gauge needle is placed alongside the pulmonary artery, and a silk suture is positioned around the pulmonary artery to keep the needle in place. The needle is removed while the suture is tied tightly to produce a constricted opening in the lumen of the artery, equal to the diameter of the needle. As the animal grows, the lumen narrows further, which results in increased RV afterload.119,120 There are currently no reports on pulmonary vascular remodeling or PH in the PAB model, but these rats do develop pressure overload–induced RVH.6,77 However, because the pulmonary artery band remains fixed, it allows researchers to study the molecular events underlying RV remodeling and test the effects of novel experimental treatments.121–125
Trichostatin, a broad-spectrum inhibitor of histone deacetylases, has been shown to worsen RVH and RV systolic pressure in PAB rats while it did the opposite in left ventricular hypertrophy.120,126 The divergent effects of the trichostatin in left ventricular hypertrophy, compared to those in RVH, highlight the possibility that remodeling of the RV may, to some extent, differ from that of the left ventricle. However, this is a poorly investigated aspect of heart failure research and beyond the scope of this review. It has been suggested that RV dysfunction in the PAB model is due to a metabolic shift from glucose oxidation to glycolysis.123,124 Also, inhibition of the glycolytic enzyme PDK with dichloroacetate has been shown to improve RV function and reduce RVH in the PAB model.123,124 The mechanisms of these improvements are thought to be the restoration of the RV repolarization and glucose oxidation.123,124 Fang et al. 124 tested the efficacy of partial inhibitors of fatty acid oxidation in the PAB model. The authors were able to show that these inhibitors increased and enhanced RV function. Only a few studies have characterized the PAB model, and more insight into its pathogenesis is needed. Nevertheless, the PAB model does have a role in the development of novel treatments that may serve as a cardioprotective therapy, to be given as complementary treatment to current PH treatments.4,122,125
Schistosomiasis model
In 2010, Crosby and colleagues 127 used the mouse model of schistosomiasis-induced PH. Schistosomiasis is one of the most common parasitic infections, together with malaria and ambiasis, with approximately 200–300 million infected people in more than 70 countries.128–130 Schistosomiasis is caused by parasites (flatworm flukes) of the Trematoda class, such as Schistosoma mansoni.129,130 The life cycle of the flatworm includes penetration of the skin, invasion of the intestine, liver, and genito-urinary system, and release of eggs in the urine or feces of the host, after which the eggs are secreted into water. The eggs hatch, and miracidiae infect the freshwater snail to facilitate their own transformation into cercardiae. The cercardiae persist in the snail, are released into the water again, and penetrate the host's skin. After penetration, the cercardiae are transformed into schistosomulae that are transported via the bloodstream to the lung, where they induce granulomas. A portion of patients with schistosomiasis develop concurrent progressive pulmonary vasculopathy that is reminiscent of idiopathic PH.129,130
In the schistosomiasis model, female C57/BL6 adult mice received a single injection of a Puerto Rican strain of S. mansoni that was a suspension of either 75–100 cercariae (for the subacute study) or 30 cercariae (for the chronic study). Female mice were chosen because they are known to develop a greater worm burden than males during chronic infection. In the subacute study, mice killed at 6, 7, or 8 weeks postinfection had a few eggs present in the lungs and no evidence of pulmonary vascular remodeling. In the chronic study, mice killed at 7, 12, 17, or 20 weeks postinfection had a greater lung egg burden and developed significant pulmonary vascular remodeling. In addition, plexiform-like lesions were observed in the pulmonary vasculature of these mice. However, in this model, no significant RVH or PH was observed. This model was then modified by infecting mice with a suspension of 30 cercariae, which caused RVH and PH after 25 weeks. 131
The model was further improved by Graham et al., 132 who infected C57/BL6 mice with S. mansoni cercariae and intravenously challenged them with S. mansoni eggs. The infection was done by placing the mice's tails in a vial containing 30–35 cercariae for 30 minutes. 132 Fifty-five days later, mice were challenged intravenously by injection of 5,000 viable eggs (suspended in 0.5 mL of sterile saline) into the tail vein. This intravenous challenge mimicked the deposition of eggs in the lung by collateral shunts, which normally form in chronically infected mice. This resulted in pulmonary vascular remodeling and PH, dependent on the upregulation of interleukin-13. 132
A therapeutic drug that has been tested in this model is praziquantel, a treatment for chronic human schistosomiasis. 131 Praziquantel increased the permeability of the parasitic membranes of adult worms to calcium ions, thereby inducing contraction that resulted in paralysis and damage of the outer tegumental surface of the adult worm. Praziquantel treatment (250 mg/kg by oral gavage) in the S. mansoni mouse model prevented PH and reversed pulmonary vascular remodeling. The authors believe that the underlying mechanism involves clearance of eggs in pulmonary vasculature and reduction of local lung cytokine expression. 131 This is a very interesting model that may contribute to further understanding of underlying mechanisms such as the role of inflammation in PH associated with schistosomiasis. The model provides an ideal platform to test novel therapeutic strategies for schistosomiasis-PH.
BMPR-2 knockout model
A genetic mutation in the BMPR-2 gene has been found in endothelial cells and PASMCs in the lungs of patients with familial or idiopathic PH.133,134 The fact that BMPR-2 plays a crucial role in the development of PH is evident from the development of PH in BMPR-2-deficient mice.135,136 In 2004, Beppu and colleagues135,136 developed a mouse model in which the BMPR-2 mutant allele of the BMPR-2 gene lacks exons 4 and 5 (which encodes the transmembrane domain and a portion of the kinase domain of BMPR-2). Mice heterozygous for this BMPR-2 mutant allele (BMPR-2+/–) survive and reproduce normally, but in PASMCs isolated from these mice, the messenger RNA (mRNA) levels of BMPR-2 are reduced by 50%. This is associated with a reduced activation of the molecules smad 1, 5, and 8 in response to BMP-2. At baseline, BMPR-2+/– mice exhibit mild PH with muscularization and thickening of the pulmonary arteries. Exposure of these mice to hypoxia (11% Fio2 for 3 weeks) results in vasoconstriction, muscularization of the small pulmonary arteries, and increased mPAP.135,136
A major shortcoming in this model is the absence of RVH and the mildness of the PH.135,136 This shortcoming has been addressed by genetically modifying the mice to express a dominant negative allele for the BMPR-2 gene in PASMCs. 137 These mice develop pulmonary vascular changes resembling the plexiform lesion as well as PH and RVH. 137 Fasudil was tested as therapeutic treatment in this model by Yasuda and colleagues,137,138 who showed that fasudil treatment (100 mg/kg/day in the drinking water) alleviates PH. This is a major breakthrough, because this study proved Rho-kinase inhibition to be successful in treating PH associated with a BMPR-2 mutation. The mechanisms by which fasudil alleviates PH in this model are thought to be independent of the smad signaling pathway. Furthermore, fasudil has also been tested and shown to be effective in patients with PH. 139 It is clear that the BMPR-2 knockout model may play a crucial role in assessing the efficacy of novel experimental treatments that may soon be used in clinical practice.
Vasoactive intestinal peptide knockout model
In 2007, knockout mice were created by deletion of the vasoactive intestinal peptide gene (VIP−/−), on the basis of the notion that VIP causes pulmonary smooth-muscle relaxation and either neutralizes or attenuates the actions of vasoconstrictors such as endothelin. 140 VIP expression is also downregulated in the lungs of patients with PH. 140 VIP−/− mice spontaneously develop moderate to severe PH with pulmonary vascular remodeling, increased muscularization of the pulmonary arteries, and RVH. In-depth investigation of the molecular mechanisms underlying the development of PH in VIP−/− mice showed that VIP deletion alters gene expression. 141 Altered gene expression includes (1) underexpression of vasodilator and antiproliferative genes, (2) overexpression of vasoconstrictor and pulmonary vascular remodeling genes, and (3) upregulation of inflammatory genes. 141 Furthermore, VIP suppresses tumor necrosis factor-alpha and interleukin-10 and promotes T-regulatory cells, which may indicate an anti-inflammatory role for VIP. 142 However, the exact role of VIP in PH has not been elucidated, and it should also be kept in mind that this model differs from human PH with regard to hemodynamic severity and histology. 140
The VIP knockout model has greatly added to our general understanding of PH but also supports the concept of exogenous VIP therapy as therapeutic strategy in PH. 143 VIP replacement therapy (500 μg/kg/daily, intraperitoneally for 3 weeks) has been shown to reverse PH and correct gene alterations in the VIP−/− mice. 143 Knowledge gained from this approach has formed the basis for a possible treatment for humans with PH. This notion is supported by the fact that the serum concentrations of VIP are decreased in patients with PH. 144 In a study done by Leuchte and colleagues, 144 20 patients with PH were treated with a single dose of inhaled aviptadil (a VIP analog, at 100 μg for 15 minutes) before right heart catheterization. Assessment of the hemodynamic parameters and blood gases displayed slight improvement of oxygenation with no side effects. 144 Further experiments may be needed in order to fully characterize the effects of aviptadil treatment. This model has been a great help in studying experimental PH.
Endothelin receptor-B knockout model
In 2001, transgenic rats with an endothelin-B (ETB)-receptor deficiency were created that develop severe PH with increased mPAP and pulmonary vascular resistance and diminished cardiac output after exposure to hypobaric hypoxia (410 mmHg barometric pressure or 76 mmHg Fio2 for 3 weeks). 145 Endothelin is a peptide with vasoactive properties and the ability to regulate vascular tone in the normal lung. 145 The actions of ET are receptor dependent, with receptors A (ETA) and B (ETB). ETA is expressed mainly in PASMCs and, if activated, leads to PASMC proliferation and vasoconstriction. On the other hand, ETB is expressed in both endothelial cells and PASMCs. Activation of the endothelial ETB causes vasodilatation via the release of nitric oxide and prostaglandin, while stimulation of the PASMC ETB causes vasoconstriction in the lungs. 145 Furthermore, in a model of BMPR-2 mutation it has been shown that, at baseline, these mice have reduced expression of macrophage-derived ETA and ETB and that antagonism of ETB results in increased endothelin levels. 30 Together these data show that reduced expression of endothelin receptors increases pulmonary endothelin levels and thereby contributes to PH. 30 It is also known that in patients with PH, endothelin levels are increased and receptor expression (ETA and ETB) is upregulated.146,147 Collectively, the data seem to suggest that the entire endothelin signaling system (endothelin and its receptors) plays an important role in PH. The research done in this model has helped in the development of endothelin receptor antagonists (such as bosentan) as a safe treatment for PH with significant clinical response. 147
Apolipoprotein-E knockout model
In 2011, in a study by Weng and colleagues, 148 apolipoprotein E–deficient (ApoE−/−) mice were crossbred with delta-Glycine-adiponectin mice to generate mice with 3-fold higher adiponectin serum concentrations. The mice with higher serum adiponectin levels had reduced PH and pulmonary vascular remodeling after induction of PH with a high dose of ovalbumin. The protein ovalbumin induces pulmonary eosinophilic inflammation in mice characterized by pulmonary vascular remodeling similar to that in humans. ApoE is a vascular protective factor known to reduce circulating oxidized low-density lipoprotein and atherogenesis in the vessel wall. 149 Furthermore, it has been reported that patients with PH have increased ApoE expression and that ApoE deficiency is linked to the development of insulin resistance.150,151 In 2007, Hansmann and colleagues 149 reported that ApoE−/− mice spontaneously develop PH with increased pulmonary artery muscularization. This is an important finding because it implicates insulin resistance and obesity as risk factors for the development of PH.149,150 The authors of this particular study also noticed that the ApoE−/− mice have reduced adiponectin levels. 149 Adiponectin is a protein, produced by the adipose tissue, with beneficial effects in insulin resistance and atherosclerosis.151,152 Subsequently, ApoE-deficient PASMCs were treated with adiponectin and displayed inhibited proliferation.151,152 All of these studies suggest that adiponectin levels may have a potential therapeutic role as a modulator of pulmonary vascular remodeling and vascular tone and may explain the relationship between insulin resistance/obesity and PH.151,152
Neprilysin knockout model
In 2009, Dempsey et al. 153 created neprilysin null (NEP−/−) mice and exposed them to either normoxia (Denver altitude) or hypoxia (18,000 feet, hypobaric chamber) for a period of 5 weeks. Neprilysin, or neural endopeptidase (NEP), is a transmembrane metallopeptidase present in the lung, heart, and peripheral blood vessels. 153 In the pulmonary vasculature, NEP is expressed in PASMCs, fibroblasts, and endothelial cells, with functions including growth and contraction. 153 In lung biopsies from PH patients, there is an approximately 70% reduction in the NEP activity, associated with reduced mRNA expression. 154 NEP−/− mice developed severe PH characterized by muscularization of the distal pulmonary arteries, thickening of the proximal media, and adventitia with RVH. 153 It was also shown that a striking increase in PASMC proliferation was corrected when isolated PASMCs were treated with exogenous NEP (0.01 μg/μL). 153 Collectively, these results highlight the importance of NEP in the pulmonary vasculature and its instrumental role in the pathogenesis of PH, although this role has not yet been fully elucidated.153–155
Interleukin-6 overexpression model
In 2009, transgenic mice overexpressing lung-specific interleukin-6 (IL-6) were created by Steiner and colleagues. 156 IL-6, a pleiotrophic proinflammatory cytokine, is produced mainly by T cells and macrophages but also by smooth muscle cells of the tunica media in the vascular wall and adventitia.38,39,156 Aside from its proinflammatory action, IL-6 modulates immune processes, hematopoiesis, and oncogenesis.157,158 In PH research, elevated levels of IL-6 have been described in both human PH and animal PH and seem to correlate well with disease severity and mortality.38,39 Investigators exposed IL-6-overexpressing mice to CHP (10% Fio2 at sea level for a period of 3 weeks). They were able to show that the mice had elevated RV systolic pressure and RVH with pulmonary vasculopathic changes. Furthermore, the mice had muscularization of the proximal arterial tree, with proliferative arteriopathy often seen in the distal arteriolar vessels of PH patients. This suggests that in PH, IL-6 promotes the development and progression of pulmonary vascular remodeling. Authors attributed this action of IL-6 to its ability to activate signaling pathways that increase endothelial cell proliferation and the expression of anti-apoptotic proteins. 156
Data generated from studies done in the IL-6 overexpression model stimulated scientific interest in IL-6 as a possible therapeutic target in PH and, in general, in the concept of anti-inflammatory therapies for the treatment of PH.38,39 In 2010, Furuya et al. 157 published a case report of a female patient with mixed connective-tissue disease and severe PH. The underlying connective-tissue disease was treated with tocilizumab, an IL-6 receptor antagonist (at a dose of 8 mg/kg every 2 weeks for 12 months). The patient responded well to treatment, with improvement of functional class (New York Heart Association), 6-minute walk distance, and hemodynamic parameters on right heart catheterization (mPAP and pulmonary capillary wedge pressure). The authors did not see any change in cardiac output and pulmonary vascular resistance. 157 Similar reports have been published on the efficacy of pharmacological IL-6 antagonism in patients with PH as well as its specific effects on PASMCs isolated from patients with PH.159–161 All of these studies underline the usefulness of the IL-6 overexpression model in studying the function of IL-6 in the development of PH and the therapeutic potential of other anti-inflammatory therapies.
Angiopoietin-1 overexpression model
In 2004, Chu et al. 162 created a rat model by overexpressing angiopoietin-1 (Ang-1). The rationale for the development of this model is based on the fact that Ang-1, a ligand secreted by smooth muscle cells, is essential for angiogenesis in utero and signals vascular endothelial cells to stimulate the proliferation of smooth muscle cells around nascent endothelial tubes. 162 Ang-1 is also known to act synergistically with VEGF to facilitate the maturation of vascular networks in vivo. Chu et al. 162 created the model by injecting 2 × 1010 genomic particles of adeno-associated virus-angiopoietin-1 (AAV-Ang-1) into the RV-outflow tract of rats while using adeno-associated virus-lacZ (AAV-lacZ)–injected rats and carrier-injected rats as controls. After 1 or 2 months, the mPAP of Ang-1-overexpressing rats was significantly increased relative to that of controls. Increased PASMC proliferation was observed within the medial layer of arterioles, with obliteration of small vessels, similar to that seen in patients with PH. Angiograms of the rat lungs displayed blunting of the small peripheral arterioles that is consistent with severe PH. 162 Ang-1 seems to play a critical role in PH, but reports in the literature seem to be controversial.163–168
Du and colleagues 164 have also shown that Ang-1 is overexpressed in the lung tissue of patients with various types of PH and that the level of Ang-1 expression is directly proportional to the severity of the PH. Karapınar and colleagues 166 showed that patients with PH associated with left heart disease (mitral stenosis) have lower serum Ang-1 levels than controls. In that study, the authors found a negative correlation between serum Ang-1 levels and severity of PH. Kümpers et al. published data showing that patients with idiopathic PH have nearly 3-fold higher plasma levels of Ang-1 than controls. In addition to these findings, Ang-1 expression remained unchanged in lungs, PASMCs, and endothelial cells isolated from patients with idiopathic PH. Thus, it can be concluded that Ang-1 plays an important role in angiogenesis and vascular remodeling, with the potential to become a therapeutic target in PH. 162 However, further studies are needed for a better understanding of the role of Ang-1 in PH. A potential angiopoietin therapy would most probably involve the whole angiopoietin signaling system (Ang-1, Ang-2, and the endothelium-specific receptor tyrosine kinase).
Serotonin transporter overexpression model
In 2004, Maclean and colleagues 169 created a transgenic mouse model by overexpressing the serotonin transporter (5-HTT). In brief, serotonin, also known as 5-hydroxytryptamine (5-HT), is a potent pulmonary vasoconstrictor and co-mitogen that is increased in the blood plasma of patients with PH.169–171 It is synthesized in endothelial cells of the pulmonary artery by the enzyme tryptophan hydroxylase-1 and can act on PASMCs and pulmonary arterial fibroblasts.170–172 Serotonin signaling occurs via cell surface receptors, and its intracellular levels are modulated by 5-HTT. 173 Expression of the 5-HTT is increased in PASMCs isolated from patients with idiopathic PH. 169
In the study by Maclean et al., 169 5-HTT-overexpressing mice were exposed to CHP (10% Fio2 for 28 days) and developed RVH and pulmonary vascular remodeling. Results from this model stimulated general interest in the testing of 5-HTT inhibitors (such as fluoxetine) as therapeutic treatment for PH. 173 In 2009, Zhu and colleagues 174 showed that fluoxetine treatment (10 mg/kg daily by oral gavage for 4 weeks) prevented PH in the MCT model and prolonged survival. These are important findings, but serotonin signaling is a complex system (consisting of 5-HT, 5-HT receptors, and 5-HTT), and all its components are upregulated in patients with PH. 171 Many 5-HT receptor antagonists have now been tested and have shown to be successful in both cell culture and animal models.171–176 Furthermore, data from Morecroft et al.177,178 suggest that 5-HTT inhibition (LY-393558, a combined 5-HT1B receptor/5-HTT antagonist) may cause pulmonary vasoconstriction, which can be inhibited by simultaneous treatment with a receptor antagonist. It is also known that components of the serotonin signaling system are coregulated by both 5-HTT and 5-HT receptors. 179 Therefore, it is important for researchers to note that targeting of both the transporter and the receptor may provide optimal therapy for PH. 179
S100A4/Mts-1 overexpression model
In 1998, Ambartsumian and colleagues180,181 created transgenic mice by overexpressing S100A4/Mts-1. In these mice, the S100A4 coding sequences were placed under control of the mouse mammary tumor virus long terminal repeat promoter (MMTV LTR).180,181 S100A4/Mts-1 is a metastasis-promoting protein that forms part of a family of calcium-binding proteins whose functions include cell proliferation, differentiation, cytoskeleton dynamics, and apoptosis.180,182 This model was initially created with the purpose of investigating the role of S100A4/mts-1 in metastatic mammary cancer.180,181 However, it was reported that approximately 5% of the S100A4/Mts-1-overexpressing mice developed pulmonary vascular remodeling similar to the plexogenic arteriopathy seen in humans with PH. 182 A reason for using this model to study PH is that in most animal models, only certain vascular changes (as seen in PH patients) are reproduced, with a lack of neointimal thickening and plexiform lesions. 182 These vascular changes are characteristic of severe PH, and in order to study it, researchers began utilizing the S100A4/Mts-1-overexpression mouse model, which displays these specific vascular changes. 182
Unlike male mice overexpressing S100A4/Mts-1, females developed plexiform lesions. 182 S100A4/Mts-1 gene expression was increased in the lungs of females, compared to those of males, and this was translated as a greater Mts-1 protein expression in distal pulmonary arteries. In addition, female mice also developed increased RV systolic pressure, while males remained unaffected. This same group of researchers isolated PASMCs from humans with PH, treated them with physiological concentrations of 17β-estradiol, and demonstrated an increase in S100A4/Mts-1 expression. These findings are important because gender differences are prominent in human idiopathic PH, and they may provide a possible explanation for these differences.183,184
Another important study done in this model involves the introduction of viral components to the model in order to establish a link between viral infection and the development of PH. 185 PH is a serious complication of HIV, and in addition, the involvement of a latent infection with the human herpes virus-8, also called Kaposi's sarcoma–associated herpes virus, has been prevalent in patients with idiopathic PH. On the basis of this notion, Spiekerkoetter et al. 184 infected 1-year-old S100A4/Mts1-overexpressing mice with the vasculotrophic virus γHV-68 (murine gamma-herpesvirus-68). Six months after the γHV-68 infection, investigators observed perivascular inflammation and occlusive neointimal formation, accompanied by significant degradation of elastin. They concluded that early viral access to the vessel wall may be a key determinant of the severity of vascular pathology following viral reactivation. 184
Furthermore, lung biopsies from children with PH associated with congenital heart defects showed increased S100A4/Mts-1 expression in PASMCs of lesions associated with neointimal formation and plexiform lesions. 182 Merklinger and colleagues184,185 investigated the underlying mechanisms for S100A4/Mts-related pulmonary vascular remodeling. In this study, S100A4/Mts-1 mice were exposed to CHP, lung tissue was analyzed with microarray, and a number of genes were shown to be differentially expressed. One of these genes was fibulin-5, a matrix component necessary for normal elastin fiber assembly. Fibulin-5 was localized in the pulmonary arteries and associated with thickened elastic lamina. This could be an underlying mechanism for the attenuation of pulmonary vascular remodeling in response to elevated pressure.185,186 The S100A4/Mts-1 overexpression model has certainly allowed researchers to investigate a very important part of the pathogenesis of PH.
CONCLUSIONS
The heterogeneity of PH in humans and the difficulties in treating it make it a very complex disorder. This has major implications for the use of animal models and the development of improved models. The validity of animal models has been largely criticized among the clinical and basic-science community, partly for good reasons. Because of our limited understanding of PH, the question is often asked, how can we model a disorder that we do not fully comprehend? This review has discussed a number of SPI animal models and highlighted their evolution into improved MPI animal models that more closely correlate with human PH. The improvement of a particular model is paramount because an animal model can be called so only if it effectively models the human disease. If it does, then it will allow researchers to develop a more clinically relevant treatment that could potentially improve the quality of life or prolong the survival of patients with PH and, if possible, cure PH. Research done with animal models has vastly contributed to our current understanding of the pathophysiology of PH, and if we continue to improve these models, they may help to develop a therapeutic breakthrough or even a cure. Animal models do not reflect clinical benefit and cannot be used as a replacement for clinical trials. A very important point that we want to highlight is that MPI animal models tend to correlate better with severe PH in humans than do SPI models. We also imply that there is a possibility that SPI models may display features of the early stages of PH in humans that are often missed because of late diagnosis. Therefore, animal models can be useful in assessing various stages of the disease progression. Nonetheless, in our opinion, data generated from animal models should not be seen as preclinical research, because of the inability to model human PH with regard to hemodynamic and histological severity. There is therefore still a long way ahead, but it is gratifying to see the great progress that has been made in PH research.
