Abstract
Keywords
The elderly receive antipsychotic (AP) medication to treat a range of psychotic, affective and behavioural conditions. However, dementia is the most common condition for which the elderly receive AP medication. Community subjects with dementia were six times more likely to be taking an AP drug than non-demented subjects, with the highest prevalence rates in the group with moderate dementia [1]. Over 90% of residents with dementia in nursing homes have at least one behavioural or psychological symptom [2] and many are prescribed AP medication [3, 4].
Despite displaying modest efficacy in randomized controlled trials (RCTs) [5, 6], AP medication has been the mainstay of pharmacological treatment for behavioural and psychological symptoms in dementia (BPSD) [7]. Many clinicians have prescribed atypical AP in place of conventional AP because of the reduced incidence of extrapyramidal side-effects and tardive dyskinesia in the former [8, 9]. Recent meta-analyses of placebocontrolled, randomized trials of atypical AP in dementia have raised concerns about the increased incidence of death or cerebrovascular adverse events [10, 11]. In response, the Committee on Safety in Medicines, UK, March 2004, issued a warning regarding the use of risperidone and olanzapine for behavioural symptoms in dementia [12]. However, there was no evidence from RCTs that conventional AP drugs were safer than the newer AP drugs. A large government-funded prospective trial comparing three atypical AP drugs and a selective serotinergic reuptake inhibitor in the treatment of BPSD is due to report in 2006 [13]. Unfortunately, no conventional AP drug was used as a comparator, despite the widespread use of haloperidol.
Retrospective database reviews have compared conventional and atypical AP medication with respect to outcomes such as death, or admission for stroke or cardiac problems. Some studies lumped all conventional AP drugs together [14, 15] or compared them with aggregated atypical AP medications [15, 16]. As receptor affinities and side-effects vary between individual AP drugs, this approach may mask individual AP differences. Others failed to make a temporal link between AP dispensing and the outcome measure [17]. The present study described the pattern of AP drug dispensing and associated (odds ratios) ORs of death in elderly veterans and war widows in New South Wales (NSW) and Australian Capital Territory (ACT) in 2001. The analysis linked dispensing and death temporally and provided associated ORs for six individual AP drugs.
Methodology
All veterans and war widows 65 years and older resident in NSW or the ACT who had been dispensed an AP medication in 2001 were recruited as subjects. Pharmacy data included dispensings occurring in the community, in residential aged care facilities and private hospitals. Public hospital dispensing, which was funded by the State, was not included. The DVA Pharmacy database is a claims-based system covering drugs that are available on the Repatriation Pharmaceutical Benefits Scheme. Claims for payment are originally received and processed for payment by the Health Insurance Commission. Date of dispensing is electronically recorded by the pharmacist dispensing the medication.
The DVA beneficiaries may be admitted to private hospitals and may access allied health and medical services without copayment from the veteran or war widow. Thus, the study population should be compared with similar populations.
In addition to AP dispensing data, antidepressant, lithium, carbamazepine, sodium valproate and benzodiazepine dispensing data were extracted. The subject's unique identifier was used to extract date of birth, date of death and gender from the administrative database. Those on AP parenteral medication (n = 13), those whose date of death preceded date of last prescription (n = 387) and those dying before 1.5.2001 (n = 151) were excluded from analysis, leaving 3284 subjects.
All subjects received a study date: either the date of death or a randomized date from 1.5.2001 to 31.12.2001. The pattern of AP and psychotropic dispensing in the 120 days preceding the study date was analysed. Table 1 shows the number of subjects dispensed each AP drug and the number of subjects in those groups dispensed no other AP drug in this time period. The number of subjects dispensed trifluoperazine, quetiapine or fluphenazine alone with respect to other AP drugs (n = 136) was too small to allow individual analysis of these three drugs, and hence they were combined. Psychotropic drugs were analysed as mutually exclusive groups: tricyclic antidepressants, all nontricyclic antidepressants, mood stabilizing medication (carbamazepine, sodium valproate and lithium), diazepam/clonazepam, and all other benzodiazepines.
Drug dispensed in 120 day window
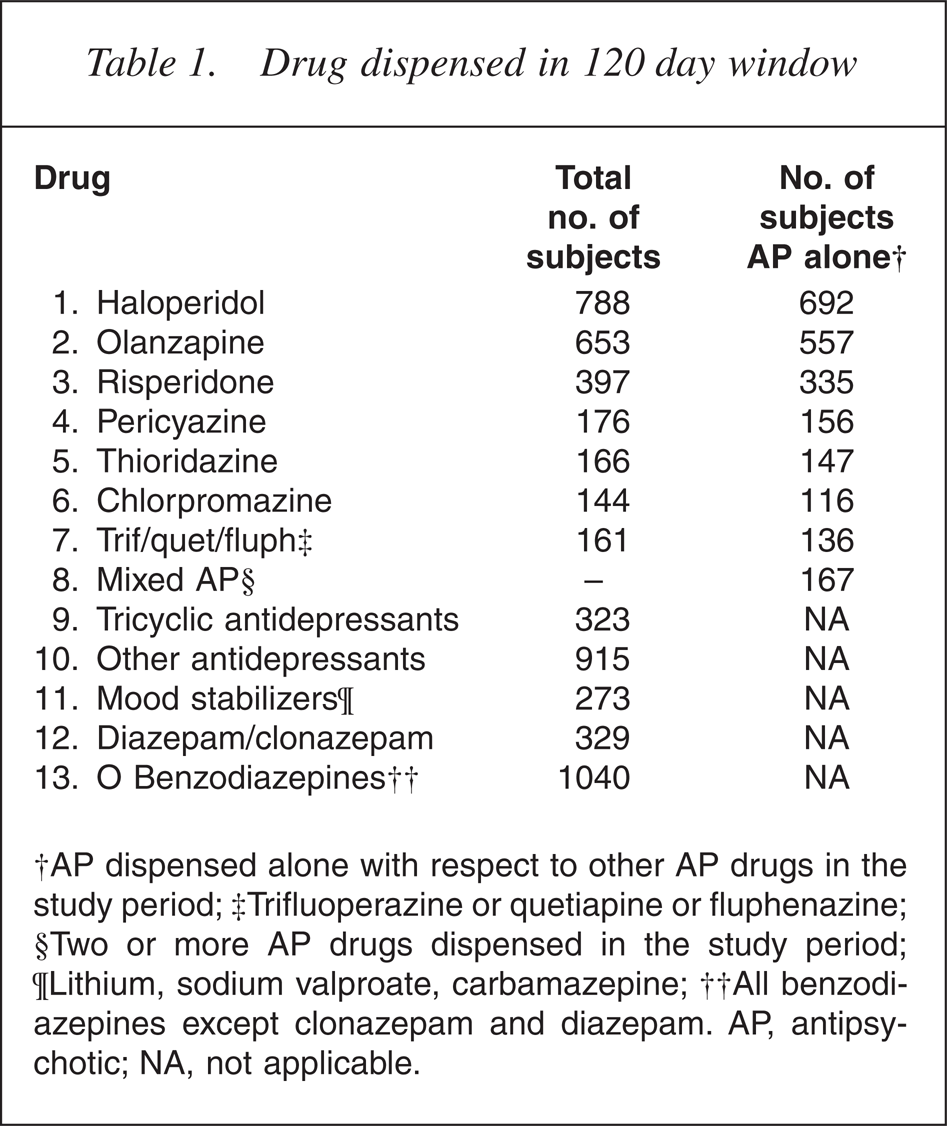
†AP dispensed alone with respect to other AP drugs in the study period; ‡Trifluoperazine or quetiapine or fluphenazine; §Two or more AP drugs dispensed in the study period; ¶Lithium, sodium valproate, carbamazepine; ††All benzodiazepines except clonazepam and diazepam. AP, antipsychotic; NA, not applicable.
The degree of coexposure of AP drugs was low, n = 167, and these subjects were classified as a separate group. Five hundred and two subjects had been dispensed AP medication in 2001 but were not dispensed either an AP or other psychotropic (OP) medication in the 120 days before their study date. This group formed a reference group against which the ORs of death for AP and OP groups were calculated.
Statistical method
Interaction indicators were constructed between each of these eight (exclusive) AP exposure variables and each of the five OP exposure variables. The analysis strategy was as follows: three logistic regression models were fit with death status (‘1’ for dead, ‘0’ for alive) as the end point or dependent variable, and various predictors:
Model 1: predictors were eight AP indicators, plus five OP indicators, plus 20 AP–OP interaction indicators (sample size n < 29) plus age and gender (df = 35);
Model 2: Model 1 was reduced by omitting any predictor that did not have a partial p-value of 0.10 or less;
Model 3 was obtained by removing from Model 2 all terms with a partial p-value greater than 0.05.
These 3 models were ‘nested’, allowing increment tests to provide reassurance that the group omissions did not conceal statistically significant effects, possibly obscured by multicolinearity. Statistical Package for the Social Sciences, version 6.1.3, was used.
The OR for risperidone, olanzapine and pericyazine were all highly significant. To test whether or not they differed from each other, a further four models were run, constraining ORs equal pair-wise among the three drugs, and constraining all three equal.
Finally, Model 4 investigated the nature of the strong haloperidol effect (an OR of 8.60: see below) by partitioning the haloperidol indicator predictor into two predictors: one indicating whether or not haloperidol was dispensed in the immediate 30 days preceding the study date, and the complementary predictor indicating whether or not haloperidol was dispensed only in the period from 120 to 31 days prior to the study date. (Note that subjects dispensed in both periods were given only the former exposure.) If this apparent high toxicity of haloperidol was related only to its use on patients already experiencing a terminal event, the effect should remain with the former predictor, but not the latter. Model 3 above was nested within a new model (Model 4) where the initial haloperidol predictor was replaced by these two complementary new predictors, and the corresponding increment tests examined whether haloperidol toxicity was different in these two periods.
Results
In this study 57.2% of subjects were male and 91.8% were 75 years or older. The death rate in the total analysis sample was 17.3% in 8 months. The death rate in the reference group was 6.4% in 8 months. Table 2 displays associated risk of death for those dispensed AP and OP medications. The risk of death associated with OPs medications only pertained to our study group, that is, those dispensed AP medication in 2001. There were no significant AP–OP interactions. The unadjusted relative risk was calculated by dividing the risk of death for each drug group by the reference group's risk of death (6.4%).
Drug-associated death rates in 8 months for AP drugs dispensed alone with respect to other AP drugs, a mixed AP group and other psychotropic drugs
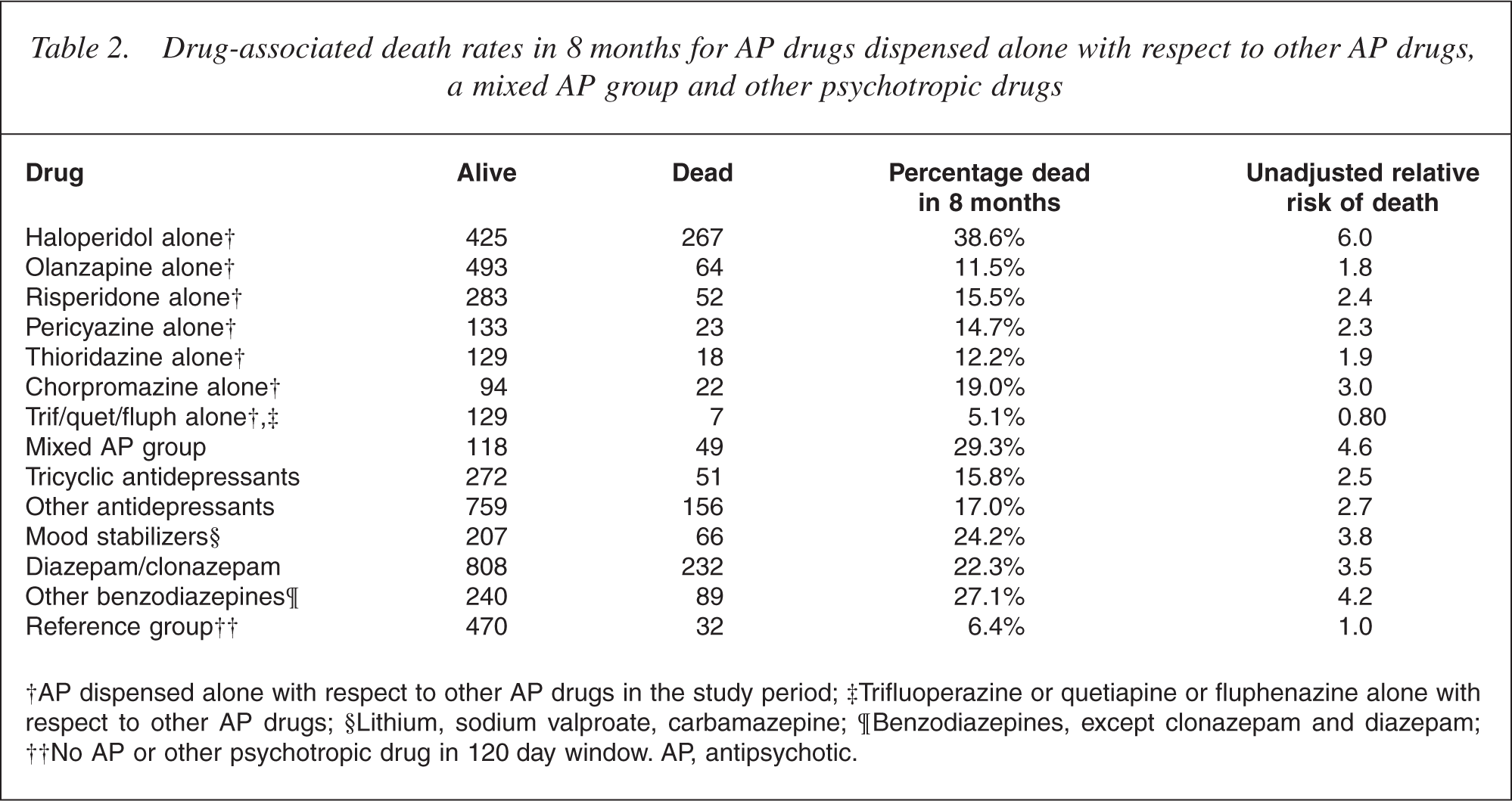
†AP dispensed alone with respect to other AP drugs in the study period; ‡Trifluoperazine or quetiapine or fluphenazine alone with respect to other AP drugs; §Lithium, sodium valproate, carbamazepine; ¶Benzodiazepines, except clonazepam and diazepam; ††No AP or other psychotropic drug in 120 day window. AP, antipsychotic.
The estimates of ORs for the final models are presented in Table 3.
AP mortality OR (model 3 and model 4)
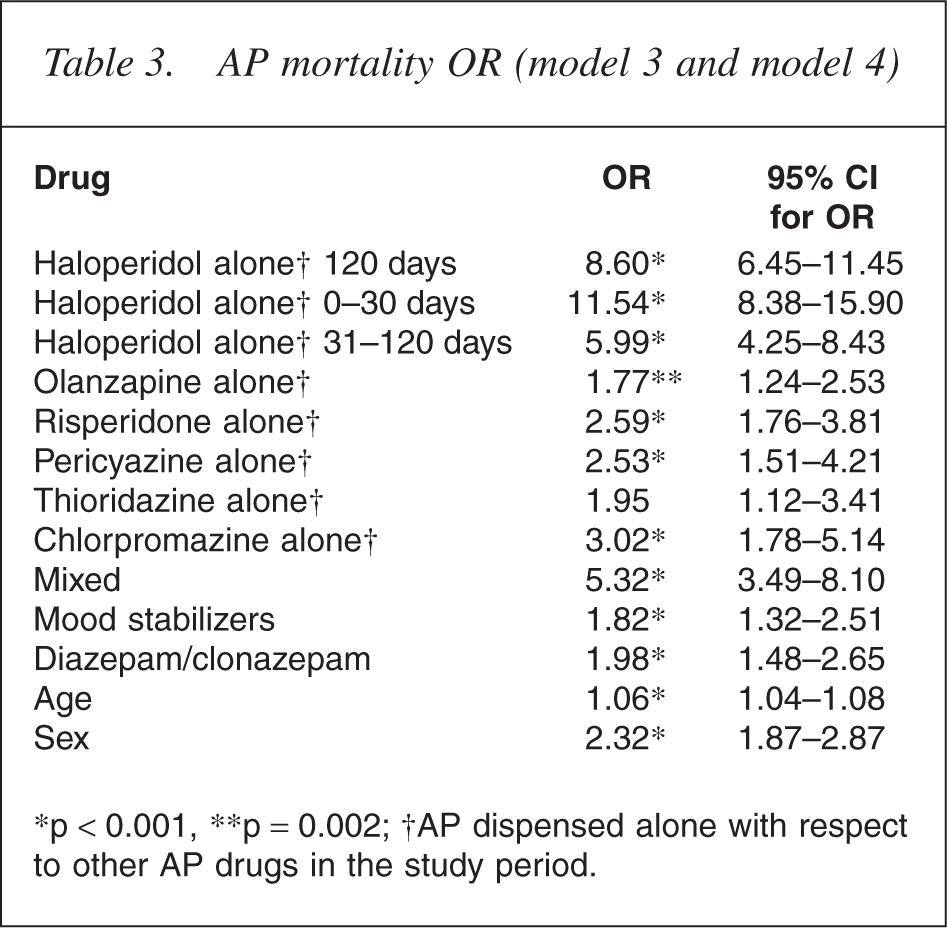
∗p < 0.001, ∗∗p = 0.002; †AP dispensed alone with respect to other AP drugs in the study period.
The OR for risperidone, olanzapine and pericyazine were all highly significant. There were no significant differences in ORs among these medications, with the difference between the olanzapine and risperidone ORs failing to reach significance (p = 0.066).
The OR for haloperidol 31–120 days was 5.99 (95% Confidence Interval [CI] = 4.25–8.43) and for haloperidol 1–30 days was 11.54 (95% CI = 8.38–15.90). The haloperidol 31–120 day OR was tested against risperidone 120 day OR, olanzapine 120 day OR and pericyazine 120 day OR and found to be highly significant in each case.
Discussion
An excess of cerebrovascular events in the treated arm of a risperidone RCT for BPSD [18] led to a number of meta-analyses of atypical AP trials [19]. The ORs for six individual AP groups in our study were significant compared with the reference group. This is consistent with the increased OR of death (1.54, 95% CI = 1.06–2.23) found by Schneider et al. in their meta-analysis [11]. Schneider et al. included 15 RCT in their meta-analysis, nine of which were unpublished. There were no significant differences in the incidence of adverse events in the published and unpublished trials, but the unpublished trials may not have established efficacy on their primary outcome measures.
Haloperidol was associated with a significantly higher OR compared with other AP drugs in our study, a finding reported in a previous veteran database review [17]. However, this association may be explained by the use of haloperidol in delirium and terminal agitation. By partitioning the haloperidol ORs into two groups, haloperidol 0–30 days and haloperidol 31–120 days, we anticipated that the latter group would contain fewer individuals who received haloperidol in the context of a terminal illness. With the application of this partitioning the OR for haloperidol 1–30 days fell from 11.54 (95% CI = 8.38–15.90) to 5.99 (95% CI = 4.25–8.43) for haloperidol 31–120 days. Haloperidol can cause extrapyramidal side-effects, falls and swallowing problems in the elderly, and has been associated with torsades de pointes in case reports [20]. This study did not explore the effects of dosage, which may be a confounding variable. Although the design of this study precluded any firm conclusions about haloperidol toxicity, haloperidol was the most commonly prescribed AP in this study and the real nature of any toxicity should be assessed by further studies. Haloperidol was also the most commonly prescribed AP drug Australia-wide for veterans and war widows 65 years and over in 2003 and 2004 (Hollis et al. 2006, unpublished).
A large NIMH funded prospective trial due to report in 2006, the Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE), will compare the efficacy and effectiveness of risperidone, olanzapine, quetiapine and citalopram in BPSD [13]. However, haloperidol was not used as a comparator as it was considered too toxic. Thus no information will be forthcoming from the CATIE trial regarding the most widely prescribed AP in elderly Australian veterans and war widows.
Even acknowledging the present study's weakness with respect to haloperidol, it is interesting that the OR for pericyazine, the fourth most commonly dispensed AP in 2001, did not differ significantly from the more expensive atypical APs. Dosage was not examined and it is possible that dispensing of lower dose equivalents favoured pericyazine. Nevertheless this was a naturalistic study reflecting the prescribing choices of doctors. Chlorpromazine may be used in palliative care [21], which may explain the higher associated OR (OR = 3.02, 95% CI = 1.78–5.14). The present authors were unable to find any reference for the use of pericyazine in palliative care or treatment of delirium from 1976 onwards. Thioridazine (OR = 1.95, 95% CI = 1.12–3.41) has been associated with prolongation of the QTc interval and had restricted prescribing indications in 2001. If doctors complied with the prescribing indications, it may be that subjects dispensed thioridazine differed from other subjects by diagnosis, that is, they were diagnosed with treatment-resistant schizophrenia, and had lower cardiac risk factors.
In the meta-analyses of Schneider et al. 2005, no significant difference among the AP drugs was detected, although the trend favoured risperidone over olanzapine [11]. In our study, the trend favoured olanzapine rather than risperidone, but failed to reach significance (p = 0.066). The assessment of comparative risk would warrant further exploration. Subjects in our study may have been less well than those accepted into RCTs, and vascular risk factors were associated with cerebrovascular adverse events in AP drug trials [18]. Male gender and age were risk factors for AP associated death in our study. Male subjects may have had increased cardiovascular risk factors or received higher dose equivalents. The study design does not allow these questions to be answered. Do age, gender, certain medical comorbidities, and particular AP drugs confer a disproportionate risk? Further studies are clearly warranted to address these important issues.
We explored the ORs for OP drugs dispensed to the AP population. Only mood stabilizers (carbamazepine, sodium valproate and lithium) and diazepam/clonazepam were associated with significantly increased ORs. Carbamazepine and sodium valproate are used in the treatment of BPSD. No RCTs or database reviews have explored the associated OR of death for those with dementia using these drugs. Clonazepam and diazepam may have been used in terminal agitation, hence the increased OR. However, in one AP meta-analysis benzodiazepine use was a mortality risk factor for those receiving olanzapine [19]. Neither the antidepressant groups nor the shorter acting benzodiazepine drugs were associated with significantly increased ORs.
Prospective atypical AP RCTs were 8–13 weeks long and hence did not assess longer-term risk. Patients may be most vulnerable in the period following commencement of the AP drug and the risk may fall over time. Database reviews using incident user designs [22] or longer RCTs may be able to quantify the risk over time. If the risk were constant, the Number Needed to Harm over 1 year with regard to CVAEs has been estimated to be 6.3 [12].
Randomized controlled trials are expensive and frequently short-term. Uncommon adverse events may not be detected. Database reviews, although lacking detailed knowledge of the subjects compared with RCTs, are relatively inexpensive and allow exploration of drug safety in those under-represented in RCTs. Thus aspects of safety can be assessed in the elderly, those with significant medical comorbidity, those receiving the medication chronically, and those with diagnoses not contained within the drug's official indications, that is, ‘off-label’ prescribing. The increased risk of coronary disease for those using high dose of Cox-2 inhibitors illustrates the importance of postmarketing studies [23]. However, the statistical methodology of cohort studies continues to be debated [24].
Two retrospective cohort studies have not shown increased incidence of admission for stroke in atypical versus conventional AP drug users [14, 25]. Another demonstrated increased incidence of sudden cardiac death in those on moderate doses of conventional AP medications when compared with non-users [26]. There were no atypical AP drug comparators in this study. Wang et al. found an increased risk of death among conventional AP users compared with atypical AP users [15]. This retrospective cohort study had many sophisticated methodological features. However, the haloperidol in the aggregated conventional group may have inflated the conventional group's risk of death. The study considered risk of death from first dispensing of an AP drug in incident users, irrespective of length or discontinuation of dispensing. It has been argued that this approach parallels an intention-to-treat analysis [24]. While acknowledging that many factors may never be known in a database review, to jettison the temporal relationship between continued drug dispensing and adverse outcome would appear to undercut one of the main hypotheses, namely, that drug ingestion is related to adverse outcomes. On discontinuation of a drug, risk attributable to the drug should diminish over a period of time, with risk of death eventually reflecting background medical comorbidity, age and gender of the subjects. Our study design allowed review of individual AP drugs and analysed information regarding dispensing in the 120 days before death.
Concern about increased mortality rates in AP drug users has been of long standing and the Report of the Royal College of Psychiatrists provides a summary of the literature and proposed mechanisms [27]. Prolongation of the QTc interval, cardiac arrhythmias, stroke, deep venous thrombosis, falls, extra-pyramidal side-effects and aspiration pneumonia have all been considered possible causes of the increased mortality rate.
This study reviewed AP drug dispensing and the associated ORs of death in elderly veterans and war widows in NSW and ACT in 2001, and thus reflected the prescribing choices of doctors. The study was sufficiently large to allow individual analysis of six AP drugs. While OP drug use was analysed, non-psychotropic drug use and measures of general medical morbidity were not controlled. The use of a reference group may have mitigated the lack of comorbid diagnostic information, although the reference group may have varied diagnostically or in disease severity compared with those who received an AP in the study period. It may be that the high haloperidol OR reflected the use of haloperidol in delirium or terminal agitation. Our study population included those without dementia, but it is likely that dementia accounted for the majority of AP dispensing in this age group [1, 4]. As questions are raised about the safety of atypical AP in dementia, lack of prospective RCTs using conventional AP drugs create the need for database reviews to provide evidence of relative safety for individual AP drugs. This study highlights areas for further examination.
Footnotes
Acknowledgements
The authors wish to acknowledge the support and funding of the Department of Veterans' Affairs. We would like to thank Mr Effie Cauchi, Acting Director of Health and Corporate Support, NSW State Office, for his ongoing commitment to this project. Any errors or omissions remain the responsibility of the authors. The authors wish to thank Professor Henry Brodaty and Professor Bob Cumming for their helpful comments on the paper.
