Abstract
Keywords
Antipsychotic medication is widely used in residential aged care facilities [1]. A recent survey in 40 Sydney nursing homes reported that 25% of residents were prescribed an antipsychotic drug [2]. In an Ontario study, 24% of residents not on antipsychotic medication on admission to the facility were commenced on this medication in the first year [3]. In addition, 13% of those receiving conventional antipsychotic drugs and 6% of those receiving atypical antipsychotic drugs were prescribed initial doses that exceeded recommendations [3]. Ninety per cent of residents with dementia have at least one behavioural or psychological symptom, with 60% exhibiting psychotic symptoms [4].
Several randomized controlled trials (RCTs) of atypical antipsychotic medication in dementia involved subjects from residential care facilities [5–8]. The mean Mini-Mental State Examination scores for subjects in these trials ranged from 5.5 to 8.8 [5–8]. A meta-analysis of the three risperidone trials demonstrated statistically significant and meaningful reductions on measures of aggression [9]. The benefit in treating psychotic symptoms was less clear. This meta-analysis of 1250 subjects did not demonstrate a reduction in score for hallucinations on the Behavioural Pathology in Alzheimer's Disease scale [10] and produced a statistically significant but small reduction of 0.7 for delusions (maximum score of 21 for the delusional subscale) [9].
RCTs that failed to demonstrate efficacy for antipsychotic medication were conducted with less impaired residents [11] or community subjects [12–14]. The Clinical Antipsychotic Trials of Intervention Effectiveness in Alzheimer's Disease (CATIE-AD) enrolled community subjects [15]. This group reported no statistically significant difference on the primary outcome measure of time to discontinuation among olanzapine, quetiapine, risperidone and placebo subjects, although the reason for discontinuation differed among the groups [15].
Recent meta-analyses of RCTs of atypical antipsychotic medication in those with dementia have demonstrated an increased risk of stroke and death [16, 17]. However, there is limited information about the risks associated with conventional antipsychotic medication. Several data linkage studies have been undertaken to assess the comparative risk of death [18, 19] and stroke [20–22] associated with atypical and conventional antipsychotic drug dispensing. While meta-analyses of RCTs compared the risk of atypical antipsychotic drugs with placebo, data linkage studies compared the risk associated with aggregated conventional and atypical drug dispensing [18, 20, 21], or dispensing of individual drugs [19, 22]. Data linkage studies involve large cohorts that include the frail elderly and those with multiple medical morbidities, thus providing information about actual dispensing in clinical settings. The US Food and Drug Administration acknowledged that conventional antipsychotic medication may not be safer than atypical antipsychotic drugs in those with dementia [23].
Carbamazepine and valproate have been considered as alternative treatments for behavioural and psychological symptoms in dementia [24, 25], although the evidence for this is limited [26, 27]. No other study known to the authors has attempted to compare risk of death associated with the dispensing of these drugs relative to the risk associated with antipsychotic medication dispensing.
This analysis was part of a larger study examining dispensing of these study drugs to all Australian war widows and veterans,≥65 years, residing in the community or aged care facility. Analyses from this study have been previously reported [19]. The present study aimed to established the instantaneous relative risk (RR) of death for Australian Department of Veterans’ Affairs (DVA) beneficiaries ≥ 65 years, who resided in an aged care facility in 2003 or 2004, and were incident antipsychotic drug, carbamazepine or valproate users.
Method
Data source and subjects
Dispensing data were extracted from the DVA claims-based pharmaceutical database. Date of dispensing and medication details were recorded by a pharmacist at point of sale and processed by Medicare Australia, formerly the Health Insurance Commission. Drugs dispensed in public hospitals are not included in this database. Approval for the study was obtained from DVA Ethics Committee and the analysis performed on de-identified data.
We identified veterans and war widows ≥ 65 years who were dispensed a study drug in 2003 or 2004. The methodology has been previously reported [19]. Subjects not dispensed a study drug in the 12 months prior to index dispensing were considered incident drug users. Subjects were not able to be enrolled more than once into the study. Those dispensed two study drugs simultaneously were analysed separately in a mixed group. Subjects were followed until death, 31 December 2004, 60 days after discontinuation of the drug, or starting a second study drug. Drug groups with fewer than 20 subjects and longer-acting parenteral antipsychotic drugs were not analysed.
Other variables
Gender, date of birth, date of death and residential status of subjects were extracted. Psychotropic drugs (antidepressant medication, benzodiazepines, non-benzodiazepine anxiolytics, lithium carbonate and antihistaminic drugs) and cholinesterase inhibitor drug dispensing data in 2003 and 2004 were extracted from the pharmaceutical database. Vascular risk factors have been linked to adverse outcomes in previous RCTs of atypical antipsychotic medication dispensing in dementia [7]. Hence, the dispensings in 2003 or 2004 of anti-arrhythmic drugs, angiotensin-converting enzyme inhibitors and receptor blockers, anti-hypertensive drugs, anti-thrombotic, anticoagulant or fibrinolytic drugs, beta-blockers, calcium channel blockers, diuretics, glycosides, vasodilators, diabetic drugs and hypolipidaemic agents were recorded for each subject. Dispensing data for drugs used in Parkinson's disease and epilepsy were extracted because these diseases were likely to be unevenly distributed among the subjects. The dispensing of morphine and drugs used in the treatment of cancer were recorded. These drugs would be associated with conditions that would, of themselves, increase the risk of death. In addition, dispensing data for Cox-2 selective non-steroidal anti-inflammatory drugs were extracted. The total number of different drugs used in the year prior to the index dispensing performed well as a measure of overall medical morbidity [28]. Data for this measure were extracted.
Statistical analysis
We assessed the risk of death associated with each study drug in two ways, namely, mortality rates per 1000 person years at risk and Cox proportional hazards models with two time intervals. In the Cox models, death was the outcome variable. Subjects were followed for 60 days after discontinuation of the study medication, starting a second study drug or the end of the study period (31 December 2004). A previous study reported higher risk in the initial period after dispensing [18] and the increased risk reported from RCTs occurred in the short term [7, 16, 17]. Thus, we constructed a second Cox model that assessed the RR of death up to 60 days after initial dispensing. In this model, subjects were censored at 60 days or on starting a second study drug. Olanzapine, the second most commonly dispensed study drug, was chosen as the referent. Although haloperidol was more commonly dispensed than olanzapine, the use of this drug in delirium and terminal agitation would have made the interpretation of comparisons with other antipsychotic drugs less straightforward. Deaths within 60 days of the last dispensing were included in the analysis. This figure was chosen to allow 30 days of use and a 30 day washout period, analogous to RCT protocols.
The details of the statistical analysis are reported elsewhere [19]. All drugs were fitted as predictors in the first model and the covariates run singly. Covariates with partial regression coefficients with p > 0.05 were omitted. The covariates were then run as multivariate predictors. Again covariates with a partial regression coefficient with p > 0.05 were omitted and the model rerun resulting in a final covariate model. Drugs were then run singly as predictors adjusted for the covariates from the final covariate model. Drugs with partial regression coefficients p > 0.05 were then omitted and the remaining study drugs were run as multivariate predictors, adjusted for the final covariate model. All omissions were tested with deviance tests between the intermediate and final model. All omissions leading to refined multivariate models were tested and supported by deviance tests against their parent model.
Results
Among DVA-entitled beneficiaries, we identified 6602 incident users of antipsychotic drugs, carbamazepine and valproate in 2003 or 2004 who resided in an aged care facility during this period. Forty-nine per cent of subjects were male. Although 35% of the group resided in an aged care facility on 1 January 2003, all, by definition, were residents at some point in 2003 or 2004. Twenty-nine per cent of the cohort died by the end of the study period. The mean age was 83.5 years.
There were sufficient subjects to analyse data for four conventional antipsychotic drugs (chlorpromazine, haloperidol, pericyazine and trifluoperazine), three atypical antipsychotic drugs (olanzapine, quetiapine and risperidone), as well as carbamazepine, valproate and the mixed group. The mean age, gender and percentage of users with some community dispensing are displayed in Table 1. The pattern of psychotropic and other drug dispensing is displayed in Tables 2 and 3.
Demographic details for RACF subjects resident in 2003 or 2004
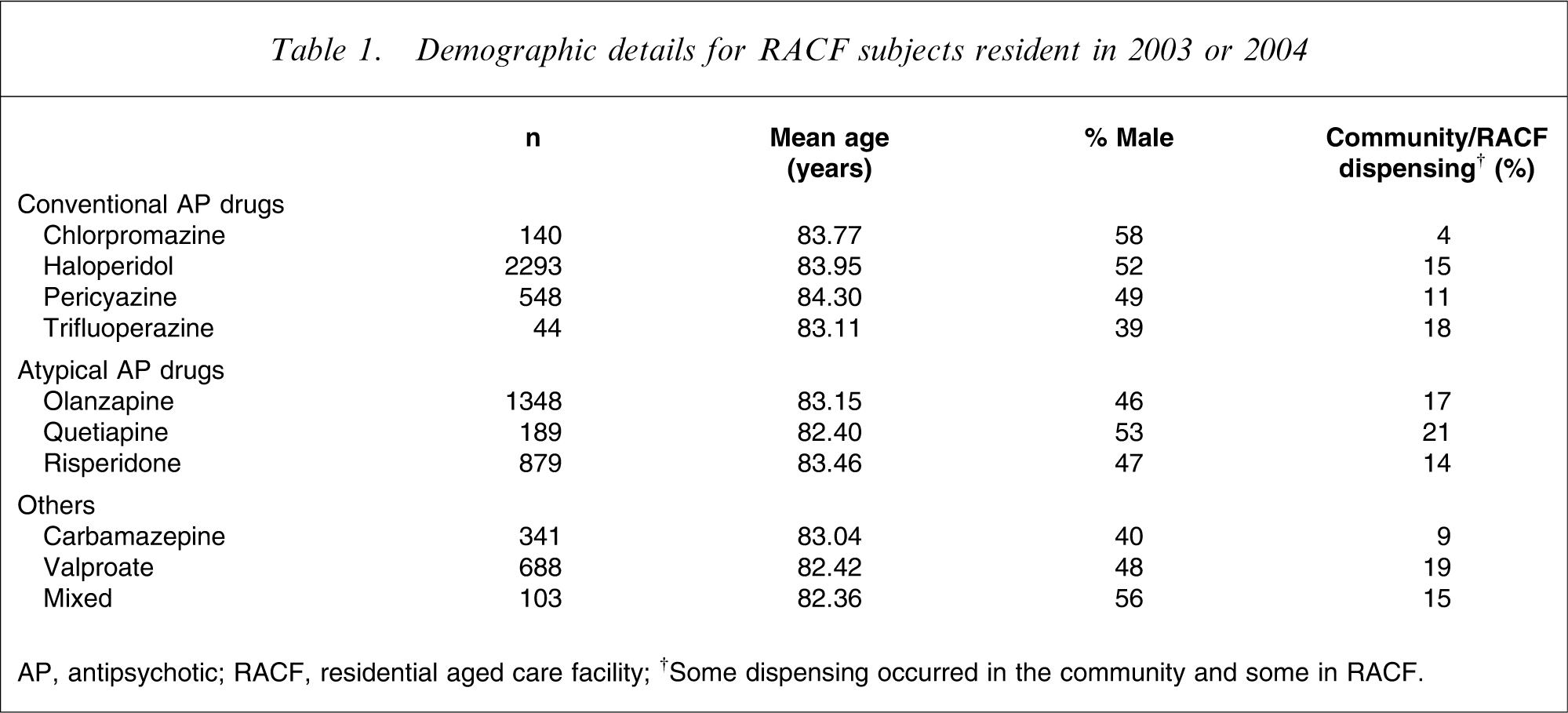
AP, antipsychotic; RACF, residential aged care facility.
†Some dispensing occurred in the community and some in RACF.
Psychotropic drug dispensing for RACF subjects resident in 2003 and 2004
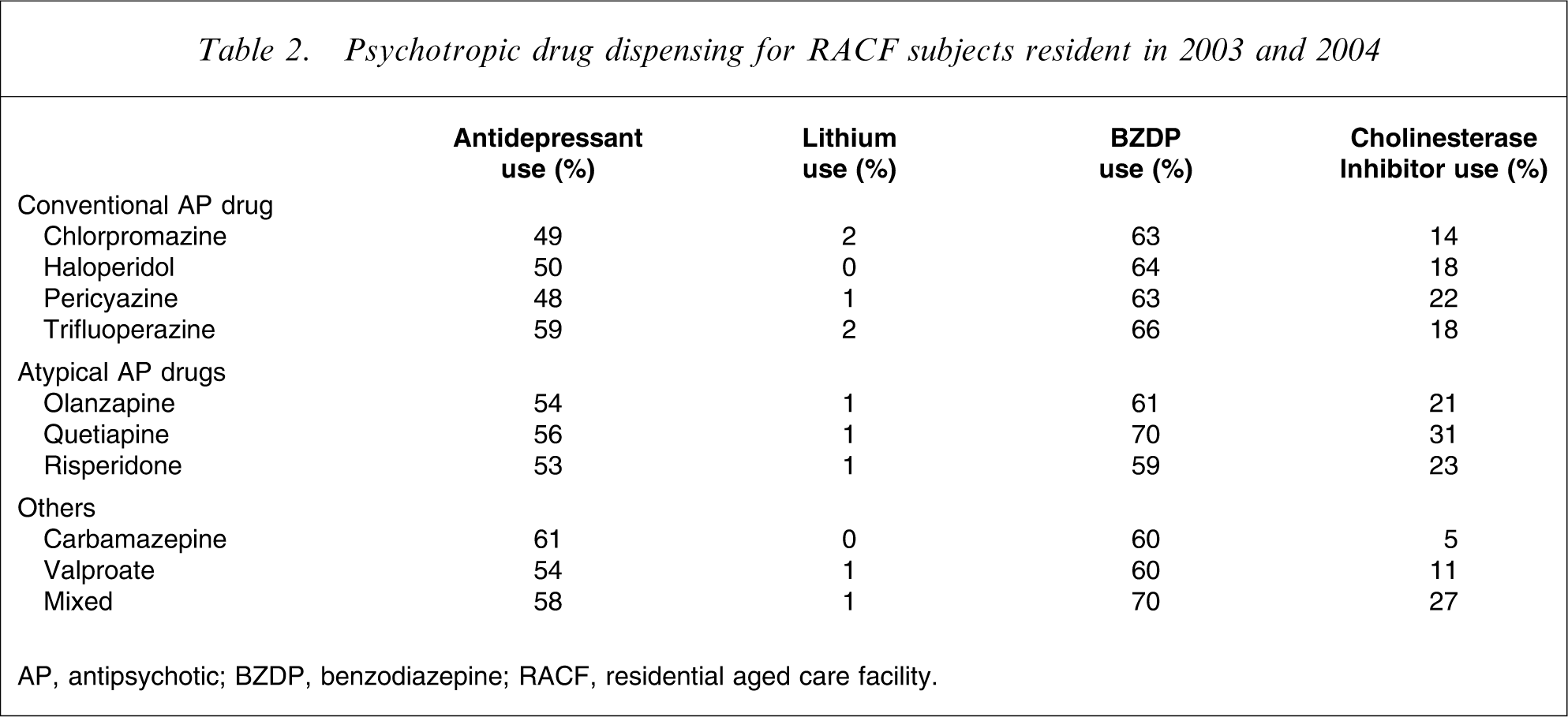
AP, antipsychotic; BZDP, benzodiazepine; RACF, residential aged care facility.
Dispensing of other medications for RACF subjects resident in 2003 and 2004
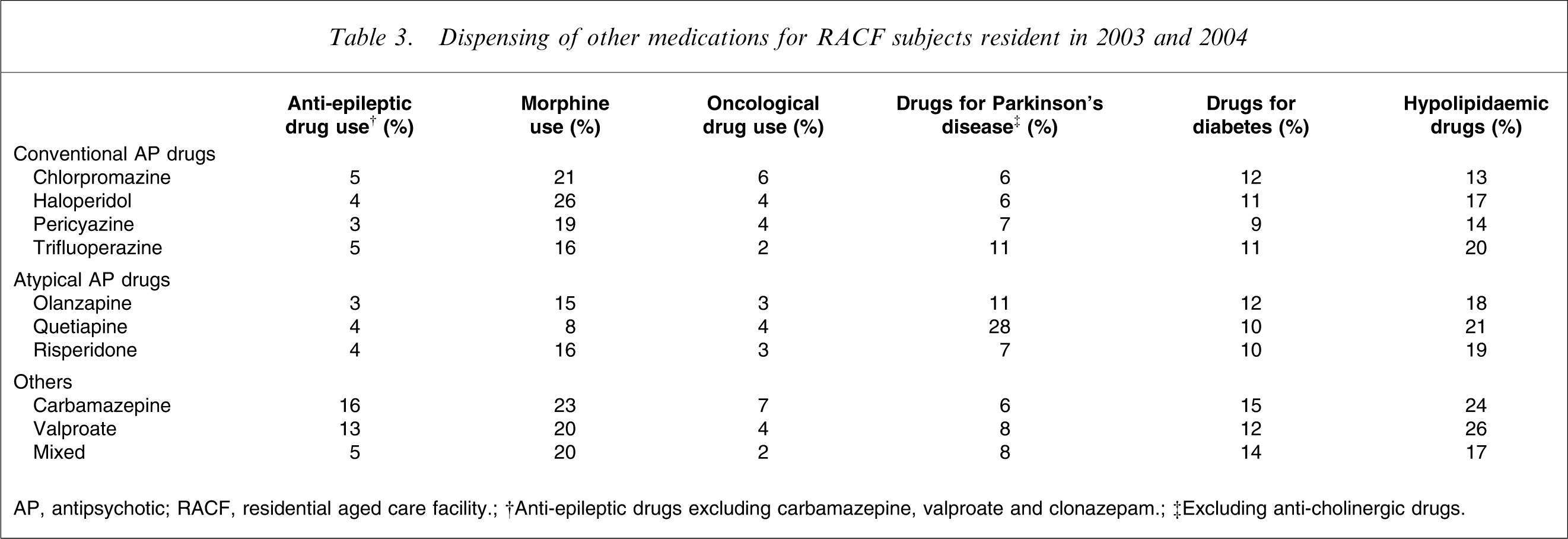
AP, antipsychotic; RACF, residential aged care facility.
†Anti-epileptic drugs excluding carbamazepine, valproate and clonazepam.
‡Excluding anti-cholinergic drugs.
The death rates per 1000 person years at risk were highest for those dispensed haloperidol and chlorpromazine. The death rates associated with these two drugs were significantly higher than for those associated with pericyazine, olanzapine, quetiapine, risperidone, carbamazepine and valproate. The total percentage who died were similar in the haloperidol, chlorpromazine and mixed groups. The lower death rate associated with the mixed group indicated that the mean period of dispensing was longer for this group than for the haloperidol or chlorpromazine groups. The results are displayed in Table 4.
Deaths per 1000 person years of exposure for RACF subjects resident in 2003 or 2004
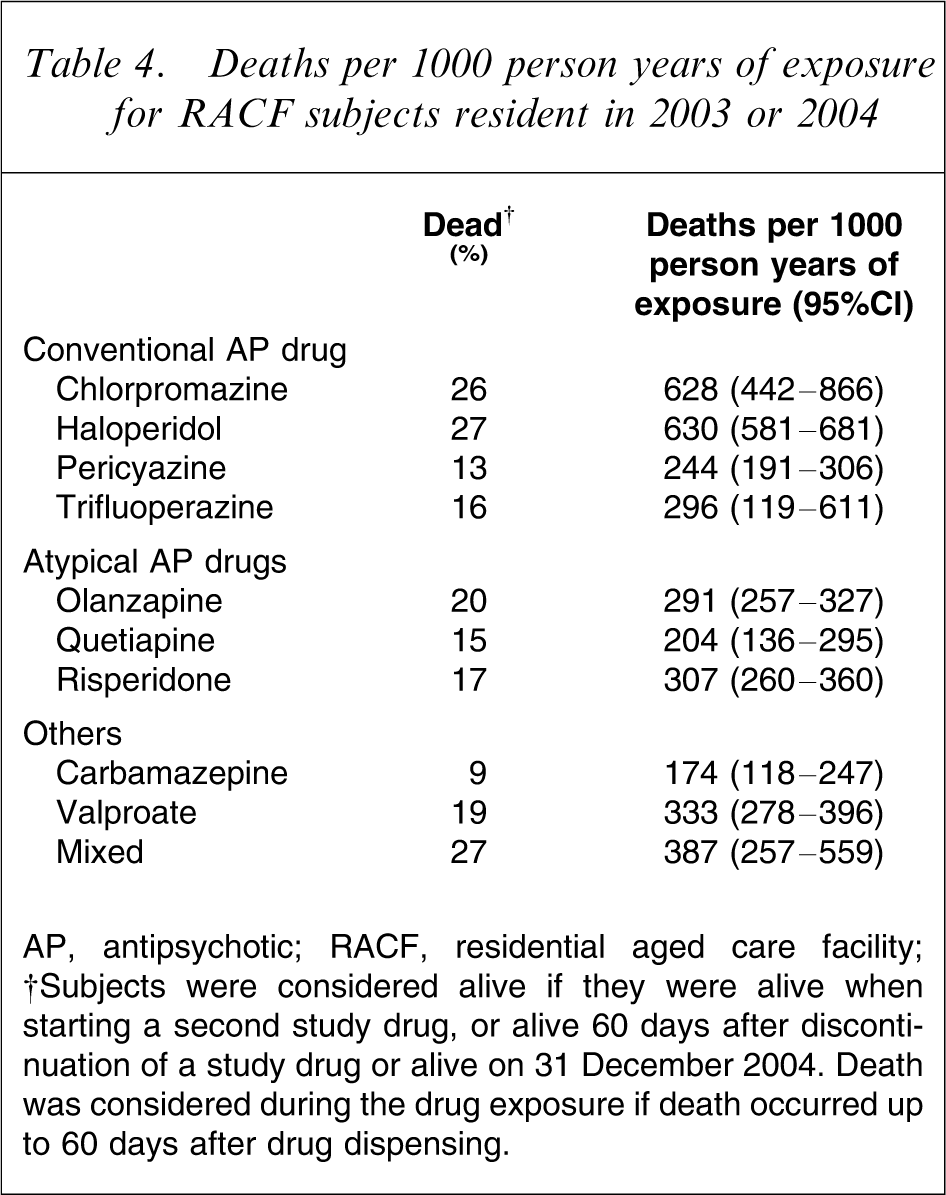
AP, antipsychotic; RACF, residential aged care facility.
†Subjects were considered alive if they were alive when starting a second study drug, or alive 60 days after discontinuation of a study drug or alive on 31 December 2004. Death was considered during the drug exposure if death occurred up to 60 days after drug dispensing.
The adjusted Cox regression models were performed with 6573 subjects because thioridazine (n = 12) and amisulpride (n = 17) groups were omitted. Statistically significant instantaneous RRs, with olanzapine users as the referent are displayed in Table 5. The instantaneous RR for those dispensed haloperidol was 1.67 (95% confidence interval (CI) = 1.50–1.84, p < 0.001) and for chlorpromazine it was 1.75 (95%CI = 1.31–2.34, p < 0.001). The RRs of death associated with the dispensing of haloperidol and chlorpromazine were higher in the regression model restricted to 60 days follow up (haloperidol RR = 2.17, 95%CI = 1.86–2.53, p < 0.001; chlorpromazine RR = 2.72, 95%CI = 1.84–4.01).
Statistically significant instantaneous risk ratios of death in RACF subjects resident in 2003 or 2004
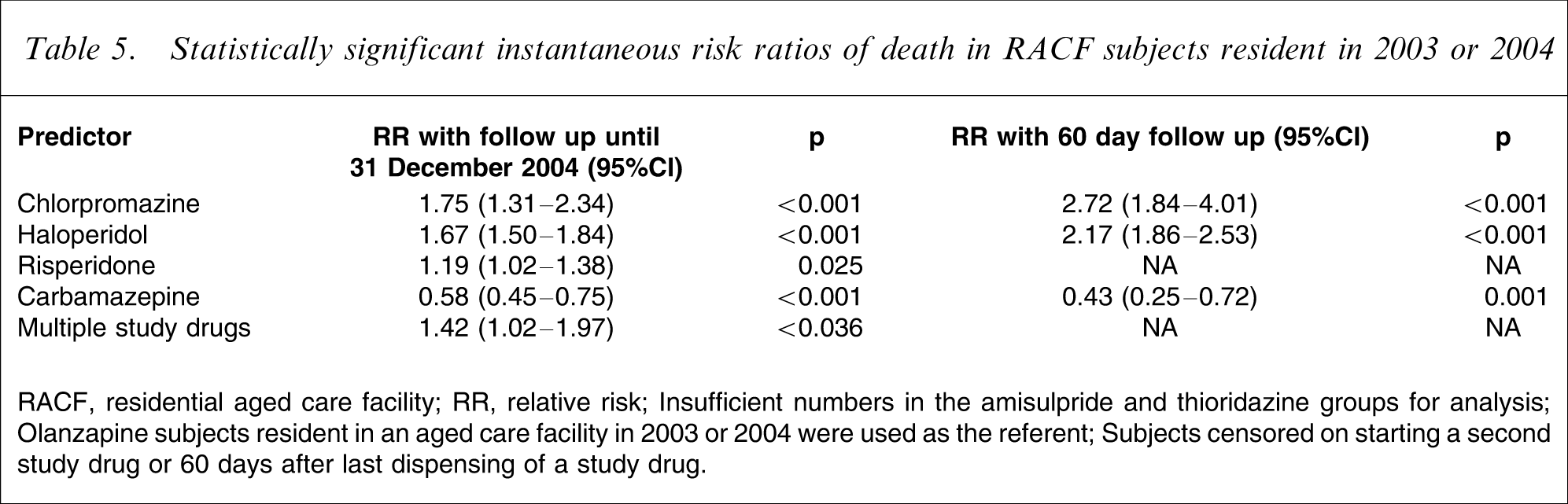
RACF, residential aged care facility; RR, relative risk.
Insufficient numbers in the amisulpride and thioridazine groups for analysis.
Olanzapine subjects resident in an aged care facility in 2003 or 2004 were used as the referent.
Subjects censored on starting a second study drug or 60 days after last dispensing of a study drug.
Discussion
In the present study haloperidol was the most frequently dispensed antipsychotic drug, followed by olanzapine and risperidone. Haloperidol and risperidone were the antipsychotic drugs most commonly dispensed to newly admitted nursing home residents in Ontario [3]. A medication chart review in 40 Sydney nursing homes in 2003 reported olanzapine and risperidone as the most commonly dispensed antipsychotic medications, followed by haloperidol [2]. In the Sydney study, the recorded diagnosis for 77% of residents with haloperidol prescriptions was dementia or cerebral disease without schizophrenia [2]. This was higher than the proportion of residents dispensed olanzapine or risperidone who had the diagnosis of dementia without schizophrenia. At the time of the Snowdon et al. study atypical antipsychotic drugs were subsidized under the Pharmaceutical Benefits Scheme for treatment of schizophrenia but not for behavioural disturbance in dementia. From the same study, six out of 11 prescriptions for chlorpromazine were for those with dementia or cerebral disease without schizophrenia [2]. By contrast, in the present study, only users who had been naive for study drugs in the previous 12 months were enrolled. Thus the proportion of subjects in the present study with schizophrenia would be lower than that in the Snowdon et al. study, which included prevalent users.
The dispensing of haloperidol and chlorpromazine was associated with an increased risk of death compared with the risk for olanzapine users (haloperidol RR = 1.67, 95%CI = 1.50–1.84, p < 0.001; chlorpromazine RR = 1.75, 95%CI = 1.31–2.34, p < 0.001). The risk was even higher in the first 60 days after initial dispensing. While adjustment was made for many covariates, the observational nature of the present study meant that it was impossible to exclude confounding by medical illnesses, such as delirium. Although reversing the underlying cause and non-pharmacological measures are the mainstay of treatment for delirium, guidelines have suggested the use of haloperidol [29]. Despite some small RCTs supporting their use [30], neither olanzapine nor risperidone has received the indication for use in delirium. However, 64% of US experts ranked risperidone as a first-line treatment for delirium, while only 41% ranked haloperidol as a first-line treatment [25].
In the larger study to which this cohort belonged, chlorpromazine was associated with an increased RR in the general incident but not prevalent groups [19]. The prevalent cohort represented a survivor group and their risk was less likely to be confounded by delirium or terminal agitation. In contrast, haloperidol was associated with increased risk in all groups analysed [19]. The RR associated with haloperidol dispensing in the general incident cohort was 2.26 (95%CI = 2.08–2.47, p < 0.01). In a further analysis, incident subjects with fewer than 31 days follow up were excluded from the Cox regression model to reduce confounding by delirium or terminal agitation. The RR associated with haloperidol in the sub-analysis excluding those with fewer than 31 days follow up was reduced to 183 but remained highly significant. The RR from the Cox regression model for the prevalent group was lower than in the incident group but was also highly significant (prevalent haloperidol users, RR = 1.38, 95%CI = 1.23–1.54, p < 0.001).
Other data linkage studies have reported an increased risk of death associated with conventional drug dispensing [18, 31]. Wang et al. compared aggregated conventional antipsychotic data with aggregated atypical antipsychotic data [18]. This study measured the RR over three time intervals but did not include a measure of dispensing continuity [18]. In the present study the associated RR was not uniform among conventional antipsychotic medication, suggesting that use of aggregated data may not provide the best assessment of risk. Like the studies of Herrmann et al. and Gill et al.[20, 21], we assessed the continuity of dispensing and censored as alive those who survived 60 days from the last dispensing.
In a further study, those admitted with pneumonia who were dispensed conventional antipsychotic medications had a slightly higher risk of death compared with those dispensed atypical antipsychotic drugs, although that study did not control for the diagnosis of dementia [32]. One previous study reported increased risk of death with risperidone use as compared with haloperidol use [33]. However, the study included non-elderly, as well as elderly, subjects and the authors postulated that risperidone was more likely to be used in frail patients. Other studies have not found an increased risk of admission with stroke for those dispensed atypical antipsychotic drugs compared with those dispensed conventional antipsychotic medications [20, 21].
Earlier data linkage studies analysed incident and prevalent users [34]. Analysis of prevalent data may underestimate the real risk of death, because those who die soon after exposure are not included. Recent studies have used incident user designs [18, 20, 21]. The RR of death associated with antipsychotic drug dispensing among prevalent users demonstrates the continuing associated risk for survivors. The study by Ray et al. compared antipsychotic medication users with non-users [34]. However, in non-randomized cohorts, users and non-users may differ in significant, unmeasured ways. The present study, like more recent studies, compared users of different types of antipsychotic medication [18, 20, 21].
In the US Food and Drug Administration meta-analysis of atypical antipsychotic drugs in dementia, excess deaths in the treated groups were caused by cardiac conditions and infections, especially pneumonia [23]. Haloperidol, risperidone, sertindole, clozapine and olanzapine all produced a dose-dependent QTc prolongation in an isolated animal heart model [35]. Furthermore, haloperidol is a potent D2 receptor antagonist and has been noted to cause extrapyramidal side-effects at doses used to treat behavioural and psychological symptoms in dementia (BPSD) [36]. Antipsychotic medication has been associated with aspiration pneumonia in those with Alzheimer's disease [37]. This may relate to the dopamine receptor antagonism causing swallowing dysfunction.
A careful meta-analysis of 15 RCTs of atypical antipsychotic medication used in dementia established an increased odds ratio (OR) of death for treated subjects (OR = 1.54, 95%CI = 1.06–2.23) [17]. Nine of these trials were unpublished at the time of the meta-analysis. The unpublished trials did not differ from the published trials in risk of death but were more likely not to have demonstrated efficacy in the primary outcome measures [17]. There were no significant differences in the ORs of death among the atypical antipsychotic drugs, although the non-significant trend favoured risperidone over olanzapine [17]. The recently published results from the CATIE-AD trial demonstrated a difference among groups of side-effects, such as sedation or extrapyramidal side-effects [15]. However, there were few serious adverse events, and no statistically significant differences among the treatment groups and placebo [15].
By including carbamazepine and valproate as study drugs we were able to establish the RR of death associated with antipsychotic dispensing without the confounding effect of carbamazepine or valproate treatment. However, this approach had some disadvantages. Some incident antipsychotic users were classified as prevalent users because they had been dispensed carbamazepine or valproate. More users were considered to have started a second study drug and were censored at that point. Those using carbamazepine and valproate were less likely to have been dispensed a cholinesterase inhibitor and possibly less likely to have received these medications for behavioural or psychological symptoms of dementia. Thus the lower RR associated with carbamazepine dispensing may reflect its use in other conditions.
The strengths of the present study are the large number of subjects and the inclusion of frail elderly users of antipsychotic medications. Like other data linkage studies, the observational design precluded randomization and hence other factors may confound the association between antipsychotic drug dispensing and death. For example, quetiapine users were more likely be dispensed drugs used in Parkinson's disease and less likely to have received morphine. Although adjustment was made for these differences, other unmeasured differences may exist among the drug groups. In the present study we adjusted for morphine use, cancer drug use and the total number of different drugs dispensed in the year prior to the initial study drug dispensing, but there may have been other unmeasured differences among the study drug groups for which we did not adjust. The total number of different medications used in the year prior to the index dispensing is a validated measure of overall comorbidity [28]. The present study, like other observational data linkage studies [18, 20–22], used dispensing rather than consumption data. However, residence in an aged care facility would suggest better compliance compared with community dwellers. It is a strength of the present study that continuation of dispensing was monitored and subjects censored 60 days after the last dispensing. Other studies did not measure continued dispensing and hence the model was not responsive to discontinuation of medication [18].
The uncertainties of observational data linkage studies limit the direct application of the present study to clinical practice. However, the increased risk associated with haloperidol and chlorpromazine dispensing in this cohort bears further scrutiny. The heterogenous risk associated with individual conventional antipsychotic drugs dispensing supports analysis of individual drug data rather than aggregated drug data. The present study supports the findings from other observational studies that atypical antipsychotic medication were not associated with an increased RR of death compared with conventional medications. Only large randomized trials will establish the RR for conventional antipsychotic drugs used in dementia with certainty.
Footnotes
Acknowledgements
The present study was funded by an applied research grant from the Department of Veterans’ Affairs. The NSW State Office of the Department of Veterans’ Affairs extracted the data. The authors are grateful for the support of Dr Graeme Killer and Skye Buckman from the Department of Veterans’ Affairs, National Office. The authors are also grateful to the New South Wales State Office of the Department of Veteran's Affairs. Any errors or omissions remain the responsibility of the authors.
