Abstract
Clinically relevant weight gain is a problem for a significant proportion of people taking some of the second generation antipsychotics [1]. Weight gain and obesity contribute to an array of medical conditions [2] as well as medication non-compliance [3] and possibility to reduced quality of life among users of antipsychotic medication [4].
Although observed clinically, and thought to be a primary contributor to antipsychotic-induced weight gain [5], changes in energy intake have been quantified in only one published study to date [6]. Gothelf et al. [6] found that in a group of 10 adolescent males who began taking olanzapine, energy intake (measured using a 2 day food record) at baseline and after 4 weeks increased by an average of 589 kcal day−1. As the weight gain observed was in excess of that predicted from the increase in energy intake, there must also have been a commensurate reduction in total or overall energy expenditure. While there has been speculation that a reduction in resting energy expenditure (REE) may contribute to antipsy-chotic-induced weight gain [5], two recent longitudinal studies found no change in REE per kilogram of body weight [7] or per kilogram of fat-free mass [6]) with antipsychotic administration.
At the most basic level, weight gain is related to an energy intake that is in excess of that expended. From a practical point of view, it is the magnitude of total energy expenditure (TEE) that is important rather than relative changes to its individual components. Regardless of the precipitating factors, when energy expenditure is lower than energy intake over the long term, weight gain and eventually obesity will result. Recent advances in measurement techniques have made it possible to reliably assess TEE in free-living conditions [8]. Previously, accurate measures of TEE were not possible outside of specialist research settings. Doubly labelled water (DLW) is the gold standard methodology used to measure TEE and hence activity energy expenditure (AEE) in free-living conditions. It is a well-established reference method for the investigation of energy metabolism and body weight regulation associated with weight gain, weight loss and obesity [8, 9]. It is an isotope dilution technique which can accurately determine energy expenditure over a period of time, usually 10–14 days. Doubly labelled water is used for the assessment of energy requirements, TEE and AEE in health and disease, to study body weight regulation and the development and treatment of obesity and to validate tools that assess energy intake and physical activity [8].
The World Health Organization (WHO) recommend that energy requirements be assessed via energy expenditure rather than energy intake [10]. Extensive databases have been developed over the past 20 years which accurately quantify energy expenditure via DLW in different population and clinical groups [11–13], yet there are no published data which accurately quantify TEE in people with schizophrenia. We decided to apply the DLW method to assess TEE and the amount of energy expended on physical activity – AEE in a group of men with schizophrenia who had been taking clozapine. As a result of the worldwide shortage of DLW at the time of this study, a control group was not used. Practical implications for clinical practice and research were interpreted from the wealth of highly accurate energy metabolism data for the general population and specific clinical groups previously published.
The purpose of this research project was to investigate energy expenditure in a group of men taking clozapine. The null hypothesis that, in men taking clozapine, TEE is similar to the level recommended by WHO for optimal health, was tested. The specific aims were to (i) measure TEE using the DLW method, (ii) determine the proportion of TEE related to physical activity, and (iii) consider the potential role of physical activity in weight gain or obesity in men taking clozapine.
Method
Men who had been prescribed clozapine for at least 6 months were recruited from The Park – Centre for Mental Health, a new residential mental health facility in Brisbane, Australia. All participants were diagnosed with schizophrenia and met the following inclusion criteria: weight stable; no serious comorbid medical condition known to affect REE; no comorbid substance misuse; capable of giving informed consent. This study was approved by the Human Research Ethics Committees at the West Moreton Health Services District and the Queensland University of Technology. All participants gave written informed consent which was independently witnessed.
Study protocol and outcome measures
All participants attended the General Health Services Clinic at The Park – Centre for Mental Health for the determination of REE, anthropometric measurement and to take the DLW. Clinical files were reviewed to determine psychiatric diagnosis, daily clozapine dose, time since commencement on clozapine and body weight at the commencement of clozapine.
Height, body mass and waist circumference (halfway between the lowest lateral border of the rib cage and the iliac crest) were measured. Body mass index (BMI) was calculated from height and weight measures. Body composition was determined by measuring total body water via the deuterium (2H2O) dilution method [14] as part of the DLW protocol.
Free-living TEE was measured using the DLW method, the gold standard for determining free-living energy expenditure which has been validated in many different groups of people in a variety of contrasting living situations [9]. The participants orally consumed a dose of DLW (0.05 g kg−1 2H2O and 0.15 g kg−1 H218O). A single urine sample was collected before the dose and 5 hours after the dose was administered. Urine samples were then collected daily for 10 subsequent days. The difference in the elimination rates of the two isotopes from the body is equivalent to the rate of carbon dioxide production which can be converted to the average daily energy expenditure using Weir's equation [15]. Urine samples and dosing mixtures were analysed using an isotope ratio mass spectrometer system (PDZ Europa, Crewe, UK) at the Energy Metabolism Laboratory at the Queensland University of Technology, Australia. Total energy expenditure was calculated using the multipoint slope-intercept method, with tracer dilution spaces calculated using the back extrapolation method [9].
Resting energy expenditure was measured using a ventilated hood system (Deltratrac II metabolic cart, Datex, Helsinki, Finland). Participants were tested immediately after waking in the morning, having fasted for at least 10 hours. Following a 10 minute adaptation phase to the hood, oxygen consumption and carbon dioxide production was continuously analysed for 30 minutes. Steady-state data were used for the analyses. ‘Steady-state’ was defined as 10 minutes of data where the coefficient of variation in the minute-by-minute data for oxygen consumption (VO2) and carbon dioxide production (VCO2) was less than 10%. The coefficient of variation of the minute-by-minute steadystate data used to calculate REE was 5.2 ± 2.3% for VCO2 and 4.7 ± 1.6% VO2. Physical activity level (PAL) was calculated as the ratio of TEE : REE. Activity energy expenditure was calculated as 0.9 × TEE − REE to account for the thermic effect of food (10% of TEE). Activity energy expenditure was corrected for the influence of body weight on energy expenditure by dividing AEE by body weight [16].
Data analysis
The data were analysed using SPSS for Windows (version 11.5; SPSS Inc., Chicago). Anthropometry, body composition, energy expenditure and physical activity data are presented as means ± SD. Pearson product moment coefficient of correlation (r) was used to determine the strength of the relationship between weight change as percentage of baseline weight (weight at the time of clozapine commencement) and energy expenditure indices. Actual TEE was compared with recommendations of TEE by WHO for optimal health (1.75 × REE) [17] by a paired t-test. Level of statistical significance was set at 0.05.
Results
Of the 43 men approached to take part in this study, eight agreed to participate. The participants were all diagnosed with schizophrenia and although, all participants were taking clozapine as their primary antipsychotic, a degree of polypharmacy was found. Participants were also taking amisulpride (1), chlorpomazine (1), citalopram (2), diazepam (1), paroxitine (1), risperidone (1), sodium valproate (1) and venafaxine (1). The mean daily clozapine dose of participants was 456 ± 143 mg, participants had been medicated for 20.5 ± 12.8 months. Summary Health of the Nation Outcomes Scales (HoNOS) score was 8.1 ± 6.0. HoNOS is a routine clinical tool used in Australia to measure mental health outcomes [18]. There was no difference in age, weight and BMI, clozapine dose and summary HoNOS score between those who chose to participate and those who did not. However, those who did not participate had been taking clozapine for longer (t = 2.31, p < 0.05). The physical characteristics, energy expenditure and physical activity of the group are shown in Table 1. Actual TEE was 21% lower and significantly different (t = 4.15, p = 0.004) to the level of energy expenditure recommended by WHO [10, 17].
Characteristics, energy expenditure and physical activity of participants
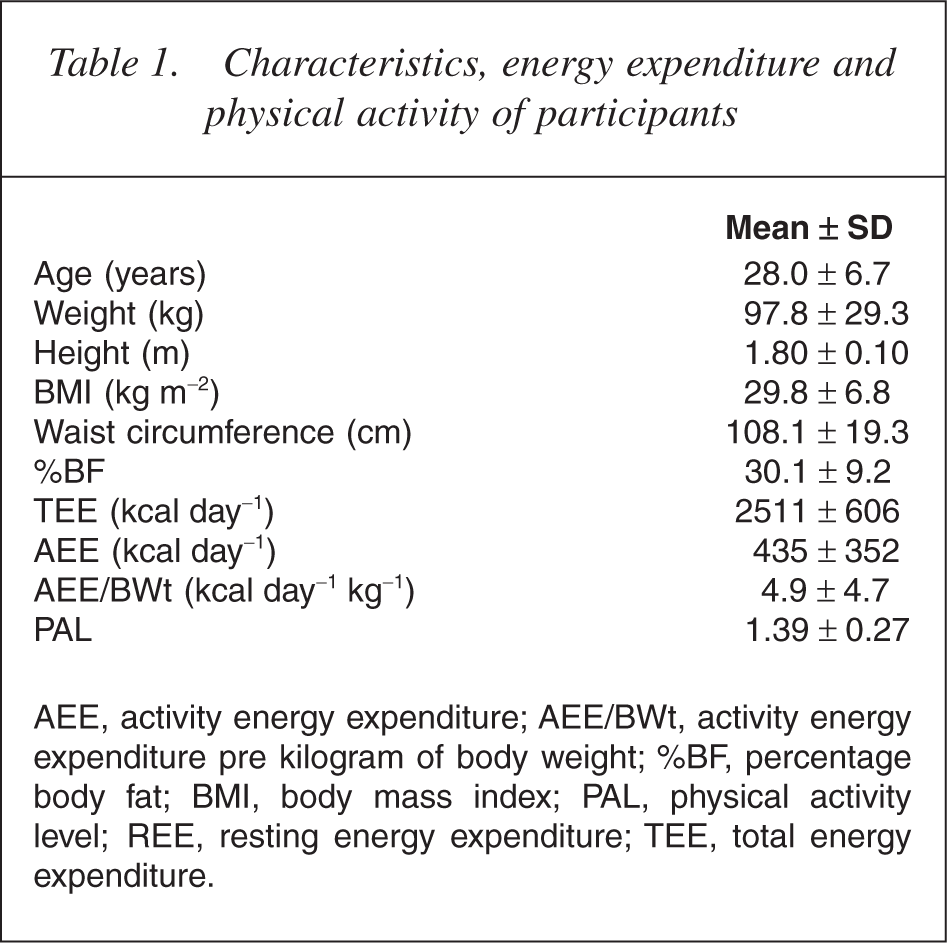
AEE, activity energy expenditure; AEE/BWt, activity energy expenditure pre kilogram of body weight; %BF, percentage body fat; BMI, body mass index; PAL, physical activity level; REE, resting energy expenditure; TEE, total energy expenditure.
Weight gain in participants was on average 8.1 ± 15.5 kg and ranged from a gain of 32 kg to a loss of 8 kg. Weight change as a percentage of initial weight varied from a loss of 10.3% of initial body weight to a gain of 29.8% of initial weight (mean 8.2 ± 16.1%). There were nonsignificant correlations between percentage change from baseline weight and AEE and PAL with AEE/BWt (AEE per kilogram of body weight) trending towards significance (r = −0.67, p = 0.06).
Discussion
This study reports on the first application of the DLW technique to measure energy expenditure in people taking antipsychotic medication. It provides the first accurate values for energy expenditure in men with schizophrenia who were taking clozapine. We have described the participant characteristics in terms of age, weight, height, BMI, body composition, waist circumference, diagnosis, severity of symptoms, clozapine dose and length of clozapine treatment to allow for comparison with other clinical groups. The participants in this study appeared to reflect the larger group of 43 men who were approached to participate, although the non-participants had been taking clozapine for longer. The average daily energy expenditure of 2500 kcal day−1 for a group of men with a BMI of 30 kg m−2 and body fat of 30% is low – 20% lower than the energy expenditure level suggested by WHO. The participants who had all been taking clozapine for more than 6 months were weight stable during the study so low energy expenditures was matched with relatively low energy intakes. Increasing energy expended on physical activity is the only way that TEE that can be increased at will. Resting energy expenditure, the largest component of TEE was also lower than predicted [19]. As energy expended on physical activity is the most variable and easily changed of the energy expenditure compartments, increases in physical activity need to be the primary venue for increases in energy expenditure.
A PAL of below 1.5 is considered to be sedentary [17]. The PAL of 1.39 in the group studied confirms reports of the sedentary lifestyle of those with psychotic illness [5]. A PAL of 1.4 equates to being relatively inactive as part of everyday life, not engaging in any exercise, and spending a predominant amount of the day sitting. Physical activity level derived from DLW, of men the same age as the participants in our study have been reported at between 1.76 and 1.79 [11, 12]. Weinsier et al. [20] found that women who maintained their body weight over 1 year had a PAL of around 1.70 while those who gained weight (+9.5 kg) over a year had a PAL of 1.55. Physical inactivity, defined by low PAL is an independent risk factor for chronic ‘lifestyle’ diseases such as cardiovascular disease and diabetes and also for weight gain and obesity [17]. This study provides objective evidence that the level of physical activity in this group of men with schizophrenia is sufficiently low to impact upon chronic lifestyle diseases and contribute to weight gain and obesity regardless of nutritional status.
Twenty-four hour calorimeter studies have also shown that a significant amount of energy can be expended in spontaneous physical activity. Some differences in energy expended by individuals in confined situations can be accounted for by substantial amounts of fidgeting. Ravussin [21] reported that energy expended on fidgeting averaged about 350 kcal day−1 ranging from 140 to 700 kcal day−1. While the mean AEE of the group in this study was approximately 450 kcal day−1, over half of the participants had an AEE of less than 250 kcal day−1. This suggests that some of the participants were engaging in very little spontaneous activity including fidgeting or postural readjustments. The low levels of AEE and PAL of many of the participants in this study means that without considerable restraint in energy intake, it will difficult for them to avoid future weight gain.
Limitations of this study include the small sample size and the lack of control group. The number of participants in this study was limited not only by the expense and limited supply of the 18O isotope, but also by the difficulties in recruiting participants. Given that the use of clozapine is restricted largely to those people with severe and persistent psychosis, desire to participate in research projects may be somewhat lower than for other clinical/ population groups. A control group consisting of people with treatment resistant schizophrenia taking a weight neutral atypical medication such as aripiprazole or ziprasidone or who were not medicated, would theoretically assist in distinguishing between characteristics of energy metabolism associated with schizophrenia and that associated with clozapine use. However, the addition of a control group would not necessarily assist to tease out these respective contributions as a small number of people with treatment resistant schizophrenia take ‘weight neutral’ medication. Ziprasidone is not currently available in Australia, the use of aripiprazole is limited in people with treatment resistant schizophrenia and those not taking atypical antipsychotics take a range of psychotrophic medications of varying weight promoting liability. Such ‘control’ groups, even when matched for age, BMI, gender and duration of illness, may reflect the heterogeneous nature of schizophrenia, specifically the differences in functioning, disability and skill level which may independently impact on energy metabolism rather than differences which could be attributed to clozapine use alone.
Implications for atypical antipsychotic-induced weight management
Energy expenditure on physical activity is important in controlling weight gain and obesity. However, the role that physical activity plays in atypical antipsychotic-induced weight gain and obesity is less clear. This study found that the relationship between current AEE per kilogram of body weight and change from baseline weight in men taking clozapine was strong although not significant (correlation coefficient of 0.70 was required for significance because of the small number of participants). Further analysis of the prospective study of Gothelf et al. [9] revealed that changes in food intake account for over half of the energy surplus required for participants to gain weight (4.8 kg over 6 weeks) while taking olanzapine, with resting energy requirements accounting for less than 10% and reductions in physical activity and in theory the thermic effect of food accounting for over one third.
Weight management strategies for people taking antipsychotic medication have primarily focused on reducing calorie intake [22]. Recently, some programs have taken a more balanced approach by also attempting to increase energy expenditure by increasing AEE [23]. From a practical point of view, a graded approach to increasing activity is important. A recent article detailed a pragmatic approach to the prevention of diabetes and weight gain associated with atypical antipsychotic usage by recommending 10–15 minutes of exercise a day rather than the standard 30 minute [24]. Ball et al. [25] reported that at the beginning of an exercise program as part of Weight Watchers, participants (who were taking atypical antipsychotics) walked an average of 160 m session−1. By the end of the 8 week program, participants were walking 1.6 km. Another important strategy in this group of people taking clozapine may be to focus on reducing inactivity rather than promoting physical activity. This has proven to be an effective long-term strategy for children in the maintenance of weight loss [26]. Given the sedentary behaviour of the study group a similar change in focus aimed specifically at reducing inactivity rather than at increasing activity may be more effective at increasing overall energy expenditure.
The sedentary nature of the group of participants in this study was reflected in PALs and energy expenditure that fell well short of those recommended by WHO [17] for optimal health and to prevent weight gain. Given the increasing recognition of the importance of sedentary behaviour to weight gain in the general community, further examination of the contributing factors (for example negative symptoms, sedation, functional abilities, skills and lifestyle) to activity levels in this clinical group is warranted. In the obesogenic environment that characterizes modern society, it is easier to match high energy expenditure with energy intake than to match low energy expenditures with energy intake to avoid weight gain. The current research suggests that weight prevention and weight loss programs for people taking clozapine and possibly other atypical antipsychotics need to focus on increasing energy expenditure. In addition to the more traditional focus on reducing calorie intake, increasing PAL and energy expended on physical activity as well a reducing inactivity could prove to be an important clinical components of strategies which aim to prevent, minimize or treat weight gain and obesity in people taking antipsychotic medications even where weight gain is primarily medicated through increased appetite.
Footnotes
Acknowledgements
This research was funded by an unrestricted grant from Novartis Pharmaceuticals as part of the CPMS (Clozaril Patient Monitoring System) program, a Queensland University of Technology Strategic Links with Industry grant and the Ipswich Hospital Foundation.
