Abstract
The search for risk factors for the development of schizophrenia has been an ongoing effort, resting on the assumption that risk factors may be avoided or pliable, thus offering some hope of amelioration or prevention of psychotic illness. Within this context, the three groups of variables postulated to contribute to schizophrenia have been arbitrarily classified as (i) demographic variables; (ii) innate predisposing or protective factors; and (iii) environmental stressors.
Of these, Bromet and Fennig [1] view family history and social class as strong confirmed risk factors, while the confirmed, potentially strong factors include age and gender, rheumatoid arthritis, season of birth and developmental complications. However, the more influential studies supporting these risk factors mainly focused on Caucasian populations and very little is known about the role of these factors, and more specifically season of birth, in indigenous African populations.
Indeed, although more than 250 studies have looked season of birth as a risk factor for the development of schizophrenia, none of these were in an African population [2, 3]. Nevertheless, these studies have almost consistently shown a winter–spring excess of 5–8%. Winter birth has been found to lead to a proportionally larger number of schizophrenic patients in later life (5–15% higher than other seasons) [3, 4]. This differential has been shown to be linked to gender [5] and where a negative family history was present [6].
Twelve Southern Hemisphere studies have been published, but a large number of methodological problems have been cited, of which matching of controls and small sample size was the most important. McGrath and Welham [7] have since published a careful meta-analysis of 10 Southern Hemisphere studies (n = 20 017 schizophrenia cases) and could not find a significant seasonality effect (no specific winter birth excess).
Season of birth has also been associated with different subtypes of schizophrenia, differences in prognosis, demographic factors and clinical presentation, that is, an excess of July births in French Kraepelin subtype schizophrenics [8], summer births in patients with deflcit schizophrenia (includes negative symptoms primary to schizophrenia) [9, 10]. Several studies found no relationship with various variables such as age of onset, marital status, total duration of hospitalization and number of hospitalizations [3, 11].
Several possible reasons for the seasonality of schizophrenia births have been proposed, including infective processes, genetic factors, obstetric complications, variations in light, environmental toxins, nutrition, climatic changes and even procreational habits of at-risk parents [2, 12]. It has also been argued that the excess could be explained by an age-incidence effect (this means that individuals born earlier in the year should be at higher risk because they are older at the time of the investigation) especially since other disorders such as mood disorders, Down's syndrome, mental retardation and diabetes have been linked to seasonality of birth. However, rejection of the strong age-incidence effect followed after a variety of studies showed (i) a winter excess even when age-incidence had been controlled for and (ii) December excesses (the first month of the northern winter) [13]. In addition, even if season of birth was associated with a higher risk for schizophrenia in a specific population group, it may only account for a small overall contribution to the risk or be a proxy for several underlying factors, such as viral infections and diet [3].
Nevertheless, given the complexity of the search for susceptibility factors for schizophrenia, a comprehensive risk profile should exist for each of the probands that partake in genetic or risk studies for schizophrenia. A comprehensive effort was launched to identify the risk factors for schizophrenia in the Xhosa ethnic group (Nguni-language African group) [14]. This study therefore investigated the seasonality of birth and its possible association with different symptom dimensions of schizophrenia in this African group.
Method
Subjects
Five hundred and forty-seven Xhosa participants with schizophrenia were able to complete the study-related assessments. Participants were recruited from the Western and Eastern Cape Provinces of South Africa. Local mental health care workers from inpatient and outpatient hospital services and community clinics who were familiar with the inclusion and exclusion criteria referred potentially suitable subjects for screening. Informed consent was obtained from participants and where necessary from their caregiver. The study complied with the ethical guidelines for research involving human participants as set out in the Declaration of Helsinki (Edinburgh, 2000) and was conducted in accordance with the written approval and guidelines of the ethics committee of the University of Stellenbosch.
Patient inclusion criteria
Male and female participants known with schizophrenia (DSM-IV) [15];
All grandparents of Xhosa ethnic origin;
At least one living parent and one living sib, or alternatively two living parents.
Patient assessment
Each participant was interviewed by a psychiatrist (D.N.) or experienced, trained research sister (I.M.) who administered the standardized Diagnostic Interview for Genetic Studies version 2 [16] in Xhosa (oral translation), the native tongue of all participants. Collateral information was gathered from hospital chart records and family members. Subjects recruited over the 6-year period were assessed together by raters during the first year of the study and thereafter regular calibration meetings were held. All information, including information on birth month, was obtained by interview.
Symptom ratings
The schedules for the assessment of positive (SAPS) and negative symptoms (SANS) were used to measure symptom severity (as part of the Diagnostic Interview for Genetic Studies). Global symptom ratings for four positive (global hallucinations, delusions, thought disorder and behavioural abnormalities) and five negative symptom domains (global affective changes, alogia, avolition/apathy, anhedonia/asociality and concentration difficulties) were used in the statistical analysis.
Educational attainment was defined as years in the educational system, and dichotomized as more than 7 years and less than 7 years. Marital status was dichotomized to indicate whether a patient has never been married or has been/is in a relationship (married, separated, widowed or divorced). Age of onset was defined as the first reported clinically significant psychotic symptomatology or first contact with psychotic services.
Control group
A Xhosa control group was used to obtain the expected birth month profile for the study population. The control group was selected from a 10% sample of the 2001 official census data of South Africa, and was matched for the period of birth similar to the study population (from 1930 to 1997) and region of birth (Western Cape and Eastern Cape), and totaled more than 6.8 million.
Climate change
As seasonal patterns are related to climate patterns and possible climate change, we looked at relevant research in South Africa. The climate change in South Africa over the past 41 years was analysed by the geography department at Wits University, using temperature data (http://www.sunsite.wits.ac.za). From 1970 onwards, the whole region has shown a general warming trend, much in the same way as the southern hemisphere as a whole [17]. However, much variability in the climate change is seen between regions in South Africa; for example, the annual minimum temperatures of the coastal cities, including the Western and Eastern Cape, are warming more than the annual maximum temperatures. Because it was clear that the patients in our study born over the last 30 years (after 1970) experienced a climate change, the data were grouped into those born before 1970 (≥30 years old) and those born after 1970.
Data analysis
Analysis of the data for birth months suggested a similarity in symptom profiles between autumn and winter months (March to August), and between spring and summer months (results not shown here). Therefore, the SANS and SAPS symptom profile for patients born in autumn or winter was compared with those born in summer or spring. The SANS and SAPS symptoms were defined as positive for those patients with a code of 2 (mild) to 5 (severe).
A logistic regression model of symptom status, including possible confounding variables such as socioeconomic status, gender, religion, education, occupation, marital status, living arrangements, number of children, age of onset and duration of disease, was fitted.
Because of the warming in the climate from 1970 onwards, birth year, categorized as younger than 30 years or older than 30 years, was also included in the model. Since some of the patients were from the same sib pair, generalized estimating equations were used to deal with the correlated nature of the data. The sib-pair responses were assumed to be equally correlated, implying an exchangeable correlation structure.
For comparison of the birth seasons of the Xhosa sample with the census control group, the four seasons were retained and a goodness-of-fit chi-square was used for comparison.
Results
Modelling of the negative and positive symptoms of schizophrenia with season of birth indicated that avolition/apathy was the only symptom complex for which there was a strong trend towards a different profile for the autumn/winter and summer/spring seasons (p = 0.066).
Figures 1 and 2 show, separately for those younger and those older or equal to 30 years, birth months for the patients. It is clear that the season of birth profiles for avolition/apathy are different for the patients older than 30 years, but not for those patients younger than 30 years, as defined by the age at interview. This result was confirmed by the model, which indicated a significant interaction of season of birth with age at interview (categorized as <30 years and ≥30 years, p = 0.014).
Birth months for the patients younger than 30 years (autumn/winter are months March to August).(□) normal: score 0 or 1; (▪) avolition/apathy: score > 1, not significant. Birth months for the patients older than or equal to 30 years (autumn/winter are months March to August). (□) normal: score 0 or 1; (▪) avolition/apathy: score < 1, p =0.03.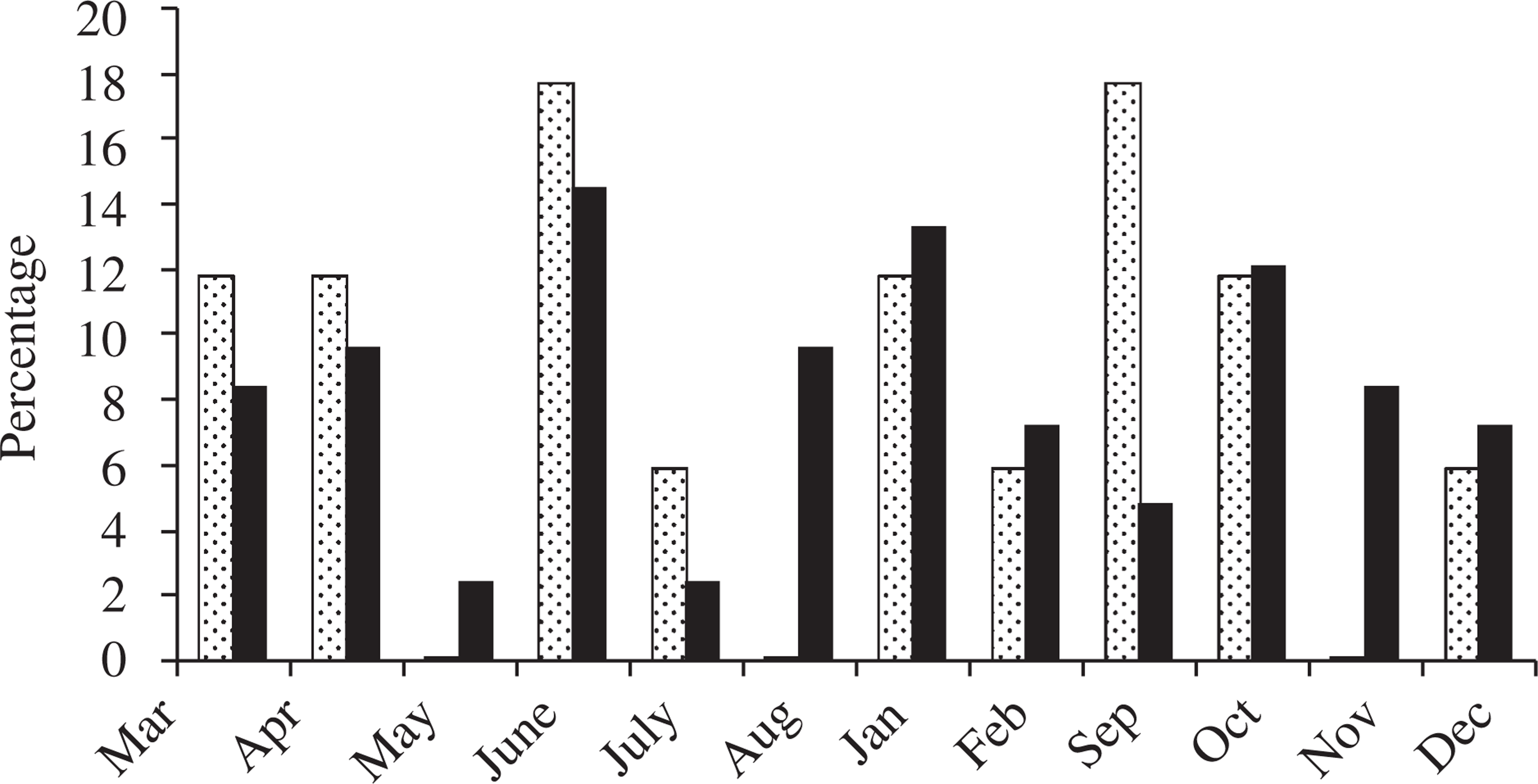
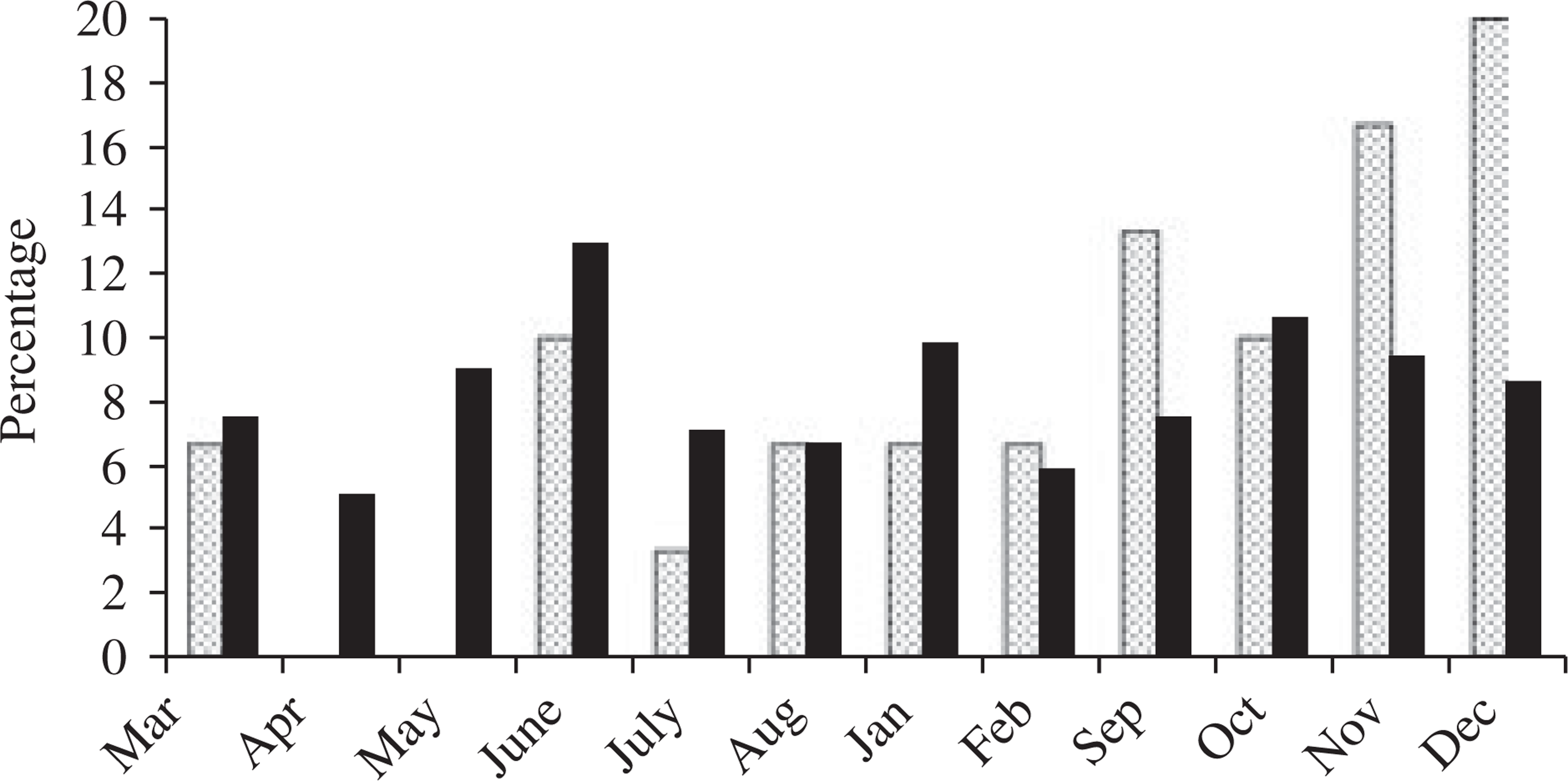
The modelling was repeated separately for the older and younger patients, and a significant result was obtained only for the older group (parameter estimate = 0.973, SE = 0.44, z = 2.22, p = 0.027).
Possible confounders for the model of the older group were then considered. Table 1 reports demographic and clinical data for each of the groups. These variables were included in the multiple regression model for the older group to check for confounding with season of birth. The significant confounders in the multiple model, marital status (p = 0.003) and work status, that is, whether a person received a welfare pension (p = 0.0001), were associated independently with avolition/apathy. The parameter estimate for avolition/apathy in the multiple model did not change compared with the parameter estimate in the univariate model (parameter estimate = 0.989, SE = 0.44, z = 2.23, p = 0.026). No interaction of marital status or work status with season of birth was observed (p = 0.67 and p = 0.75, respectively).
Demographic and clinical variables divided according to their season of birth and age at interview

∗The p-value relates to the significance of the confounders in the univariate model. SD, standard deviation.
From Table 1, it can be seen that the birth rates for older patients born during autumn/winter and spring/summer are 46% and 54%, respectively. The comparable rates in the 2001 census data are 51% in autumn/winter and 49% in spring/summer. Figure 3 gives the birth rates for the four seasons. This shows a 5% decrease of births in autumn and a 4% increase of births in spring for our sample compared with the 2001 census. Therefore, the seasonal birth rates in our sample differed to those in the general Xhosa population (χ2 = 9.0, p = 0.03).
Birth season for Xhosa patients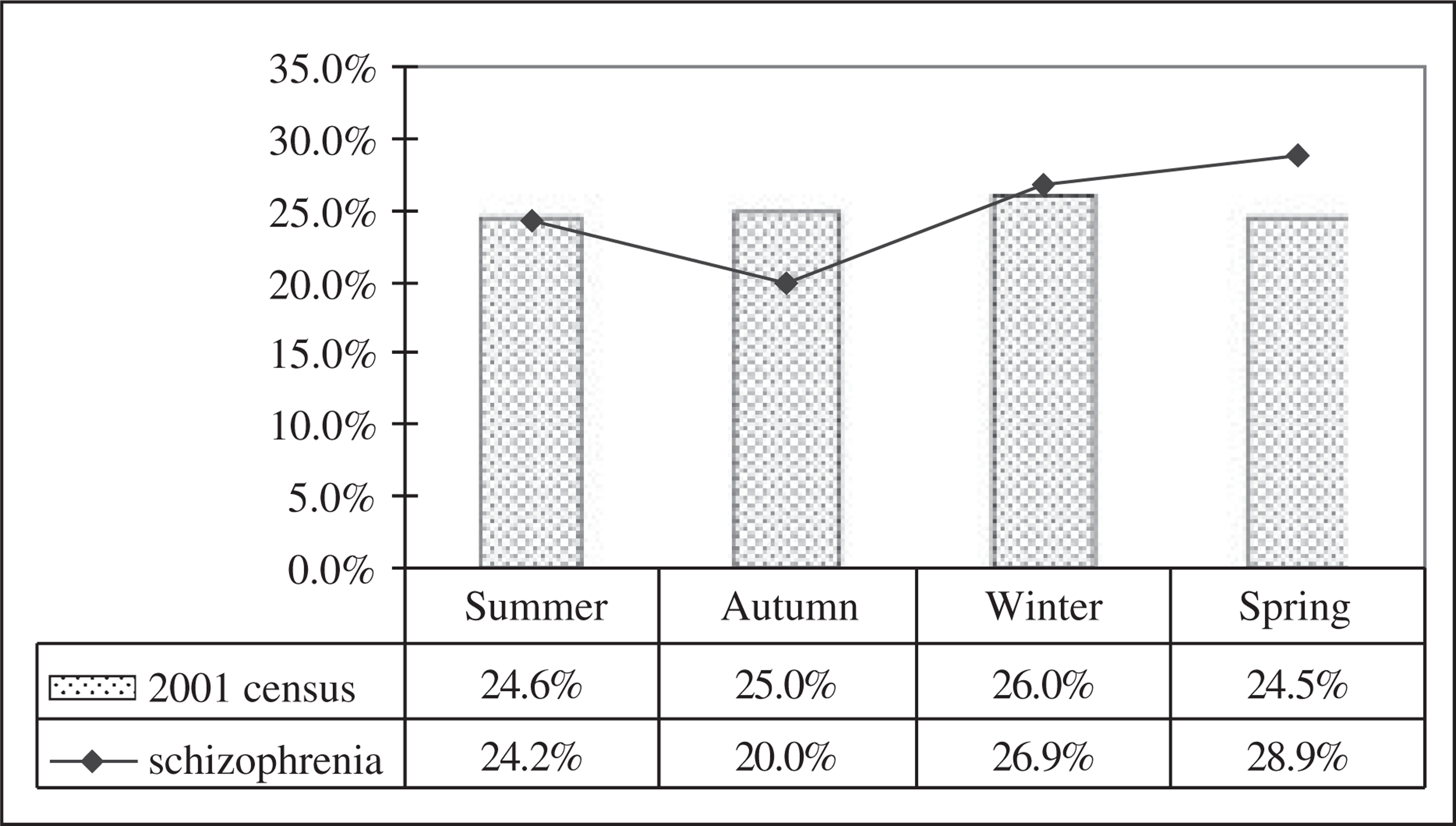
Discussion
Our analyses showed that avolition/apathy occurs nearly three times as often among autumn/winter-born patients older than 30 years as among summer/spring-born patients older than 30 years (OR = 2.68; 95% CI = 1.13–6.42). But why does this result only hold for older patients? One explanation could be the general warming trend across the country, especially in the coastal cities, including the Western and Eastern Cape from 1970 onwards. Although this was a secondary analysis for the study, the study results show clear trends, but with marginal significance. And although the external control group was very large and representative of the Xhosa population, it would be more appropriate to use a control group matched for factors other than age, birth region and ethnicity. The birth region is also only a proxy for where the people lived for most of their lives.
Although this is the first report in an African population to suggest a possible relationship between specific negative symptoms and season of birth, Caucasian studies have reported similar findings [3, 10, 18]. Higher levels of anhedonia have also been reported (although not consistently) in patients born 1 month after a winter season with a high rate of infections [3]. Troisi et al. [18] reported that female patients born in winter and early spring had higher negative and anergia Positive and Negative Syndrome Scale scores while men born in other seasons had higher scores on the anergia factor.
Gender was neither a confounder, nor an effect modifier in the relationship of avolition/apathy with season of birth, as previously found by Troisi et al. [18]. Because only about 11% of the sample were female, it is possible that the female sample was too small to detect a significant interaction.
It is unlikely that all winter-born schizophrenics represent a seasonal subtype. Most likely, only a small minority of the winter-born patients belong to the putative subtype whereas the majority of them would be cases of the nonwinter-born subtype, simply born in winter [19].
Our study showed a spring excess of 4%. The fact that the birth information was obtained by interview could impact on the quality of the birth data. The birth months show an unusual peak for the month of June. This phenomenon also occurs in the 2001 census data (see Fig. 4) and could possibly be explained by the data-handling methods of the Department of Home Affairs that allocates fictitious birth dates to individuals with unknown birth dates.
Birth month for Xhosa patients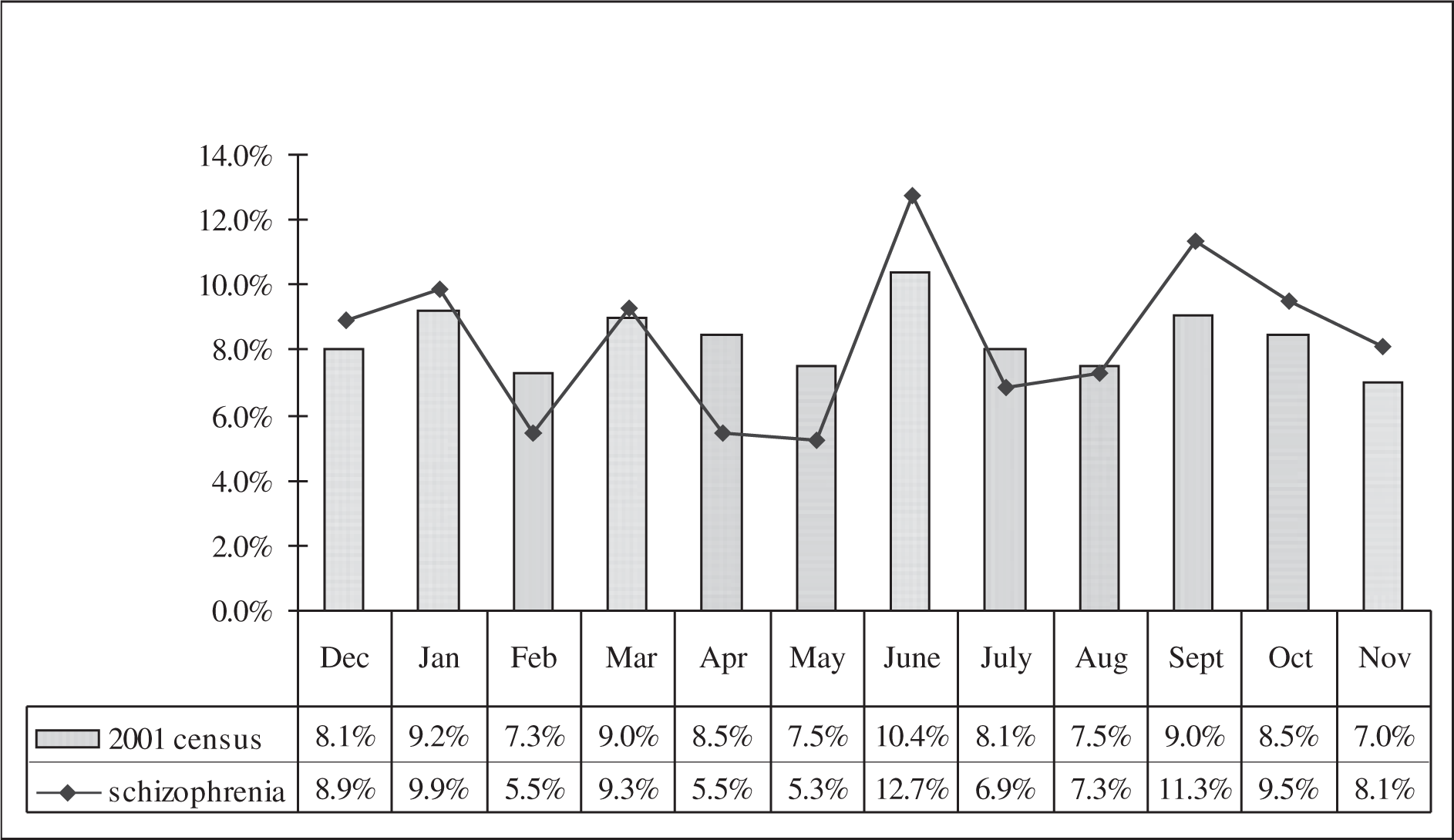
Because of the large familial component of schizophrenia, the possible association with a higher risk for schizophrenia, or at least for a subtype of schizophrenia in the Xhosa population, may account for only a small overall contribution to the risk.
Finally, this study furthers our knowledge about the seasonal birth effect on an indigenous Xhosa population in the Southern Hemisphere. In future, research studies in Africa can possibly investigate the role that temperature changes, and in particular the general warming trend, play in the mechanism of seasonal birth effect and schizophrenia.
