Abstract
Over recent years, there has been increasing interest in self-mutilation as a behaviour that is distinct from suicide attempts [1]. Most patients who report self-mutilation describe it as a behaviour to relieve tension, and with no intent to die. Traditionally, it has often been linked to borderline personality disorder, and one of the criteria for borderline personality disorder is ‘recurrent suicidal behaviour, gestures or threats, or self-mutilating behaviour’. Self-mutilation is most common during adolescent and young adult years, and is relatively uncommon in middle age or older. Briere and Gil [2] have reported that self-mutilation occurs equally among males and females, and has a prevalence of 4% in the general population and 21% in clinical samples. However, in two samples of high-school students, reported rates of self-mutilation have been as high as 13.9% [3] and 21.4% [4]. Selfmutilation has also been associated with childhood abuse and neglect, and with dissociation, guilt, rejection, boredom and emotional distress [1, 2].
Self-mutilation is also a recognised clinical feature of Lesch–Nyhan syndrome, a rare X-linked recessive metabolic disorder with hyperuricemia, although the most common self-mutilatory behaviour is self-biting of lips or fingers, followed by head banging [5]. Selfmutilating behaviour has also been described in laboratory animals, with suggestions that this reflects a genetic predisposition [6].
It is now widely recognised that antidepressant responsiveness may vary between children and adults. Thus in children and adolescents the tricyclic antidepressants are not effective, while fluoxetine is effective [7–9]. We have recently reported that in young adults with depression (aged 18–24 years), fluoxetine is superior to nortriptyline and clomipramine is superior to desipramine [10, 11]. We have also reported that in young adults, but not in older adults, the T allele of G protein β3 (GNβ3) is associated with a differential response to fluoxetine over nortriptyline [12]. We were thus interested in whether this T allele is associated with other aspects of depression that are agerelated. As self-mutilation is an age-related behaviour, we were interested in exploring its association with the T allele. Furthermore, we hypothesized that if there was an association, it would be stronger in young depressed patients than in depressed patients of all ages. Finally, we were interested in whether such an association would remain significant when two variables traditionally associated with self-mutilation, borderline personality disorder and childhood abuse, are also included as risk factors.
Method
Patients
The depressed patients in this study were recruited for a 5-year prospective outcome study, with initial randomization to fluoxetine or nortriptyline as the first antidepressant drug. More details on the clinical characteristics, personality features and predictors of response to the initial antidepressant are available in other papers [10,13–15]. In brief, patients needed to be suffering from a current major depressive episode as the principal diagnosis and be suitable for treatment with fluoxetine or nortriptyline. Psychiatric exclusion criteria were minimized but included severe alcohol or drug dependence if it was the principal diagnosis, a history of schizophrenia or mania (hypomania was allowed) and severe antisocial personality disorder. At the time of entry to the study, patients were required to be free of all psychotropic medications for a minimum of 2 weeks, except an occasional hypnotic for sleep. After giving written informed consent to participate in this research study, which had been approved by the Canterbury (New Zealand) Ethics Committee, patients reported for a detailed clinical and neurobiological assessment.
Assessment
All patients were assessed and treated by a psychiatrist or a senior psychiatric registrar. Patients were interviewed using the Structured Clinical Interview for DSM (SCID) [16], and were rated with the Montgomery–Asberg Depression Rating Scale (MADRS) [17], the Hamilton Depression Rating Scale (HDRS) [18] and the Mental State Examination for Melancholia [19]. The SCID had been extended to cover all of the DSM-III-R and DSM-IV melancholic and atypical depression criteria. At the end of the mood section of the SCID we had added additional questions about lifetime suicide attempts and selfmutilation. For self-mutilation, the initial probe question was whether they had ever harmed themselves to relieve tension. If they answered yes to the screening question, they were then asked about methods, how often, how recently and about their mood state when self-mutilating.
At the time of their baseline assessment, patients completed the screening self-report questionnaire (SCID-PQ) for Axis II personality disorder symptoms. Approximately 6 weeks later, the treating psychiatrist using results from the SCID-PQ and their clinical knowledge of the patient assessed then for personality disorder(s) using the SCID-II [20].
Childhood neglect was assessed using the care scale of the Parental Bonding Instrument (PBI) [21]. Maternal and paternal care scores were averaged, and neglect was defined as being in the lower quartile for parental care. Childhood abuse was assessed during an independent interview conducted by a research nurse who systematically asked about a variety of experiences, including childhood physical and sexual abuse. After each screening question, details were collected about the nature of the abuse, the number of times abuse had occurred, the relationship to the perpetrator and the age at which abuse had occurred. For this paper, childhood sexual abuse was limited to abuse involving physical contact. More details on the assessment of neglect and abuse, and their associations with avoidant and borderline personality disorders are available in other papers [13, 22].
Genotyping
DNA was extracted from peripheral blood samples by the guanidium isothiocyanate method of Ciulla et al. (1988). As previously described [12], to genotype the 825C<T polymorphism we developed a novel, allele-specific, polymerase chain reaction (PCR) assay based on the amplification refractory mutation system [23].
Statistical analyses
For statistical analyses, the data were transferred from a relational database to the statistics package, SYSTAT. The primary approach to examine the associations between risk factors and self-mutilationwas by logistic regression. Initially, each potential risk factorwas examined in a univariate manner. Formultivariate logistic regression, all variables that were significantly associated with self-mutilation in univariate analyses were initially included. In a stepwise manner, any variables that did not remain significantly associated with self-mutilation were deleted.
Results
Of the 195 depressed patients who were entered into this treatment trial, 24% (46/195) reported a history of self-mutilation. Further breakdown of the self-mutilation and suicide attempt histories of these patients shows that 14% (27) had histories of both self-mutilation and suicide attempts, 10% (19) had a history of self-mutilation but no history of a suicide attempt and 21% (40) had a history of a suicide attempt but no self-mutilation. In males, 18% (15/84) had a history of self-mutilation, while 28% (31/111) of females had such a history.
In depressed patients aged 18–24 years, 42% (30/72) had a history of self-mutilation. For patients aged 25 years or older who had an onset of their first major depressive episode before the age of 25 years, 20% (12/60) had a history of self-mutilation. For those patients aged 25 years or older who had their first depressive episode at 25 years or later, only 6% (4/63) gave a history of self-mutilation.
Of the 46 who had self-mutilated, the most common method was wrist cutting (31), followed by head banging (16), self-hitting, biting or scratching (15), self-burning (7) or inserting sharp objects (3). The vast majority had self-mutilated on multiple occasions. Only 4 of the 46 ever reported self-mutilation when not depressed. Of the 46, four had self-mutilated during the week prior to presentation and 15 within the past month. For 20, the last episode of self-mutilation was more than a year prior to the current presentation.
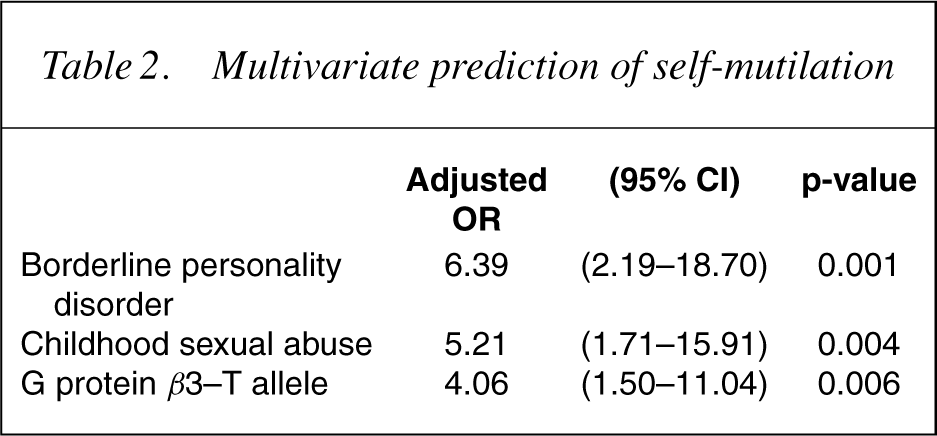
While we had a complete data set on the clinical variables and childhood abuse and neglect, personality disorder diagnoses were only available on 183 patients, and genotyping data were only available on 157 patients. The genotype frequencies were 50% (79) CC, 43% (69) TC and 7% (11) TT. From Table 1, which shows the univariate associations of putative genetic, developmental and personality risk factors for self-mutilation, it can be seen that the presence of the T allele of GNβ3, childhood sexual abuse, childhood physical abuse, any personality disorder and borderline personality disorder were all significantly associated with an increased risk. Table 2 shows that in a multivariate logistic regression, borderline personality disorder, childhood sexual abuse and the T allele of GNβ3 were all independently predictive of self-mutilation.
Univariate associations of genetic, developmental and personality risk factors for self-mutilation
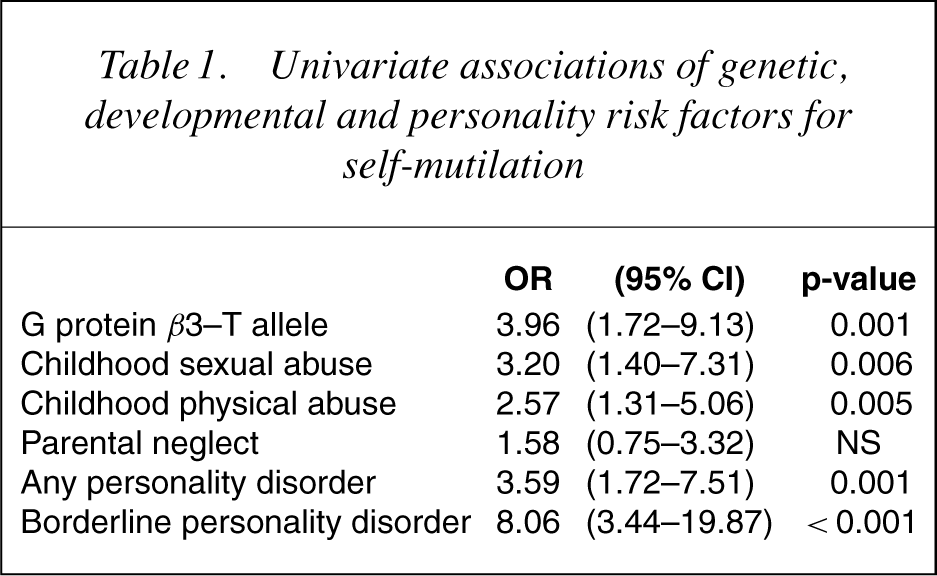
Multivariate prediction of self-mutilation
We examined the changes in anti-HSP autoantibody levels between patients in acute mania and patients in remission (Table 2). The total serum anti-HSP90 autoantibody levels were significantly increased in patients in acute mania compared with those in remission (26.35 ± 21.17 ng mL−1 vs 21.52 ± 16.74 ng mL−1, p = 0.002, paired ttest). As shown in Table 2, the differences were also noted in both medicated and unmedicated subgroups (p < 0.05, paired t-test).
Of the 157 patients in this study, 152 described themselves as of European descent. In Christchurch (New Zealand), this is largely from the United Kingdom. Three described themselves as Maori (Polynesian), all three had a T allele and one had borderline personality disorder. Two described themselves as Asian, one of who had a T allele. Repeating the statistical analyses excluding the five non-Europeans had minimal impact on the results.
In Table 3, which shows the univariate associations of the same risk factors in the young depressed sample, it can be seen that the odds ratios are higher for all risk factors in this young sample, compared with the sample as a whole.
Univariate associations of genetic, developmental and personality risk factors for self-mutilation in young (18–24 years) depressed patients
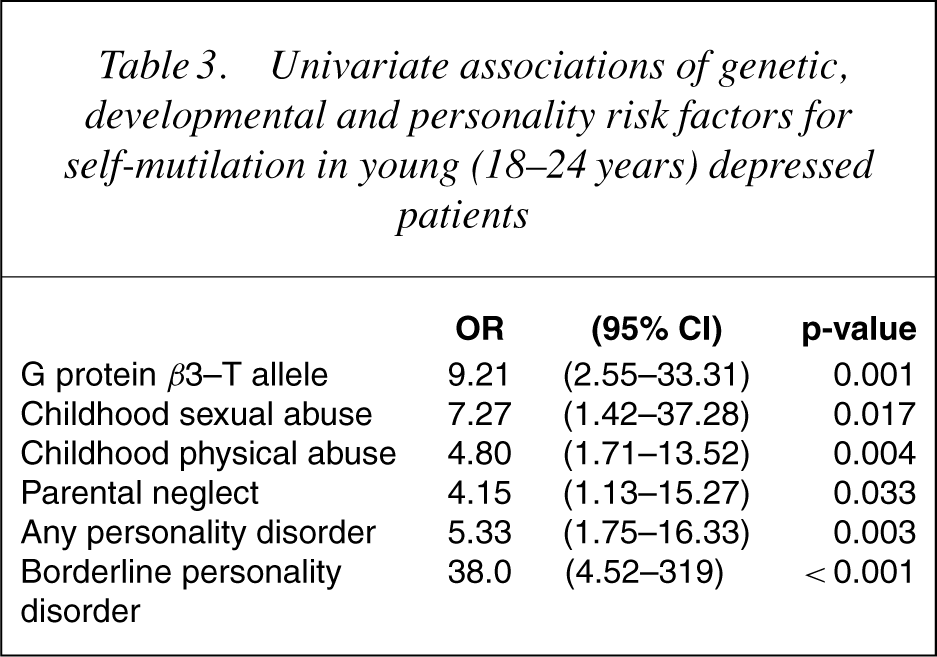
Table 4 presents the same data, but in an alternative form, and shows the probability of a history of self-mutilation on the basis of the presence or absence of child sexual abuse and the T allele of GNβ3. Whether one looks at all ages, or just the young depressed patients, it is clear that the probability of self-mutilation is very low in the absence of both childhood sexual abuse and the T allele, but very high when both risk factors are present. The presence of just one of the risk factors is associated with an intermediate risk. There were no significant interactions between genotype and the other risk factors for self-mutilation. The odds ratio between the T allele of GNβ3 and suicide attempts was not significant (OR=1.04).
Probability of self-mutilation by a history of sexual abuse and G protein β3 polymorphisms

Discussion
In this study, 24% of depressed patients reported a history of self-mutilation, most commonly cutting. The rate of self-mutilation is even higher in younger depressed patients, and much lower in older depressed patients with a late (25 years or older) age of their first depressive episode. For the vast majority, this occurred when they were depressed and happened on multiple occasions. Our findings that borderline personality disorder and childhood sexual abuse are risk factors for self-mutilationwere expected, based upon current understanding. However, we have shown that the T allele of GNβ3 is also a risk factor for self-mutilation, and that this remains a significant risk factor, evenwhen the impact of childhood sexual abuse and borderline personality disorder are accounted for. Furthermore, as hypothesized the association of these three risk factors are even stronger if we limit the sample to young (18–24 years) depressed patients. Indeed, the odds ratio between the T allele of GNβ3 is 3.96 in the sample as a whole, but increases to 9.21 in our young depressed patients.
G proteins consist of α, β and γ subunits and are involved in relaying signals via adenyl cyclase and phospholipase C, into intercellular post-synaptic responses caused by neurotransmitter binding to post-synaptic receptors. The T allele (C825T) of GNβ3 is located in exon 10, affects splicing and is associated with a deletion of 41 amino acids, altered structure and enhanced signal transduction to a variety of stimuli, including an adrenergic stimuli [24, 25]. Outside of psychiatry, this T allele has been associated with hypertension, obesity and increased antihypertensive response to thiazide diuretics [26–28].
The mechanism by which this T allele of the GNβ3 may increase the risk of self-mutilation is unclear, in part because we understand so little about the neurobiology of self-mutilatory behaviours. Borderline personality disorder patients who self-mutilate have a significantly reduced perception of pain, whichmay be state independent but which is further elevated during stress [29]. It has also been suggested that the ‘analgesia’ during self-injury in borderline patients is related to both neurosensory and attitudinal/ psychological abnormalities [30]. Furthermore, the pain threshold in such patients may be correlated to EEG theta activity [31]. Others' studies suggest that selfmutilation, as with other ‘impulsive’ behaviours, may be linked to decreased serotonergic function as assessed by neuroendocrine challenge tests [32, 33]. Finally, decreased superoxide dismutase activity, and the associated excessive generation of free radicals may also be part of the neurobiology of self-mutilation [34].
While we have again reported significant associations between borderline personality disorder and childhood sexual abuse with self-mutilation, it is worthy of note that only 28% of those who self-mutilated had experienced childhood sexual abuse. Similarly, only a minority of those who self-mutilated met criteria for borderline personality disorder. Indeed, Table 4 suggests that either childhood sexual abuse or the T allele of GNβ3 markedly increases the risk of self-mutilation, and that the presence of both risk factors increases the risk yet again. It is also of note that we found no significant associations between the polymorphisms of GNβ3 and either borderline personality disorder or childhood sexual abuse.
In this paper, we have reported for the first time a significant association between the T allele of GNβ3 and the risk of self-mutilation in depressed patients, and that this association is even stronger in young depressed patients. Furthermore, this genetic association remains a significant risk factor for self-mutilation when variables such as borderline personality disorder and childhood sexual abuse are taken into account. Like all genetic associations in psychiatry, this finding needs to be replicated. However, if the finding is replicated, it may potentially provide clues to understanding the neurobiology of selfmutilation.
Footnotes
Acknowledgements
This research was funded by the Health Research Council of New Zealand. We thank all patients and clinicians who contributed to this study, and give special thanks to Robyn Abbott for patient care and Isobel Stevens for study coordination and data management.
