Abstract
Depression is common in individuals with a variety of medical illnesses, with estimated prevalence rates varying from 20% to 50% [1], [2]. Studies in people living with HIV/AIDS have shown that up to 50% of outpatients have significant depressive symptoms [3–7]. The cause of the high prevalence of depression among people living with HIV/AIDS, despite significant advances in the medical treatment of HIV/AIDS, remains uncertain.
It has been suggested that people livingwith HIV/AIDS who suffer from depression may comprise two groups: thosewith primary (idiopathic) depression and thosewith a secondary or ‘organic’ mood disorder [8]. The primary group will include individuals who experience depression for reasons that are similar to medically well (and non-HIV/AIDS) populations. These include factors such as a family history of affective disorders, personality style and experiences of adverse life events. By contrast, secondary depression implies that the mood disorder is in some way provoked by HIV/AIDS. This is consistent with the concept of secondary depression initially proposed by Robins and Guze [9] and extended by Klerman and Barrett [10] to describe affective states secondary to, or associated with, concomitant systemic medical diseases or drug reactions. Treisman et al. [8] suggest that in some people living with HIV/AIDS, depression may be the result of the neurotropic effects of the virus on the subcortical brain areas. This would represent a manifestation of HIV central nervous system (CNS) involvement as part of the progression toward HIV-associated dementia, due to the same neural areas being affected in HIV infection and depression. Work by this group [11] showing an increase in depressive symptoms in the months before the development of AIDS supports their hypothesis. The possibility of a brain reservoir of resistant HIV virus due to poor blood–brain penetrance of antiretroviral drugs is of important practical concern with regard to the choice of highly active retroviral therapy with higher blood–brain penetrance.
In this study, we aimed to assess whether distinct subtypes of depression can be related to the presence/absence of various psychological, social or cognitive factors. Psychomotor dysfunction and cognitive impairment in people living with HIV/AIDS have been found to be indicative of pathological brain processes due to the neurotropic effects of the virus. We hypothesized that: (i) depression with cognitive impairment occurring in the context of advanced HIV disease and increasing immunosuppression is indicative of a distinct ‘organic’ type depression (secondary depression); and (ii) in a ‘non-organic’ depression type, personality, past history and family history of depression, psychosocial situation and illicit substance use would be important aetiological factors.
Method
Subjects for this study were recruited from patients attending two general hospital infectious diseases outpatients' clinics, a sexual health centre clinic and a general practice clinic with a high HIV/AIDS patient caseload. Approval for the study was obtained from the Human Research and Ethics committees for the Royal Melbourne Hospital, the Alfred Hospital and the Department of Human Services. Patients were eligible for the study if they met the following criteria – aged 18 years and older, HIV seropositive and able to read and write in English. Patients meeting the criteria were informed by their treating physicians about the study and upon agreeing to participate, met with the research assistant on site who gave a full description of the study and obtained written informed consent. Patients' sociodemographic data were obtained by the research assistant and patients were then given a set of self-rated scales to complete at home and mail back. Appointments were also made for them to return, usually within 2 weeks, to complete the structured clinical interview and a battery of neuropsychological tests. The participant's treating physician also completed forms providing information on the patient's medical (e.g. stage of illness, viral load, t-cell count and duration of illness) and psychiatric history (e.g. any current or past psychiatric illness and any family history of psychiatric illness). Adherence to antiretroviral medication was measured through completion of a self-report compliance questionnaire in which patients were asked to report how many days in the last 4 days they had missed taking their antiretroviral medication.
When patients returned to complete the structured clinical interview and neuropsychological tests, they also completed the cannabis use screen (CAUSE) which was developed by the Dandenong Psychiatric Research Centre (T. Rolfe, Personal communication) to measure quantity, frequency, method and type of use of cannabis.
To assess severity of depressive symptoms, we used the self-report Beck Depression Inventory (BDI) [12]. The BDI has been used extensively in depression studies and has been shown to have high validity and reliability in measuring depressive symptoms. Patients also completed the Medical Outcomes Study Short-Form Questionnaire (MOS SF-36) [13], which is a widely used instrument to measure functional wellbeing. It has been reported that severity of depression is associated with poorer functioning, regardless of type of depression [14]. The revised NEO Personality Inventory (NEO-PI-R) [15] was used to measure personality traits including neuroticism, conscientiousness, extraversion, agreeableness and openness to experience. The structured clinical interview for DSM-IV axis I disorders (SCID; [16]) was administered to determine the presence of a depressive disorder. Patients who met the criteria for a current ‘major depressive disorder’ on the SCID were then administered a mental state rating scale the CORE [17] to subtype depression as melancholic or non-melancholic. The CORE is a behavioural sign-based approach (as opposed to a symptom-based diagnostic system) validated to reliably identify melancholic depression, a subtype of the depressive conditions, by measurement of features of psychomotor disturbance. The psychomotor features of melancholia have been suggested to result from dysfunction in frontal subcortical neural networks [18].
Neuropsychological assessment was performed using three measures: the Hopkins HIV Dementia Scale [19], the Grooved Pegboard (GPB; [20]) and the Cambridge Automated Neuropsychological Test Battery (CANTAB; [21]). Premorbid intelligence quotient (IQ) was assessed using the National Adult Reading Test (NART; [22]).
The Hopkins HIV Dementia Scale was developed to screen for indications of HIV dementia by assessing memory-registration and recall, attention, psychomotor speed and construction. Patients are scored out of 16, with a score of 10 or less suggestive of dementia. The GPB test is a measure of psychomotor speed. Subjects are timed on the time it takes to insert 25 pegs into grooves in a board using first the dominant then the non-dominant hand.
The CANTAB is a computerized neuropsychological test battery shown to be sensitive to cognitive changes in a wide range of brain disorders including HIV-related cognitive impairment [23–25]. Four tests chosen from this battery for this study were: spatial working memory (SWM), stockings of Cambridge (SOC), attentional set shifting (IED) and reaction time (RTI). These tasks tap into the brain's executive functioning capability, the key area of function, which has been shown in previous research [25] to be affected in HIV-positive individuals.
The NART is a literacy test validated to measure premorbid IQ. Subjects are asked to read 50 words. This test was used to assist in delineating cognitive impairment from pre-existing intellectual disability.
Statistical analyses
The total available data (n=174) was used in each analysis, rather than a maximum subset (n=129), which has valid data on all the measures. Before analysis all variables were checked for normality, using normal plots, as well as skewness and kurtosis measures and transformed if necessary. For the descriptive data, median values have been reported wherever there was the potential problem of extreme values skewing the mean values. A preliminary analysis aimed to identify predictors or correlates of depressive symptoms. A selection of ‘typical’ correlates as well as some HIV-specific indicators was included in this analysis. The dependent variable was (square-root-transformed) BDI scores.
Results
Sample-completers versus non-completers
Study completers and non-completers sociodemographic and medical characteristics
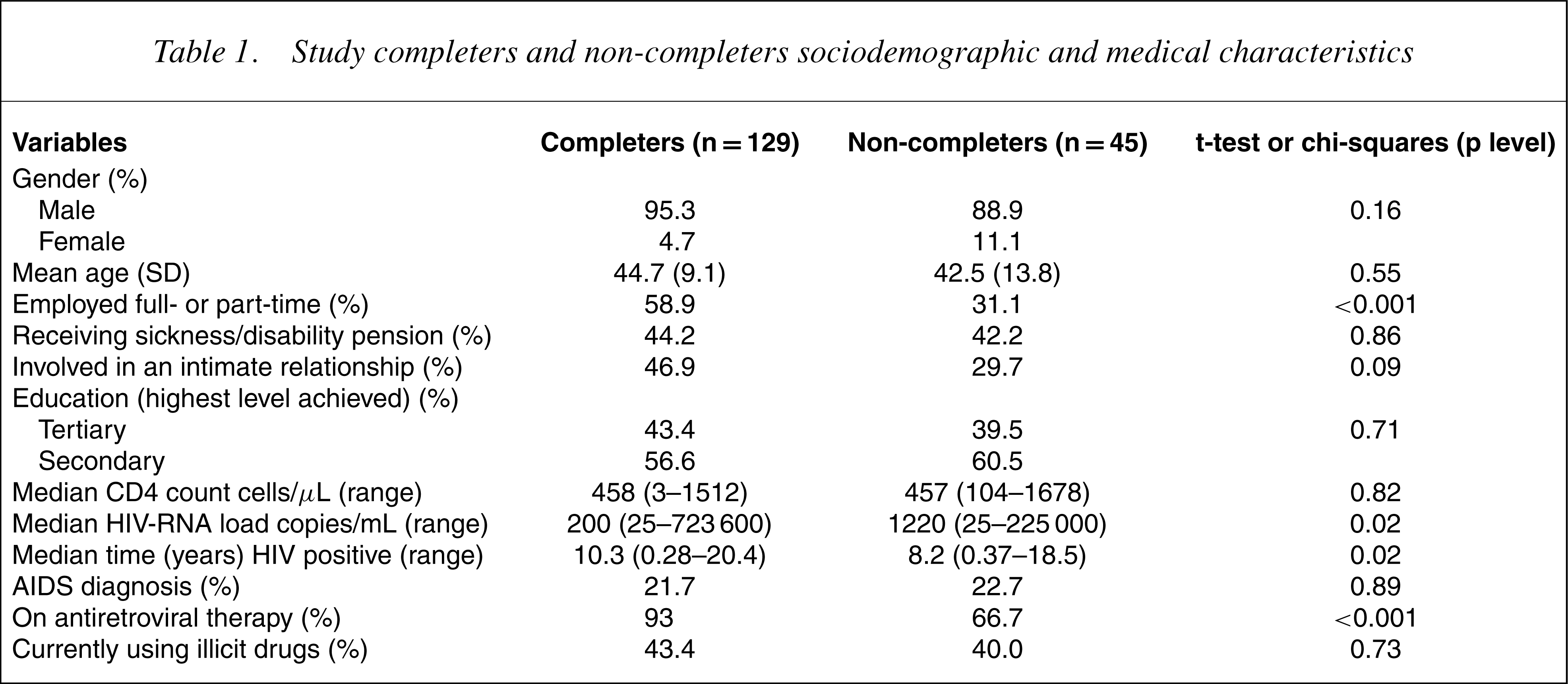
As shown in Table 1, the completers were significantly more likely to be on antiretroviral therapy, had lower HIV-RNA loads and had been diagnosed with HIV for a significantly longer period of time. They were also more likely to be employed compared to the non-completers.
Prevalence of depression measured on Beck Depression Inventory and structured clinical interview for DSM-IV
Approximately, one-third (34.8%) of participants who completed the BDI scored above the cut-off commonly used for the identification of depression [1] (i.e. ≥14). The mean (SD) BDI score of the whole group was 12.3 (8.2).
Thirty-five (27%) of those who completed the SCID met criteria for a current mood disorder. A variety of specific mood diagnoses were made including major depressive disorders (8.5%), dysthymic disorder (9.3%), mood disorder due to a general medical condition (4.6%), substance induced mood disorder (0.8%), bipolar affective disorder (1.5%) and depressive disorders not otherwise specified (2.3%). Of note, none of the participants who met criteria for a current major depressive disorder at the SCID interview were found to have depression with melancholic features according to the CORE assessment.
Five of the 35 individuals with a current mood disorder had also experienced a mood disorder in the past. The SCID interview identified another 33 (26%) individuals who had experienced a mood disorder in the past but who were not currently depressed. Throughout the remainder of the paper mood disorders are reported as a single category (MD) and data analyses are based on this.
The BDI scores and SCID diagnoses had a significant relationship with each other (F(1,134) =7.994, p=0.005). Participants who met criteria on the SCID for a current mood disorder had a higher BDI (mean=12.3) than those who did not meet criteria (mean=10.8), although agreement between these measures for categorical diagnosis was not exceptionally high. In the rest of the paper the exploration of the nature of depression will be discussed using BDI scores.
Personality traits
The NEO-PI was used to assess personality in five domains: neuroticism, extraversion, openness, agreeableness and conscientiousness. The mean scores (SD) scores for each of the domains were: neuroticism= 93 (24), extraversion=106 (22), openness=119 (20), agreeableness= 122 (17) and conscientiousness=114 (21). Compared to norms derived from a gender-matched group [15] participants in this study scored highly in neuroticism, in the average range for extraversion, openness and agreeableness and in the lowrange for conscientiousness.
Illicit substance use
At baseline, 75 (58%) participants reported current use of illicit substances. The majority of these (n=59) were using cannabis, of whom 22 were using it on a daily basis. Further assessment of cannabis use using the CAUSE indicated that the majority (n=30) had been using cannabis for a period greater than 5 years. Most (n=20) reported using less than 1 gram per week.
Medication adherance
Assessment ofmedication adherence revealed themajority of participants (78%) did not miss any days ofmedication in the last 4 days. Only two participants reported missing 3 out of the last 4 days of medication.
Cognitive impairment
On the Hopkins Dementia Scale, a score of 10 and less has generally been considered suggestive of HIV dementia but must be confirmed by further neuropsychiatric assessments. In our study sample, while only nine participants (7%) scored 10 or less to meet the criteria for a diagnosis of HIV dementia on the Hopkins Dementia Scale, other forms of neuropsychological testing revealed a higher prevalence of cognitive deficits.
Psychomotor slowingwas evident on the GPB and impaired reactiontime test in the CANTAB battery. On the GPB test, the mean time (in seconds) for the group to complete the test using the dominant hand was 80.4 (SD=19.2) and for the non-dominant hand was 85.5 (SD=17.4). Overall, this group took longer to complete the GPB task compared to a set of derived norms from 360 healthy volunteers [20] in which males at all age levels took a mean time (in seconds) of 69.9 (SD=12.5) to complete the task using the dominant hand and 75.4 (SD=13.6) using the non-dominant hand. This effect remained even when using median times for this sample group (dominant hand: 76 seconds; non-dominant hand: 83 seconds).
On the CANTAB, each individual's results were standardized against a set of norms matched for age and sex but not HIV serostatus. This allowed for a z-score to be generated. A single sample t-test was then performed to compare the z-scores of the current study sample with the normative data (Table 2).
Comparison of study participants CANTAB test results with normative data (single sample t-test)
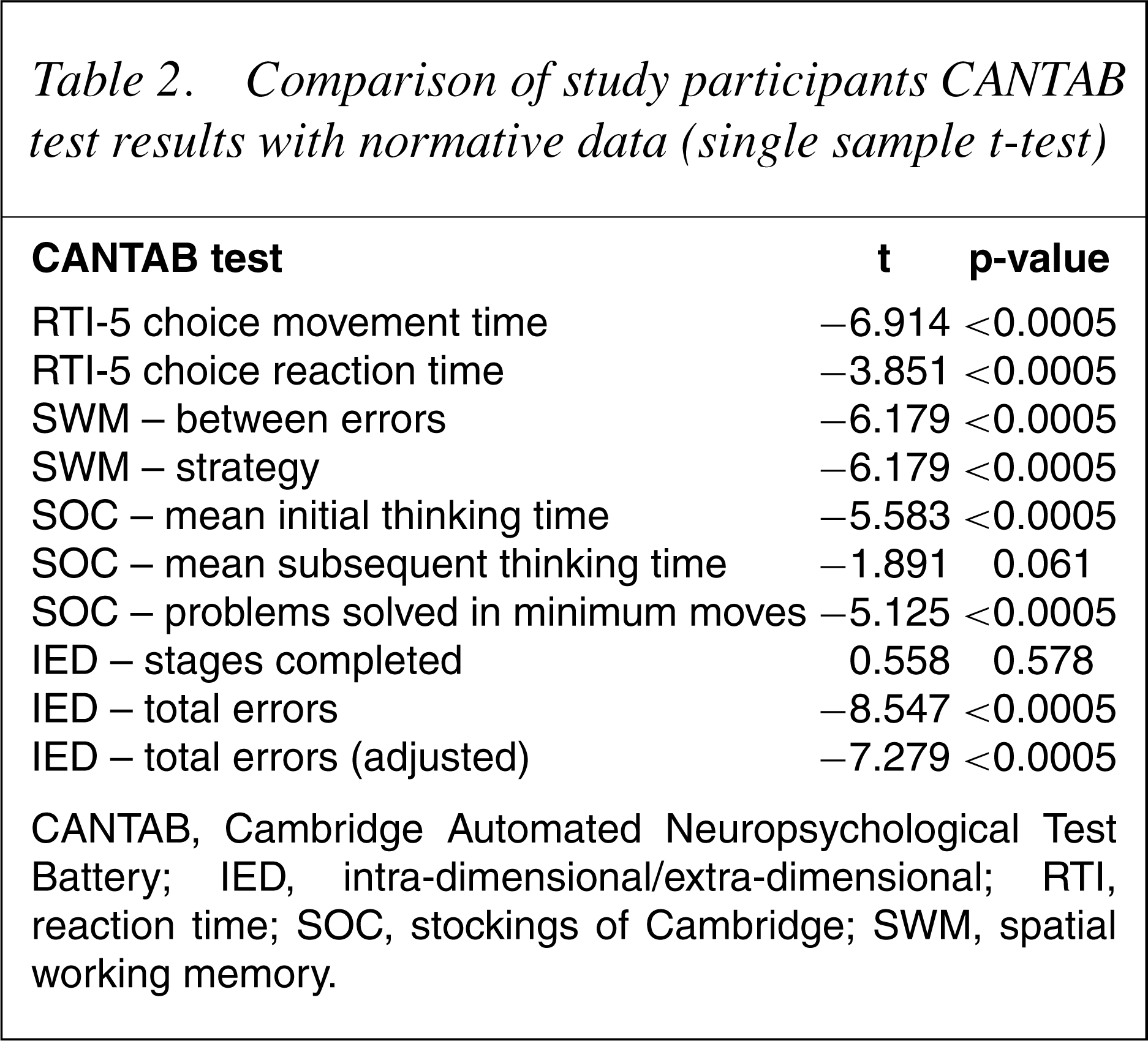
CANTAB, Cambridge Automated Neuropsychological Test Battery; IED, intra-dimensional/extra-dimensional; RTI, reaction time; SOC, stockings of Cambridge; SWM, spatial working memory.
Predictors of depressive symptoms
The predictors of depressive symptomatology included the NEO-PIR personality factors of neuroticism, extraversion and conscientiousness. Neuroticism had a strong association with depressive symptomatology (r=0.69, p<0.01). It should be noted that this relationship is in addition to the above-average neuroticism score of the patient group. Conscientiousness (r=−0.41, p<0.01) and extraversion (r=−0.36, p<0.01) both had a negative but weaker relationship with depressive symptomatology. The anovas showed that depression was influenced by past contact with mental health services (F(2,110) =5.44, p<0.01), current relationship status (F(1,132) =4.21, p<0.05) and living arrangements (alone or with partner, F(1,134) =4.95, p<0.05). Patients with past contact with mental health services, not currently in a relationship or living alone, had significantly higher levels of depression than other patients. However, it was not possible to determine whether contact with mental health services pre- or post-dated the onset of their current episode of depression. Those in full-time paid-employment had somewhat lower BDI scores, but this difference (slightly less than three points) was not significant (F(1,134) =5.68, p=0.069).
There were trends for the influence of a family history of psychiatric problems and current illicit substance use on depressive symptomatology, but these did not reach significance. It is of note that levels of illicit drug use in this group are increased compared to use in the general community, as reported in the Australian National Drug Strategy Household Survey [26]. As seen in Table 1, almost half the study cohort reported current use of illicit drugs.
To explore whether apparent depressive symptoms might be a sideeffect of medical problems (i.e. somatic symptoms of advanced disease), correlations between BDI scores and the HIV-specific measures of viral load, CD4 cell count and number of years HIV seropositive were examined. None of these correlations reached significance. There was a trend for higher depression scores to be associated with more missed days of antiretroviral medication, but a univariate anova showed that this difference was not statistically significant (F(4,120) =0.695, p=0.597). Because of smaller numbers in some of the cells, the groups were collapsed into two groups: missed medication versus no missed medication. The latter had a higher mean BDI score (14.12, SD=8.05) compared to the former (11.16, SD=7.79), however, the difference was not significant (F(1,116) =2.105, p=0.150).
In sum, there were no significant effects of the medical variables, age and IQ on depressive symptoms. There were too few females in the study sample to enable accurate assessment of effect of sex and the observed mean difference was less than one BDI point.
Effects of sex, age and IQ on BDIwere also explored. The largest correlation was between IQ and BDI, which only reached r=−0.01 (NS). The relationship between depressive symptoms and a broad range of cognitive functions measured by the CANTAB, Hopkins HIV Dementia Scale and the GPB were examined. The correlation between the BDI and the Hopkins HIV Dementia Scale was non-significant, reaching only (r=−0.06). Most CANTAB subscales had negative correlations with the BDI; however, only two subscales (SWM between errors and SWM strategy) showed significant correlations with the BDI. Calculating correlations between the cognitive items of the BDI and the CANTAB measures showed roughly the same pattern of results as the full scale BDI except SOC (mean initial thinking time) which also reached the 0.05 significance level. Results from the GPB test were used to classify participants to cases (if their performance using either the dominant or non-dominant hand fell into the 5th percentile described in Ruff and Parker's study [20]), or non-cases. The GPB ‘cases’ for either hand were slightly more depressed than non-cases; however, these differences were not significant for the dominant (F(1,126) =0.001, p=0.993) or the non-dominant hand (F(1,126) =0.178, p=0.674).
Impact of depression on functioning
The relationship between depression and physical and social functioning was measured in eight domains on the SF-36 questionnaire. They were: physical functioning, role-physical, bodily pain, general health, vitality, social functioning, role-emotional and mental health. Pearson's correlations were computed to examine the relationship between BDI scores and functioning. All the eight SF-36 subscales (z-scores) had significant negative correlations (p<0.001) with the BDI scores, suggesting that increased depressive symptoms were related to lowered physical and social functioning. The largest correlation with the BDI scores was for mental health (−0.727). The lowest was for physical functioning (−0.261).
McHorney et al. [27] suggested that the SF-36 has a higher-order two-factor structure, which broadly relates to two aggregate summary scores; the physical component summary (PCS) and the mental component summary (MCS). These two scores were computed as described by Ware and colleagues [13]. There was a moderate negative correlation between BDI scores and PCS (r=−0.291, p<0.001) and a strong negative correlation with MCS (r=−0.657, p<0.0005). Again, depressive symptoms are broadly related to decreased mental and social functioning, with a weaker, but still significant, relationship with reduced physical functioning.
Discussion
There was a high rate of depression in this group of HIV-seropositive individuals with one-third of the group scoring above the cut-off score on the BDI, indicative of depressive illness and 27% meeting DSM-1V criteria for a depressive disorder. Contrary to our expectations, we did not identify two distinct subtypes of depression in this group of people living with HIV/AIDS. As with medically well cohorts, we found depression was associated with higher levels of neuroticism, a past history of psychiatric disorder and aspects of the current psychosocial situation. Family history and illicit drug use were also linked with the presence of depression. Specifically, we found no significant evidence for a distinct subtype of ‘organic’ depression when we tested for correlations between BDI scores and cognitive measures.
The group as a whole showed cognitive deficits of psychomotor slowing and executive dysfunction as evident on the GPB and CANTAB tests. However, only a minority met the criteria for HIV dementia on the Hopkins Dementia Scale. Although it could be argued that these deficits in cognitive functioning were due to depression, Sahakian et al. [25] still found these same deficits in their asymptomatic HIV-seropositive group, even when they specifically excluded those subjects with a significant level of depression. Spatial working memory was the only area where there was significant correlation between the BDI scores and cognitive scores in the current study. Other groups have also shown a decline in working memory associated with HIV infection [28], [29]. Working memory is considered to be subserved by the lateral prefrontal cortex – the same area that has been shown to be involved in the pathogenesis of major depression. One could postulate that the lateral prefrontal cortex is also affected in HIV infection, resulting in the symptoms of depression and working memory deficits.
Consistent with a large body of evidence showing high levels of functional disability due to depression, participants who reported higher levels of depressive symptoms showed impairment in domains of functioning related to social, physical, general health and overall wellbeing. There was no obvious link between depressive symptoms (BDI) and adherence to treatment regimens (missed HIV medications), although there were weak trends in the data. Some studies have reported that depression is one of the barriers to adherence to treatment [30], [31], but the relationship is complex and must also consider factors extrinsic to the individual patient [32].
Overall, our findings suggest that, at least in ‘well’ cohorts, depression in individuals with HIV is not a distinct subtype. Rather, it appears to occur in individuals with a range of well-recognized risk factors and to be associated with disability, which is determined by severity of depressive symptoms. Social supports appear to be protective factors against depressive symptomatology in this study group, highlighting the importance of encouraging community support programs/groups for people with HIV/AIDS and facilitating active engagement of the HIV positive individuals with significant others.
The results of this study must be considered within certain methodological limitations. First, our sample was relatively ‘well’ from a medical point of view: normal CD4 count; low viral load; over 50% used; and only onefifth with a diagnosis of AIDS. This is in marked contrast to the groups of people living with HIV/AIDS previously studied [1–5],[7] and in whom the presence of an ‘organic’ dementia has been suggested. Second, the majority of the patients who participated in this study were treated with combination antiretroviral therapy, including drugs with good CNS penetration and thus the potential to prevent or delay any neuropathic effects of the virus. This is in contrast to earlier studies [1–5],[7] undertaken before the availability of highly active retroviral therapies. Taken together, these factors may have acted to obscure any differences which may exist. Third, we cannot exclude the possibility that selection of participants may have biased our sample. Although not documented, therewas anecdotal evidence that doctors were protective of their patients and less likely to ask patients whom they considered ‘too unwell’ or ‘too depressed’ to participate in the study. Examination of our hypothesis that there are two subtypes of depression in people living with HIV/AIDS may require recruitment of samples with more advanced disease and/or longitudinal follow-up of current medically well cohorts.
Conclusions
In this study of people livingwith HIV/AIDS, over onethird of the subjects scored above the BDI cut-off score for depression but none met the criteria for melancholic depression using the CORE. Up to one-third showed impaired performance on specific cognitive tests of executive function and psychomotor speed, but only a small number of subjectsmet criteria for HIV-related dementia. There was no evidence for a distinct subtype of ‘organic’ depression. Rather, depression was associated with personality style, past psychiatric history and psychosocial circumstances. As has been shown with other populations, there was a high degree of functional impairment associated with higher levels of depressive symptoms. This may exacerbate the level of disability due to underlying medical illness and underscores the need for identification and effective treatment. Early intervention, prevention and treatment approaches in people living with HIV/AIDS should be modelled on those used in other depressive samples.
Footnotes
Acknowledgements
This project was funded by the Mental Health Branch, Department of Human Services, Victoria, Tender Brief No. T1176. The authors are grateful for the support of the physicians and administrative staff of the Melbourne Sexual Health Centre, the Royal Melbourne Hospital Victorian Infectious Diseases Unit, the Alfred Hospital Infectious Diseases Unit and the Prahran Market Clinic.
