Abstract
Background
Bipolar disorder as a recurrent affective illness produces significant distress and dysfunction, ranking among the top 30 causes of worldwide disability [1]. The unique clinical challenges posed by this chronic illness include various acute phases of mood instability: depressive, hypomanic, manic and mixed episodes. Varying patterns of mood disturbance as well as periods of increasingly unstable or fluctuating mood, such as seen in rapid-cycling patients, provide a wide spectrum of affective pathology that requires intense and careful psychopharmacologic treatment in order to seek therapeutic balance.
The treatment of acute episodes of bipolar disorder, particularly when mood is above baseline (manic/ hypomanic), is relatively straightforward and often successful using antipsychotics, anticonvulsants, lithium, benzodiazepines or electroconvulsive therapy (ECT). Similarly, although there are greater unmet needs in the treatment of bipolar depression than for bipolar mania, acute depressive episodes have known and effective treatments, including antidepressant medications, ECT, and more recent evidence supports the anticonvulsant lamotrigine.
The majority of research studies on the treatment of bipolar disorder have focused on the management of these acute episodes. The efficacy of treatments for acute episodes has been established, replicated and accepted widely through the application of various research methods. Similar rigour has not been applied to date to the investigation of effective preventative strategies for bipolar disorder.
Prevention of acute bipolar episodes through maintenance phase of treatment is critical in order to improve the poor long-term outcomes that exist for bipolar patients. Mood stabilizers have emerged as the cornerstone of prophylactic treatment in bipolar disorder, with lithium being the first known effective agent, supported by first generation maintenance studies conducted and published during the 1960s and 1970s.
The need for alternatives to lithium for maintenance treatment of bipolar disorder is clear. An estimated 20–40% of patients do not respond sufficiently to lithium [2], [3]. Undesirable side-effects, adverse reactions, a narrow therapeutic index and the increased risk of toxicity with overdose can make lithium therapy less than ideal formany patient. Efforts to investigate the potential utility of anti-epileptic drugs have been ongoing because initial reports showed favourable results for valproate in 1966 by Lambert et al. [4] and for carbamazepine in 1978 by Ballenger and Post [5]. However, these agents also cannot be considered ‘ideal mood stabilizers’ because their use is too often complicated by side-effects, toxicity and refractoriness. There is also a relative paucity of controlled, double-blinded data to fully support the use of anticonvulsants for bipolar maintenance therapy. Only recently has an atypical antipsychotic, olanzapine, showed relapse prevention efficacy in bipolar disorder.
The aim of this paper is to review maintenance therapies in bipolar disorder by examining available data, particularly from double-blinded, placebo-controlled studies. This is done while acknowledging that there is little consensus on the preferred or required method of bipolar maintenance studies and that study designs have evolved greatly since the 1970s, especially in areas of enrolment procedures, randomization schemes, specific outcome measures used, statistical analyses and regulatory changes [6]. Also, there is now an increased understanding of various types of bipolar disorder and unpredictable states such as rapid-cycling, dysphoric mania and iatrogenic cycle acceleration that did not exist when early maintenance studies were designed and carried out.
Lithium
Early studies consistently reported lithium to be more effective than placebo for bipolar prophylaxis. Examination of eight early controlled bipolar I prophylaxis studies that allowed for cumulative analyses of relapse rates supports the superiority of lithium over placebo for maintenance treatment [7–14] (Table 1). Thirty-one per cent of lithium-treated patients (n=226) experienced a relapse during a 5- to 24-month period of maintenance compared to 76% of placebo-treated patients (n=225) (Table 1). Placebo-treated patients also had an almost threefold increased rate of manic relapses compared to lithium maintained patients (59% placebo vs 22% lithium). The combined data from these early controlled studies show a twofold reduction in depressive relapses in lithiumtreated patients compared to those on placebo (16% lithium vs 33% placebo).
Early controlled lithium studies for bipolar disorder prophylaxis
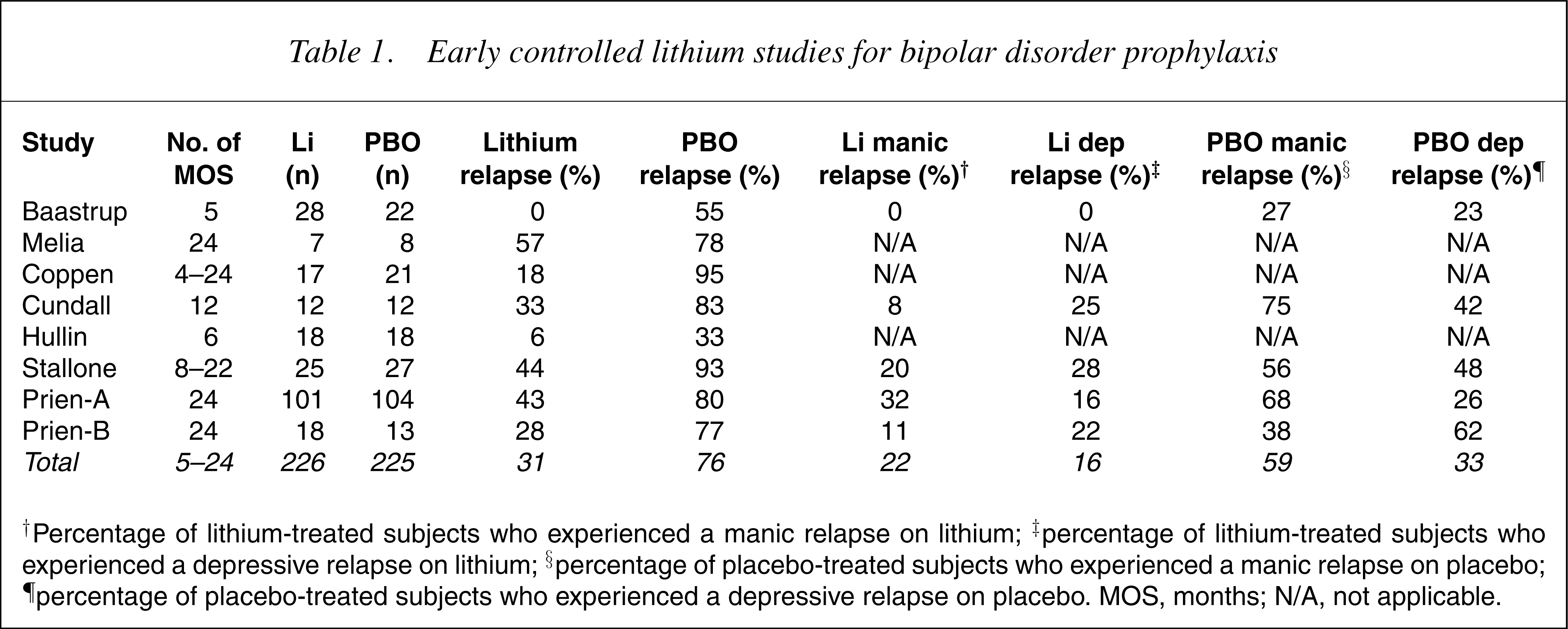
†Percentage of lithium-treated subjects who experienced a manic relapse on lithium; ‡percentage of lithium-treated subjects who experienced a depressive relapse on lithium; §percentage of placebo-treated subjects who experienced a manic relapse on placebo; ¶percentage of placebo-treated subjects who experienced a depressive relapse on placebo. MOS, months; N/A, not applicable.
A larger and more heterogeneous analysis of 28 studies of various design by Baldessarini et al. [15] yielded similar positive results for lithium prophylaxis. Yielding 2985 different subjects, their analyses posited an overall crude ratio of 3.2-fold lower recurrence risk with lithium. Additional benefit was reported for patients on lithium prophylaxis in terms of reduced hospitalization by 82%, reduction of manic or hypomanic recurrences by 64% and of depressive episodes by 46%. Lithium-treated patients experienced fewer days of illness per year and a 25% reduction in the average duration of any recurrent episodes while on lithium prophylaxis. When data was extracted to compare bipolar I (n=218) versus bipolar II (n=142) patients on lithium prophylaxis, long-term lithium treatment was found to reduce morbidity in both of these bipolar subtypes. Frequency of episodes decreased from 1.54 to 0.82 episodes per year (47% reduction) in bipolar I patients and even more in bipolar II patients, from 2.28 to 0.79 episodes per year (65% reduction). Depression was also prevented by lithium, more successfully for bipolar II patients who experienced 54% fewer episodes of depression per year than the 35% reduction seen in bipolar I.
However, many of these early studies were published before 1975 and used discontinuation study designs that involved patients with a known positive response to lithium being abruptly discontinued from lithium if randomly assigned to the non-treatment or placebo group of the maintenance phase of the study. This design may inflate the preventative effect of lithium because of the known effect of rapid lithium discontinuation, which increases early relapse into depression or mania [16]. Additionally, early lithium studies may have lacked specification of diagnostic criteria, not differentiated unipolar from bipolar depressed patients and reported response rates as a percentage of only those patients completing the study while failing to report reasons for premature discontinuations. Modern method would better substantiate or refuse the conclusions from these earlier studies.
Despite these limitations, lithium remains the best studied bipolar maintenance therapy. A 2004 meta analyses of nine placebo-controlled studies (n=770) by Burgess et al. concluded that lithium is effective in the prophylaxis of bipolar disorder relapse [17]. This report supported results from an earlier analysis of 19 blinded, randomized, controlled trials of prophylaxis in 865 patients by Davis et al. in 1999 that found 74% recurrence rate on placebo compared to just 29% on lithium [18]. Controlling for the possible effect of lithium withdrawal relapse inflating efficacy as discussed earlier, Davis reported that lithium significantly reduces relapse by 50% in bipolar patients, which further supported the conclusion that maintenance with lithium is effective. Dosing guidelines for lithium maintenance therapy have often suggested serum levels between 0.8 and 1.2 mmol/L. Gelenberg et al. studied 94 bipolar patients in a randomized, double-blind, prospective trial using standard dosing to achieve blood levels of lithium between 0.8 and 1.0 mmol/L versus ‘low’ dosing to levels between 0.4 and 0.6 mmol/L [19]. They reported that there was a 216 times greater risk of relapse using the ‘low’-dose strategy for prophylaxis compared to the standard dosing. Lithium side-effects were more frequent when standard dosing was used. However, these results may have been affected by a study design that called for an abrupt reduction of lithium by 50% for those patients randomized into the ‘low’-dosing group who had lithium on board in the standard range. These patients accounted for nearly all of the relapses in the ‘low’-dose group. The effect of lithium discontinuation, particularly if rapidly or abruptly stopped, needs to be considered in any bipolar maintenance study design or interpretation.
Recent naturalistic studies have suggested that a significant proportion of bipolar patients have inadequate responses to lithium and have also reported high dropout rates. Harrow et al. reported that 40% of lithium-treated patients experienced a manic episode during a mean 1.7-year maintenance treatment period, a result equivalent to those patients on no medication [20]. Lithium treatment has also been reported to lower the risk of relapse for only the first 32 months of prophylaxis therapy [21]. Other large, open, prospective naturalistic studies report good outcomes in patients with bipolar disorder on lithium maintenance in approximately one-third of patients [22], [23]. Again, additional controlled studies using modern methods have been needed for some time to better understand the efficacy of lithium maintenance.
The Bowden et al. 2000 study [24] provided randomized, blinded and placebo-controlled data that supported both divalproex and lithium as effective maintenance therapies in bipolar I patients. This was the first maintenance study to use modern methods, including enrolment of patients during an index manic phase and tapering lithium over 2 weeks instead of rapid discontinuation. Although the primary outcome measures in this study did not distinguish between the two ‘active’ compounds and placebo and neither active treatment separated from placebo in terms of the primary outcome measure, there were secondary measures of note. Lithium extended the time until relapse by 55% compared to placebo. However, divalproex was more successful than lithium in extending the duration of prophylaxis as well as preventing deterioration in depressive symptoms and Global Assessment Scale (GAS) scores.
Bowden and Calabrese have also examined the efficacy of lithium as maintenance therapy in bipolar I patients over an 18-month period using modern methods while comparing lamotrigine to lithium. A recently completed 18-month maintenance study using a placebo-controlled, double-blind method compared lamotrigine and lithium in recently manic or hypomanic patients with bipolar I disorder [25]. This multicentre study enrolled 349 patients who were manic/hypomanic or manic/hypomanic within 60 days of the screening visit into the openlabel phase lasting 8–16 weeks during which lamotrigine was prescribed as an adjunctive or monotherapy (100–200 mg/day) and other psychotropics were discontinued gradually after stabilization and before entry into the double-blind phase. One hundred and seventy-five of these 349 patients were randomized to maintenance treatment with lamotrigine 10–400 mg/day (n=59); lithium titrated to serum levels of 0.8–1.1mEq/L (n=46), or placebo (n=70). On the primary outcome measure of time until additional pharmacotherapy was required for treatment of a mood episode, both lithium and lamotrigine were superior to placebo (p=0.003 and p < 0.02, respectively). The median time until one-quarter of the patients experienced an emergent mood episode was 72 weeks for the lamotrigine-treated group, 58 weeks for lithium-treated subjects and 35 weeks for those receiving placebo. Lamotrigine, not lithium, was superior to placebo in terms of time until discontinuation for any reason (p=0.03, lamotrigine vs placebo; p=0.7, lithium vs placebo). Lithium, but not lamotrigine, was superior to placebo at prolonging the time to a manic, hypomanic or mixed episode (p=0.006, lithium vs placebo; p=0.280, lamotrigine vs placebo). However, lamotrigine showed significantly greater efficacy for prolonging time to a depressive episode compared with lithium (p=0.015, lamotrigine vs placebo; p=0.167, lithium vs placebo). The results of this controlled maintenance study support the use of lithium prophylaxis for mood episodes above baseline for bipolar I patients, while indicating a more favourable preventive efficacy of lamotrigine for recurrent depressive episodes in these patients.
Calabrese et al. have recently completed the first placebo-controlled, double-blind 18-month maintenance study of lamotrigine and lithium in recently depressed bipolar I patients [26]. Their multicentre study enrolled 966 patients into the open-label phase using the same protocol as the aforementioned Bowen study to obtain 463 bipolar I patients with most recent depressive episode in remission to randomize one of five maintenance treatment groups: lamotrigine (50, 200 or 400 mg/day), lithium (titrated to serum levels of 0.8–1.1mEq/L) or placebo. Reproducing the results of the Bowden 2002 study, both lamotrigine and lithium were significantly superior to placebo at delaying time to intervention for any mood episode (p=0.029, lamotrigine 200 and 400 mg/day vs placebo; p=0.029, lithium vs placebo). In the present study, however, both lamotrigine and lithium were superior to placebo in the analysis of overall survival in the study. The emergence of total depressive episodes across all groups outnumbered emerging mania or hypomania by an almost 3:1 margin. Confirming the findings of differential prophylactic efficacy observed in the Bowden 2002 study, Calabrese et al. also detected lithium's superiority for delaying time to a manic or hypomanic episode, while lamotrigine, but not lithium, was superior to placebo in terms of delaying time to intervention for depressive episodes.
In a meta-analysis of the data from the lamotrigine bipolar trials noted earlier [25], [26], Goodwin and Geddes reported that lithium was significantly more effective than placebo in preventing relapse in patients with bipolar disorder [27]. They observed that overall lithium had significant efficacy against manic relapse with a less substantial and not significant statistically, effect against depressive relapse.
Carbamazepine
A meta-analysis by Davis et al. in 1999 on mood stabilizers in the prevention of recurrent affective disorders included 10 double-blind, randomized studies comparing carbamazepine to lithium [18]. In 572, patients followed in maintenance treatment between 1 and 3 years, 55% of carbamazepine-treated patients versus 60% of lithiumtreated patients had experienced amajor affective relapse. This difference was not statistically significant.
Early studies by Ballenger and Post [5] and Okuma [28] noted positive prophylactic effects of carbamazepine in bipolar disorder compared with placebo. Subsequent maintenance studies comparing carbamazepine with lithium produced variable findings. Both these studies permitted rescue medications to manage breakthrough mood episodes. Two studies found that the two treatments were equally efficacious [29], [30]; however, other studies supported superior efficacy of lithium prophylaxis [31–33].
Placidi et al. reported that both carbamazepine and lithium were effective in two-thirds of patients over a 3-year period in a total of 83 patients [30]. However, a higher dropout rate was observed in patients in the lithium cohort who exhibited mood incongruent psychotic features. Carbamazepine was also reported to display no significant improved efficacy compared with lithium among patients with mood-incongruent illness, comorbidity, mixed states and bipolar II disorder [34].
Compared to carbamazepine, lithium was reported to ‘significantly lengthen the time in remission’ [33]. Small et al. reported that all carbamazepine-treated patients dropped out during the first 24 weeks of a 2-year prophylaxis study; lithium dropouts occurred over the first year [32]. Coxhead et al. also observed a high rate of early dropout but posited that this may have been because of a precipitous withdrawal of lithium, which was used to stabilize all patients before entering the maintenance phase of the study [31]. Rescue medications for breakthrough mood episodes were also allowed in these comparative studies of carbamazepine versus lithium prophylaxis.
Although many open studies support the conclusions that carbamazepine has an effective role as an alternative to lithium in the maintenance treatment of bipolar disorder, they also suggest that it may lose some prophylactic efficacy over time [35], [36]. A quantitative metaanalysis by Dardennes et al. [37] in 1995 included four randomized, double-blind, controlled studies comparing carbamazepine with lithium in the prophylaxis of bipolar disorders [29–31],[33]. Their findings argued that carbamazepine has not yet been conclusively shown as an effective bipolar prophylactic treatment, particularly in patients not selected for treatment on the basis of cycle length or history of lithium response or non-response. Carbamazepine may safely and successfully reduce the number of mood episodes, hospitalizations, cumulative affective morbidity and required lithium dosage when used as augmentation in lithium-refractory bipolar patients, but this report requires further study [38].
Kleindienst and Greil have recently reported an openlabel randomized clinical trial that focused on the differential efficacy of carbamazepine and lithium for bipolar maintenance treatment over a 21/2-year period in 171 bipolar patients [39]. In bipolar I patients (n=114), lithium was superior to carbamazepine; this greater efficacy for lithium was also reported in the 67 patients identified as having a classical subtype of bipolar I disorder characterized by the absence of mood-incongruent delusions and comorbidity. The carbamazepine-treated classical bipolar patients had a significantly higher rate of hospitalization compared to the lithium group (62% vs 26%). Conversely, there was a trend favouring carbamazepine over lithium for prophylaxis in patients with a history of mood-incongruent delusions and comorbidity. When dropout, re-hospitalization and interepisode morbidity were taken into account, the response rate remained higher for lithium prophylaxis compared to carbamazepine [40]. Kleindienst also analysed the impact of episode sequence on efficacy and found that patients with amania–depression-free interval sequence did better with lithium [39]. Lithium also showed a positive impact on reducing suicidal behaviour; carbamazepine did not [39]. These reports and general expert consensus are consistent in stating that carbamazepine is inferior to lithium in the maintenance treatment of bipolar disorder [41].
There may be a synergistic effect with combination prophylactic therapy for bipolar disorder as suggested by a 3-year cross-over study that was randomized and double-blinded to compare carbamazepine, lithium and the combination of carbamazepine and lithium [2]. Fiftytwo subjects were randomized to receive lithium or carbamazepine prophylaxis for 1-year, crossed-over to the other drug for the second year and then given both drugs in combination in the third year. Although the differences were not statistically significant, the percentage of patients who had marked or moderate improvement on the Clinical Global Impressions (CGI) scale favoured the combination therapy (55.2% combination vs 33.3% lithium vs 31.4% carbamazepine). It was also noted that patients with a history of rapid-cycling did significantly better (p < 0.05) on lithium plus carbamazepine than on monotherapy (56.3% combination vs 28.0% lithium vs 19.0% carbamazepine).
Valproate
Lambert and Venaud performed a comparative study of valproate (formulated as valpromide) versus lithium in the prophylaxis of unipolar and bipolar disorders [42]. This 18-month open randomized study did not use the double-blind method because of concerns about the necessity of administering repeated doses of lithium to adjust the dosage, thus rendering the double-blindmethod meaningless. The 150 subjects were randomized to valproate or lithium at 27 different centres. There were no significant differences between groups. Valproate was administered at a set dose of 1200 mg/day throughout the study while the dosage for lithium was adjusted for each subject as a function of clinical results and serum lithium levels. Measured or targeted blood levels for either valproate or lithium were not reported.
In the valproate group (n=95), there were 39 episodes of recurrent mood episodes or 0.51 episodes per subject. The lithium group (n=79) experienced 42 recurrences or 0.61 episodes per subject. This difference was not statistically significant, although it represents a 20% lower rate of new episodes among valproate-treated patients compared with lithium-treated patients during this 18-month open, randomized study. The nature of recurrent episodes observed while on either studies maintenance drug tended to suggest that although both valproate and lithium prevented recurrence of both manic and depressive phases, they both showed slightly less ability to prevent depressive episodes.
Secondary outcomes were also not significantly different between the two treatment groups, although it was noted that valproate was better tolerated than lithium. This randomized maintenance study therefore suggested that valproate's efficacy in bipolar prophylaxis was at least equal to lithium and was better tolerated.
Bowden et al. conducted the first double-blind, randomized, controlled maintenance study in bipolar I disorder that followed patients from an index manic episode through a subsequent 52-week maintenance phase [24]. There were 571 patients enrolled in the open phase of treatment during which the investigator provided discretionary treatment of the manic episode with the exception of depot neuroleptics and ECT. This open phase lasted 3 months or less with entry into randomization with scores of 11 or less on the Mania Rating Scale (MRS), 13 or less on the Depressive Syndrome Scale (DSS) and more than 60 on the GAS on two consecutive occasions at least 6 days apart.
Recovery criteria were met by 372 patients allowing entry into the randomized, placebo-controlled, double blind, parallel-group study. Subjects were randomized to divalproex, lithium or placebo in a 2:1:1 ratio. Patients that were randomized off their divalproex or lithium and onto the other agent or placebo had the drug gradually tapered off during the first 2 weeks of maintenance treatment. Study drugs were administered three times per day and dosed to serum trough concentrations of 71–126 ug/mL for valproate and 0.8–1.2 mmol/L for lithium.
The results of this study used an intent-to-treat analyses for efficacy on data from 187, 90 and 92 patients in the divalproex, lithium and placebo groups, respectively, with no significant difference found in time to recurrence of any mood episode during maintenance therapy. The mean durations of survival in maintenance treatment for the three groups were 198, 152 and 165 days, respectively. On this measure there was a significant difference between the divalproex and lithium-treated groups favouring divalproex (p=0.015). Specifically examining the time to either a manic episode or a depressive relapse for the three treatment groups favoured divalproex compared to lithium on several secondary outcome measures, including less deterioration in depressive symptoms and GAS scores. Early termination for intolerance or noncompliance during the study also favoured divalproex (22%) over lithium (35%) (p < 0.02).
The Cochrane Database of Systematic Reviews recently reviewed the available research evidence to clarify the justification of the increasing use of valproate, especially in the US, for bipolar disorder prophylaxis [43]. Of 19 trials detected by their search criteria, the Bowden et al. 2000 study [24] was the only one that met their inclusion criteria for a randomized, controlled trial comparing valproate with placebo or other mood stabilizers for specific use in maintenance therapy. In their analysis although valproate may be more efficacious than placebo in the prevention of mood episodes, ‘the effect is not robust to the choice of analysis and the true efficacy of valproate therefore remains uncertain’ [43].
In a subsequent post-hoc analysis of the Bowden et al. 2000 study, Gyulai et al. reported that divalproextreated patients had less worsening of depressive symptoms than lithium-treated patients during maintenance [44]. Divalproex decreased the probability of depressive relapse in bipolar disorder, particularly in those patients who had previously experienced an antimanic response and in those patientswith amore severe course of illness.
Lamotrigine
Lamotrigine is an anticonvulsant of the phenyltriazine classmost often used as add-on or monotherapy of partial seizures that has been reported to possess antidepressant and mood-stabilizing properties [45–47]. The spectrum of suggested activity for lamotrigine, including cases involving treatment-resistant forms of bipolar depression and rapid-cycling, has made the anti-epileptic agent the target of more intensive study as a potentially unique maintenance treatment option for bipolar disorder.
The initial lamotrigine maintenance study published by Calabrese et al. in 2000 was a double-blind, placebo-controlled study in rapid-cycling bipolar disorder [48]. Lamotrigine was added to the treatment regimens of 324 patients meeting DSM-IV criteria for rapidcycling bipolar disorder during the initial open-label phase. One hundred and eighty-two of these patients stabilized and were randomly assigned to the double-blind maintenance phase after being stratified for bipolar I or II disorder. These stabilized patients were tapered off other psychotropic treatment and randomly assigned to either lamotrigine monotherapy or placebo in a 1:1 ratio for the 6-month maintenance phase.
The primary outcome measure was time to additional pharmacotherapy for emerging symptoms of a mood episode. Secondary outcome measures included survival in study, percentage of patients stable without relapse for 6 months and changes in GAS and Clinical Global Impressions – Severity of Illness scale (CGI-S) scores.
Before stabilization, 57% of the patients were depressed, 20% were hypomanic or manic, 18% were euthymic and 5% had mixed states. Treatment groups (n=182) entering the randomized phase of the study were similar with regard to demographics, past psychiatric history and current psychiatric state. Seventy-one per cent were classified as having bipolar I disorder. The average daily dose of lamotrigine was 288±94 mg, with a range of 100–506 mg/day.
During the 6-month maintenance phase of the study, 49 placebo patients (56%) and 45 lamotrigine-treated patients (50%) required treatment of an emerging mood episode with additional pharmacotherapy. There was no statistical significance between these two treatment groups on the primary outcome measure of time to additional pharmacotherapy – a median of 12 weeks for placebo versus 18 weeks for lamotrigine. When overall survival in study was analysed (withdrawal from study for any reason, including additional pharmacotherapy), the difference between the treatment groups became significant (p=0.04), with lamotrigine at 14 weeks median survival versus only 8 weeks for placebo.
However, greater differences arose when the data were stratified according to diagnoses of bipolar I disorder or bipolar II disorder. For bipolar I patients, there were no significant differences between lamotrigine- and placebo treatment groups on measures of time to additional pharmacotherapy or survival in study. For bipolar II patients, there was a trend toward significance (p=0.07) detected between lamotrigine and placebo groups on time to additional pharmacotherapy; this trend grew to reach statistical significance (p=0.015) favouring lamotrigine over placebo on survival in the study. Medial overall survival during maintenance therapy for these rapid-cycling bipolar II patients was 15 weeks for lamotrigine and 4 weeks for placebo. The percentage of patients stable without relapse for the entire 6 months of the randomized phase was significantly greater (p=0.03) in the lamotrigine group (41%) than in the placebo group (26%). This statistically significant advantage for lamotrigine held up to diagnostic stratification for the bipolar II subtype only, with 46% of bipolar II patients on lamotrigine monotherapy stable without relapse after 6 months compared to only 18% of placebo-treated bipolar II patients. Similarly, secondary outcome measures returned trending or statistically significant and favourable differences between lamotrigine and placebo maintenance treatment for bipolar II but not bipolar I patients: reduced CGI-S scale scores at weeks 6 and 12 (p=0.10) and improvements in GAS scores at weeks 3, 6 and 12. These results suggest that lamotrigine may be a useful treatment for rapid-cycling bipolar II patients.
The Bowden and Calabrese 18-month maintenance studies [25], [26] comparing lamotrigine, lithium and placebo reviewed earlier provide further support for the use of lamotrigine as a mood stabilizer, perhaps complementary to the effects of lithium. In recently manic or hypomanic patients with bipolar I disorder, both lithium and lamotrigine were superior to placebo at delaying the time until additional pharmacotherapy was required for treatment of a mood episode (p=0.003 and p < 0.02, respectively). Lamotrigine showed significantly greater efficacy for prolonging time to a depressive episode compared with lithium (p=0.15, lamotrigine vs placebo; p=0.167, lithium vs placebo). The results of this controlled maintenance study support the use of lamotrigine prophylaxis of mood episodes below baseline for bipolar I patients. In recently depressed patients with bipolar I disorder, both lamotrigine and lithium were significantly superior to placebo at delaying time to intervention for any mood episode (p=0.029, lamotrigine 200 and 400 mg/day vs placebo; p=0.029, lithium vs placebo). Lamotrigine and lithium were superior to placebo on the analysis of overall survival in the study, while lamotrigine, but not lithium, was superior to placebo in terms of delaying time to intervention for depressive episodes. This study concludes ‘lamotrigine is an effective treatment for bipolar disorder with a spectrum of efficacy that provides particular benefits in the treatment of depressive symptomatology’.
In the combined analysis of the data from the lamotrigine bipolar trials [25], [26], Calabrese et al. confirmed that overall lamotrigine was significantly more effective than placebo in preventing relapse in patients with bipolar disorder, while showing primary effectiveness against depression [49].
Olanzapine
In a comparator study of olanzapine versus divalproex sodium over a 47-week period of maintenance treatment of 251 patients with bipolar disorder, the mean improvement in MRS scores was significantly greater in the olanzapine-treated group [50]. Although there were no significant overall differences between the two treatments in the rates of symptomatic remission and subsequent relapse over the entire 47-week period, the olanzapine treated patients experienced a significantly shorter median time to remission of acute mania compared with the divalproex-treated group (14 days vs 62 days, respectively).
A placebo-controlled study evaluating the preventive efficacy of olanzapine added to either lithium or valproate has recently been published [51]. The results from this 18- month maintenance study found that among patients who experienced syndromic remission from an acute manic episode following the addition of olanzapine to lithium or valproate, those remaining on the combination therapy sustained symptomatic remission longer than those on lithium or valproate monotherapy. There was no reported difference in the duration of syndromic remission.
Data from the first placebo-controlled, double-blind study to examine the efficacy of olanzapine monotherapy in relapse prevention in bipolar I disorder has been evaluated [52]. The results from this 48-week study found that the median time to symptomatic relapse into any mood episode after open-label stabilization of acute mania with olanzapine was significantly longer among patients continuing to receive olanzapine than in placebo-treated patients (174 days vs 22 days, respectively). This study suggests that olanzapine may prevent bipolar I relapse into any mood state for significantly longer periods of time than placebo.
Recently unpublished data comparing olanzapine with lithium in relapse prevention of bipolar disorder from a randomized double-blind controlled 12-month trial have been presented [53]. Among patients achieving initial symptomatic remission from acute mania with openlabel combination therapy with olanzapine and lithium, those randomized to olanzapine monotherapy were significantly more likely to remain in remission than those in the lithium monotherapy group. Olanzapine was significantly more effective than lithium in the prevention of manic and mixed episodes; both olanzapine and lithium were comparable in preventing depressive relapse.
Evidence-based medication maintenance therapy in bipolar I disorder
The available data particularly support the use of lithium, lamotrigine and olanzapine monotherapies for relapse prevention in bipolar I disorder. Lesser evidence exists for valproate and less still for carbamazepine. Generally, all indicators point to improved prevention using a combination of these mood stabilizers, particularly in patients with a more severe illness course.
With the notable exception of lamotrigine, there is a paucity of placebo-controlled data to guide maintenance therapy decisions after remission from an acute depressive episode in bipolar disorder. Given the described modest effects of lamotrigine in preventing manic relapse, the majority of clinicians agree that lamotrigine maintenance is typically best used as part of a combination medication strategy in bipolar I patients with a propensity for depressive relapse. It may be considered as monotherapy maintenance in bipolar II disorder, however, given the particularly depressive burden of this bipolar subtype.
Patients in remission from a bipolar depression episode, thought to be responsive to a combination of traditional antidepressant plus a mood stabilizer, present a difficult challenge because of concerns that antidepressants may destabilize the course of illness. Reports suggest that in patients acutely responsive to antidepressants, discontinuation of the antidepressant may more likely lead to depressive relapse [54]. These data are derived from nonrandomized studies. Clinically individualized treatment plans should allow for the careful continuation of antidepressant medications into maintenance phase for responsive patients with bipolar disorder who are concurrently receiving appropriate mood stabilizer and who do not have a history of rapid-cycling. If antidepressants must be used for patients with a past history of rapid-cycling, serious consideration of a more abbreviated duration of treatment with the antidepressant must be made to minimize the risk of inducing cycle acceleration.
If atypical antipsychotics have been used as part of a successful acute treatment for bipolar depression they may be considered for discontinuation 3–6 months after remission, with continuation of the mood stabilizer. Presently, olanzapine has the best evidence among atypical antipsychotics for maintenance treatment, including prevention of depressive relapse. However, given ongoing concerns about weight gain and other possible side-effects with long-term atypical antipsychotic use, first-line maintenance should continue with lithium, lamotrigine or valproate.
The majority of data in existence and those reviewed here for maintenance therapy of bipolar disorder pertains to treatment after a manic episode. Although lithium, valproate, carbamazepine, lamotrigine and olanzapine all have data to suggest defined roles beyond initial antimanic use and into maintenance phase, the majority of patients should continue with the regimen that brought about remission. In the majority of patients, this will be a combination of medications, typically a mood stabilizer plus an atypical antipsychotic. The reviewed study by Tohen et al. [51] supports the practice of continuing the antipsychotic along with the mood stabilizer because those patients on this combination achieved a more sustained symptomatic remission. Again, consideration of dose reduction and discontinuation after 3–6 months of treatment with the atypical antipsychotic and continued mood stabilizer must be made because of concerns associated with all atypical antipsychotics in long-term use.
Since the majority of patients with bipolar disorder require, or are placed on, some form of combination long-term medication therapy, there remains a need to obtain further data on the various permutations possible in order to more accurately and safely guide clinical treatment. It is not uncommon to review patient-care plans for bipolar maintenance that include three or more concurrent mood stabilizers, antipsychotics and/or antidepressants. Recognition of these issues and the clear void of data applicable to ‘real-world’ long term management of patients with bipolar disorder warrant the design and implementation of well-controlled, double-blinded and placebo-controlled studies to help address the unmet need faced by clinicians and their patients.
