Abstract
The few studies available that compare neuropsychological function in different bipolar disorder states indicate that these patients have a number of cognitive difficulties [1–5]. Other studies have demonstrated that bipolar patients show distinct alterations of cerebral blood flow during the different states of their disease. However, there are only a few studies examining neuropsychological dysfunction and neuroimaging data in bipolar patients [6–9], and to our knowledge this is the first study reporting on the relationship between these two markers.
Cognitive deficits have been found in selective [10] and sustained attention, in around 75% of bipolar patients, suggesting a role as markers of neuropsychological vulnerability for bipolar disorder [11]. As regards mnesic functions, some recent studies have shown learning and verbal memory dysfunctions in euthymic bipolar patients [3],[11–15]. However, it has been suggested that subthreshold depression may influence mnesic functions [14], [16], [17].
As regards executive functions and verbal fluency, the findings in bipolar disorder are not consistent [12], [18]. Some studies indicate worse performance in frontal functions [12], [14] and lower verbal fluency in stable bipolar patients [14], while others did not find significant differences in verbal fluency between bipolar patients and healthy controls [12].
Finally, as regards psychomotor function, patients with melancholic depression usually show psychomotor slowness [19], while distractability appears to speed movement in manic patients, suggesting that psychomotor function is state-dependent [12], [20].
Neuroimaging studies are controversial but the pathophysiology of affective disorders appears to be related to the frontal lobe, subcortical structures and the limbic system [21]. In bipolar and unipolar patients, most positron emission tomography (PET) and single photon emission computed tomography (SPECT) studies have described patterns of abnormality in cerebral blood flow and/or glucose metabolism (22], and also show an increase in radiodrug reuptake in the temporal lobes, which is more evident in patients with mania. Studies performed during the manic phase indicate left-right asymmetry [23], with less perfusion in the right and basal temporal cortex than in the left and dorsal regions. Gyulai et al. [24] found that in 12 rapid cycling bipolar patients (four without medication and eight receiving lithium carbonate), radiodrug distribution was also asymmetric in the anterior part of the temporal lobes in both the depressive/dysphoric phases as well as in the manic/hypomanic phases, being symmetric in the euthymic phases. Based on these results, the presence of a state-dependent ‘temporal dysfunction’ was suggested for bipolar disorder.
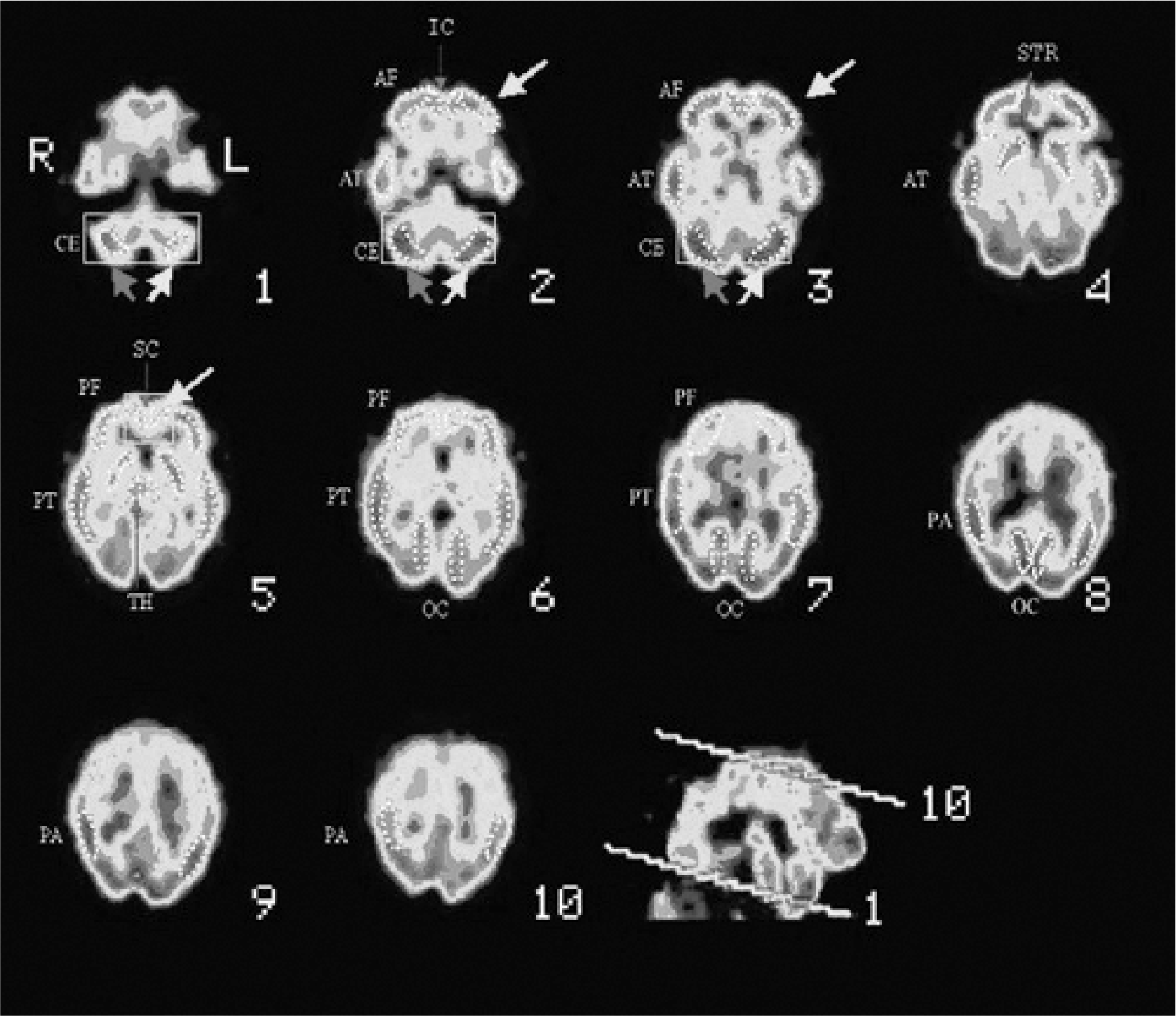
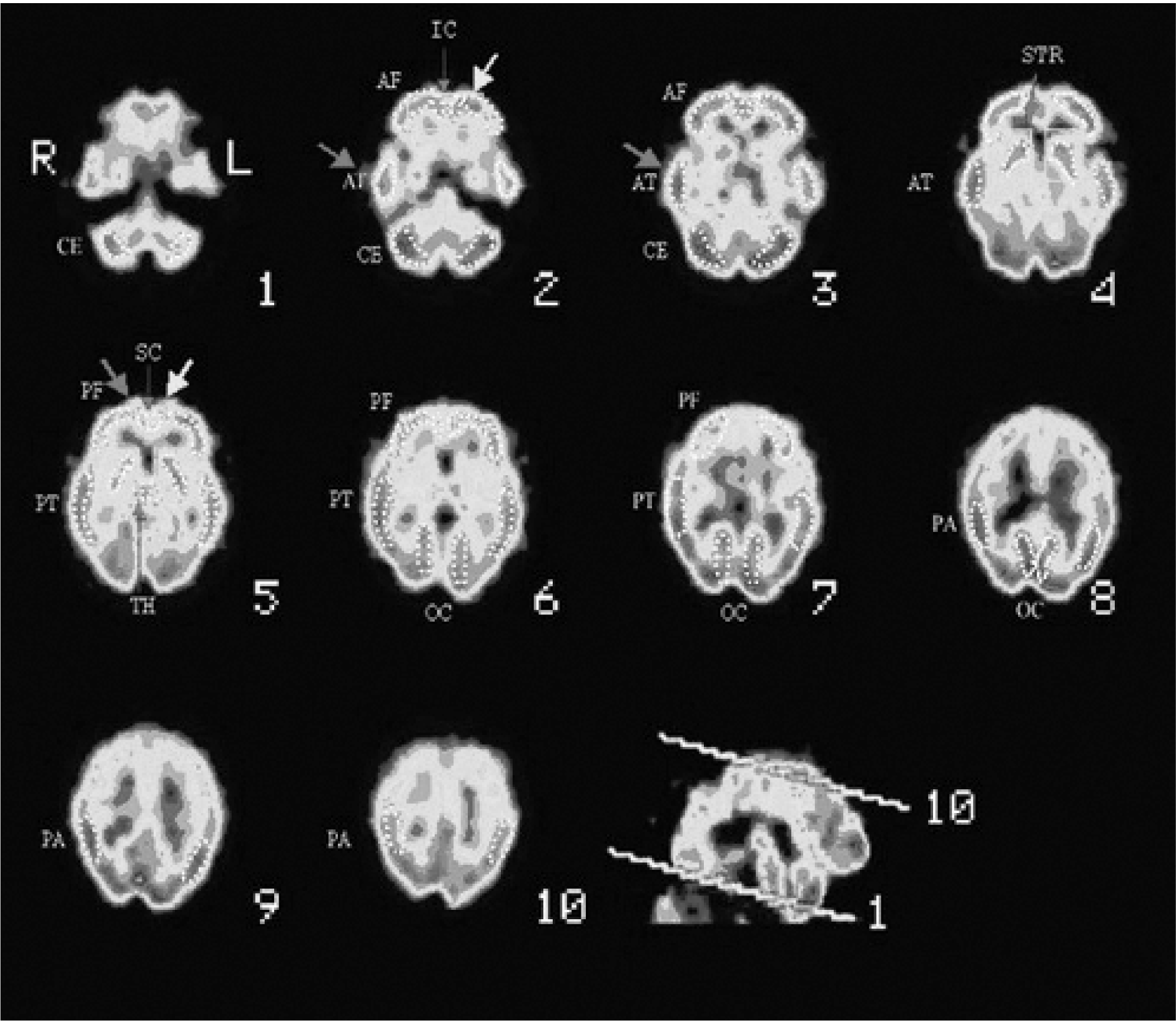
Cerebral perfusion SPECT in bipolar disorder. Frontocerebellous oblicuous planes. Regions of interest (ROIs) applied in the study. CE, cerebellum; AF, anterior frontal; PF, posterior frontal; IC, anterior cingulate; SC, posterior cingulate; AT, anterior temporal; PT, posterior temporal; PA, parietal; OC, occipital; STR, striate; TH, thalamus. Cerebral perfusion SPECT in bipolar disorder: depressive episode. CE, cerebellum; AF, anterior frontal; PF, posterior frontal; IC, anterior cingulate; SC, posterior cingulate; AT, anterior temporal; PT, posterior temporal; PA, parietal; OC, occipital; STR, striate; TH, thalamus. Cerebral perfusion SPECT in bipolar disorder: manic episode. CE, cerebellum; AF, anterior frontal; PF, posterior frontal; IC, anterior cingulate; SC, posterior cingulate; AT, anterior temporal; PT, posterior temporal; PA, parietal; OC, occipital; STR, striate; TH, thalamus.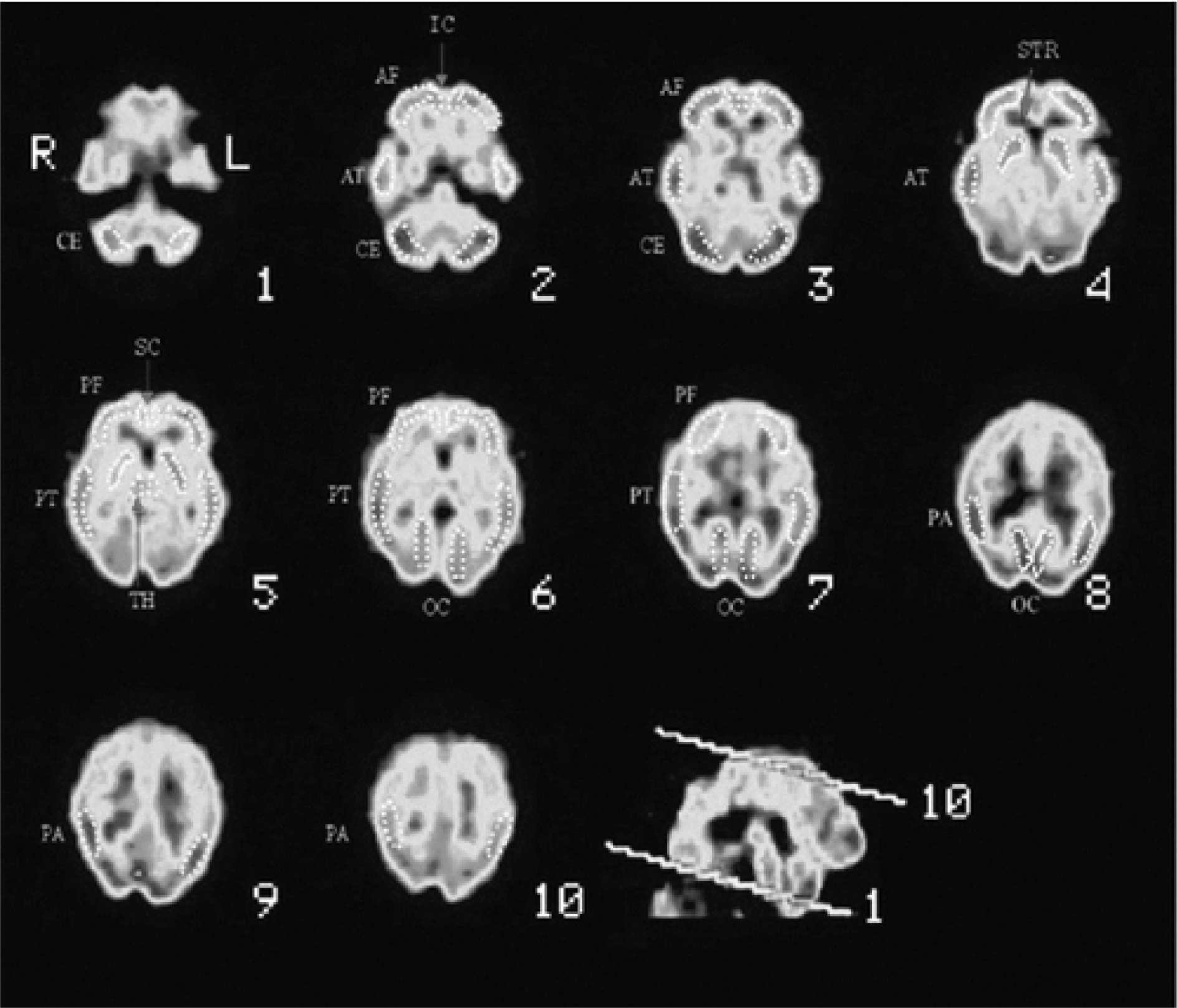
The aim of this study was to determine the possible alteration in neuropsychological functioning and cerebral blood flow in patients with bipolar disorder and to establish a relationship, as yet not formulated, between these markers and the psychopathology of the disorder itself.
Method
Subjects
The study was carried out in a cohort of patients recruited from the Bipolar Disorders Program of the Hospital Clínic at the University of Barcelona (Spain).
The study included 43 patients fulfilling the Research Diagnostic Criteria (RDC) [25] for bipolar disorder with the standardized interview Schedule for Affective Disorders and Schizophrenia (SADS) [26]. Patients were classified according to their clinical state by means of RDC criteria and cut-off scores on the Young Mania Rating Scale (YMRS) and Hamilton Depression Rating Scale (HDRS): (i) patients with mania (n = 7, 23.3%) (YMRS ≥ 20); (ii) patients with hypomania (n = 8; 26.7%) (YMRS ≥ 12); (iii) patients with depression (n = 12, 40%) (HDRS ≥ 17); and (iv) euthymic patients (n = 3, 10%) (YMRS ≤ 7 and HDRS ≤ 7). The exclusion criteria included the presence of any severe mental or physical disease (apart from bipolar disorder), mental retardation, current history of alcohol dependence or other psychoactive substances or pregnancy. For the control group we recruited six subjects among non-blood relatives of the patients included in the study. They were healthy subjects with no personal psychiatric history, and were age, sex and education-matched to the patient group.
The study was presented and approved by the Ethics and Research Board of the Hospital Clínic of Barcelona (Spain). Prior to inclusion in the study, both patients and healthy control subjects were informed and gave written informed consent to participate in the study.
Study design
The first patient assessment included the collection of general demographic and clinical data by two independent psychiatrists. Neuropsychological assessment was performed within 1 week of the diagnostic assessment. In manic, hypomanic or depressed patients the SPECT was undertaken during the first 24–48 hours after diagnosis. In euthymic patients, SPECT was carried out during the first week after diagnosis. To avoid possible bias related to differences in pharmacological treatments received, both SPECT and neuropsychological assessments were performed prior to administering any medication.
General assessments
For each cohort we determined the following clinical parameters at baseline: (i) Global Assessment Functioning (GAF); (ii) Positive and Negative Symptom Scale (PANSS) [27], [28] as a measure of psychotic symptoms with its three subscales (positive, negative and general psychopathology); and (iii) Hamilton Depression Rating Scale (HDRS) [29] to measure depressive symptoms; and (d) the validated Spanish version [30] of the Young Mania Rating Scale (YMRS) [31].
Neuropsychological assessment
We used the following neuropsychological tests: Vocabulary subtest of the Wechsler Adult Intelligence Scale (WAIS) [32] to measure estimated premorbid intelligence; Digit subtest of WAIS [32] to assess attention and working memory; Wisconsin Card Sorting Test (WCST) [33] to determine executive functions; the Stroop Test [34] and Trail Making Test [35] to measure attention processes, also using the latter to evaluate psychomotor functioning; California Verbal Learning Test (CVLT) [36] and Logical Memory and Visual Reproduction subtests of the Wechsler Memory Scale (WMS-R) [37] to measure memory processes and the Controlled Oral Word Association Test (COWAT) to assess phonetic verbal fluency (FAS) [38].
Functional neuroimaging
All the cerebral SPECT studies were performed in the Department of Nuclear Medicine of the Hospital Clínic of Barcelona. The cerebral SPECT technique involved the administration of 99mTc-HMPAO and all studies were evaluated by two experts in nuclear medicine.
We undertook semiquantitative analysis of the SPECT of healthy controls and euthymic, manic and depressed bipolar patients. We analysed 26 anatomical regions of interest (ROI), traced for each hemisphere in 10 oblicuous or standardized frontocerebellous planes of 2 pixels in thickness. The shape and the placement of the ROI involved taking normal T1 images as a standard reference. The ROI sites (all symmetrical) were as follows: anterior frontal region, 4 right and 4 left; anterior temporal region, 3 right and 3 left; posterior temporal region, 4 right and 4 left; parietal region, 2 left and 2 right; occipital region, 3 left and 3 right; basal ganglia, 2 left and 2 right; thalamic region, 1 left and 1 right; cerebellous hemisphere, 3 right and 3 left. ROIs were not traced to delimit white substance because of its scarce reuptake and resolution, and hence the low diagnostic power of SPECT in these regions. The analytical parameters of the SPECT included regional reuptake indices (RRI), relative perfusion indices (RPI) and asymmetry indices (AI) from the reuptake obtained in each of the different ROIs: cerebellum, frontal lobe, cingulate, temporal lobes, parietal lobes, occipital lobes, striate and thalamus.
The indices were calculated with the following formulae:
RRI = ROI/Reference ROI; Reference ROI = brain; RPI = ROI/Reference ROI; Reference ROI = brain; AI = (right RRI – left RRI)/(left RRI – right RRI).
Statistical analysis
For comparisons between patients and controls we applied Student's t-test for normal distribution (Shapiro-Wilk statistical method), and the Mann–Whitney U-test when the distribution was not normal. Analysis of variance or Kruskal–Wallis test were used according to the normal distribution of the variables under study. To quantify the degree of correlation among the quantitative variables, we used the Spearman's rank correlation coefficient.
Statistical analysis was carried out using SPSS statistical package version 10.0 [39] with a level of statistical significance of 0.05.
Results
Patient disposition
Forty-three patients out of the 85 initially interviewed were included in the study after confirming diagnosis and applying inclusion and exclusion criteria. In these 43 patients, we performed cerebral SPECT evaluations in 39 patients in some phase of their disease, and 30 patients were assessed with the neuropsychological battery. Thus, data of cerebral SPECT and the neuropsychological explorations in the same episode were obtained in a total of 26 patients out of the 43 patients included in the study.
As regards the six control subjects that were also included in the study, we performed cerebral SPECT evaluations in five, and neuropsychological explorations in three of them.
Characteristics of the sample
Table 1 shows the general demographic and clinical data of the patients. No statistically significant differences were observed in either the age or the level of education between the group of patients and the control group (Student's t-test = −0.67; p = ns; Mann–Whitney U-test = 18.5; p = ns).
Demographic and general clinical characteristics of the patients included in the study
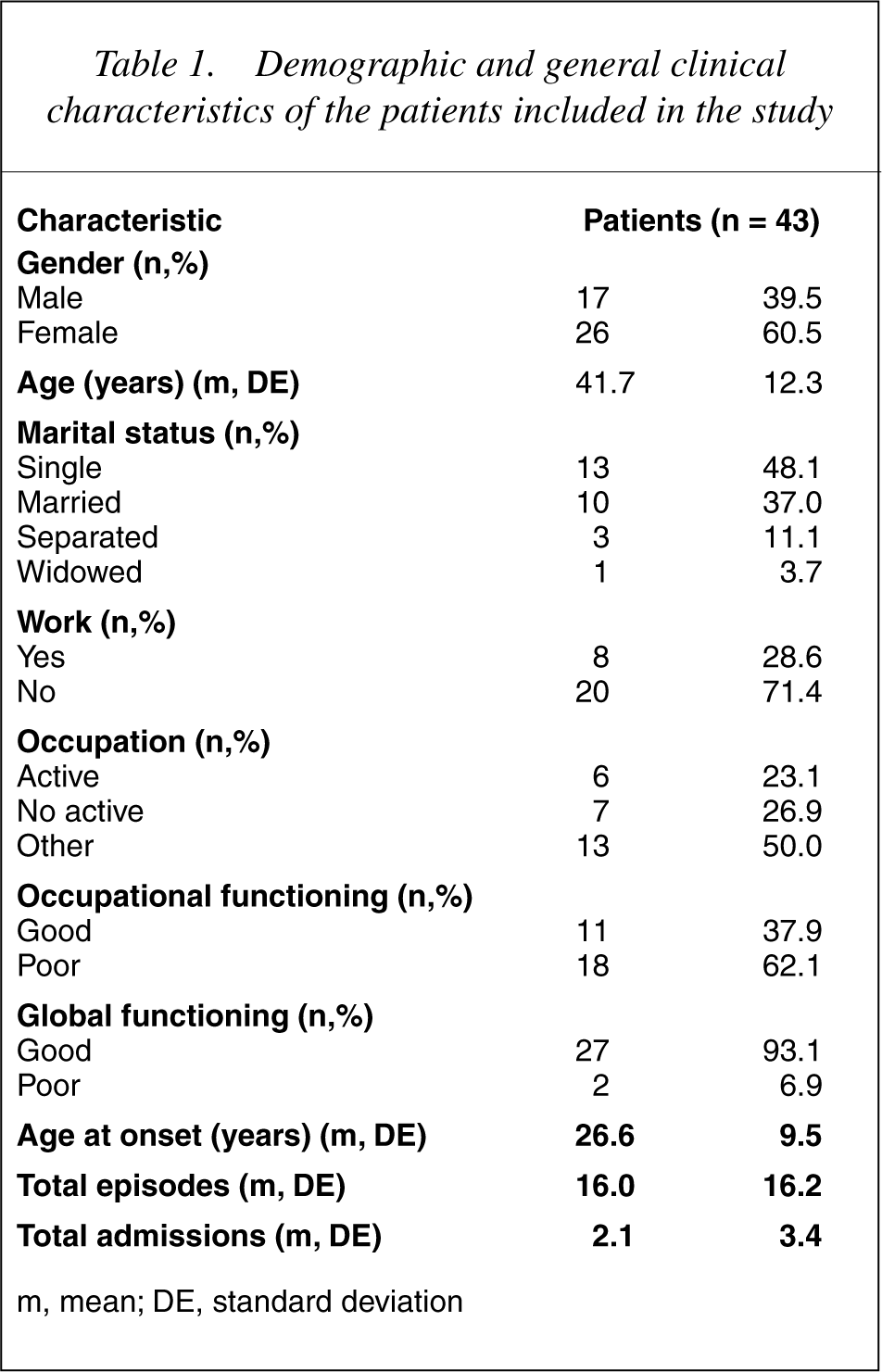
m, mean; DE, standard deviation
Table 2 shows the results of clinical evaluations and psychosocial functioning assessments of the patients. In the PANSS scale analysis, depressive patients showed higher scores in negative subscale and total score, while manic patients showed greater predominance of positive symptoms. Higher scores were observed in euthymic patients compared to their depressive and manic/hypomanic counterparts, as measured by the GAF scale (Kruskal–Wallis test = 7.23; p < 0.05).
Clinical assessments and rating scales
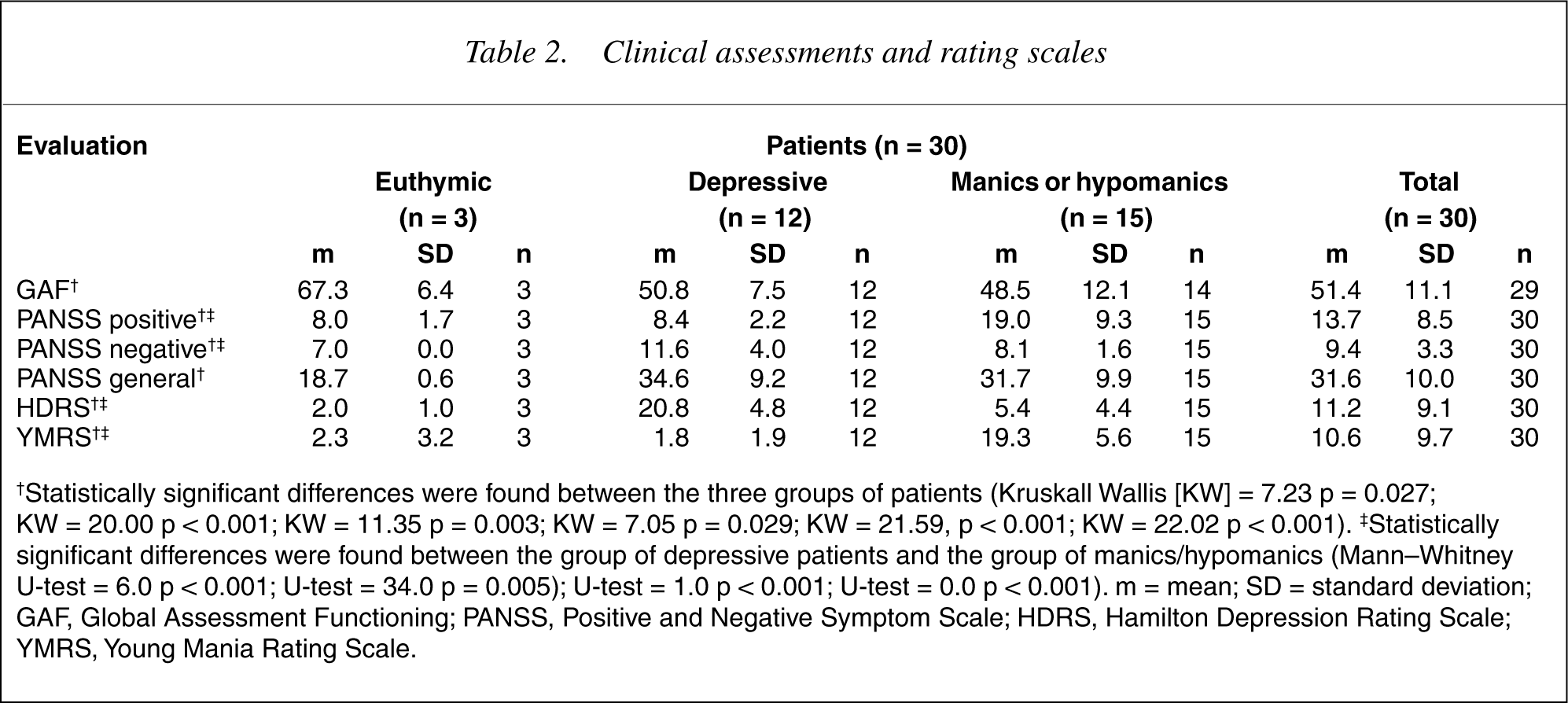
†Statistically significant differences were found between the three groups of patients (Kruskall Wallis [KW] = 7.23 p = 0.027; KW = 20.00 p < 0.001; KW = 11.35 p = 0.003; KW = 7.05 p = 0.029; KW = 21.59, p < 0.001; KW = 22.02 p < 0.001). ‡Statistically significant differences were found between the group of depressive patients and the group of manics/hypomanics (Mann–Whitney U-test = 6.0 p < 0.001; U-test = 34.0 p = 0.005); U-test = 1.0 p < 0.001; U-test = 0.0 p < 0.001). m = mean; SD = standard deviation; GAF, Global Assessment F unctioning; PANSS, Positive and Negative Symptom Scale; HDRS, Hamilton Depression Rating Scale; YMRS, Young Mania Rating Scale.
Correlations between cognitive functions and cerebral blood flow
Table 3 presents a summary of the findings of correlations between cognitive functions and cerebral blood perfusion. In general, the greatest statistically significant correlations between the different cognitive dysfunctions evaluated and regional hypoperfusion were those obtained with perfusion analysis expressed by the RRI calculated according to the area of reference. Whereas the greatest statistically significant correlations with respect to the relationship of impairment in cognitive functions and regional hyperperfusion were those obtained in the analysis with the RPI calculated with cerebellum reuptake as the reference. Neither analysis presented discordant significance. We also determined the correlation between the AI for each ROI and the score obtained with every neuropsychological test.
Correlation analyses between cognitive function and cerebral blood flow
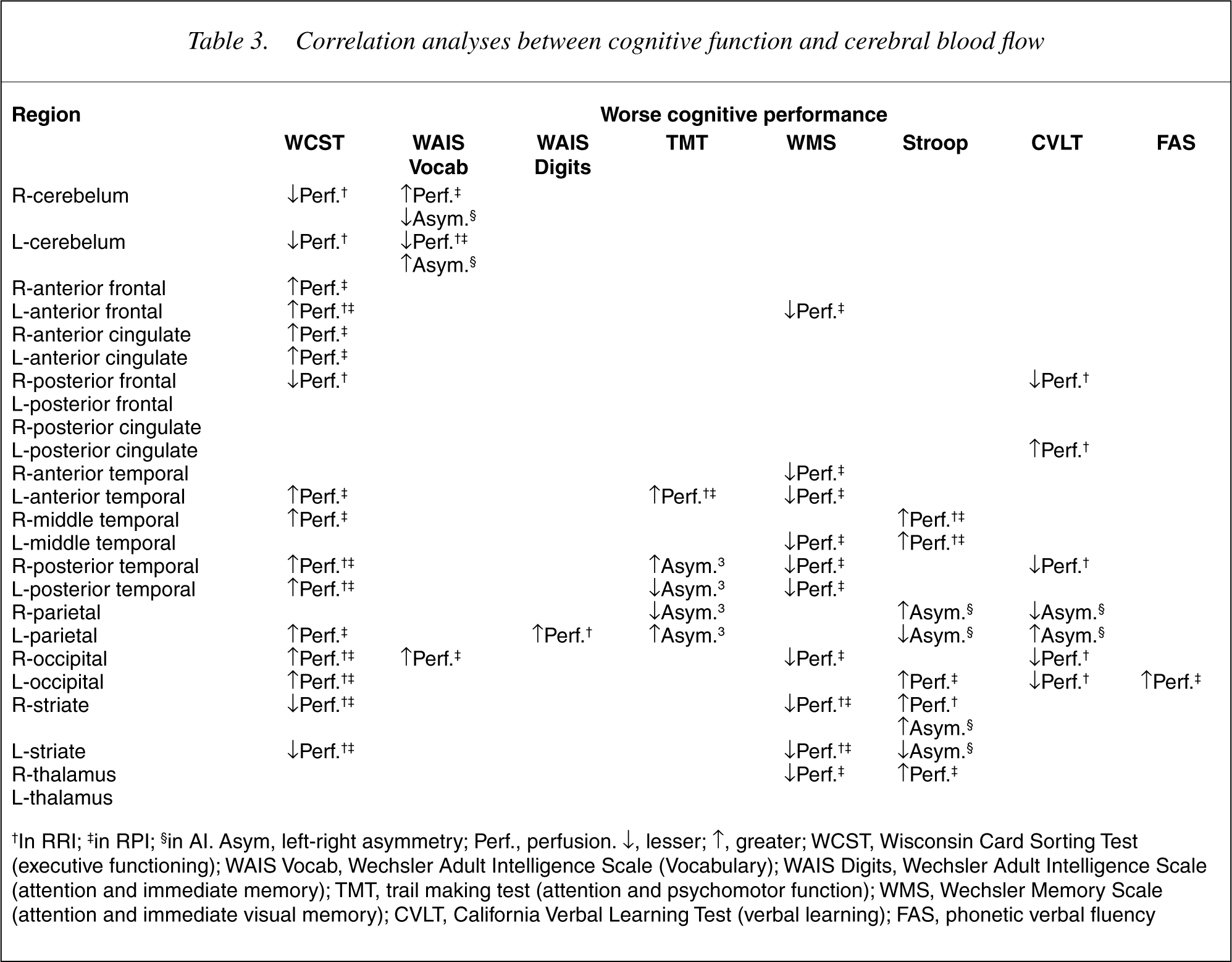
†In RRI; †in RPI; §in AI. Asym, left-right asymmetry; Perf., perfusion. ↓, lesser; ↑, greater; WCST, Wisconsin Card Sorting Test (executive functioning); WAIS Vocab, Wechsler Adult Intelligence Scale (Vocabulary); WAIS Digits, Wechsler Adult Intelligence Scale (attention and immediate memory); TMT, trail making test (attention and psychomotor function); WMS, Wechsler Memory Scale (attention and immediate visual memory); CVLT, California Verbal Learning Test (verbal learning); FAS, phonetic verbal fluency
In attention and working memory, as assessed by the Digit subtest (WAIS), we observed a statistically significant correlation among lower scores on this test and lower RRI in the left parietal region (Spearman's Rho = p < 0.05). Similarly, as regards attention processes and psychomotor speed assessed by the Trail Making Test performance was worse with both analysis of correlations from the RRI as well as the RPI. Therefore, greater change in attention and psychomotor speed were positively correlated to greater perfusion in the left anterior temporal region (Spearman's Rho = p < 0.05). Moreover, the greater the change in psychomotor speed, the greater the left-right asymmetry in the posterior region of the temporal lobe (Spearman's Rho = p < 0.05). Poor performance on the Stroop test was related by means of RRI analysis to a greater perfusion in the middle right and left temporal regions and the right striate. In the RPI analysis this parameter was related to greater perfusion in the middle and anterior temporal regions, occipital lobe, thalamus and the right striate and to greater AI for the parietal regions and the striate (Spearman's Rho = p < 0.05).
In relation to memory, those patients that showed worse performance in Logical Memory Visual Reproduction subtests (WMS-R) also obtained lower RRI values (less perfusion) in the cerebellum and the anterior frontal region (Spearman's Rho = p < 0.05), greater perfusion in the right and left striate (Spearman's Rho = p < 0.05) and lower RPI values (less perfusion) in the left anterior frontal region, in the anterior and posterior regions of the temporal lobe, in the middle temporal region of the left hemisphere, in the right and left striate and in the right region of the occipital lobe and the thalamus (Spearman's Rho = p < 0.05).
Patients with impaired performance in verbal learning function (CVLT) showed less perfusion as evaluated by the RPI in the posterior frontal regions and posterior cingulate (Spearman's Rho = p < 0.05) and a greater perfusion evaluated with the same RPI in the occipital region and an inferior left-right AI (Spearman's Rho = p < 0.05).
Finally, in phonetic verbal fluency (FAS) a statistically significant correlation was only observed between perfusion in the left occipital region evaluated with the RPI. Thus, the greater the perfusion the worse the verbal fluency.
Discussion
To date, few studies have reported the use of neuroimaging techniques as diagnostic markers, and their correlation with neuropsychological tests in bipolar patients. The most important finding from this study is that cognitive dysfunction in bipolar disorder is associated with the redistribution of blood perfusion across different regions of the brain.
However this study has limitations, namely: the crosssectional design, which restricted internal validity; the reduced sample size due to the high cost of the resources and time required for carrying out the assessments; and incomplete neuropsychological and SPECT data due to non-compliance of some patients, especially during the acute clinical state. Our main finding, consistent with previous studies, that of worse performance in memory, executive and attentional tasks (Stroop) is related to a greater perfusion in the striate region. Other studies [40–42] have suggested that striate structures are related not only to memory, but also to attentional tasks.
Patients with a poor performance in executive functions, logical memory subtest and verbal learning (CVLT), also showed low perfusion in the frontal region – suggesting hypofrontality. A decrease in the prefrontal cortex CBF is the most frequent finding in both unipolar and bipolar depression. In the latter, anomalies have been reported in temporal CBF, although these are not as clear as in unipolar depression. I Implication of the limbic system remains controversial, however, it appears increasingly likely that the pathophysiology of affective disorders involves the frontal lobe, substructures of the temporal lobe such as the hippocampus and the amygdala, subcortical structures and the limbic system [21],[43–45].
Poor performance on executive and psychomotor functions, was related to greater perfusion in the anterior temporal region. It has been suggested that the middle temporal lobe and the thalamus may work together to establish long-term memory, whereby projections to the frontal lobe would provide the pathway by which information is transformed into action [46]. In the present study, worse memory performance (Logical Memory Delayed Recall of the WMS) was related to lower perfusion in the temporal and anterior frontal regions, and this appears to confirm the association between delayed recall and the temporal and frontal lobes. On the other hand, there was a reciprocal relationship between performance of executive and attention tasks (STROOP) and increased perfusion in the middle temporal region. This result partially supports the findings of a previous study where performance in attention tasks pointed to middle temporal function [47]. The worse the memory performance (WMS) and verbal learning (CVLT), the lower the perfusion in the posterior temporal region, suggesting that temporal lobe structures are implicated in declarative memory tasks such as the CVLT. The medial temporal lobe is considered essential for memory encoding and consolidation, but potential frontal dysfunctions can also be revealed on the CVLT, such as retrieval deficits [48]. This relationship has already been reported in bipolar patients by Van Gorp [12], [13], [15] and in the general population [49].
We found that a worse performance in verbal learning and executive functions implied a higher perfusion in the cingulate region. This result suggests the involvement of the cingulate gyrus [50].
We also observed that a worse performance in executive function was related to a lower perfusion in the cerebellum. The role of the cerebellum in executive functions is probably due to the motor, graphic and attention abilities required. Although it has recently been suggested that cerebellum neurones may project towards different cerebral areas that modulate mood [51], our study did not find any association between perfusion levels and mood scales, and so our findings do not support this hypothesis.
The results of our study suggest that the bipolar brain manifests a distinct perfusion pattern characterized by striate asymmetry that is different from healthy subjects, and in keeping with the findings from recent MRI studies [52], [53]. This alteration in the striatum cerebral perfusion may help to explain the manic symptomatology of bipolar patients and their executive, attention and memory dysfunctions. Nonetheless, cerebral perfusion in manic patients follows a more complex pattern with marked disturbances in other regions such as the frontal and anterior temporal region and the cingulate. It is also worth noting that in bipolar patients, hypoperfusion in the posterior right frontal region may be linked to severe psychotic symptoms and that poor executive function (WCST), is consistent with findings in patients with schizophrenia [43], [45], [47].
The results of our study help to confirm current hypotheses that implicate functional anomalies in frontal-subcortical structures, the cerebellum and limbic system in bipolar disorder [6], [21]. However, further studies with larger samples are required to corroborate the findings of the present study and to firmly establish neuropsychological and cerebral blood flow dysfunctions in bipolar disorder [54]. This would enable the development of a comprehensive model for the understanding of the pathophysiology of bipolar disorder and the development of specific treatments.
Footnotes
Acknowledgements
We thank Neus Cerdà, Statistician (Biométrica, Barcelona, Spain).
Financial support: this study was partially supported by a FIS grant N°00/1069 from the Ministerio de Sanidad y Consumo, Spain, by the Fundació Marató de TV3 (Barcelona, Spain) and by the Stanley Medical Research Institute (Bethesda, Maryland, US).
