Abstract
This year sees the fiftieth anniversary of one of the landmark achievements in scientific history. In their article in Nature, 25 April, 1953 [1], James Watson and Francis Crick proposed the double helical structure of deoxyribonucleic acid (DNA) consisting of two chains held together by purine and pyrimidine bases. The story of their journey towards the double helix has been well documented, most notably in Watson's own account [2]. It is a story filled with drama and controversy that continues to fuel debate. This, at least in part, is because their model depended heavily on work by Maurice Wilkins and Rosalind Franklin at King's College, London. Wilkins shared his data with Watson and Crick and was later a joint recipient with them of the Nobel Prize. However, Wilkins also gave Watson and Crick access to Franklin's fine quality X-ray crystallography pictures that provided vital clues. Franklin herself probably never authorized this and although her results were published in the same issue of Nature as those of Watson and Crick and Wilkin's group, she later died prematurely and did not share in the glory of becoming a Nobel Laureate.
Controversies aside, the discovery of the double helical structure was a defining moment for genetic research that has impacted on a diverse range of disciplines, including psychiatry. In a famously understated sentence in their paper Watson and Crick remarked: ‘It has not escaped our notice that the specific pairing we have postulated immediately suggests a possible copying mechanism for the genetic material.’ They thus established a framework that would develop into the field of molecular genetics, providing the impetus for the later discoveries of messenger RNA, transfer RNA, the genetic code and leading, ultimately, to the triumphs of the human genome project.
Genetics is now one of the most active and promising areas of modern psychiatric research. Advances are beginning to be made that potentially allow an understanding of the causation of psychiatric disorders at the molecular level. However, it should not be forgotten that there has often been scepticism about the idea that genes influence susceptibility to psychiatric disorders and there has even been downright hostility. Providing evidence to support the notion that genes play a role in psychopathology has depended on classic quantitative genetics, the basic designs of which predate the molecular era.
Pre-molecular genetics
The term ‘genetics’ did not exist until it was invented by Bateson in 1905 and indeed genetics only came into being as a branch of biological science in the early 20th century on foundations laid separately by Gregor Mendel and Francis Galton in the late 19th century. Nevertheless, the observation that psychiatric disorders tend to run in families is an ancient one. The first example that we have been able to find of an attempt to study this systematically comes from the archives of the Bethlem Royal Hospital in London where the front sheet on case notes dating from 1823 required the doctor to answer the question ‘whether hereditary?’ Of course it could well be that certain types of behaviour, normal or abnormal, cluster in families because of shared environmental effects rather than shared genes. The bedrocks of psychiatric genetics have therefore been the two types of so-called ‘natural experiments’, twin studies and adoption studies, which have enabled the separating out of the effects of genetic and environmental influences on the developmental disorders. Adoption studies have been widely used in such disorders as schizophrenia, alcoholism and antisocial behaviour. Twin studies have been even more widely used providing evidence for a significant genetic component in many disorders, including unipolar depression [3], bipolar affective disorder [4] and schizophrenia [5]. A major trend over the last two decades has been to apply more sophisticated statistical models to attempt to estimate genetic, shared environmental and non-shared environmental effects. For example, Cardno and Gottesman [5] carried out a metaanalysis of five recent twin studies on schizophrenia and reported that the heritability, or proportion of variants accounted for by genes, is as high as 80–85%. Although this leaves as much as 20% of variance to be explained by environment, this seems to be entirely of the nonshared type. Figure 1 summarizes the results of other recent twin studies showing the estimated contribution of genetic and environmental factors for various behavioural disorders.
Estimates of genetic and environmental effects from recent twin studies.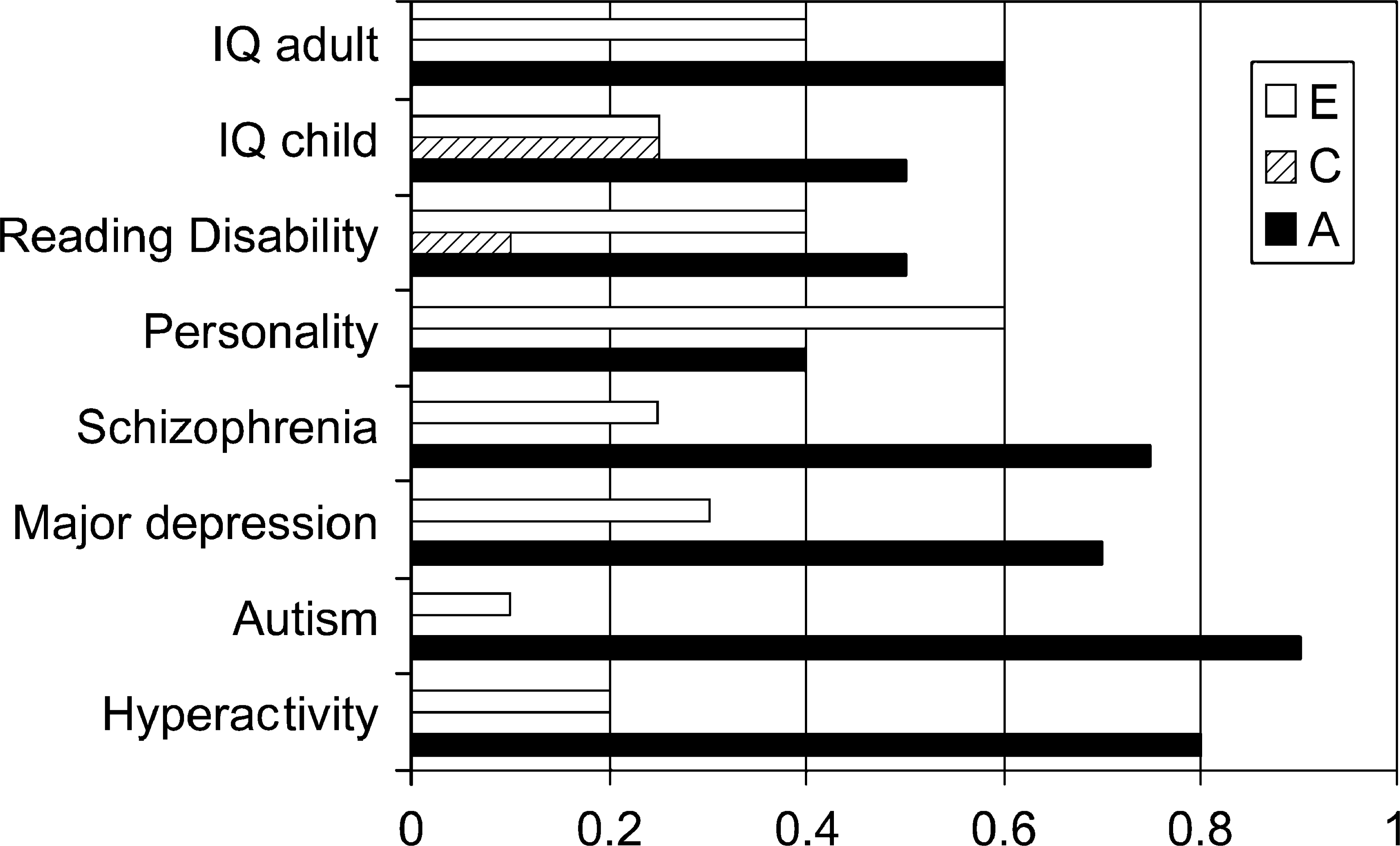
Given that quantitative studies provide consistent and convincing support for a genetic contribution to psychiatric disorders the question arises: can we go beyond merely stating that genes play a role and move on to locating and identifying genes and discovering how their effects arise? The modern approach to this question is a set of methods called positional cloning.
Positional cloning
The basic process of positional cloning is illustrated in
Fig. 2. Genetic markers evenly spaced throughout the genome (i.e. the 23 pairs of chromosomes) are studied in families containing multiple individuals affected by the disorder. Assortment of a particular marker locus with the disease, a phenomenon called linkage, indicates that the marker and a disease susceptibility locus are close together on the same chromosome. Thus linkage mapping is used to identify a chromosomal region that contains a susceptibility gene and various methods are then used to narrow down the region until the gene itself is found. Thereafter variations that confer vulnerability to the disease can be identified. The approach is most straightforward with single gene disorders that have regular Mendelian modes of inheritance. For example, in early onset familial Alzheimer's disease that shows an autosomal, dominant mode of inheritance, three genes have been identified in which mutations bring about the disorder. These are the amyloid precursor protein (APP) gene and the genes called presenilin 1 and presenilin 2 [6, 7]. Unfortunately the application of the classic linkage approach to other psychiatric disorders has been more problematic. Although early successes were claimed both in bipolar affective disorder [8] and schizophrenia [9], neither of these could be replicated.
Process of positional cloning to find a gene that confers susceptibility to a disorder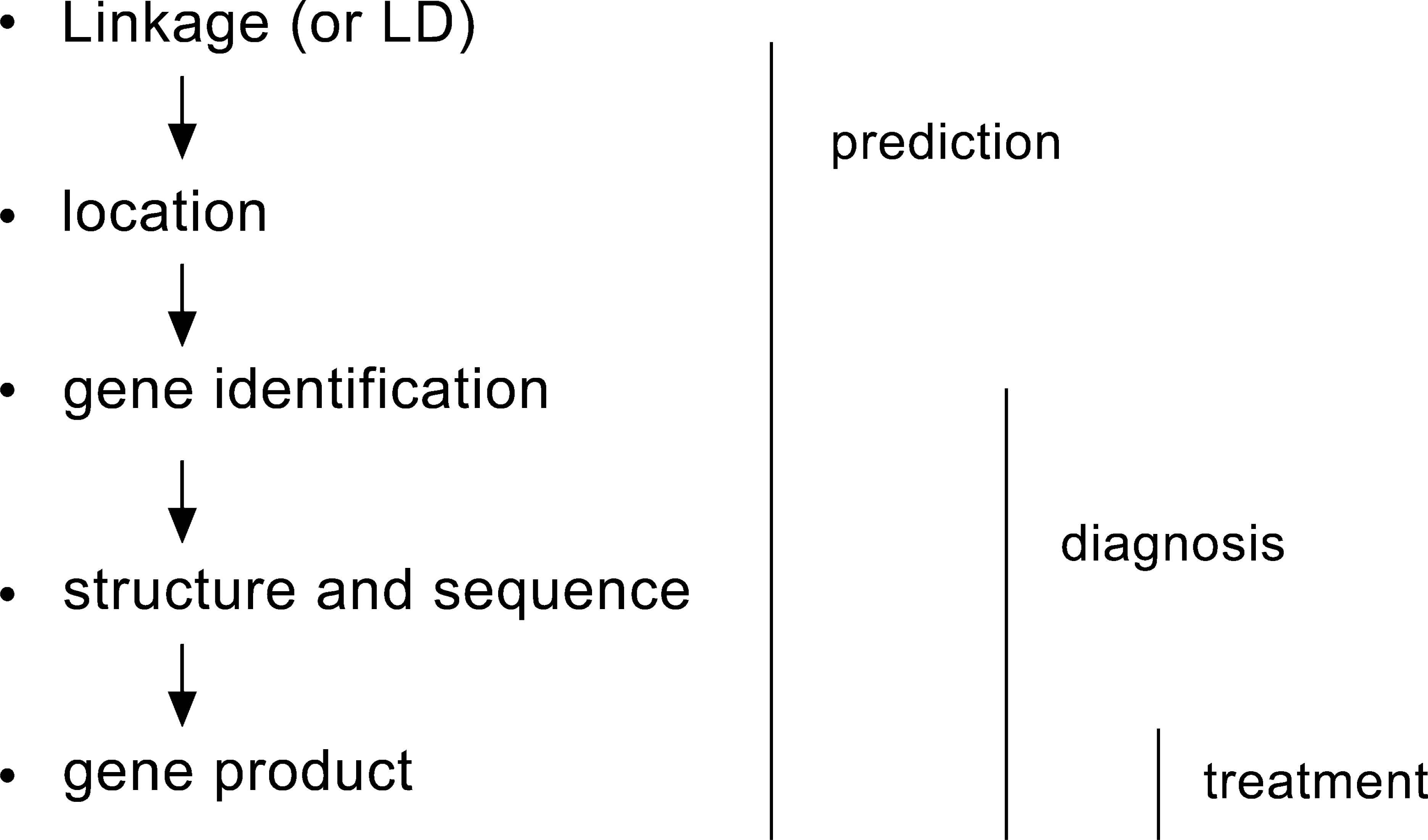
The main failing of the classic linkage approach is its assumption that highly familial forms of disorder result from genes of major effect. While this is proven to be the case for Alzheimer's disease, major gene forms of schizophrenia or bipolar disorder appear to be rare or nonexistent. Instead, the pattern of inheritance is more complex, involving multiple genes as well as multiple environmental factors. Genes in such complex systems are often called quantitative trait loci (QTLs) since they are likely to result in dimensions (quantitative continua) representing liability to disorders (qualitative dichotomies) that only manifest when a certain threshold is exceeded [10]. The goal of QTL research is to identify the genes that make contributions of varying effect size to the variance of a complex trait. Since some of the genes may make only small contributions, linkage approaches have yielded results that have appeared inconsistent and confusing. This has been particularly true in studies of bipolar disorder and schizophrenia where, as we have noted, early results that gave scope for optimism subsequently met with non-replication.
Fortunately, a clearer pattern is emerging. A metaanalysis of whole genome scans for linkage [11] found persuasive evidence of linkage regions in schizophrenia on chromosomes 8p, 13q and 22q. The chromosome 13q and 22q regions were also implicated in bipolar disorder. This may seem surprising to those brought up on the orthodox view that schizophrenia and bipolar disorder are genetically distinct but it fits well with a twin study analysis [12] suggesting a genetic overlap between schizophrenia and bipolar disorder and specific genetic factors that contribute to each of these two Kraepelinian syndromes. Other linkage data point to a region on 6p in schizophrenia [4]. The chromosome 22 region has also been implicated in studies of patients with microdeletions of this chromosome who have high rates of a schizophrenic-type psychosis [13].
Work has attempted to refine these regions using an approach called linkage disequilibrium mapping. This is done within families or by comparing cases and controls where the goal is to find a marker allele or haplotype (a set of alleles carried as a ‘block’ on the same chromosome) that differs in frequency between cases and controls. Linkage disequilibrium stretches over much smaller distances than linkage; where it is found, it indicates that the marker is either extremely close to or actually within a gene that effects susceptibility to the disorder. Four promising and novel ‘positional candidates’ [4] have been identified in this way. These are neuregulin-1 on chromosome 8p–p21, dysbindin on chromosome 6p22.3, proline dehydrogenase which maps to chromosome 22q11 and a new protein G72 located at chromosome 13q34. G72 has been shown to interact with another gene, D-amino acid oxidase (DAAO). Interestingly, the gene products of all of these candidates appear to be involved in glutamatergic transmission in the brain [14]. Evidence supports involvement of a gene which maps to a linkage region on chromosome 1q, regulator of g protein signalling-4 (RGS4). RGS4 is involved in signalling via metabatropic glutamate receptors and other g protein coupled receptors including dopamine D2 receptors. There has also been renewed interest in chromosome 22q candidate gene catechol-O-methyltransferase (COMT) because of the findings both of a variant that affects frontal lobe function [15] and a haplotype that has been implicated in a large scale case control study of schizophrenia [16].
The extent to which the chromosome 13q and 22q candidate genes may be involved in bipolar disorder is under study. Meanwhile, a gene that maps to 22q11, g protein kinate-3 (GRK3) has emerged as another promising candidate in linkage disequilibrium studies of bipolar disorder [17]. Association studies in mood disorders have also focused on candidates implicated by the mode of action of antidepressants. In particular, variants in the serotonin transporter gene or in its promotor region have been associated with mood disorders in a number of studies [18].
Promising results have also emerged in childhood psychiatric disorders, an area which until recently was almost a ‘genetic free zone’. Examples include linkage studies implicating a number of regions in childhood autism, particularly a region on chromosome 7 [19]. Regions on chromosomes 6p and 15q have been implicated both by linkage and by linkage disequilibrium studies in reading disability [20] and candidate genes such as the dopamine DRD4 receptor and the dopamine transporter (DAT1) have been associated with attention deficit hyperactivity disorder [21].
The next 50 years?
The future of psychiatric genetics looks bright. As more knowledge is gained about the genetic basis of psychopathology, progress can be made not only in an improved understanding of pathophysiology but also in the development of new and more specific drug treatments. It is also likely in the not too distant future that DNA testing will be used to predict patient responses to drugs both in terms of symptom alleviation and susceptibility to side-effects. Work in this area has already begun and there is preliminary evidence that such prediction is possible. For example, response to atypical antipsychotic drugs appears to be influenced by genotype at the gene for serotonin receptor 5-HT2a [22] and response to serotonin reuptake inhibitors is influenced by variation at the serotonin transporter gene [23].
Genetic research also has the potential to reduce the stigma associated with psychiatric disorders. As a case in point, the progress made in understanding the genetic aetiology of Alzheimer's disease has done much to improve public perception of this disorder. As more genetic evidence emerges for other psychiatric conditions like schizophrenia, perhaps these too will become more accepted in society.
The completion of the sequencing of the human genome [24] is another source of optimism for the future of psychiatric genetics. With access to detailed knowledge of the ordering of all 3 billion letters of DNA in the genome, researchers are provided with a revolutionary new tool in the search for genes in psychopathology. For example, association studies will be able to identify QTLs more readily through the use of detailed marker maps based on hundreds of thousands of singlenucleotide polymorphisms (SNPs) within known genes or their regulatory regions.
But finding genes is not the end of the story. Once they are located, researchers are then faced with the puzzle of how the genes translate into the overt behaviours and experiences of psychiatric disorders. This is the common aim of two parallel research fields: the bottom-up approach of functional genomics and the top-down approach of behavioural genomics [25]. Functional genomics focuses on the investigation of gene products, their structure and expression. Three major areas in this field have emerged in recent years: gene manipulation, gene expression profiling and proteomics [26]. Behavioural genomics focuses on the behaviour of the entire organism and examines issues such as how the effects of specific genes unfold in behavioural development and how they interact and correlate with experience. Together, these two approaches promise to make important contributions to our knowledge of gene function. It is a huge challenge and one that will no doubt keep researchers busy for many years to come.
Despite the past achievements and promising future of genetic research in psychiatry, there still remain major misunderstandings and misconceptions. For some, there is temptation to oversimplify the role of genes in mental illness, and adopt a one-gene-one-disorder viewpoint. This is reinforced by repeated media headlines announcing the discovery of the gene for various human traits and behaviours such as aggression, intelligence, criminality and even bad luck. Such exaggerated and misleading reports misrepresent the true scope of psychiatric genetics. Researchers acknowledge that genes represent risk factors, and not fated outcomes. Whether we develop a psychiatric disorder like depression or schizophrenia is not solely determined by our genetic make-up alone. We are not ‘doomed from the womb’. The most striking demonstration of this fact is that the concordance rates for schizophrenia among identical twins is not 100%. Clearly, environmental factors are of major importance and this is recognized within modern genetic research. There is increasing interest in the way genes and environment co-act or interact to increase vulnerability to psychopathology and important evidence is emerging. For example, Kendler et al. [27] found evidence favouring an interaction between genes and life events in unipolar depression. They reported that the risk of depression following a threatening event was about twice as high in subjects at high genetic risk than in those at low genetic risk.
In summary molecular psychiatric genetics is beginning to come of age and molecular medicine as a whole has come a long way since Watson and Crick's landmark discovery. With continuing advances in knowledge and technology, genetic research is a dynamic and exciting field that promises to revolutionize our understanding of psychopathology. The next 50 years should see post-genomic psychiatry coming into its own but it should not be forgotten that the translation into improved patient care ‘will depend as much on the traditional disciplines of clinical description and epidemiology and our ability to combine these with genetic, psychological and sociological approaches in the integrative science of psychiatry’ [28].
