Abstract
The enhancement of surface acidity for tungsten-doped CeTiOx was investigated through electronic structure. Tungsten doping increased the ratio of Ce3+ from 12.0% to 29.6%, which was confirmed by XPS. The doped tungsten was present as W5+ in a ratio of 71.5%. In O K-edge XAS spectra, increase in
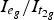 indicated the formation of oxygen vacancies through tungsten doping. The decrease in W 5d-eg peak intensity demonstrated the occupation of the W 5d orbital by electrons formed at the oxygen vacancies. The enrichment of W5+ was owing to transfer of electrons to 5d-eg orbital of tungsten rather than to cerium ions. The acid sites were analysed using FTIR and NH3-TPD. The peak intensity of Brønsted acid sites and amount of NH3 desorption increased from 66.2 to 136.1 µmol g−1 with tungsten doping. The doped tungsten formed Brønsted acid sites and led to enhanced surface acidity and catalytic activity.
indicated the formation of oxygen vacancies through tungsten doping. The decrease in W 5d-eg peak intensity demonstrated the occupation of the W 5d orbital by electrons formed at the oxygen vacancies. The enrichment of W5+ was owing to transfer of electrons to 5d-eg orbital of tungsten rather than to cerium ions. The acid sites were analysed using FTIR and NH3-TPD. The peak intensity of Brønsted acid sites and amount of NH3 desorption increased from 66.2 to 136.1 µmol g−1 with tungsten doping. The doped tungsten formed Brønsted acid sites and led to enhanced surface acidity and catalytic activity.
Get full access to this article
View all access options for this article.
