Abstract
Spectroscopic strategies that substantiate periodic oscillations in low rates of NADH oxidation exhibited by ECTO-NOX proteins at the animal and plant cell surface are described. Both continuous display and discontinuous rate determinations exhibit the oscillations but continuous displays lack sufficient resolution to discern details. A procedure is documented where rates are determined by least squares analyses of traces recorded over 1 min at intervals of 1.5 min. These traces recapitulate the continuous displays but offer an opportunity to reliably estimate changes in reaction rates over short time intervals not afforded by the continuous traces. Results from previously used rate determination over 5 min intervals are included for comparison. Turbidity is identified as the major contributor to losses in resolution. Even highly purified NOX preparations tend to aggregate to form turbid suspensions. With turbid suspensions, double beam or dual wavelength instrumentation where the sample is placed immediately adjacent to the photomultiplier tube are required to reduce losses in resolution from turbidity. Also required are high levels of synchronous ECTO-NOX function. Blue or red (plants) light, small molecules (i.e., melatonin) and autosynchrony alone or in combination were used to synchronize the oscillations. Special problems posed by preparations containing more than one ECTO-NOX form and where the different ECTO-NOX forms do not cross entrain are discussed.
Keywords
INTRODUCTION
ECTO-NOX proteins are cell surface oxidases with protein disulfide-thiol interchange activity. The constitutive (CNOX) forms of the two activities alternate to generate oscillations with very precise and characteristic period lengths of 24 min (Morré, 1998; Morré and Morré, 1998; Morré et al., 1999b; Pogue et al., 2000; Chueh et al., 2002a, b). Site directed mutagenesis analysis of a cloned cancer ECTO-NOX with a 22 min period length designated tNOX (Morré and Morré, 1998; Morré et al., 1999b; Pogue et al., 2000; Chueh et al., 2002a, b) demonstrate that NOX proteins are cellular oscillators that provide a biochemical basis for the biological clock (Morré et al., 2002a). COS cells were transfected with cDNAs encoding tNOX proteins having a period length of 22 min or with C575A or C558A cysteine to alanine replacements having period lengths of 36 or 42 min. Such transfectants exhibited 22, 36, or 40 to 42 h circadian patterns in the activity of glyceraldehyde-3-phosphate dehydrogenase, a common clock-regulated protein, in addition to the endogenous 24 h circadian period length. The fact that the expression of a single oscillatory ECTO-NOX protein determines the period length of a circadian biochemical marker (60 X the ECTO-NOX period length) provided compelling evidence that ECTO-NOX proteins are the biochemical core oscillators of the cellular biological clock.
In keeping with their proposed clock function, the period lengths of all ECTO-NOX proteins are independent of temperature (temperature compensated). Whereas NOX activities double with every 10°C rise in temperature (Q10 = 2), period length is invariant with a Q10 of 1 (Morré and Morré, 1998; Pogue et al., 2000; Wang et al., 2001; Morré et al., 2002a), a hallmark of the biological clock (Edmunds, 1988; Dunlap, 1996). Also implicit in the evaluation of any periodic or oscillatory phenomena is the need for a high level of synchrony (entrainment). ECTO-NOX proteins auto-synchronize (auto-entrain) in solution (Morré et al., 2002b). Red (plants) or blue (plant and animal) light entrains with organisms, tissue explants and cells but not with membranes or molecules (ECTO-NOX proteins lack chromophores) (Morré et al., 1999a; 2002c; Morré and Morré, 2003). Also, small molecules such as melatonin will entrain the constitutive ECTO-NOX (CNOX) with a 24 min period of both plants (unpublished) and animals (Morré and Morré, 2003). Entrainment is a second hallmark of the biological clock (Edmunds, 1988; Dunlap, 1996).
Among the natural electron donors for the oxidative activity of ECTO-NOX proteins are hydroquinones [coenzyme Q in animals (Kishi et al., 1999), vitamin Kj hydroquinone in plants (Bridge et al., 2000)]. Natural acceptors are molecular oxygen (Morré et al., 1998a) and protein disulfides (Chueh et al., 1997).
NADH or NADPH are artificial donors. Their use has been predicated entirely on convenience and the historical precedent of ECTO-NOX discovery based on NADH oxidation (Morré, 1998). Specific activities are low, in the nanomole/min/mg range, as a consequence of very small turnover numbers for NADH oxidation determined to be 200 to 500. Yet in well-synchronized NOX preparations, NADH oxidation rates oscillate between rapid and slow to create reproducible and statistically significant recurring patterns within each period. In this report, spectroscopic approaches to rate analyses are described that validate the basic oscillatory phenomenon associated with ECTO-NOX proteins. Our approaches to the measurement of low rates of NADH oxidation based on least squares slopes of spectrophotometric traces are illustrated along with statistical evaluations of the reproducibility of the oscillatory patterns.
METHODS
Spectrophotometric Assay of NADH Oxidase
NADH oxidase activity was determined as the disappearance of NADH measured at 340 nm in a reaction mixture of 25 mM Tris-Mes buffer (pH 7.2), 1 mM KCN and 150 μM NADH at 37°C and continuous stirring (Morré et al., 1995) using paired Hitachi U3210 UV-visible spectrophotometers (Hitachi Instruments, San Jose, CA) or a SLM Aminco DW-2000 spectrophotometer (Milton Roy Company, Rochester, NY) in the dual wavelength mode of operation with continuous recording and 430 nm as reference. “While these instruments are capable of recording data electronically, chart recorder output was the means of equipment use. The instruments were employed in double-beam (Hitachi U3210) or dual wavelength (SLM 2000) modes with the reference path empty. Data were recorded continuously for periods of up to 102 min over intervals of 1 or 5 min. After each 1 min measurement interval, the chart paper was returned to the starting position, the baseline was offset by a convenient increment, and recording was restarted exactly 1.5 min after the previous trace was initiated. Timing was with a digital clock. Uncertainty in restart interval was likely less than 1 second, with no cumulative timing error. The two Hitachi U3210 instruments were directly adjacent to each other and shared a common thermostating bath. Assays were initiated by addition of NADH with a starting concentration of 150 μM.
For comparison, data also were obtained using a Hewlett Packard 8452 Diode Array Spectrometer with 2 nm resolution (now Agilent Technologies, Palo Alto, CA) or with a Spectronics UV500 Spectrophotometer (Thermo Electron Scientific Instruments, Rochester, NY). For the Hewlett Packard 8452 Diode Array Instrument, data were recorded at 1 s intervals using ASCII commands transmitted through the HPIB interface with Hewlett Packard's DOS drivers. Temperature was not controlled either for the Hewlett-Packard 8452 instrument or for the Spectronics UV500 instrument.
Widi the Hitachi U3210 and SLM Aminco DW2000 spectrophotometers, temperatures were controlled by a circulating water system. The bath temperature was set to 37°C. Temperatures recorded were 35°C at the cuvette of the first Hitachi U3210 instrument, 34°C at the cuvette of the second Hitachi U3210 instrument and 31°C upon return to the water bath. “With the SLM spectrophotometers, the cuvette temperature during assay also was 35°C. Cuvettes were typically at room temperature when loaded and normally temperature equilibrated for 20 min before beginning the assays.
Statistical Analysis
Results were analyzed using fast Fourier transform and decomposition fits. Period length was determined from user-defined fast Fourier transforms in SigmaPlot 5.0 (SPSS). Reproducibility of oscillatory decomposition fits patterns were evaluated by time series (decomposition) analyses (Foster et al., 2003). The model type was additive. A trend line was first fitted to the data. The data were then smoothed by subtracting a centered moving average of length equal to the length of the period determined by Fourier analysis. Finally, the time series were decomposed into periodic and error components. The decomposition fits were used to validate the periodic oscillatory pattern and to demonstrate that minor intervening fluctuations also recurred within each period as part of a reproducible pattern. The decomposition fits used MINITAB®, a statistical package.
Data Reduction Methods
From the SLM Aminco DW2000 and Hitachi U3210 spectrometers, reaction rate was determined from the least squares linear slope of the absorbance vs. time chart data. In practice this was accomplished by measuring rise vs. run of the linear absorbance vs. time line best fitting the data. Typically, slopes were > 10 absorbance standard deviations per min for the SLM Amico DW2000 and normally >3 absorbance standard deviations per minute for the Hitachi U3210. Despite the fact that some slopes were within the noise envelope of the traces especially with the Hitachi U3210, slopes were estimated to an accuracy of ±0.015 nmoles/min compared to the actual rates recorded at 1 to 10 nmoles/min.
RESULTS
Figure 1A is a continuous trace of a well synchronized single culture of CHO cells determined over 96 min using the Aminco SLM 2000 spectrophotometer to illustrate the oscillatory activity characteristic of the ECTO-NOX enzymes from a continuous trace. Intervals of rapid activity were interspersed with intervals of lesser activity. The period length was about 24 min.

Decrease in A340 from the Aminco SLM 2000 spectrophotometer as a measure of consumption of NADH over 96 min for a well-synchronized (light) culture of CHO cells. (A) Complete reaction mixture. Periods of rapid oxidation of NADH alternate with periods of much slower NADH oxidation. (B) Reaction mixture lacking cells. (C) Reaction mixture lacking NADH.
No oscillations were observed with NADH alone in the absence of cells (Fig. 1B) or with cells alone in the absence of NADH (Fig. 1C). In the absence of cells, a slow rate of non-enzyme-catalyzed NADH oxidation was observed. In the absence of NADH, the absorbance increase was due to light scattering as turbidity increased.
Oscillations in the rate of oxidation of NADH by CHO cells illustrated in Figure 2 also with, a period length of 24 min become more obvious when analyzed over 5 min (Fig. 2A, B) or over 1 min at intervals of 1.5 min (Fig. 2C, D) using the Aminco SLM 2000 in the dual wavelength of operation with reference at 430 nm. Both measuring and reference wavelengths pass simultaneously through the same cuvette so that errors due to electronic fluctuation and turbidity changes, for example, were minimized. Panel A illustrates rates determined over 5 min as would be done for an average determination of specific activity. Since the rates were not steady state, rates from a number of such traces (usually 3 to 5) normally were averaged where determination of an average rate was a primary consideration. With 5 min traces linked front to back, the oscillatory pattern shown in B was revealed, which was similar to that observed with the continuous recording shown in Figure 1A.

Oscillatory oxidation of NADH by 106 CHO cells using the Aminco SLM 2000 in the dual wavelength mode of operation illustrating the three methods of spectroscopic analyses employed in previous studies. (A) Activity analyzed for successive intervals of 5 min each. Traces are arranged from top to bottom and from left to right (see Fig. 4D). (B) The 5 min assays aligned as a continuous trace. (C) Activity analyzed over 1 min at intervals of 1.5 min. Traces are arranged from top to bottom and from left to right (see Fig. 4D). (D) Slopes calculated from C. Assays were for 70 min. The period length was 24 min (arrows). HeLa cells contain a background activity approximately equal to that of the oscillatory activity that is proteinase K susceptible and does not oscillate (Sun et al., 2000).
For more detailed evaluations, we have used determinations of rates over one min at intervals of 1.5 min, as shown in Figure 2C. The measured slopes when represented graphically in Figure 2D illustrate maxima separated by intervals of 24 min. With the SLM 2000, the width of the trace was such that when slopes were estimated to the nearest 0.1 mm, measurement errors were ±0.015 nmole/min and measurement errors greater than ±1 mm (±0.05 nanomoles/min) would have been virtually impossible to introduce. These assays all were for 70 min. HeLa cells contained a background activity as seen in Figure 2D approximately equal to that of the oscillatory activity. The background activity was susceptible to digestion with proteinase K and did not oscillate (Morré and Morré, 2000). The oscillatory NOX activities are resistant to protease digestion, including digestion with proteinase K (del Castillo-Olivares et al., 1998).
A double peak oscillatory pattern is typically observed with plasma membranes from stem sections of dark-grown soybean seedlings and analyzed using the SLM 2000 (Fig. 3). The plasma membranes were solubilized with 0.5% Triton X-100 to minimize turbidity. Detergent treatment, however, introduced a non-oscillatory background NADH oxidase activity nearly equal to the oscillatory NADH activity. The background activity introduced by detergent treatment derives from the inner surface of the solubilized plasma membrane vesicles (Morré and Morré, 2000) and was susceptible to digestion with proteinase K. These preparations were well synchronized by light treatment of the soybeans prior to harvest of the plant material and show two maxima in NADH oxidase activity with the second maximum (double arrow) following the first (single arrow) by about 9 min (Fig. 3B).

Oxidation of NADH by a well-synchronized preparation of soybean plasma membranes solubilized in Triton X-100 to reduce turbidity. The Aminco SLM 2000 in the dual wavelength mode of operation was used. (A) Continuous trace over 108 min. (B) Activity analyzed over 90 min from slopes determined over 1 min at 1.5 min intervals as illustrated for Figures 2C and 2D. Since the vesicles were solubilized, a major contribution to background comes from a NADH oxidase of the internal plasma membrane surface that does not oscillate (Morré and Morré, 2000).
Much of the previous work, especially with plants, compared measurements using two side-by-side Hitachi U3210 dual beam spectrophotometers with air as reference. The most challenging measurements were with turbid untreated preparations of plasma membrane vesicles (initial absorbances between 1 and 2). Continuous traces exhibited more noise than those with the SLM 2000. Figures 4A and 4B compare a continuous trace of a soybean plasma membrane with a starting absorbance of 1.4 generated in one of the two machines with data obtained from analyses of individual rates determined over 1 min at 1.5 min intervals in B as illustrated in E. A regular pattern of rapid rates alternating with very slow rates were observed with both data sets. These intact plasma membranes have a very low background rate and the rate of NADH disappearance in between the maxima actually reached zero (Fig. 4B). Panel C illustrates a series of 18 5-min traces assembled end to end for the same plasma membrane preparation assayed in one of the two machines subsequent to the data of A and B but timed to be in phase. The individual traces are shown in D. The sequence with which the traces were acquired is given by the letters a — r. The same sequence was followed for Figures 2A, 2C and 4E (top to bottom, left to right). These types of traces are those most commonly used to measure effects of inhibitors and other parameters affecting NOX activities. Examples of sequential 5 min assay segments where activity in m and f exhibit little or no activity are encountered as well. A minimum also was encountered at the end of i and the beginning of j, which would be detected with 1 min traces as a rate near zero. Figure 4E gives examples of sequential 1 min traces measured at intervals of 1.5 min as for Figure 4B and Figure 4E Despite the width of the traces, slopes even with these relatively turbid samples were determined with an accuracy and reproducibility sufficient to reliably reveal details of the oscillatory pattern (Fig. 4B). A further illustration is provided in Figures 4F and 4G where the two instruments again were used in parallel. Figure 4F shows the results of the 1 min analyses at intervals of 1.5 min, whereas in G the same preparation was analyzed based on 18 consecutive 5-min scans as in C and D, except that the rates were determined sequentially without interruption. Both methods revealed a similar oscillatory pattern of oscillations.

Oscillatory oxidation of NADH by a crude preparation of isolated, intact plasma membrane vesicles from soybean measured using paired Hitachi U3210 dual beam spectrophotometers with air as reference. The solutions were very turbid (initial absorbance of 1.4) and represent a worst case scenario at the limits of the capability of the instrument. (A) Continuous trace over 90 min with alternating rapid and slow rates of NADH oxidation. (B) An identical aliquot of the same preparation as in A but analyzed in parallel using the second Hitachi instrument based on rates determined over 1 min at 1.5 min intervals. (C) An assembly of 18 assays of NADH oxidation each over 5 min determined on the same preparation sequentially but in phase with A and B. (D) Examples of 5 min traces used in the generation of C. These preparations, largely lacking background activity and relatively well synchronized by light, exhibit periods where activity slows or stops between periods of rapid activity. The period length is 24 min. The 5 min assay segments labeled f and m exhibit little or no activity. A minimum is also encountered at the end of i and the beginning of j. (E) Examples of rates determined over 1 min at intervals of 1.5 min. (F) Analyses of E. (G) Sequential 5 min traces as in D except determined sequentially without interruption. All assays were for 90 min.
To illustrate the reproducibility of the pattern of oscillations, decomposition fits for two full periods of each of the experiments of Figures 1–4 are summarized in Figure 5 along with three measures of the accuracy of the statistically fitted values. The MSD (mean standard deviation) comparing all four data units analyzed averaged 26%. Differences between maxima and minima were highly significance (p < 0.001). Also evident from the decomposition fits were the double peak pattern of Figure 3B (Fig. 5B) and of Figure 4F (Fig. 5D) compared to the single peak patterns of Figure 2B (Fig. 5A) and Figure 4B (Fig. 5C).

Decomposition (time series) analyses of the oscillatory patterns of NADH oxidation of Figures 1 to 5. (A) Decomposition fit of data of Figure 2D. The data consist of a pattern of repeating oscillations, all conforming to a 24-min period length (arrows). (B) Decomposition fits (open symbols, dashed lines) for two full cycles of data of Figure 3B. (C) Decomposition fits for data of Figure 4B. (D) Decomposition fits for data of Figure 4F. Period lengths were determined by Fourier analysis. The decomposition fits show the reproducibility of the patterns of oscillations. Three measures of the accuracy of the statistically fitted values are provided in the insets.
With assays of A340 from the HP8452A diode array spectrophotometer, the variation in rates mostly fell within the envelope of machine variation, even with CHO cells treated with Triton X-100 to reduce light scattering (Fig. 6). However, periods of enhanced activity separated by periods of reduced activity similar to that of Figure 1, although incompletely resolved, were discernable. In B — D, three preparations of CHO membranes were analyzed on 3 consecutive days. The preparations were synchronized at the beginning of each experiment by the addition of 1 μM melatonin (Morré and Morré, 2003) to phase the period. Following adjustments in rates due to the melatonin addition, a steady state was reached in which alternating rapid and slow rates (arrows) were marginally resolved in the same general location in each of the three traces. The preparation of Figure 6D was treated with Triton X-100 and centrifuged to further reduce turbidity.
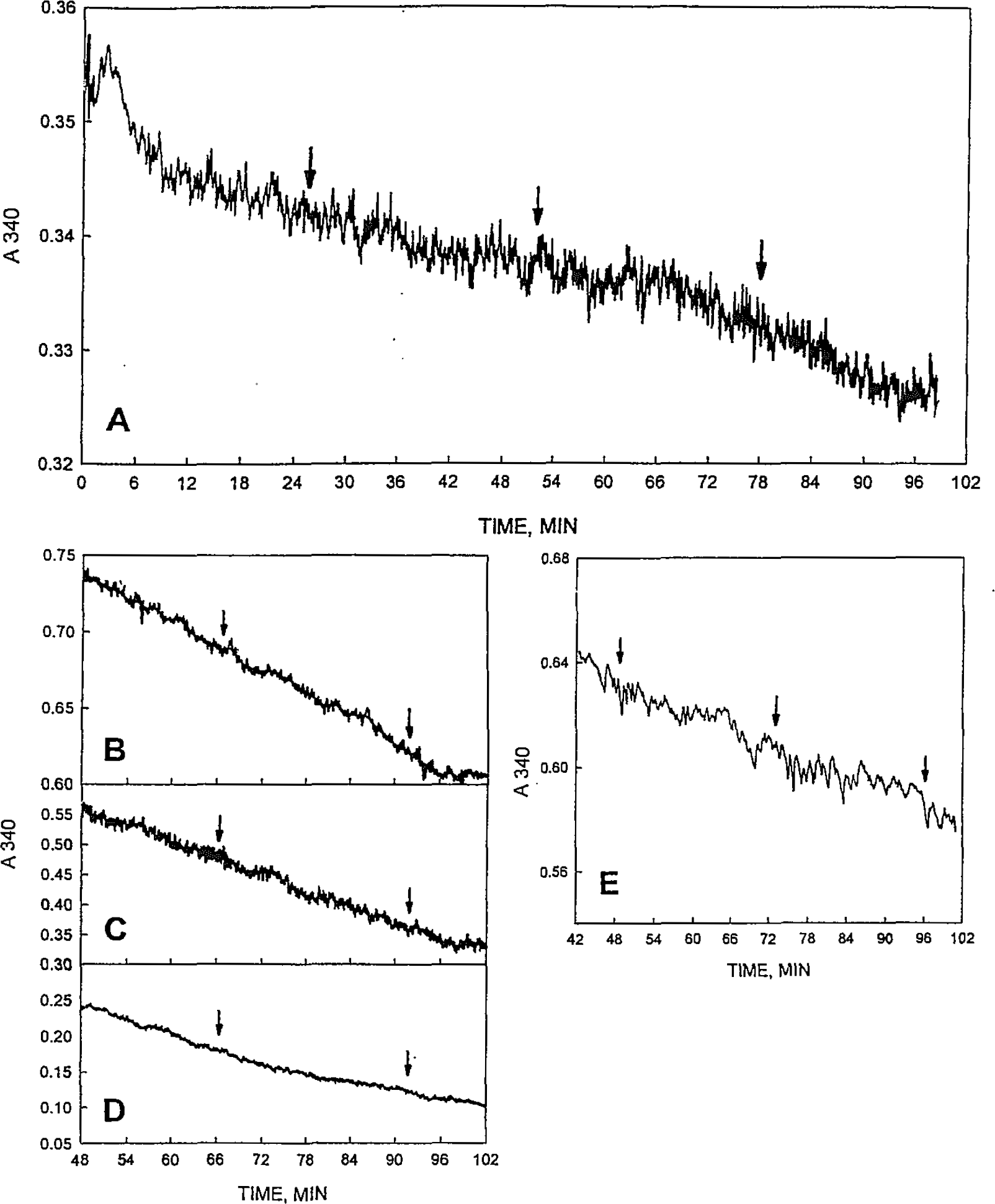
Assays of A340 from the HP8452A diode array spectrophotometer observing consumption of NADH. Arrows are spaced at 24 min to coincide with at least one interval where rapid rates are separated by a slow rate. (A) CHO microsomes dissolved in 0.5% Triton X-100 to reduce turbidity. (B-D) Three different preparations of CHO membranes in which synchrony was induced by addition of 1 μM melatonin at t = 0. D was with 5% Triton X-100 added followed by centrifugation to remove floating lipids. A double peak with a 24 min period similar to that of Figure 3 was seen at the arrows in all three repetitions on subsequent days with different preparations. (E) A skunk cabbage spadix plasma membrane preparation comparable to the soybean plasma membrane preparation of Figure 3A.
The only plant preparation analyzed using the HP8452A diode array instrument was a plasma membrane preparation from skunk cabbage (Symplocarpus foetidus) spadices (Fig. 6E). Despite an initial absorbance 0.4 that of the soybean preparations of Figures 3 and 4, alternating rapid and slow rates of NADH oxidation were not clearly resolved above the scatter in the absorbance recordings (arrows separated by 24 min coincide with rapid rates of NADH oxidation). Thus instrument variation largely precluded the use of the single beam diode array instrument for analyses of NOX periodicity from continuous traces with turbid samples. Line segment analyses were not attempted with data from this instrument.
Results, although not equivalent to the SLM Aminco DW-2000, were better when obtained using the commercially available Spectronics UV500 (Thermospectronics, Rochester, NY) (Fig. 7). This diode array dual beam instrument with an end on photomultiplier did resolve periods of rapid and slow oxidation of NADH separated by intervals of 24 min with preparations synchronized by melatonin addition. The samples were unstirred, at room temperature and without temperature control.

Assays of A340 for the Spectronics UV 500 spectrophotometer observing consumption of NADH. (A) Continuous trace over 105 min of a preparation containing 106 intact CHO cells where synchrony was achieved by addition of 1 μM melatonin at t — 12 min. Arrows spaced at 24 min coincide with intervals where rapid rates of NADH oxidation were observed. (B) As in A except for a preparation of plasma membranes of dark grown soybean hypocolyls (150 mg total protein). Melatonin (1 μM.) was added at t = −12 min. (C) Data of B displayed as successive 1.5 min segments. (D) Analysis of C giving rates determined by least squares analyses of the 1.5 min segments shown in C.
The traces obtained using the Spectronics UV500 were marred by random discontinuities or spikes often in clusters (Figs. 7A, B). These were especially evident in the data of Figure 7C comparing individual 1.5 min traces comparable to Figure 2C. Despite these discontinuities, measurements of best fit slopes did reveal the characteristic NOX pattern of oscillations obtained with the SLM Aminco DW-2000 and the Hitachi instruments (Fig. 7D).
Also providing useful information were the absorbance values recorded at 10 sec intervals using the Beckman DB200 spectrophotometer (Beckman Instruments, Palo Alto, CA). These data were analyzed to determine best fit slopes at intervals of 1.5 min. Oxidation rates were calculated from the formula N — (N + 1)/2 for each 1.5 min interval to generate data comparable to that of Figure 4F (not shown).
DISCUSSION
In a series of papers, we have demonstrated that the constitutive ECTO-NOX activity, CNOX, supports oscillatory behavior with a period length of 24 min (Morré, 1998; Morré and Morré, 1998; Pogue et al., 2000; Wang et al., 2001; Chueh et al., 2002a,b; Morré et al., 2002a–c). The focus of this paper has been to examine critically the spectrophotometric methods used to investigate these oscillatory reactions of whole cells and plasma membranes. ECTO-NOX activities can be measured conveniently with whole cells as NADH is an impermeant substrate.
It is important that all reactants were equilibrated at the spectrophotometers' temperature prior to measurement of the oscillatory patterns. During temperature equilibration, sample warming contributed significantly to absorbance values. Near 34°C, the mean temperature used, a 1 degree change in temperature results in a density change of 0.03% (Lide, 1992). For turbid samples (A ≡ 1), this corresponds to an absorbance change of 3 × 10−4.
For NOX assays, a grating instrument with an end-on window photomultiplier tube along with the double beam stability offered by this type of instrument is preferred. For example, the smooth signal generated by the Aminco SLM 2000 offers an unequivoval demonstration of oscillatory behavior with continuous tracings and an opportunity for unambiguous fitting of lines slopes to discontinuous traces.
The continuous tracings provided by the Aminco SLM DW2000 and Hitachi U3210 recording spectrophotometers have been analyzed variously to elucidate details of the oscillatory patterns. One method developed over several years of experimentation is to estimate rates from line slopes fitted to continuous traces obtained over 1 min at 1.5 min intervals. The 0.5 min intervals between measurements are sufficient to allow for chart repositioning and instrument restart. The rise over run ratio allows for accurate measurement of slopes determined over one min with a precision of ±0.015 nmoles, approximately equivalent to instrument and/or thermal variation (variation in trace width) for the Aminco SLM DW2000. The actual rates estimated were in the range of 1 to 10 nmoles/min, 50 to 500 times the precision with which the line slopes were determined. This ratio represents the power of the line slope method of minimizing instrument or thermal variation as a significant source of measurement error. Maxima and minima are readily discerned and data are obtained that permit statistical evaluations not possible from continuous traces. For example, the maxima in NADH oxidation of well synchronized preparations frequently may be resolved into two maxima rather than a single maximum. In addition, minor oscillations coinciding with maxima in protein disulfide thiol interchange also maybe discerned (Sun et al., 2000).
From the rate data, period lengths were verified by fast Fourier analysis. The method we have adapted for statistical validation of the reproducibility of rate data is time series analysis (Foster et al., 2003) where decomposition fits compare successive oscillatory patterns of period length determined by Fourier analysis. The decomposition fits served to evaluate the reproducibility of the oscillatory patterns by generating a predicted or forecasted oscillatory pattern and by comparison to the predicted pattern yielded mean average percentage error (MAPE), a measure of the periodic oscillation, mean average deviation (MAD), a measure of the absolute average deviations from the fitted values, and mean standard deviation (MSD), the measure of standard deviation from the fitted values.
Implicit is the need for a high degree of synchrony in order to visualize periodic oscillations in NOX activity. The simple proof of this statement rests in the observation that unsynchronized NOX preparations as a population do not generate discernable oscillations with a regular period. The same preparations, however, as a population will generate oscillations once synchronized (Morré et al., 2002b,c) under identical chemical and thermal conditions as for the unsynchronized preparations.
Unlike chemical oscillators, ECTO-NOX oscillations are inherent in the protein itself and not dependent on the chemical environment or temperature per se (period length is independent of temperature). These factors primarily influence absolute rates and, to a much lesser degree, resolution. The greatest contributors to a lack of resolution are turbidity and particle settling. Stirring is essential to eliminate the latter. Turbidity is difficult or impossible to eliminate in ECTO-NOX preparations. Improved resolution with the SLM DW2000 and the Spectronics UV500 instruments results as turbidity influences are minimized by the end on photomultipliers. Double beam dual wavelength with A340 subtraction from the same cuvette to reduce the turbidity component is an aid with very turbid samples but most of the data generated here were with a single wavelength. A reference in the reference cell lacking NADH, for example, only exacerbates the turbidity problem so that the routine reference of choice is air. Turbidity may arise from vesicles (membrane preparations), lipid micelles (disrupted vesicles) and protein aggregates (solubilized and purified ECTO-NOX proteins). Purified ECTO-NOX proteins tend to aggregate to form insoluble (and inactive) amyloid rods under most standard conditions of assay (del Castillo-Olivares et al., 1998; Kelker et al., 2001). Addition of detergent is of no direct benefit and may actually aggravate the situation since detergents enhance aggregation of NOX proteins (Morré et al., 1998b).
Diode array instruments are excellent for scanning. Full spectrum incident light is selectively absorbed by the sample and transmitted. However, for situations other than for clear aqueous samples of 1 cm path length, their utility is limited. With NOX proteins it is rarely possible to provide a clear sample. With a turbid sample, polychromatic light is scattered. The transmitted light is no longer focused and may not even reach the detector. Within a conventional diode array instrument, the sample is placed too far from the detector to avoid serious interference from light scatter.
Thus, with the Hewlett Packard 8452 single beam diode array spectrophotometer, variations due to light scatter, even in well-synchronized preparations, were sufficient to prevent observation of statistically significant deviations in reaction rates of the continuous traces that would distinguish them from single experiential or linear decay. When the diode array data were numerically differentiated using the Savitsky-Golay algorithm presuming local linearity and with averaging times ranging from 11 sec to 121 sec (Savitsky and Golay, 1964), no departures from linearity could be discerned above the variations due to light scatter. Unfortunately the system was not thermostated, so that convective fluctuations may have contributed as well to the activity fluctuations. Sound statistical practice requires that one regress the long-term trend and then determine if the residuals still contain significant information in excess of random noise. This has been done, for example, with data from the Beckman DB200 where individual data points collected at intervals of 10 sec over 1.5 min intervals were subjected routinely to least square analyses.
Unfortunately, spectrophotometers of the Aminco SLM 2000 design are no longer available commercially. The only instrument we were able to identify with an end-on photomultiplier and dual beam stability was the Spectronics UV 500 (Thermo Electron Scientific Instruments, Rochester, NY). This instrument, appropriately modified to achieve both stirring and temperature control, should fulfill the basic requirements for either continuous or discontinuous measurements of ECTO-NOX activities. An alternative for discontinuous measurement with non-turbid samples of low specific activity would be to average absorbance values recorded at 10 sec intervals over 1.5 min and determine rates by numerical averaging (subtracting the values at N + 1 from those at N).
Synchrony of ECTO-NOX (CNOX) preparations may be achieved using red (plants) or blue (plants and animals) light (Morré et al., 1999a; 2002c) or a small molecule such as melatonin (Morré and Morré, 2003; Figs. 6 and 7). In addition, ECTO-NOX proteins autosynchronize (autoentrain). Two CNOX preparations out of phase with each other, when mixed, initially generate a complex pattern which is the summation of the two patterns at the time of mixing (Morré et al., 2002b). However, after 24 h, even at reduced temperatures, the two preparations function synchronously with a single 24 h period.
The possibility that the oscillatory behavior of the NOX protein is a manifestation of a classic Belousov-Zhabotinsky (BZ) reaction observed far from equilibrum is considered unlikely since frozen preparations of NOX proteins continue to oscillate for several years with no change in period length or amplitude. BZ oscillations are chemical reactions, highly dependent on amounts and ratios of reactants and on temperature (Scott, 1994). In contrast to systems exhibiting BZ oscillation, the NOX oscillations occur over a wide range of substrate concentrations, pH and temperatures. Unlike BZ oscillations, the alteration of two activities, NADH hydroquinone oxidation and protein disulfide-thiol interchange, is the basis for the periodic behavior (Morré, 1998). Temperature independence of period length (Morré and Morré, 1998; Pogue et al., 2000; Wang et al., 2001; Morré et al., 2002b) is unknown for BZ oscillations.
The structural basis for the oscillations appears to be local alternations in β-structure and α-helix as determined by Fourier transform infrared spectroscopy or circular dichroism (Morré and Morré, 2003). These regular periodic changes in protein secondary structure do not require the presence of substrate and seem to serve as an underlying basis for the recurring series of conformational changes.
Two characteristics of the NOX protein, a period length independent of temperature (Morré and Morré, 1998; Pogue et al., 2000; Wang et al., 2001) and entrainment (the ability to respond to environmental signals such as light and melatonin to phase the oscillations) (Morré et al., 1999a; 2002b,c; Morré and Morré, 2003), clearly set the NOX oscillations apart from other periodic phenomena and fulfill two of the more important criteria for NOX proteins to be the biochemical basis of the biological clock (Morré et al., 2002a). The relationship between the NOX period and the circadian day length (60 X the NOX period) has been shown by site-directed mutagenesis (Chueh et al., 2002b; Morré et al., 2002a). A relationship between the regular oscillations of activity of the NOX proteins and the circadian clock was shown in COS-1 cells (SV-40-transformed African monkey kidney cell line) stably transfected with NOX proteins having period lengths longer or shorter than 24 min. A truncated form of tumor-associated NOX (ttNOX) with a period length of 22 min, a C575A replacement of ttNOX with a period length of 36 min, or a C558A replacement of ttNOX with a period length of 42 min generated circadian days of 22, 36 or 42 h respectively (Morré et al., 2002a). As an endogenous biochemical marker to monitor the circadian rhythm, the activity of glyceraldehyde-3-phosphate dehydrogenase (GAPDH), a common clock-regulated house-keeping protein, was used.
