Abstract
Hot-dipped galvanised (HDG) steel and Zn5Al-coated steel were exposed for 6 years at different atmospheric weathering sites worldwide. The mass loss of the coatings has been measured after 1, 2, 4 and 6 years of exposure. In addition, corrosion products have been determined using scanning electron microscopy, X-ray diffraction and FTIR spectroscopy. It is shown that Zn5Al is better than HDG-steel independently on the exposure duration and location. The mass loss of Zn5Al is also less dependent of the environmental parameters (such as chloride deposition, relative humidity and temperature) than HDG-steel. The ratio of performance after 6 years of exposure varied from 1.3 to 5.1 with the mean value of 3.1. The analyses of the corrosion products indicate the formation of protective Zn/Al - 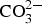 layered double hydroxide (LDH) and sulphate-containing corrosion products of Zn and Al after 6 years of exposure. A mechanism explaining the superior properties of Zn5Al is proposed.
layered double hydroxide (LDH) and sulphate-containing corrosion products of Zn and Al after 6 years of exposure. A mechanism explaining the superior properties of Zn5Al is proposed.
Introduction
Steel surfaces coated with thin continuously applied hot-dipped zinc coatings or zinc alloy coatings provide superior corrosion properties compared to uncoated steel surfaces. The zinc coating works as an efficient barrier which protects the steel surface. It also provides galvanic protection at defects in the coating or where the steel substrate is uncoated, such as at cut-edges or at defects extending through the zinc coating. Furthermore, the corrosion of the zinc coating in the atmosphere will result in the formation of compact corrosion products [1–4] which adhere to the steel surface and will act as a barrier protecting the substrate from corrosion [5,6]. Both for bulk zinc material and zinc-coated steel, the atmospheric corrosion often leads to localised corrosion attacks of the zinc [2 4]. Corrosion attacks which may penetrate locally through a 20 µm hot-dip galvanised steel (HDG) coating down to the steel surface have been observed already after ½ year exposure in a corrosive marine environment [4].
The corrosion rate of the zinc coating can be largely reduced by alloying with aluminium or aluminium and magnesium. Zinc coatings alloyed with 5 wt-% aluminium and 0.05% mischmetal (lanthanium and cerium) e.g. Zn5Al with commercial name Galfan™ with improved corrosion properties were introduced for nearly 30 years ago. It was shown that Zn5Al had significantly lower corrosion rates compared to HDG in severe marine and industrial sites after 5 years of exposure [7]. In addition, at least two times, lower corrosion rates were observed in cyclic modified salt spray tests [8]. After 10 years outdoor exposure tests in marine, industrial and rural sites, Zn5Al alloy-coated sheet steel presented at least twice better corrosion resistance compared to HDG [9]. However, a ratio of improvement 3-4 was observed after exposure in a severe marine atmosphere after 7 years [10]
In contrast to hot-dipped galvanised steel with low Al content, Zn5Al coating has a multiphase structure, e.g. a two-phase structure with a zinc-rich proeutectoid phase surrounded by a eutectic phase (≈5 wt-%) consisting of β-aluminium (5-25 wt-% Al) and η-zinc lamellas (<5 wt-% Al) [11,12]. Studies of the initial corrosion stages of corrosion of Zn5Al indicate that the eutectic phases are preferentially attacked while the zinc-rich proeutectoid phase was more resistant [8]. Zhang et al. [13] studied the initial atmospheric corrosion of Zn5Al coating in detail and found that the corrosion product formation starts at the η-Zn phase adjacent to the β-Al phase. The propagation of the corrosion of Zn5Al coating in a marine atmosphere resulted in the formation of zinc and zinc/aluminium corrosion products such as Zn6Al2 (OH)16CO3·4H2O, Zn5Cl2(OH)8·H2O (Simonkolleite) /Zn2Al(OH)6Cl·H2O (Layered double hydroxide (LDH)) and NaZn4SO4(OH)6Cl·6H2O (Gordaite).
Elvins et al. [12] reported that Zn5Al coatings with identical compositions but different microstructures can show different corrosion behaviour in NaCl solution. Other studies have indicated a ratio of improvement in the range 1.7-3.5 over HDG for a Zn5Al coating with a coarser microstructure while improvement was better (2.8-3.8) for a fine microstructure [14].
However, despite the technological importance, there is a lack of atmospheric corrosion data from field exposures published for Zn5Al coatings. The corrosion data available in the literature are from 2 to 3 decades ago and data are lacking from a wider range of environments and from different climatic regions. Furthermore, there is no complete understanding of the corrosion mechanisms under atmospheric weathering conditions for Zn5Al-coated steel.
The objectives of this study were to investigate the long-term corrosion behaviour of Zn5Al and hot-dip galvanised steel during exposure in a wide range of climatic conditions worldwide and to study the nature of the corrosion attack and the formation of corrosion products after different times of weathering under atmospheric conditions.
Experimental
Materials
Metallic coatings.

Reference carbon steel (e.g. DC01, EN 1.0330) and pure zinc (Z1 grade) were exposed for 1 year at each field site to determine the corrosivity class according to ISO 9223 [15].
Exposure sites
Locations and characteristics of exposure sites.
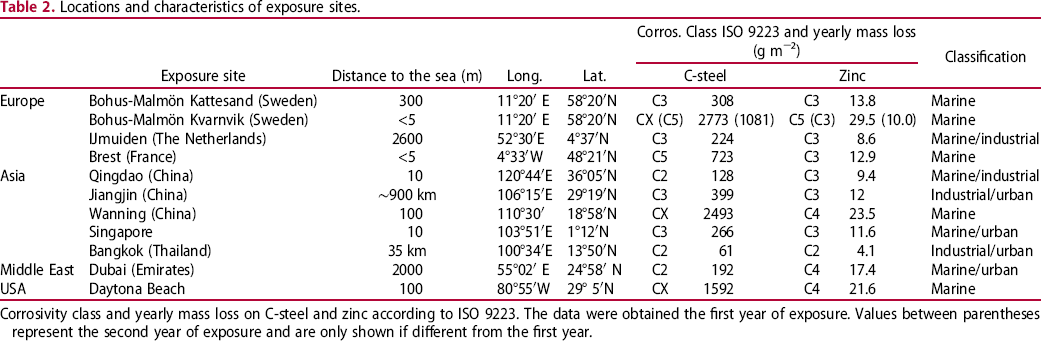
Corrosivity class and yearly mass loss on C-steel and zinc according to ISO 9223. The data were obtained the first year of exposure. Values between parentheses represent the second year of exposure and are only shown if different from the first year.
Mean temperature, relative humidity, chloride deposition and SO2 concentration at different field sites.
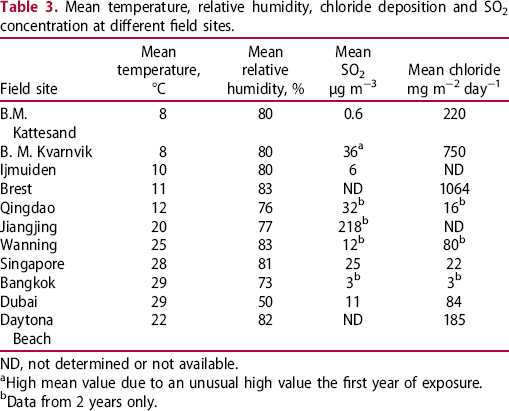
ND, not determined or not available.
aHigh mean value due to an unusual high value the first year of exposure.
bData from 2 years only.
For all sites, samples have been exposed at the inclination of 45° to the horizontal and facing south or the corroding source (sea). Samples were withdrawn after 1, 2, 4 and 6 years of exposure.
Weight loss measurements
Three replicates were collected for each material after 1, 2, 4 and 6 years of exposure. Before pickling, the samples were brushed in order to remove non-adherent corrosion products and rinsed in flowing water. The corrosion products were removed by pickling in a saturated glycine (NH2CH2COOH) solution according to ISO 8407:2009 [18]. The pickling was performed by repeating the cleaning step to obtain dissolution curves which could be extrapolated to obtain the mass loss following the ISO 8407 standard.
SEM–EDS investigations
FEG–SEM investigations were performed using a LEO 1530 with Gemini column, upgraded to a Zeiss Supra 55. An EDS detector, 50 mm2 X-Max Silicon Drift Detector (SDD) from Oxford Instruments was used for the chemical analysis. Cross-sections of the samples were prepared by mounting the samples in two components epoxy resin and polishing the samples in ethanol.
Analysis of corrosion products
Analysis of corrosion products on the metal surfaces was performed using Infrared reflection absorption spectra (IRRAS) using a Bruker Vertex 70 spectrometer with Harrick's Seagull multi-reflection accessory, with p-polarised light (KRS-5 wire grid polariser) and an angle of incidence of 10°. A Bruker FM beamsplitter was used for measurements in the region 200-4000 cm−1 using a resolution of 8 cm−1. An Au-mirror was used as background for the FTIR-measurements.
FTIR imaging was made with the Bruker Vertex 70 spectrometer and a Hyperion 3000 microscope equipped with 64 × 64 focal plane array detector (FPA). Reflection measurements were performed using a 15× objective. The measurements were performed averaging 500 scans at a resolution of 8 cm−1 and making a ratio of the single beam spectra against a background spectrum of a gold mirror.
A Bruker AXS D8 Discover with a Cu-anode a SolX detector with energy window for Cu Kα χσΙνσΣταρτ and a Göbel mirror on the detector side was used for X-ray diffraction (XRD) analysis. Measurements were performed at 5° angle of incidence.
Results
Mass loss measurements
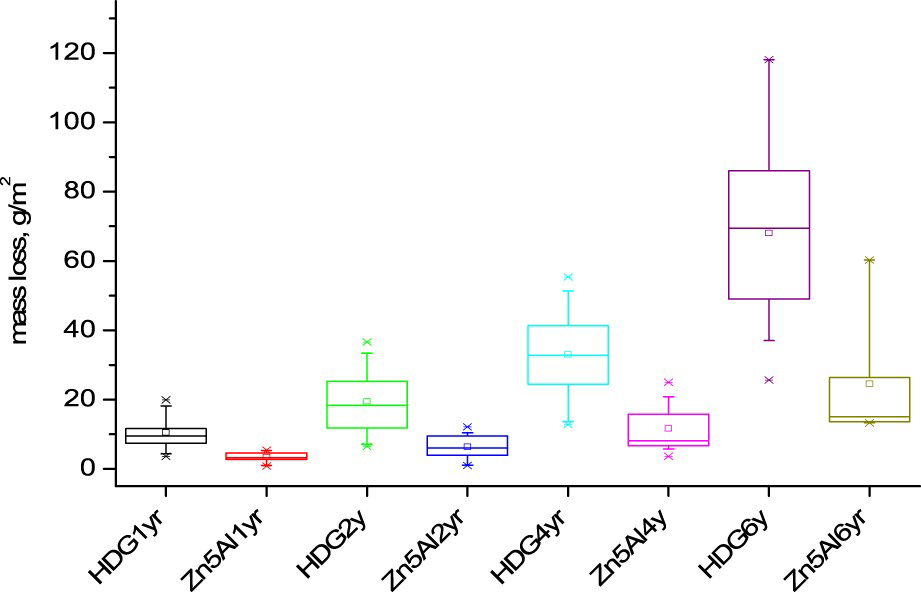
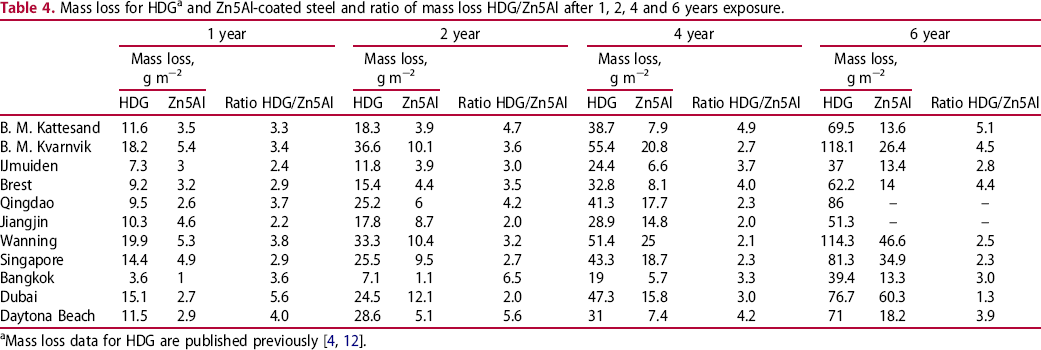
The mass loss of Zn5Al was consistently lower for all exposure sites and exposure times compared to HDG, see Table 4. Figure 1 shows that the ratio of the mass loss of HDG and Zn5Al decreased from about 3-6 to about 2-4 when increasing the exposure time. The important decrease in the ratio measured at Dubai between year 1 and 2 and between year 4 and 6 may be related to important sand storms occurring during these periods. Indeed, it is possible that this will favour some erosion of the coating leading to a contribution to the overall mass loss. However, the mean value of the ratio between the mass losses of HDG and Zn5Alfor all sites was ∼3 after 4 and 6 years and in the range of 3.6/3.9 after 1 and 2 years. It should be noticed that rather similar values were also found when considering only marine corrosion sites. The variation in the mass loss data is illustrated by the box plots in Figure 2 in which the size of the boxes is larger for HDG for all exposure times, indicating that HDG is more sensitive to the exposure conditions compared to Zn5Al which shows less variations between different exposure sites. Actually, the median value of the data for Zn5Al coating shows that the mass loss values for these samples are clustered close to the lower limit of the box. The median value for the HDG material is in the centre of the box showing that the mass loss values are evenly spread for the different exposure sites.
Ratio of the mass loss for HDG versus Zn5Al after 1, 2, 4 and 6 years of exposure for each site as well as the mean ratio for all sites and the pure marine sites separately. Box-chart plots of the mass loss for HDG and Zn5Al-coated steel materials after 1, 2, 4 and 6 years of exposure. All data are included as individual results for all field stations. Mass loss for HDGa and Zn5Al-coated steel and ratio of mass loss HDG/Zn5Al after 1, 2, 4 and 6 years exposure.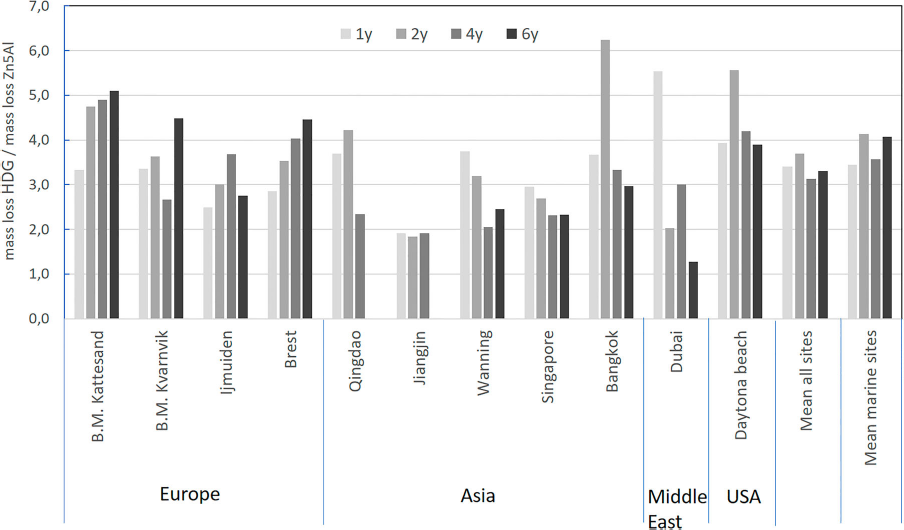
The variation of the data increased with time for both materials, but this was more pronounced for HDG reflecting the higher sensitivity to the exposure conditions for this coating. The mass loss versus time of exposure for some selected sites is shown in Figure 3. The corrosion is quite linear with time for both materials, but there is a tendency for an increase in the corrosion rate between 4- and 6-year exposures for HDG, especially at the corrosive site of Wanning. This could be associated with the consumption of a large part of the metallic zinc coating after 4 years exposure. At shorter exposure times, the corrosion rate is determined by the corrosion of the zinc layer itself, provided there are no defects through the zinc layer. After prolonged exposure, the zinc layer is partly consumed down to the steel surface [4]. The steel surface is covered by zinc corrosion products in these areas, but the presence of defects and cracks in the corrosion product layers makes it possible that the cathodic reactions can take place at the steel surface which could increase the corrosion rate of the remaining zinc layer on the samples. As seen from the SEM–EDS investigations below, large areas of the HDG coating were consumed after 6 years of exposure. For Zn5Al coating, the corrosion also resulted in the penetration of the coating layer after 6 years, but a larger part of the metallic coating was remaining compared to HDG.
Mass loss for HDG and Zn5Al and coated steel versus time for selected exposure sites.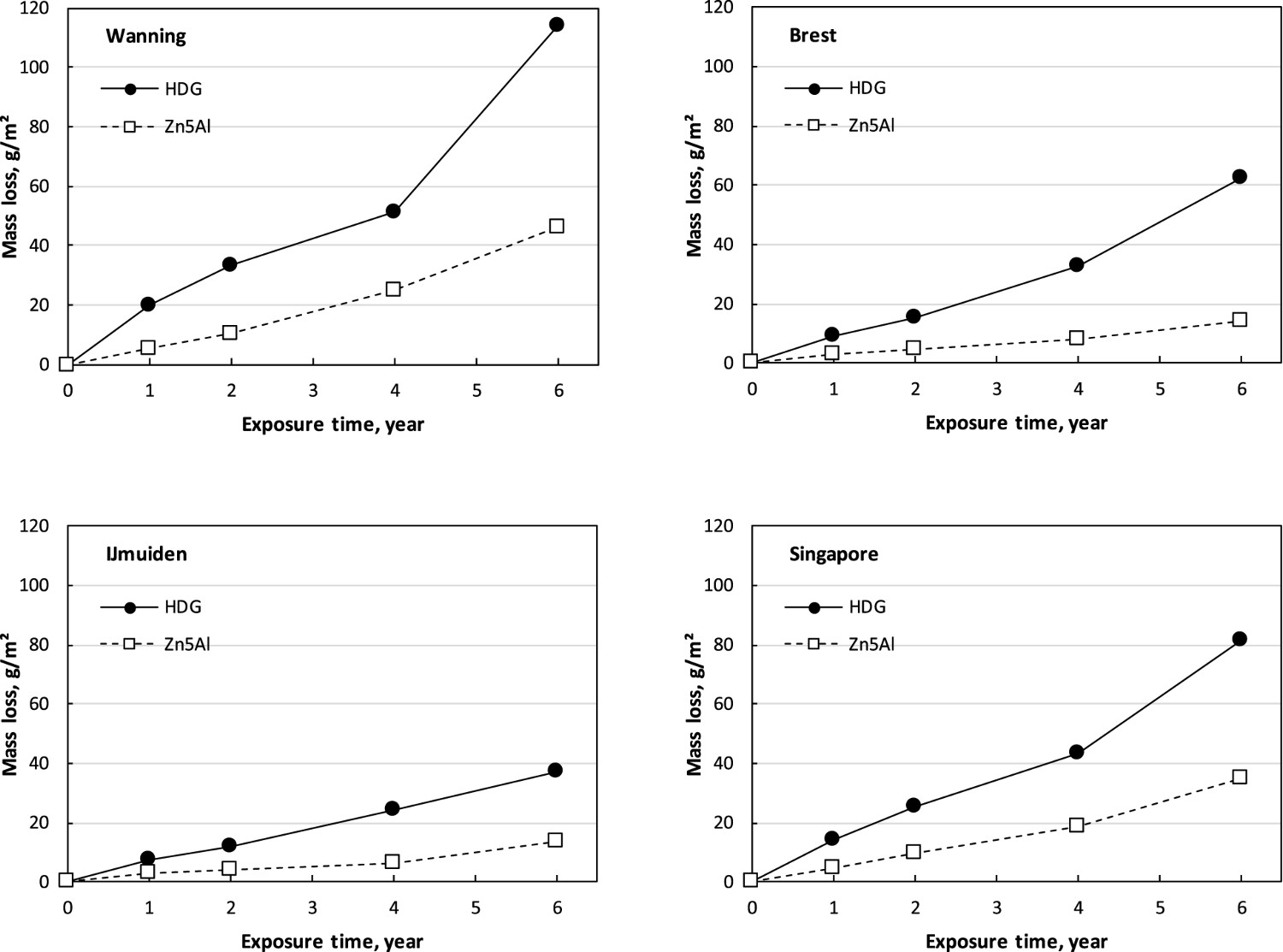
SEM–EDS investigations of cross-sections
In order to get information about the nature of the corrosion attacks of the coating layers, SEM–EDS studies were made on selected samples. Figure 4 shows typical SEM images of cross-sections of Zn5Al-coated steel exposed at the sites in Brest and Qingdao, while the corresponding images of HDG are shown in Figure 5. The corrosion attack of the Zn5Al coating is largely localised to some areas on the samples and seems to be initiated in the eutectic while the zinc dendrites are more stable as seen for ZnAl alloys with similar composition in previous works [9,12]. The corrosion attacks on HDG resulted in larger areas where the zinc coating is completely converted to corrosion products which covered the steel surface. However, also for HDG, the corrosion attack was localised and penetrated the coating already after ½ year of exposure [4]. The corrosion products offer some protection of the steel surface by a barrier effect while the remaining metallic zinc will offer cathodic protection of the steel substrate. This may also increase the rate of corrosion of the remaining zinc coating as indicated by the increase in mass loss for HDG between 4 and 6 years of exposure.
SEM images of cross-sections of Zn5Al after 6 years of exposure in Brest and Qingdao. SEM images of cross-sections of HDG after 6 years of exposure in Brest and Qingdao.

The SEM–EDS elemental images in Figures 6 and 7 for Zn5Al exposed 6 years in Brest and Qingdao show local corrosion attacks in the eutectic phases with the presence of oxygen and sulphur in the attacked zones. The corrosion attacks propagate in a zone close to zinc dendrites and sulphur content was higher at the bottom/front of the localised corrosion attack, indicating that there is a driving force for transport of sulphur due to the formation of local electrochemical cells. This is similar to ZM-coated steel (zinc aluminium magnesium) which show the formation of localised corrosion zones in eutectic regions with the formation of sulphate and chloride containing corrosion products [19]. For the Zn5Al coating, the contribution from chloride seems to be small as judged form the weak chloride signal in the EDS images. The results also indicate some accumulation of aluminium in the corrosion products, probably due to the formation of sulphate and carbonate-containing basic salts of aluminium and/or zinc.
SEM–EDS maps of Zn, Al, O, S and Cl for a cross-section of Zn5Al-coated steel exposed 6 years in Brest. SEM–EDS maps of Zn, Al, O, S and Cl for a cross-section of Zn5Al-coated steel exposed 6 years in Qingdao.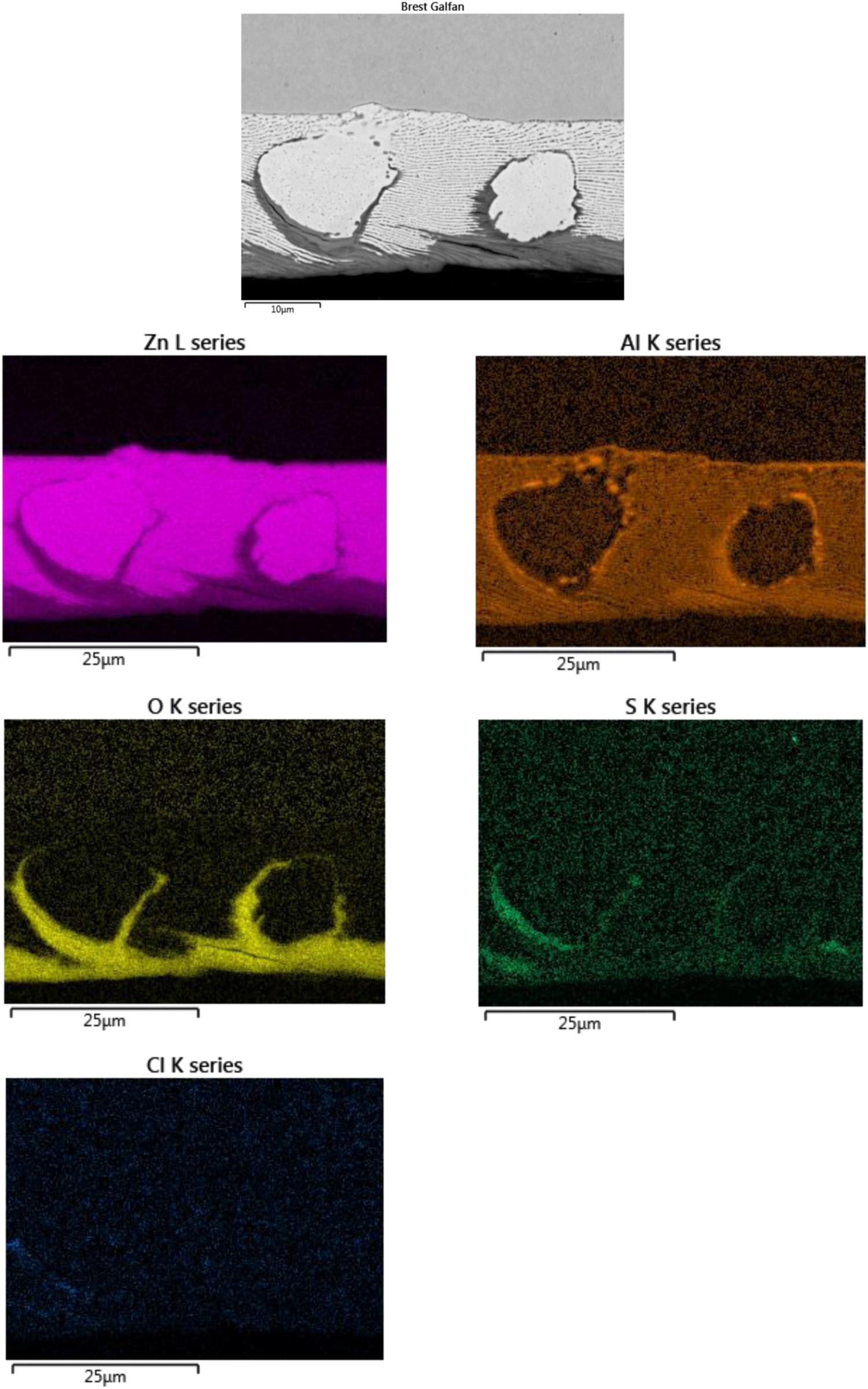

Corrosion product analysis
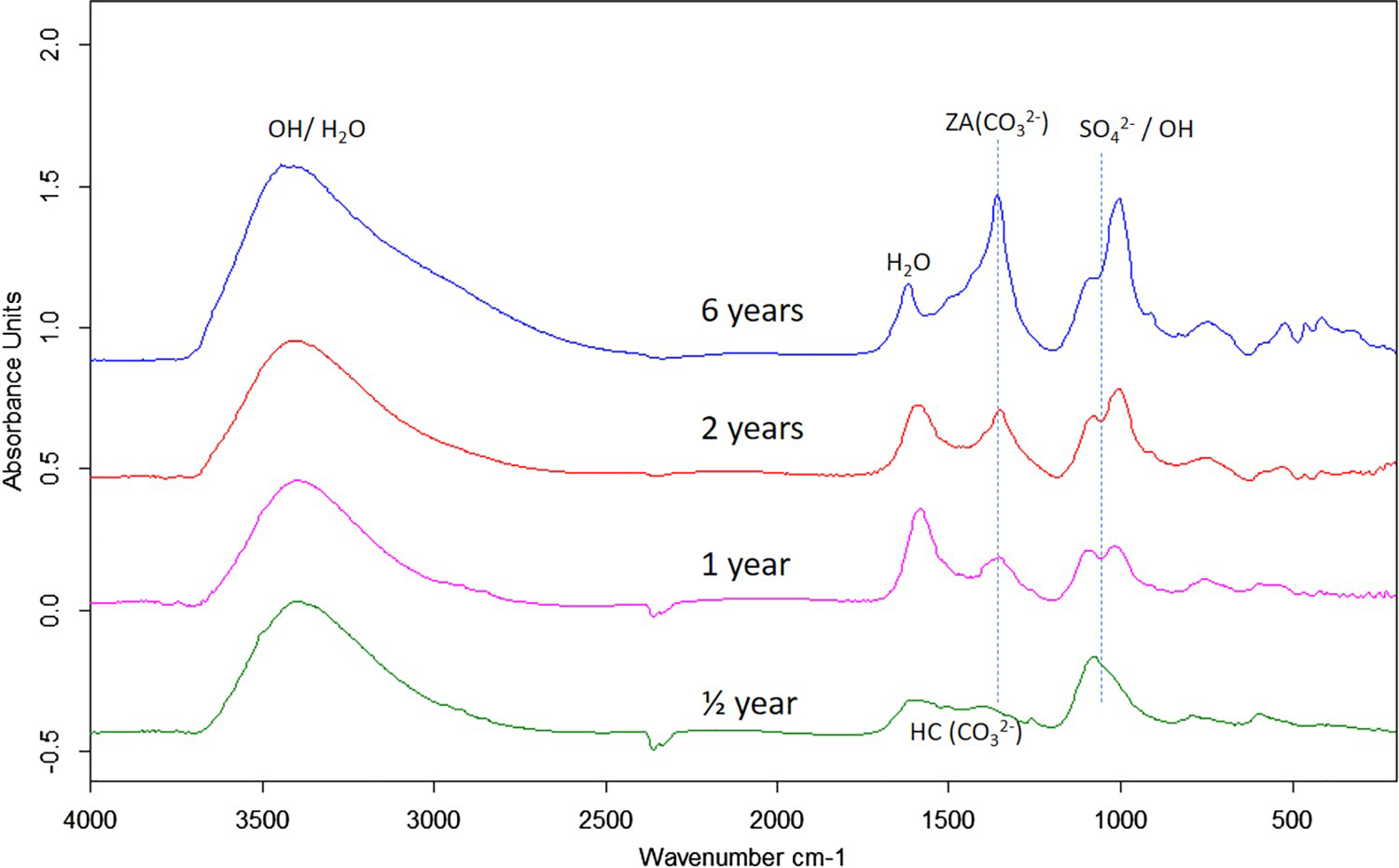
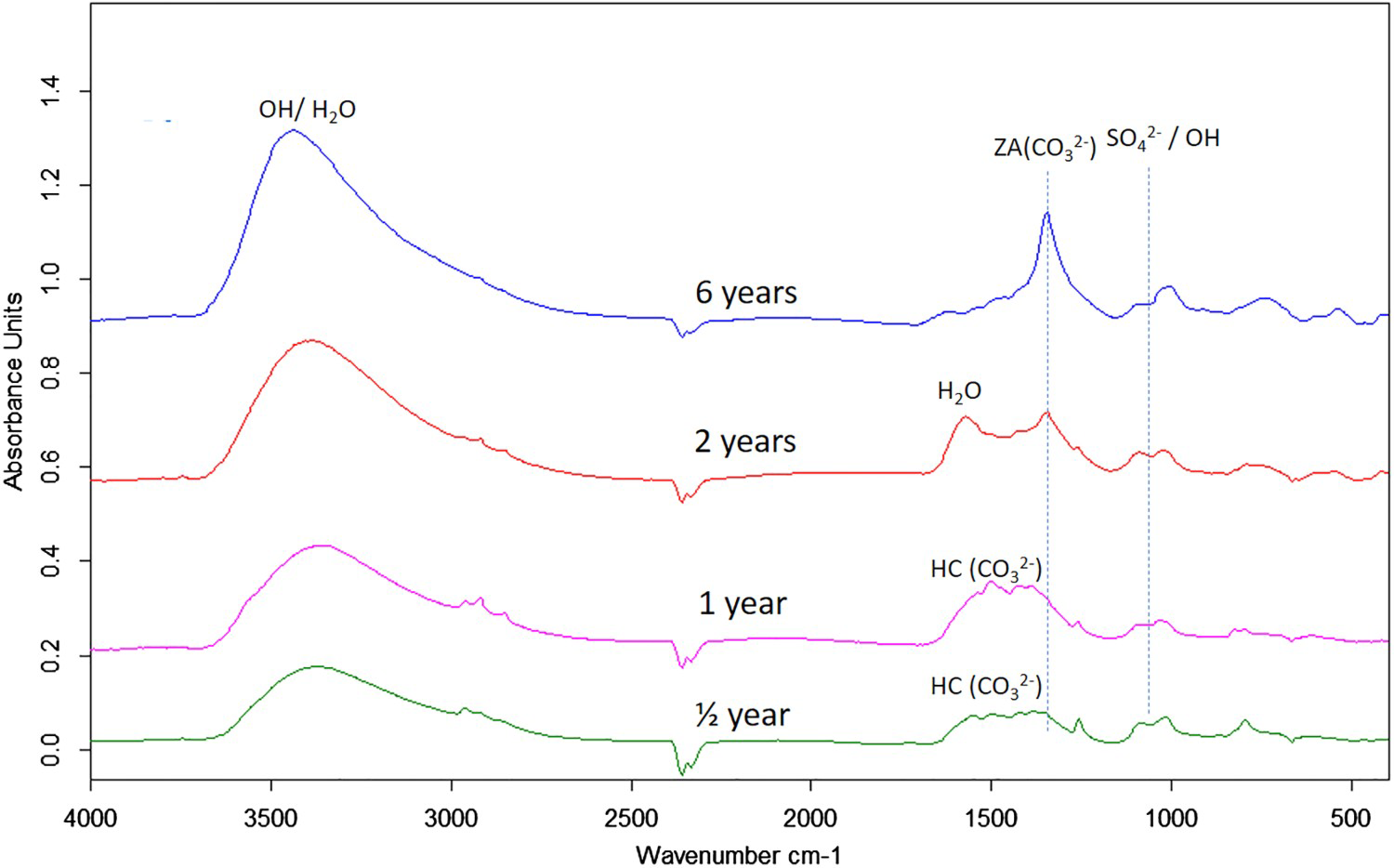
Corrosion products analysis performed by XRD, FTIR spectroscopy and FTIR imaging indicate that the dominating corrosion products on Zn5Al-coated steel after 6 years of exposure in Brest and Qingdao are zinc corrosion products such as zinc hydroxy carbonate (Zn5(OH)6(CO3)2) and zinc hydroxy sulphate (such as Zn4(OH)6 SO4·3H2O) and Zn/Al compound, such as the LDH, Zn2Al(OH)6(CO3)1/2 ·zH2O. Table 5 shows possible Zn, Al and Zn/Al corrosion products formed Zn5Al and HDG during atmospheric corrosion under the present conditions. The XRD analysis suggests that the compounds in Table 6 are present the corrosion products for HDG and Zn5Al coatings after 6 years of exposure in Brest, Qingdao and Jiangjin. FT-IRRAS spectra obtained after 0.5, 1, 2 and 6 years of exposure are shown in Figures 8 and 9 for the sites in Brest and Qingdao. Assignments of bands are based on reference spectra published earlier [4,20,21]. The spectra after longer exposure times are dominated by bands from the characteristic asymmetric stretching vibration of FT-IRRAS spectra of Zn5Al exposed 0.5, 1, 2 and 6 years at Qingdao. FT-IRRAS spectra of Zn5Al exposed 0.5, 1, 2 and 6 years at Brest. FTIR-FPA images of the intensity of Possible corrosion products, their chemical formula and abbreviations. Suggested corrosion products on HDG and Zn5Al from XRD after 6 years exposure.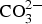 from a Zn/Al-
from a Zn/Al- 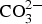 LDH, such as Zn2Al(OH)6(CO3)1/2 · zH2O, around 1360 cm−1 which originates from intercalated carbonate ions in the LDH. There are also a strong band from OH− and H2O in the region 2600-3500 cm−1 and strong bands between 900 and 1150 cm−1. The latter with contributions from Al/Zn-
LDH, such as Zn2Al(OH)6(CO3)1/2 · zH2O, around 1360 cm−1 which originates from intercalated carbonate ions in the LDH. There are also a strong band from OH− and H2O in the region 2600-3500 cm−1 and strong bands between 900 and 1150 cm−1. The latter with contributions from Al/Zn- 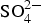 and Al/Zn-OH, probably in the form of hydrated aluminium and zinc hydroxy sulphates. It is also possible that poorly crystalline Al(OH)3 and/or AlOOH are formed, which would contribute to bands in the region 900-1100 and 2600-3500 cm−1. The SEM–EDS images show accumulation of sulphur in corroded zones with higher content at the bottom, which is consistent with the formation of sulphate-containing corrosion products. The images also indicate an increase in the Al content in the corroded zones which implies that aluminium is bonded in the corrosion products. The IRRAS spectra obtained after different exposure times show that Zn2Al(OH)6(CO3)1/2 ·zH2O becomes more significant after longer exposure times. For 0.5- and 1-year exposure in Brest, the carbonate bands are probably mainly due to the presence of zinc hydroxy carbonate, while the contribution from Zn2Al(OH)6(CO3)1/2 ·zH2O is dominating after 2 and 6 years of exposure. For the Zn5Al coating exposed in Qingdao, the Zn/Al-
and Al/Zn-OH, probably in the form of hydrated aluminium and zinc hydroxy sulphates. It is also possible that poorly crystalline Al(OH)3 and/or AlOOH are formed, which would contribute to bands in the region 900-1100 and 2600-3500 cm−1. The SEM–EDS images show accumulation of sulphur in corroded zones with higher content at the bottom, which is consistent with the formation of sulphate-containing corrosion products. The images also indicate an increase in the Al content in the corroded zones which implies that aluminium is bonded in the corrosion products. The IRRAS spectra obtained after different exposure times show that Zn2Al(OH)6(CO3)1/2 ·zH2O becomes more significant after longer exposure times. For 0.5- and 1-year exposure in Brest, the carbonate bands are probably mainly due to the presence of zinc hydroxy carbonate, while the contribution from Zn2Al(OH)6(CO3)1/2 ·zH2O is dominating after 2 and 6 years of exposure. For the Zn5Al coating exposed in Qingdao, the Zn/Al- 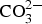 LDH is the dominating carbonate product already after 1-year exposure. FTIR imaging of the surfaces of Zn5Al exposed in Qingdao for 6 years in Figure 10 illustrates the local distribution of corrosion products associated with local corrosion attacks on the coating surface.
LDH is the dominating carbonate product already after 1-year exposure. FTIR imaging of the surfaces of Zn5Al exposed in Qingdao for 6 years in Figure 10 illustrates the local distribution of corrosion products associated with local corrosion attacks on the coating surface.
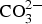
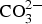 and
and 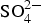 /OH bands for Zn5Al after 6 years of exposure in Qingdao.
/OH bands for Zn5Al after 6 years of exposure in Qingdao.

Discussion
The mass loss measured for the Zn5Al coating was in the range of 13-60 g m−2 after 6 years of exposure, whereas the corresponding mass loss for HDG was between 37 and 120 g m−2. The ratio of the mass loss of HDG to Zn5Al was ∼3 which is in line with previous investigations [9], but also rather similar to ZM coatings with Al (and Mg) content in the range 2-3.5 wt-% [19,22]. The improvement was generally slightly better for the marine exposure sites, although the variation was considerable between different sites. It should be noticed that no direct correlation was found between the relative performance of Zn5Al compared to HDG and the corrosivity of the sites (see, for instance, the site of Bangkok (C2) and all sites in C3 and C4). This also hold true when trying to correlate the chloride deposition and the level of improvement. Rather similar ratios of improvement were found at all marine sites despite the large variability in chloride deposition (see a comparison of Table 3 and Figure 1). However, the site of Jiangjin with a very high SO2 deposition shows a clear different tendency compared to all other sites, indicating that very high SO2 levels may significantly decrease the level of improvement of Zn5Al. This is in line with previous results reported for ZM-coated steel in SO2 environments [23]. It was observed that the variations in the mass loss between different exposure sites were smaller for Zn5Al compared to HDG which indicates that the former coating is less influenced by variations in exposure conditions for the different sites. This is most likely linked to the rapid formation of protective corrosion products (see discussion below).
The corrosion leads to a type of localised corrosion attack which is initiated in the eutectic phase on Zn5Al and which penetrated through the whole coating layer to the steel surface after 6 years of exposure. This is also the case for HDG and ZM-coated steel (with 2% of Al and 2% of Mg) and local penetrations of zinc coatings after longer exposure times at atmospheric weathering sites have reported in before [4].
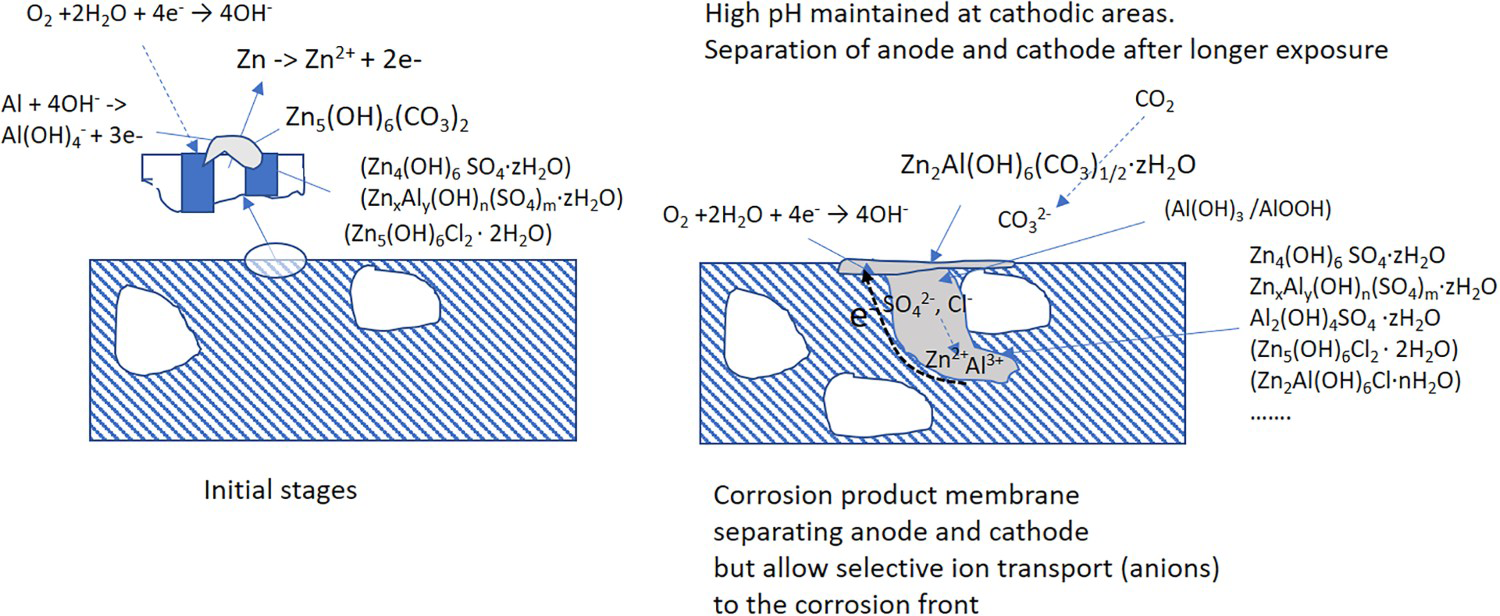
The corrosion attacks on the Zn5Al are preferably located to the eutectic phases giving rise to localised attacks which extend to the steel surface. Corrosion products which contain Zn/Al - Schematic illustration of the initial and advanced stages of atmospheric corrosion of Zn5Al in a marine atmosphere.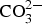 LDHs and hydrated basic sulphates of zinc and aluminium are formed in the attacked zones. The formation of Zn/Al-
LDHs and hydrated basic sulphates of zinc and aluminium are formed in the attacked zones. The formation of Zn/Al- 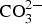 LDHs is observed for Zn5Al-coated steel after longer exposure times and the better corrosion resistance of Zn5Al is most likely related to the formation of this phase. The initiation and propagation of the corrosion attacks on Zn5Al in a marine atmosphere is illustrated schematically in Figure 11. It is suggested that the corrosion attacks are initiated at the eutectic phases with the Zn-rich phases attacked preferentially as indicated in [13]. In the atmosphere, zinc-rich corrosion products are dominating with the formation of zinc hydroxy carbonate. When the corrosion propagates in the eutectic areas, the corrosion products filled zones are formed which separate the anodic processes in the bottom of the corrosion pits from the cathodic reactions which probably take place in the parts of the corrosion attacks which are close to the surface of the coating with access to oxygen (and carbonate). Ionic transport through the corrosion products (anions towards the anodic region) will allow the maintenance of the local electrochemical cell which leads to the local penetration of the coating. The cathodic reactions taking place in the outer parts of the coating leads to an increase in the pH which promotes dissolution of Al and the precipitation of Zn/Al-
LDHs is observed for Zn5Al-coated steel after longer exposure times and the better corrosion resistance of Zn5Al is most likely related to the formation of this phase. The initiation and propagation of the corrosion attacks on Zn5Al in a marine atmosphere is illustrated schematically in Figure 11. It is suggested that the corrosion attacks are initiated at the eutectic phases with the Zn-rich phases attacked preferentially as indicated in [13]. In the atmosphere, zinc-rich corrosion products are dominating with the formation of zinc hydroxy carbonate. When the corrosion propagates in the eutectic areas, the corrosion products filled zones are formed which separate the anodic processes in the bottom of the corrosion pits from the cathodic reactions which probably take place in the parts of the corrosion attacks which are close to the surface of the coating with access to oxygen (and carbonate). Ionic transport through the corrosion products (anions towards the anodic region) will allow the maintenance of the local electrochemical cell which leads to the local penetration of the coating. The cathodic reactions taking place in the outer parts of the coating leads to an increase in the pH which promotes dissolution of Al and the precipitation of Zn/Al- 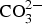 LDH. The dissolution of zinc from the outer parts of the coating will also promote the formation of the Zn/Al-
LDH. The dissolution of zinc from the outer parts of the coating will also promote the formation of the Zn/Al-  LDH. The improved corrosion resistance compared to a pure zinc coating is thus probably related to the formation of a dense and compact layer of Zn/Al-
LDH. The improved corrosion resistance compared to a pure zinc coating is thus probably related to the formation of a dense and compact layer of Zn/Al-  LDH in the outer parts of the coating which will decrease the overall cathodic reaction. It should be noticed that as Zn5Al seems to be less sensitive to environmental conditions (see Figure 3) and in particular to chloride deposition, it seems that the mechanism described above is also taking place even at low chloride deposition.
LDH in the outer parts of the coating which will decrease the overall cathodic reaction. It should be noticed that as Zn5Al seems to be less sensitive to environmental conditions (see Figure 3) and in particular to chloride deposition, it seems that the mechanism described above is also taking place even at low chloride deposition.
Conclusions
The results from the investigation of the atmospheric corrosion of Zn5Al at exposure sites in Europe, Asia, Middle East and the USA allow the following conclusions:
The corrosion performance of Zn5Al coating was always better compared to HDG after 1, 2, 4 and 6 years of exposure to different worldwide environments. The ratio of improvement of Zn5Al to HDG was in the range 1.3-5.1 after 6 years, while the mean value for all sites was 3.3. The improvement was higher for the pure marine sites (4.1). The data for the mass loss for Zn5Al showed less variations compared to HDG for the different field station which can indicate that Zn5Al is less sensitive than HDG for differences in the environmental conditions The long-term corrosion performance of Zn5Al is characterised with localised corrosion attacks preferentially in the eutectics. The improved long-term performance of Zn5Al is probably linked to the formation of Zn/Al -
 LDH and sulphate-containing corrosion products of Zn and Al.
LDH and sulphate-containing corrosion products of Zn and Al.
Footnotes
Disclosure statement
No potential conflict of interest was reported by the author(s).
