Abstract
The corrosion behaviour of Titanium carbonitride (Ti(C,N)) films grown by chemical vapour deposition was analysed in artificial sea water environment. From potentiodynamic polarisation curves, two passivation zones were detected, which originated from an initial oxidation of TiC and TiN to TiO2 followed by growth of the TiO2 layer upon increased polarisation. X-ray photoelectron spectroscopy analyses verified the mechanism by detecting a gradual decrease in Ti(C,N) peaks accompanied by a gradual increase of oxidised Ti (e.g. TiO2). It was likewise found that carbon in TiC mainly decomposes into carbonate species while the nitrogen in TiN remains elemental and likely escapes as nitrogen gas. Accordingly, Ti(C,N) behaves like a superposition of TiC and TiN with their individual oxidation behaviour, resulting in a highly corrosion resistant material.
GRAPHICAL ABSTRACT
Highlights
Corrosion properties of CVD grown Ti(C,N) film Two passivation zones detected in the polarisation curve TiC and TiN oxidises to TiO2, TiCO3 and N2
Introduction
Titanium carbides and nitrides have been used as wear protective hard coatings for decades. A major field of use is the metal cutting industry, where coatings of TiC, TiN and Ti(C,N) are routinely fabricated by either chemical vapour deposition (CVD) or physical vapour deposition (PVD) [1–4]. These hard coatings are also interesting for other applications, such as medical implants, where the excellent chemical stability and low toxicity of the coating are required [5–7].
CVD is routinely used to grow uniform and dense coatings of Ti(C,N) with excellent mechanical properties [8,9]. The technique offers a high step coverage and is therefore particularly adapt to coat complicated geometrical shapes and porous substrates. The technique has for instance been used to coat steel substrates, due to the moderate temperature needed to grow Ti(C,N) thin films (i.e. <830°C) [1]. Insights into the chemical stability can be obtained by investigating the electrochemical properties and more specifically the corrosion properties in different environments. While plenty of corrosion studies of TiC and TiN can be found in the literature [10–14], Ti(C,N) has been significantly less investigated [1,7,15].
Recent advances in the development of highly oriented Ti(C,N) thin films have revealed superior mechanical properties [16,17], compared to the state of the art in Ti(C,N) coatings grown by CVD [17]. Cutting fluid for coolant and chip removal is commonly used in many cutting operations. As such, it is likely that the protective coatings typically applied to the tools (e.g. Ti(C,N)) will come in contact with the cutting fluid under severe conditions (e.g. high temperature and high pressure). These conditions put the material under both mechanical and chemical stress, possibly causing rapid oxidation of the material. The present study aims to investigate the chemical stability of the Ti(C,N) thin film in a simulated sea water environment by studying the corrosion properties of the material. The corrosion mechanism and the corrosion products will be investigated by electrochemical analyses coupled with surface and chemical characterisation (i.e. atomic force microscopy (AFM), scanning electron microscopy (SEM) and X-ray photoelectron spectroscopy (XPS)).
Material and methods
Synthesis and sample preparation
The deposition and characterisation of the CVD Ti(C,N) coating used in this study are described in a previous work [16]. The CVD deposition was performed for 155 min at a growth rate of 0.67 µm/h using the following parameters: T = 830°C, P tot = 8.0 kPa, P TiCl4 = 0.26 kPa, P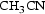 = 0.04 kPa, P
= 0.04 kPa, P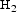 = 0.7 kPa and P
= 0.7 kPa and P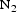 = 7.0 kPa. Ti(C,N) films were grown on single crystal c-sapphire substrates, as these substrates are insulating and thus do not contribute to the electrochemical response during corrosion measurements. The composition of the coating was Ti(C0.6,N0.4)0.8[16]. TiC and TiN grown by CVD onto sapphire substrates were used as references; deposition parameters for the reference TiC and TiN coatings can be found in Table A1.
= 7.0 kPa. Ti(C,N) films were grown on single crystal c-sapphire substrates, as these substrates are insulating and thus do not contribute to the electrochemical response during corrosion measurements. The composition of the coating was Ti(C0.6,N0.4)0.8[16]. TiC and TiN grown by CVD onto sapphire substrates were used as references; deposition parameters for the reference TiC and TiN coatings can be found in Table A1.
Characterisation
The surface morphology was analysed before and after corrosion measurements by high-resolution scanning electron microscopy (HR-SEM, Zeiss Merlin). The surface roughness and effective surface area were investigated before and after the corrosion measurements by AFM (PSIA XE150) operated in non-contact mode and in a 5 × 5 µm scan area.
The chemical environment of the corrosion products was investigated by XPS using a PHI Quantera II Scanning electron spectroscopy for chemical analysis (ESCA) microprobe with monochromatic Al Kα radiation and a spot size of 100 µm. Before the spectral acquisition, a gentle Ar+ etch of 200 eV was carried out for 1 min in order to remove contaminants from handling the sample in air. To avoid charging of the samples, neutralisation by both electrons and low energy Ar+ ions was used during the measurement.
Corrosion measurements
Potentiodynamic polarisation measurements were carried out to evaluate the corrosion properties of the coatings. All electrochemical analyses were performed in 3.5 wt-% NaCl aqueous electrolyte using a VersaSTAT4 (Princeton Applied Research) potentiostat/galvanostat. A three-electrode cell composed of the sample as the working electrode, a Pt wire counter electrode and saturated Ag/AgCl reference electrode was used. The exposed area for all samples was 0.196 cm2. Initial open circuit potential measurements for 1 h were followed by polarisation at −1.5 V for 300 s, after which linear sweep voltammetry (LSV) was used to scan from −1.5 to +1.5 V at 1 mV/s. The corrosion potentials (E corr) and the passivation potentials (E pass) were extracted from the polarisation curves.
Results and discussion
Corrosion properties of Ti(C,N)
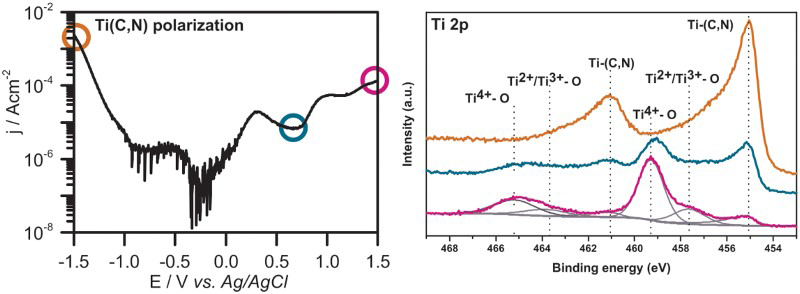
Potentiodynamic polarisation curves measured for the Ti(C,N) coatings are shown in Figure 1. From the corrosion potential (i.e. potential of zero net current) at about −0.3 V, a steady current increase was observed until maximum at 0.32 V was reached. At higher potentials the current decreased until a second maximum appeared at 1.05 V. This behaviour indicates two reactions passivating the surface in different potential regions. To understand the origin of these passivating reactions, reference samples of CVD grown TiC and TiN thin films were analysed and the resulting polarisation curves are shown in Figure 1. Titanium carbide showed a similar polarisation behaviour as Ti(C,N) with the notable difference in the first peak position (i.e. at 0.51 V for TiC) as well as two additional minor passivation peaks at higher potentials (i.e. 0.73 and 1.16 V). A higher passivation current was then observed in the region 0.5-0.9 V, compared to Ti(C,N). Titanium nitride, on the other hand, showed a steady current increase up to 1.10 V, after which the current density remained constant to finally rise after 1.3 V. The Ti(C,N) polarisation curve is therefore, most probably, a result of both TiC and TiN oxidation and passivation [13,14]:
Potentiodynamic polarisation curves for Ti(C,N), TiN and TiC thin films in 3.5 wt-% NaCl electrolyte. Points where samples were stopped for XPS analyses are marked.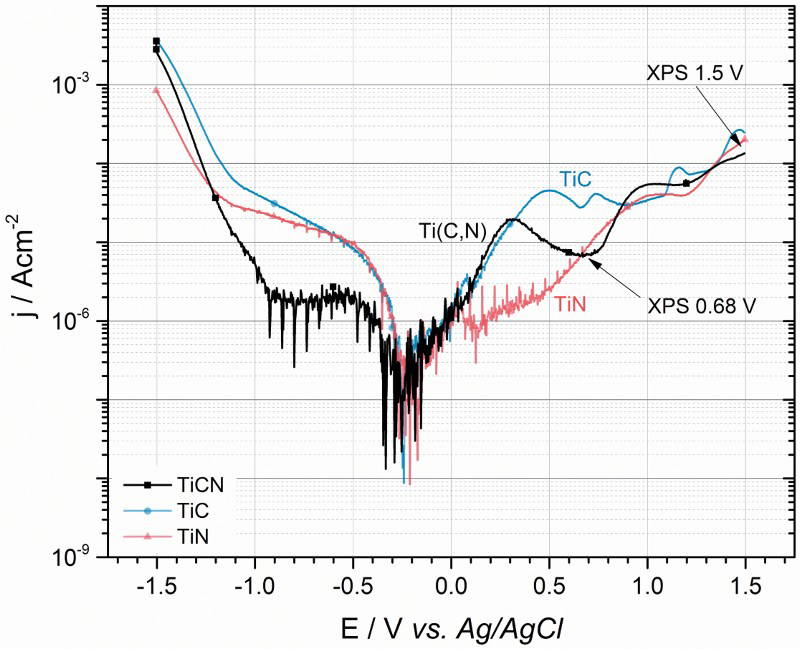
Corrosion properties of CVD grown Ti(C,N), TiC and TiN thin films in 3.5 wt-% NaCl electrolyte.

XPS analysis of corroded Ti(C,N)
In order to get a better insight on the corrosion mechanism of Ti(C,N), XPS was used to analyse Ti(C,N) films at different degrees of polarisation. In Figure 2(a–d), high-resolution core level spectra of Ti 2p, C 1 s, N 1 s and O 1 s are presented for the corroded Ti(C,N) samples stopped at 0.68 V and 1.5 V during the LSV experiment, as well as for the pristine Ti(C,N) reference sample. The core-level spectra for the reference sample agrees well with what has been reported in the literature on Ti(C,N) thin films [18–20]. Significant differences were observed in the XPS spectra of the corroded samples, compared to the reference sample. In the Ti 2p spectra, the Ti(C,N) peaks at about 455.1 and 461.0 eV decreased in intensity upon increasing polarisation (Figure 2(a)). New peaks also appeared at higher binding energies at about 459.3 and 465.2 eV, indicating an oxide environment on the surface, most probably corresponding to TiO2[21]. However, a distinct peak shoulder below 458 eV was also observed for the fully polarised sample (i.e. 1.5 V), indicating the presence of Ti with oxidation number below +4 [22].
XPS spectra of core levels of (a) Ti 2p, (b) O 1 s, (c) C 1 s and (d) N 1 s for the Ti(C,N) samples that, during the polarisation curve, were stopped at 0.68 and 1.5 V, respectively, and for the pristine reference Ti(C,N) sample.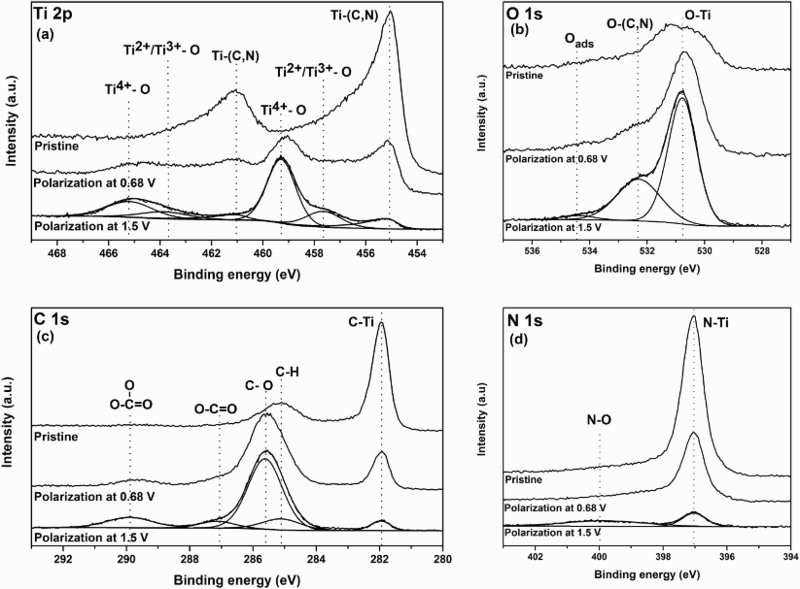
In the C 1 s, a peak at about 282.0 and 285.1 eV can be observed for all samples, which most probably correspond to a carbide environment and adventitious carbon, respectively [20,23]. As the Ti(C,N) samples were polarised, the carbide contribution decreased in intensity and new peaks at higher binding energies occurred (∼285.7, ∼286.7 and ∼289.9 eV). The latter is most likely corresponding to carbonate species and the former to oxidised carbon species of various degree of oxidation [23,24]. In the N 1 s spectrum, a peak at about 397.0 eV can be observed for all samples, which is most likely attributed to a nitride environment and follow a similar trend as the carbide peak in the C 1 s spectra, i.e. decreasing with increasing polarisation [18,24]. This is also in agreement with the observed decrease of the Ti(C,N) peaks in the Ti 2p spectra. Furthermore, in the N 1 s spectrum, there is a relatively small and broad feature for the fully polarised sample at about ∼400 eV, corresponding to oxidised N containing species [25]. In the O 1 s spectra, the O-Ti contribution at about 531 eV is increasing in intensity with increased degree of polarisation, which confirms the assumption of a more oxide character for the Ti(C,N) surface upon increased polarisation [19,20]. Additionally, a clear shoulder was observed in the O 1 s spectra at the high binding energy side of the O-Ti peak, most probably corresponding to an O-(C,N) environment [25–27]. Also, a small contribution at about 534.4 eV in the O 1 s spectra can be observed, that most probably could be ascribed to adsorbed water [28].
The XPS analysis of Ti(C,N) films confirms the assumption that the film is gradually corroded according to the individual TiC and TiN corrosion mechanisms. In particular, detection of oxidised carbon species on the surface after full polarisation confirms the previous hypothesis that carbonate species can be formed during oxidation of TiC. However, the analysis cannot exclude CO2(g) as a secondary reaction product. The TiN peak evolution during semi polarisation (i.e. 0.68 V) indicates that initially an oxide surface layer is formed, which attenuated the TiN signal. After full polarisation (i.e. 1.5 V), the TiN peak shape is altered and the intensity decreased, which could be interpreted as a two-step oxidation process.
Surface morphology
The surface morphology was analysed after the corrosion measurements by SEM and AFM, and compared to the pristine surface, as is shown in Figure 3. The smooth film surface was composed on sub-micrometre-sized crystals that generated an effective surface area gain of 8.8% (i.e. effective surface area 27.2 µm2 compared to the 25 µm2 footprint area analysed). After full polarisation to 1.5 V (i.e. LSV scan to 1.5 V), no significant change in surface morphology or roughness was observed and likewise no signs of pitting corrosion. As expected, the high corrosion resistance of TiC and TiN are maintained in CVD grown Ti(C,N) films in the artificial sea water environment.
Surface morphology (a and c) before and (b and d) after corrosion analyses imaged by (a–b) SEM and (c–d) AFM.
Conclusions
The corrosion properties of CVD grown Ti(C,N) films were investigated through electrochemical analyses coupled with XPS analyses of the corroded surface. Two distinct passivation zones were observed for Ti(C,N) with peaks at 0.32 and 1.05 V which aligned with reference measurements of TiC and TiN films. XPS measurements confirmed the gradual oxidation of TiC and TiN, in Ti(C,N), to TiO2 by analysing the corrosion product after each passivation peak. Carbon oxidation, in TiC, was also detected as carbonate species were found on the surface. Unlike TiC, the main corrosion product of TiN is elemental nitrogen that most likely leaves the surface as N2(g). Also, titanium species of lower oxidation state than +4 were detected, possibly originating from compounds having a composition between TiO2 and TiO. The corrosion measurement did not produce any noticeable difference in surface morphology or roughness, as confirmed by SEM and AFM. As such, it can be concluded that CVD-grown Ti(C,N) films are highly resistant to corrosion in artificial sea water and undergo an individual passivation of TiC and TiN to TiO2.
