Abstract
Polypyrrole (PPy) was prepared on the mild steel substrate by electrochemical polymerisation in the solution containing pyrrole monomer and succinic acid. The mild steel surface could be passivated before and during electropolymerisation by molybdate. The morphology and structure of the PPy film were studied with SEM. The typical cauliflower structure of PPy was observed. Raman and IR spectroscopy showed that the obtained PPy was in an oxidised state. The thermal stability of PPy was investigated by the thermal gravimetric analysis, showing that PPy was stable at higher than 480°C. The electrochemical property of the PPy film was performed by open circuit potential, polarisation curves (I/E), and electrochemical impedance spectroscopy. The corrosion behaviour of mild steel (CT3) with PPy film in solution NaCl 3% was studied.
This paper is part of a supplementary issue from the 17th Asia-Pacific Corrosion Control Conference (APCCC-17).
Keywords
Introduction
Conducting polymers (CPs) have been paid much more attention to a new kind of material since Chemical Communications published the seminar paper of Hideki Shirakawa, Alan MacDiarmid and Alan Heeger on polyacetylene. After that, a large number of papers, patterns on CPs could be seen. Polyaniline (PANi), polypyrrole (PPy) and polythiophene (PT) have been known as common CPs for many application adhesives, antistatic film, lithium batteries, sensor, drug delivery, and corrosion protection [1–7]. Among them, corrosion protection of metals with CPs has been an interesting and challenging approach. It is said that they can reduce the corrosion current of steel and other metals [6–8].
CPs have often been used as an anticorrosion coating. They could be electropolymerised directly on the surface metal even the active metal such as mild steel and aluminium. The easier way for many applications is to prepare formulated solution of these polymers and cover on the metal surface [9–12]. The mechanism of corrosion protection with CPs has still been controversial. The mechanism depends much on a specific setup experiments. Some authors proposed that CP could play a role as mediation of electron transfer, as oxygen reduction catalyst. Anodic protection and barrier effect have been discussed much. They were used to explain the corrosion protection mechanism in many cases [8,9]. In many cases, CPs can decrease the corrosion rate of active materials, but that significant advances are necessary to increase the protection time, especially if the polymer films have small defects.
Using dopant anions to improve the property of CPs has been known. Dopants could be introduced into the polymers chemically or electrochemically. The doping/releasing in/from a polymer of dopant anions during oxidation/reduction would provide the self-healing ability of polymer. The polymer could protect corrosion site when needed [8]. The dopant anions were present in solution containing a pyrrole monomer, oxalic acid and dodecylbenzensulfonic acid resulting nanocomponents could be formed expectedly on alloy Cu–Zn or only Zn electrochemically [13,14]. Consequently, the PPy could protect these metals from corrosion [15,16]. In the presence of ferrocyanate doped in the CPs, PPy could deposit on Mg, Zn without difficulties. That is the reason why the adhesion was improved. Some authors deposited PPy on mild steel in KNO3, K2C2O4, or Na2SO4 solutions. The PPy film was adherent and compact since the PPy could establish the covalent bonds with mild steel [17–21]. Troch-Nagels et al. [22] synthesised PPy in Na2SO4 solution on mild steel that did not need any further treatment]. Beck et al. [23] tried to improve the corrosion protection of PPy on mild steel with oxalate dopant as well.
One of the most important applications of PPy is the corrosion protection of metals. Lehr and Saidman synthesised PPy film as two-layer film. The inner PPy layer was doped with molybdate and nitrate anion. The outer PPy layer was fabricated in sodium bis (2-ethylhexyl)sulfosuccinate (AOT) [24,25]. Moreover, PPy nanocomposite could be synthesised electrochemically. Carbon nanotube and chitosan were used as the modifier in nanocomposite as well.
In this publication, the PPy film was synthesised in succinic acid solution and applied as a protective corrosion coating for the CT3 mild steel. The corrosion protection given by succinate dopant is investigated by the open circuit potential (OCP) – time measurements and potentiodynamic curves. In the presence of molybdate, a strongly adherent film could be formed directly on the mild steel by electropolymerisation without any dissolution of metals.
Experiment
Chemicals
Pyrrole (Py) monomer (Merck) was distilled under nitrogen before using, Na2MoO4·2H2O and succinic acid (C4H6O4) were purchased from Merck and used without any further purifying.
Preparation of PPy films
Mild steel CT3 (TISCO company, Thai Nguyen, Vietnam) was purchased in market with a size of 30 mm × 30 mm × 1 mm and used as substrate (C: 0.16%; Mn: 0.62%; Si: 0.15%; P: 0.010% and S: 0.042%, according to GOST 001-2001-TCVN 1656-75 standard). The substrates were mechanically ground with SiC paper to 2000 grit, degreased in ethanol, washed carefully in distilled water and dried in air.
Concentration of the solutions used in electropolymerisation.
Before polymerisation of pyrrole, the mild steel substrate was passivated. The mild steel sample was polarised in solution Na2MoO4 0.1 M (without pyrrole monomers) at +0.5 V (vs. SSE) for 36 min, washed with distilled water and then kept in a nitrogen atmosphere. A PPy film was formed on the surface of the mild steel with constant current at 0.9 mA cm−2 for 60 min. After the synthesis, the PPy film was washed many times with distilled water and dried at 50°C in reduced vacuum conditions.
Characterisation of the PPy films
Thermal gravimetric analysis (TGA) was performed by Ghimashu-50H with a scan rate of 10°C min−1 in atmospheric condition.
The chemical structure of PPy was characterised by Raman and FT-IR spectroscopies. Raman spectra were recorded by Laser Raman Spectrophotometer (Ramalog 9I, U.S.A.). FT-IR spectroscopy was done by Prestige -21 (Shimadzu).
EDX spectra and SEM images were obtained with SEM-HITACHI-4800 under the energy of 10 keV.
Corrosion protection test
The OCP, Tafel curves and electrochemical impedance spectroscopy (EIS) were tested in solution NaCl 3% on Zennium (Zaehner, Germany). EIS measurements were carried out on the PPy films at various DC potentials (referred here after as E dc) by applying 5 mV AC on the DC potential at frequencies ranging from 100 kHz to 0.1 Hz.
Results and discussion
Electropolymerisation of PPy
In a succinic acid medium, the PPy film was not successfully obtained if Na2MoO4 was not added in the solution for polymerisation. In order to get the PPy films, the steel surface was passivated in the solution Na2MoO4.
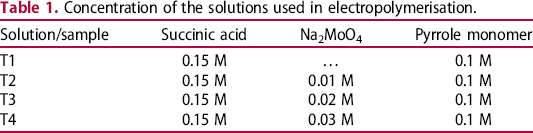
The mild steel substrate was polarised in solution Na2MoO4 0.1 M for 36 min at +0.5 V (vs. SSE) before electropolymerisation [4]. The change of current density is shown in Figure 1. The current density reduced very fast. After several minutes the current density reduced from 150 µA cm−2 to zero (actually nearly zero). After more than 10 min the current density was plateau (Figure 1). The low current density could be explained by the appearance of the passivated layer.
The polarisation curve – time of the mild steel in 0.1 M Na2MoO4 solution at +0.5 V.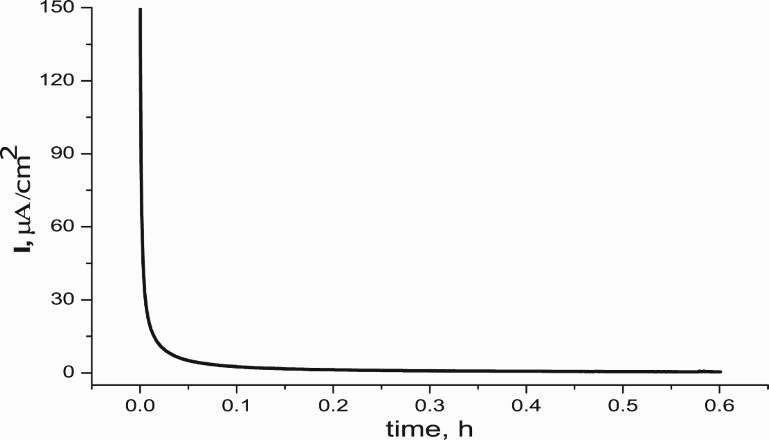
PPy film on the mild steel was polymerised at 0.9 mA cm−2 in the solution containing 0.15 M succinic acid and 0.1 M pyrrole monomer. In the case of without passivation, the changes of potential during polymerisation were presented in Figure 2. As shown in Figure 2, the potential of polymerisation of PPy to prepare T1 sample was high (about +2.25 V). In the presence of MoO4 2−, the potential of polymerisation was lower (at 0.65 V, in the cases of T2, T3 and T4, see Figure 2).
The current–time curves of electropolymerisation of PPy indifferent solutions.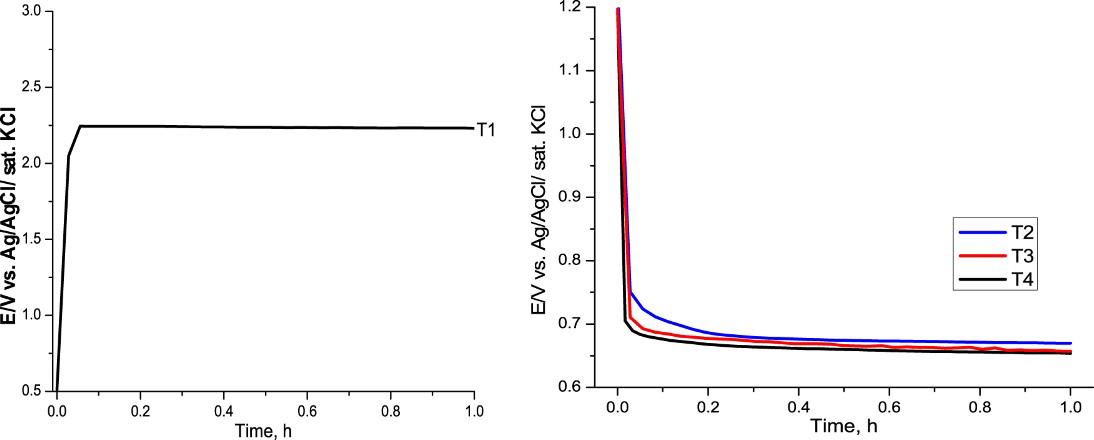
SEM images
The PPy films formed on the mild steel were rather homogenous and compact. This can be seen from the SEM images in Figure 3. The typical cauliflower structure of PPy film on mild steel is observed. It is similar to the other publications [26,27]. However, sample T1 was rather rough (see Figure 3(c)). In the presence of MoO4 2−, the surfaces of PPy films were more homogenous (see Figure 3(d–f)). MoO4 2− anion has been known as corrosion inhibitor. They have passivated the steel electrode surface after polarisation (see Figure 1), so that the polymerisation of pyrrole became easier. Therefore, pyrrole monomers were oxidised on steel electrode as an inert electrode. The film was formed homogenously.
SEM pictures of mild steel without passivation (a), with passivation (b), PPy formed in solutions T1 (c), T2 (d), T3 (e), and T4 (f).
In comparison with bare steel (Figure 3(a)), the thin layer was seen in the passivated steel surface (Figure 3(b)). This passivated layer could be a reason to form the more adherent PPy film on the CT steel.
Raman spectroscopy of PPy
Figure 4 shows the Raman spectra of the PPy-succinic and PPy-succinic-molybdate layers, respectively. The Raman peaks of the PPy doping anion succinate, molybdate layer in Figure 4 were assigned as follows: the peaks at 1569 cm−1 (T1, T4, T2), at 1577 cm−1 (T3) to the stretching of conjugative backbone C−C=C, the peaks at 1369 cm−1 to N−C ring stretching, and the peaks at 1049 cm−1 to −N−H in-plane bending [28,29]. The peaks at 966 cm−1 were due to the ring deformation and −C−H out of plane deformation [30], respectively. All characteristics peaks of PPy could be observed. It concluded that PPy was in an oxidised state.
Raman spectrum of PPy measured at 514 nm with 1 mW laser power.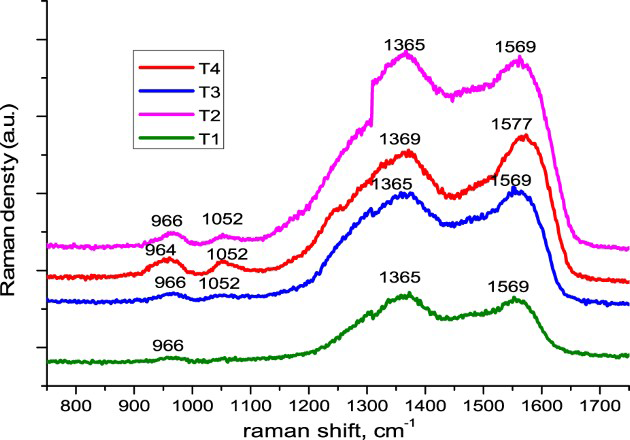
IR spectroscopy
IR spectroscopy of PPy films is shown in Figure 5.
IR spectra of PPy T1 (a) T2, T3, and T4 (b).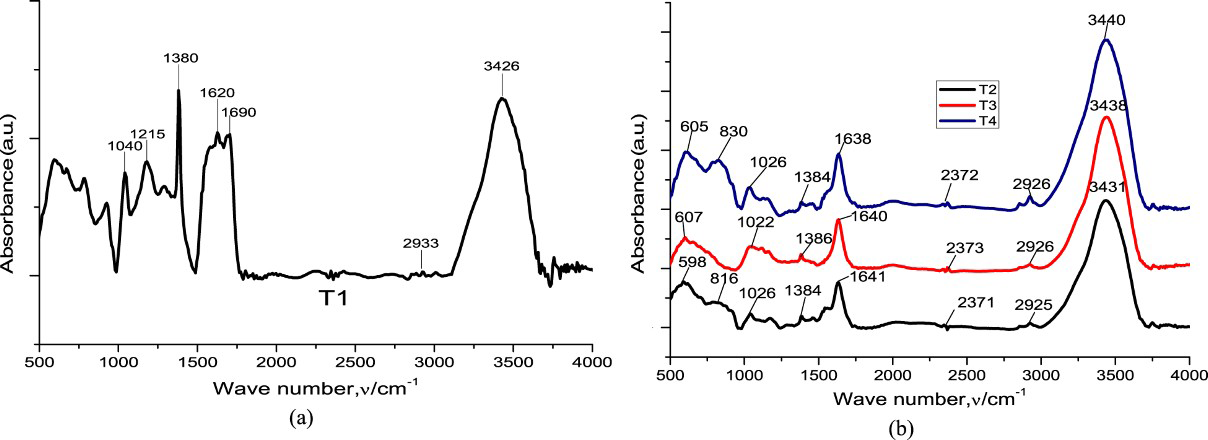
The examination of figures shows that the sample contains all the main characteristics band of PPy. The peak at 3445 cm−1 is assigned to the presence of N–H stretching vibrations. The incorporation of the anion in the PPy is evidenced by the peaks 2926 and 1640 cm−1 assigned to –CH2 and –C=O stretch in succinate anion. The characteristic absorptions peak of the PPy ring was observed at 1558 cm−1. The peak at 1384 cm−1 was assigned for stretching C–N bond. The peaks at 1026 and 820 cm–1 are attributed to the =C–H in-plane and out-plane stretching vibration, respectively. The FT-IR spectral results confirm the formation of PPy-succinic.
TGA/DTA analysis
In order to know the thermal stability of PPy, thermogravimetric analyzer measurements were carried out. The experiment was in atmosphere pressure, temperature rate 10°C min−1. The TGA and DTA are shown in Figure 6.
TGA and DTA analysis results of samples T1, T2, T3, and T4.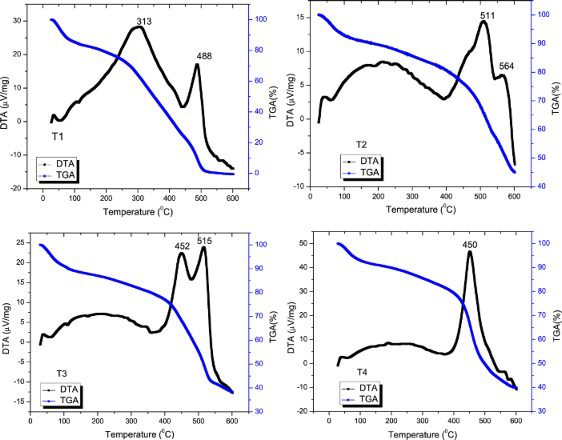
The weight loss observed at temperature below 100°C can be ascribed to expulsion of moisture, oligomers as well as the unreacted monomer. The degradation of PPy was observed between 275 and 600°C. The sample degraded fast at the temperature range of 260-500°C (T1) and 430-550°C (T2, T3, T4). After thermal decomposition at 600°C, the residual masses of T2, T3, T4 were 45.19, 38.3, and 39.78%, respectively. It maybe the inorganic compounds in PPy. Owing to the mobility of molybdate, doping level in PPy could not the same in all the samples. However, the sample T1 was decomposed completely at 500°C. The DTA curve has exothermic peaks with two peaks at 313 and 488°C (T1), 511 and 564°C (T2), 452 and 515°C (T3) or a single peak at 450°C (T4). The exothermic peaks were caused by the oxidative degradation process of PPy. These two exothermic peaks indicate that PPy in T1, T2, T3 was not uniform, the small peak degraded first, while the larger ones degraded later.
EDX spectra
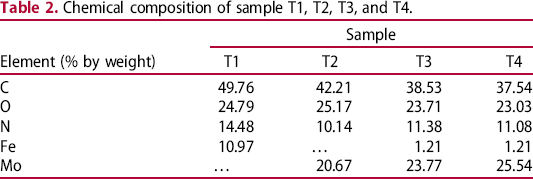
It could be seen that molybdenum in the EDX spectra (Figure 7b–d) showing that molybdate was incorporated in the PPy film during polymerisation of pyrrole. The amounts of elements in these samples are shown in Table 2. In Figure 7(a), the presence of molybdenum helped the PPy films to be more stable than one without molybdate (Figure 6). Peak of iron cation could be observed on the EDX spectra. Steel substrate was dissolved during electrochemical polymerisation so that they were detected. In the case of sample T1, amount of iron was high (10.97% by weight) showing that in the absence of molybdate, steel was very strongly dissolved during electrochemical polymerisation. In the presence of molybdate in the polymerisation solution, the dissolution of steel electrode was almost inhibited. It led to the amount of iron in the samples being very small (see Figure 7 and Table 2). Even, iron could not be detected in sample T2. The amount of molybdenum increased from sample T2 to sample T4 due to the increasing of molybdate in the polymerisation solution (Table 2).
EDX spectra of samples T1 (a), T2 (b), T3 (c), and T4 (d). Chemical composition of sample T1, T2, T3, and T4.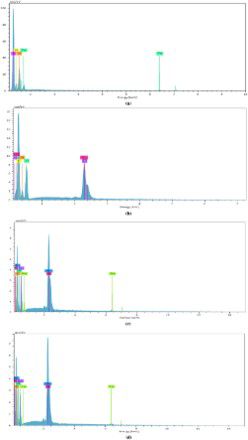
Electrochemical corrosion test
Change of OCP
After forming on mild steel substrate, the PPy was immersed in 3% NaCl solution for OCP testing. Bare mild steel sample after passivation in 0.1 M Na2MoO4 solution was also kept in 3% NaCl solution for comparison. Obtained OCP curves were shown in Figure 8.
OCP of mild steel CT3, T1, T2, T3, and T4 in solution NaCl 3%.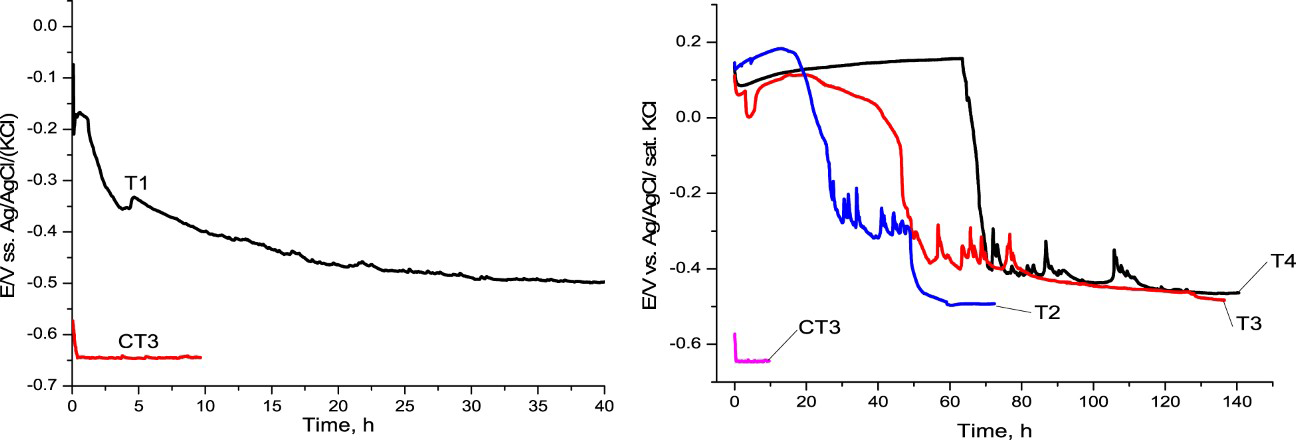
Although mild steel CT3 was passivated with molybdate, OCP could not improve in NaCl solution. The iron substrate was corroded at −0.645 V. OCP of sample T1 was more positive. It took 50 h to reach the corrosion potential of iron. OCP of samples T2, T3, and T4 have been prolonged 72.4, 136.5, and 140.5 h for T2, T3, and T4, respectively. The difference in the amount of molybdenum in the samples maybe the reason for the difference of OCP extending duration. It is concluded that PPy doped molybdate and succinate could have improved the OCP in the NaCl solution.
Tafel plots
In Figure 9, Tafel plots of sample T1, T2, T3, and T4 are presented. It could be seen that although a passive layer of molybdate was formed on CT3 mild steel, its OCP was in corrosion range of mild steel (Figure 9, see sample CT3). Corrosion current and potential were 1.29 × 10−6 A cm2, −0.645 V, respectively. A passive layer of molybdate alone could not improve corrosion protection.
Tafel plots of CT3, T1, T2, T3, and T4 in solution NaCl 3%.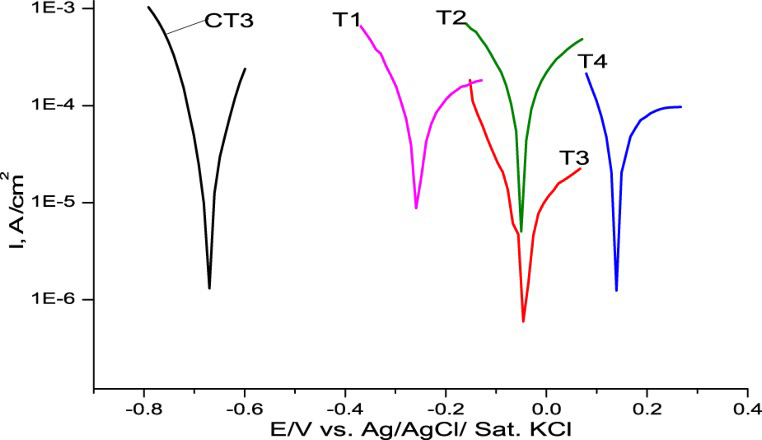
Corrosion potential and current of CT3, T1, T2, T3, and T4 in solution NaCl 3%.

Electrochemical impedance spectroscopy (EIS)
For sample T1, the impedance of PPy was about 120 Ω at the beginning, the rest potential was 0.09 V (Figure 10(a)). The anion exchange process could take place between chloride and succinate. The observations show that the film resistance was high. However, the rest potential of PPy film was −0.106 V after 2 h espousing in solution NaCl 3%. The film resistance was 110 Ω. That means the conductivity of the film was increased. The anion exchange process could reach the equilibrium state. After 17 h, the film could not protect the mild steel any more. The rest potential was fallen down −0.509 V. The PPy was in a reduced state. Succinate anions had released from the PPy films.
Impedance spectroscopy of PPy sample T1 (a), T2(b), T3(c), and T4(d) in solution NaCl 3%.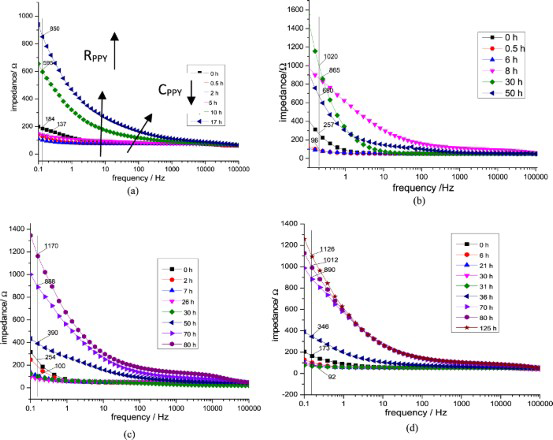
From Bode plot (Figure 10(a)), it can be concluded that the resistance of PPy increased and the capacitance decrease slightly, indicating there was the anion exchange between chloride and succinate anion. After 2 h exposure in 3% NaCl, the rest potential was −0,106 V. After 17 h, the PPy film could not protect iron any more.
For sample T2, T3, and T4, the behaviour of EIS were the same (Figure 10). The resistance of PPy films increased during the immersion time. The PPy films changed from oxidised state to reduced one. In comparison with sample T1, resistances of T2, T3, and T4 were higher. It could be explained by the presence of molybdate in the PPy films (Figure 10b–d). It means that the corrosion rate of sample T2, T3, and T4 were slower than that of sample T1. The corrosion rate is calculated by the following equation:
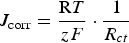
The results of EIS seem to be in agreement with the results of OCP and Tafel plots.
Conclusions
The PPy film could be formed successfully on the mild steel with a constant current in a solution containing succinic acid and sodium molybdate at different concentrations. Molybdate and succinate dopants were incorporated in PPy electrochemically. The mild steel substrate should be passivated before electropolymerisation. The cauliflower structure of PPy was seen with the SEM. Oxidised state PPy could be obtained in these synthesis conditions. PPy was thermally stable more than 450°C. In the presence of molybdate and succinate dopants, the corrosion protection ability of PPy improved significantly in NaCl 3% solution.
