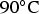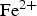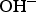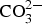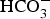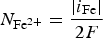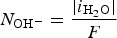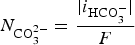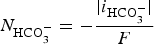Abstract
In the frame of conceptual design for the long-term management of high level radioactive waste defined by Andra (France), it is assumed that carbon steel (C-Steel) overpack would be mainly affected by general corrosion. As it is widely accepted that precipitation of a corrosion product layer (CPL) can limit the corrosion rate, the aim of this work is to simulate the growth of a thick FeCO3 CPL and its impact on the evolution of the corrosion rate. We use a 1D finite element method numerical model based on the resolution of Nernst–Planck equation in free potential conditions. Two numerical models of the evolution of the corrosion rate of low alloy steel in anoxic conditions are developed. These models take into account the growth of a porous CPL assuming either a constant and homogeneous porosity or a varying and heterogeneous porosity and results are compared to experimental data.
This paper is part of a supplement on the 6th International Workshop on Long-Term Prediction of Corrosion Damage in Nuclear Waste Systems.
Keywords
Introduction
Owing to their long-term activities, French high level radioactive waste will be stored in a deep Callovo-Oxfordian (Cox) argillite during several thousand years. The COx was identified as a suitable rock in terms of stability, low permeability and good mechanical resistance. The waste plant dimensioning and its durability are managed by the French national radioactive waste management agency (Andra). In this context, experimental and modelling studies are carried out to assess the behaviour of carbon steel overpacks containing the primary waste packages, more especially in terms of corrosion rate evolution during the disposal period [1]. Thus, modelling approach is used not only to simulate short-term (underground laboratory tests) experiments, but also to predict the long-term behaviour of the steel overpacks.
Since about nearly three decades, some studies using numerical simulation have been proposed in the literature to predict the general corrosion rate evolution of carbon steels. Because most of these studies were related to oil and gas industry, acidic CO2 (and/or H2S) environments were considered in flowing conditions [2 -10]. Other studies are referred to basic or neutral porous environments such as concrete or clays in the context of radioactive waste disposal [11–16]. In some of these studies, numerical simulation was implemented to predict the behaviour of carbon steels using either a semi-empirical [2,3,11,12] or a mechanistic [4–10] approach.
In a previous study carried out in the context of radioactive waste disposal, the estimation of the corrosion rate evolution of carbon steel has been performed based on a numerical approach in which the corrosion product layer (CPL) growth was taken into account in terms of a porosity decrease [17]. This study also permitted to identify the suitable kinetics parameters to be used in this context. In fact, the role of the CPL has been highlighted and implemented in their models by other authors [18–20]. For instance, F. King used a fixed porosity coefficient, characteristic of the CPL, in the transport equations [16,21,22]. Van Hunnik used the concept of scaling tendency (ST) to account for the effect of the CPL in a time-dependent model [19]. In a modelling study relative to the corrosion of carbon steel [20], this ST factor depends on the ratio of corrosion rate and precipitation rate of corrosion products and then evolves during the simulation. Then, the ST factor is supposed to be an indicator of whether CPL is protective or not. Furthermore, the chemical nature of the CPL has also been considered to carry out the effect of the CPL on the corrosion rate of carbon steel [23,24].
Nesic also defined a porosity coefficient for the CPL function of time and distance from the steel surface [20,25]. A similar approach leading to a heterogeneous distribution of the CPL will be presented in this paper.
Actually, the purpose of this paper is to demonstrate that the definition of the porosity of the CPL influences strongly the results in terms of corrosion rate evolution calculated with a mechanistic corrosion model. Two approaches will be presented in this work. The first one considers that the CPL behaves like a diffusion barrier with a constant and homogeneous porosity. On the contrary, in a second approach the porosity of the CPL is function of time and distance from the metal surface.
The simulation results will be compared with experimental measurements over several months of the corrosion rate of carbon steel coupons in a closed deaerated porous medium.
Experimental feedback
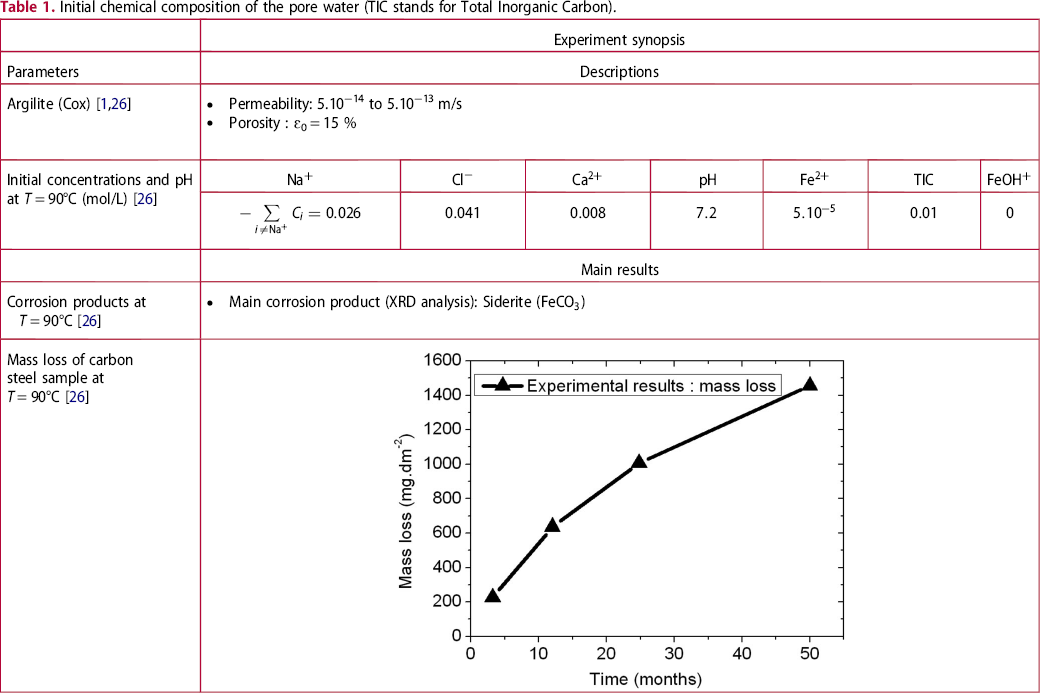
Initial chemical composition of the pore water (TIC stands for Total Inorganic Carbon).
In the following section, we present the numerical model that will be used to simulate this experiment. Simulation will be conducted only for T = 90°C.
Model and governing equations
The mass conservation for any chemical species considered in the medium is given by
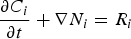
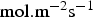 ), Ci the concentration (
), Ci the concentration (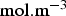 ) and Ri the production/consumption rate of species i due to chemical reactions.
) and Ri the production/consumption rate of species i due to chemical reactions.
Excluding the convection term, in dilute solutions, the flux is expressed as follows:
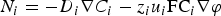
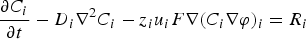
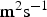 ), the charge number and the ionic mobility (
), the charge number and the ionic mobility (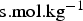 ) of species i. φ is the electric potential in the solution (V), F the Faraday constant (F = 96485
) of species i. φ is the electric potential in the solution (V), F the Faraday constant (F = 96485 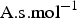 ). The ionic mobility ui can be expressed through the Nernst–Einstein equation:
). The ionic mobility ui can be expressed through the Nernst–Einstein equation: 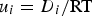 , where T is the absolute temperature (K) and R the universal gas constant (R = 8.314
, where T is the absolute temperature (K) and R the universal gas constant (R = 8.314 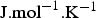 ).
).
Then, the electroneutrality condition is assumed in order to get as many equations as varying (i.e. the concentrations and the potential):
Progressive meshing. Meshing of subdomain of 2 cm. Electrochemical reactions at the metal surface with corresponding kinetics laws. ii stands for the current density, αi stands for the apparent charge transfer coefficient, ii stands for the apparent exchange current density, Vm the potential of the metal and 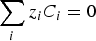
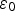 , is equal to 0.15 [1,26]. The pore water contains the following chemical species:
, is equal to 0.15 [1,26]. The pore water contains the following chemical species: 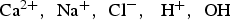
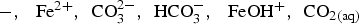 . Siderite (
. Siderite (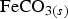 ) is assumed to be the only solid that can precipitate forming the CPL [26,27]. According to the experimental results,
) is assumed to be the only solid that can precipitate forming the CPL [26,27]. According to the experimental results, 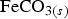 is the main mineral expected to precipitate [26]; however, our numerical model could be adapted to integrate other minerals. The initial composition of the pore water is summarised in Table 1. The electrochemical reactions occurring at node N1 (metal surface), i.e.
is the main mineral expected to precipitate [26]; however, our numerical model could be adapted to integrate other minerals. The initial composition of the pore water is summarised in Table 1. The electrochemical reactions occurring at node N1 (metal surface), i.e. 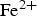 oxidation,
oxidation, 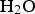 and
and 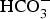 reductions, are reported in Table 2 with their corresponding kinetics law.
reductions, are reported in Table 2 with their corresponding kinetics law.


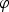 the electric potential in the solution.
the electric potential in the solution.
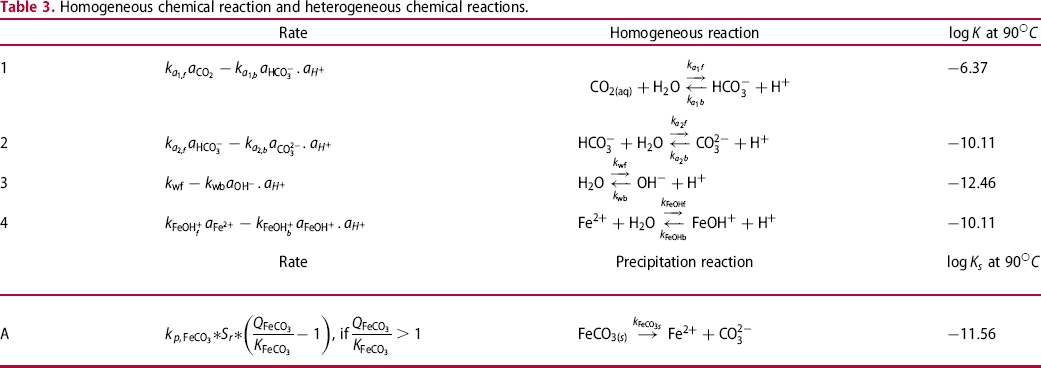
Homogeneous chemical reaction and heterogeneous chemical reactions.
Summary table of all the parameters: values and units.
Flux for electroactive species at node N1.
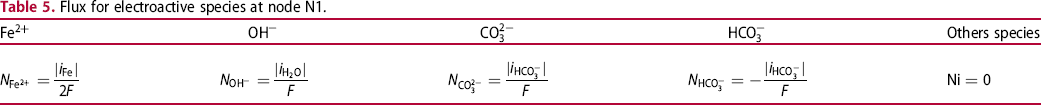
Two methods are used to simulate the effect of the CPL on the corrosion rate. In a first step, a homogeneous CPL with a fixed porosity grows on a steel surface in contact with a Cox argillite. In the second approach, porosity is supposed to evolve during the CPL growth, resulting from the amount of precipitated siderite (CPL grows in density as well as thickness).
It must be noted that a minimal CPL thickness has been fixed at t = 0 s to allow the simulation to start. We suppose, in fact, in both models that the nucleation period of the CPL is fast (non-limiting) in comparison with the growth period.
In numerical simulation, for each time step, when the siderite becomes under-saturated at the external face of the CPL, the following x-coordinate is assumed to be the clay environment. Hence, for each time step we obtain two domains: the CPL and the clay environment. Thus, the thickness  of the CPL is obtained by calculating the saturation level all along the x-coordinate.
of the CPL is obtained by calculating the saturation level all along the x-coordinate.
We define the total porosity by
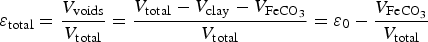
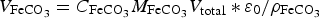 and
and 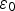 the porosity due to the argillite.
the porosity due to the argillite.
Finally,
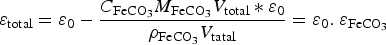
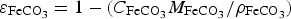 the porosity due to the precipitated siderite in a medium of porosity equal to 1.
the porosity due to the precipitated siderite in a medium of porosity equal to 1.
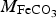 is the molar weight of siderite
is the molar weight of siderite 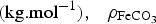 , its density (
, its density (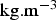 ) and
) and 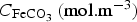 its concentration.
its concentration.
This coefficient takes into account the Cox porosity (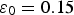 ) and the porosity resulting from the precipitation of siderite
) and the porosity resulting from the precipitation of siderite 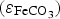 . It is constant in the first case, and is function of the amount of precipitated siderite in the second case. Fluxes at the metal surface and diffusion coefficients are function of the total porosity according to
. It is constant in the first case, and is function of the amount of precipitated siderite in the second case. Fluxes at the metal surface and diffusion coefficients are function of the total porosity according to
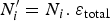
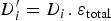
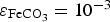 and
and 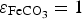 outside of the CPL i.e. in the external porous domain corresponding to argillite (
outside of the CPL i.e. in the external porous domain corresponding to argillite (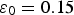 ). The precipitation term
). The precipitation term 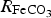 is assumed to contribute to the growth of the CPL.
is assumed to contribute to the growth of the CPL.
Second approach – heterogeneous CPL with a varying porosity: In this case, the expression of the porosity coefficient is obtained with Equation (9) [20,25].
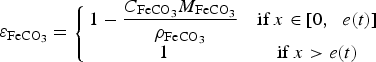
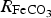 contributes to the growth of the CPL and the evolution of porosity inside the CPL.
contributes to the growth of the CPL and the evolution of porosity inside the CPL.
Values of the parameters used in the numerical model.

Result and discussion
Homogeneous CPL with constant porosity
In Figure 2 is given the total porosity profile for four different times. It can be observed that the CPL starts to grow at t = 1.25.105 s corresponding to approximately 1, 45 days.
Profile of the total porosity for four times. First approach – homogeneous CPL with constant porosity.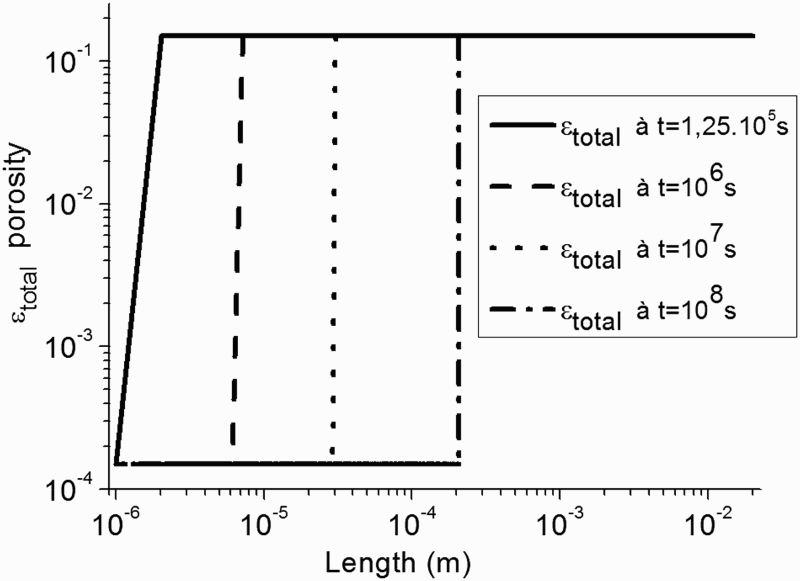
It is worth noting that at t = 1.25.105 s, the slope is imputed to the transition from a mesh to another mesh. As shown in Figure 2, the thickness of the CPL is equal to 200 µm at t = 108 s (deduced from the transition of porosity value observed at t = 108 s).
In Figure 3, the evolution of the corrosion rate and mass loss of the metal are given.
Evolution of the corrosion rate and mass loss of the metal assuming a homogeneous CPL with constant porosity.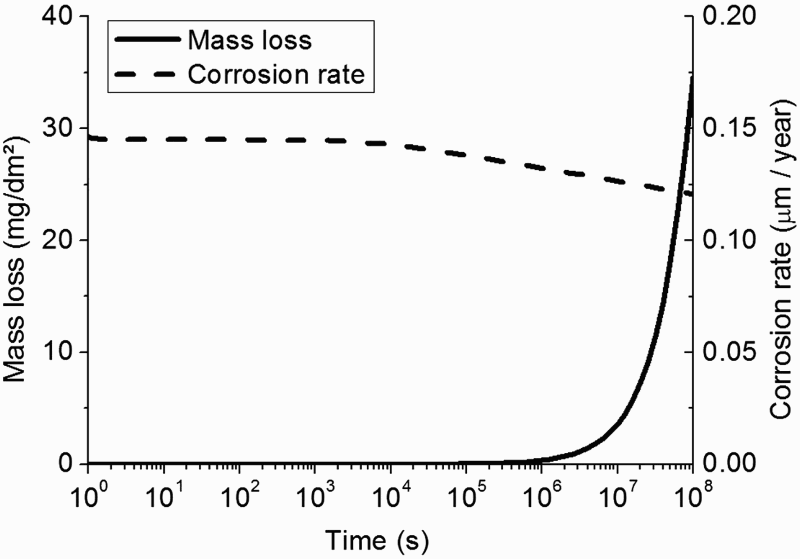
As soon as precipitation starts, the corrosion rate is quite constant and is function solely of the porosity at short time. However at long time, the increase of the calculated pH in the CPL next to the metal surface (quite constant at short time, and then increase from t = 2.106 s to t = 108 s) leads to a slight decrease of the corrosion rate.
Heterogeneous CPL with varying porosity
As shown in Figure 4 when the porosity is assumed to be dependent on the amount of precipitated siderite (Equation 9), we observe obviously that the value of porosity inside the CPL is not constant.
Porosity profile due to precipitation of FeCO3(s) for four different times assuming a heterogeneous CPL with a varying porosity.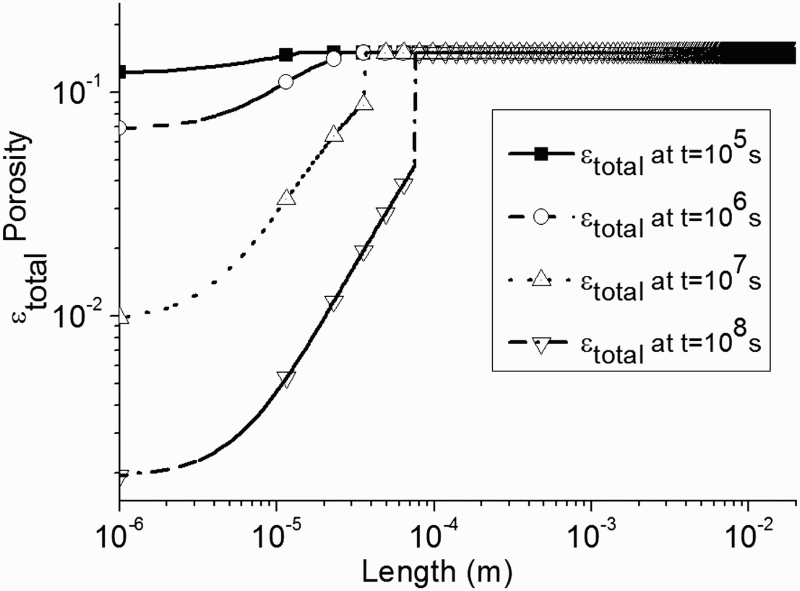
In Figure 5 are given the evolution of the corrosion rate (µm/year) and mass loss (mg/dm²).
Evolution of corrosion rate in µm year−1 and mass loss of metal in mg dm−² assuming a heterogeneous CPL with a varying porosity.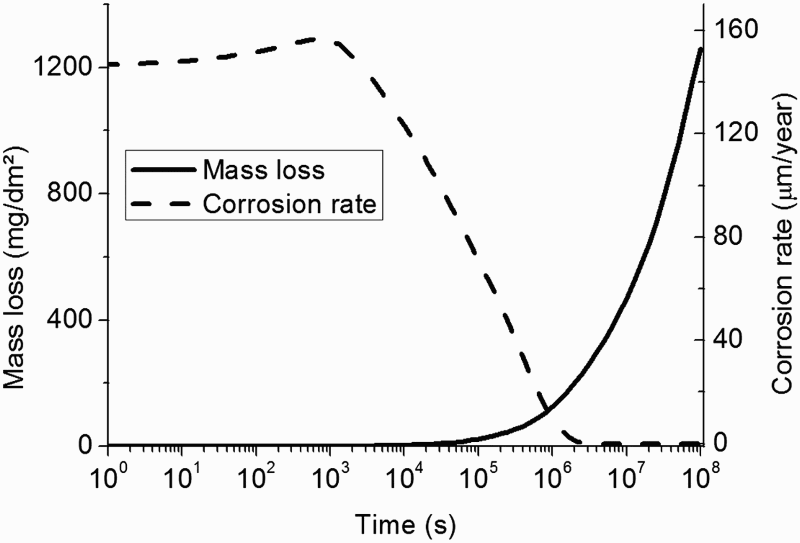
The corrosion rate varies from 150 µm.year−1 to 3 µm.year−1. In fact, initially the porosity at the metal surface is close to 0.15; hence, the transport of species is not affected by the precipitation of siderite. Then, the porosity decreases resulting from the formation of corrosion product, with the effect to decrease both the active metal surface and the transport of aqueous species, and consequently the corrosion rate.
In Figure 6 the evolution of the experimental and simulated mass loss at T = 90°C is compared.
Mass loss of low alloy steel coupons in contact with saturated argillite in anoxic conditions. Results are given for temperature T = 90°C up to 50 months. Comparison of numerical results at 90°C with experimental data [26].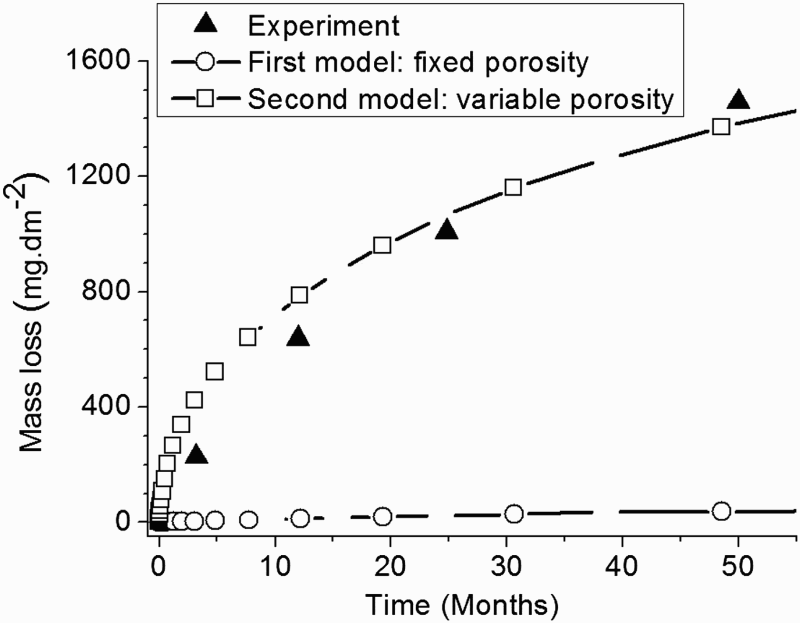
The simulated curves are referred to the different approaches developed previously, i.e. accounting for i) a CPL with a fixed and homogeneous porosity, ii) a CPL with a varying and heterogeneous porosity.
As shown in Figure 6, there is a good agreement between the second approach and the experiment [26]. In fact, the mass loss at t = 24 months and at t = 50 months are respectively equal to 1024 mg/dm² and 1400 mg/dm². These values are close to the experimental data, i.e. 1006 mg/dm² after 24 months and 1456 mg/dm² after 50 months.
In contrast, with the first model where a fixed and homogeneous porosity is considered, a steady state is observed and this leads to a constant corrosion rate. This behaviour does not reflect the experimental data showing a significant decrease of the corrosion rate.
In both cases, the corrosion rate is function of the fraction coverage of FeCO3 on the metal surface. In the first case, the fraction coverage is constant because the porosity is constant (and uniform) within the CPL. In this condition, a low decrease of the corrosion rate can be observed most probably due to the increase of the pH. In the second case, the porosity (and then the fraction coverage on the metal surface) varies in a large extent but remains larger in comparison with the previous case. In this condition, the saturation of FeCO3 is higher near to the metal surface leading to a thinner CPL.
In the following section, a parametric study is performed in order to understand what controls under both assumptions (varying and fixed porosity) the evolution of the corrosion rate, and thus the mass loss of the metal. In Figure 7 are summarised the results obtained for two additional cases of fixed porosity, considering (a) Evolution of the pH at metal surface (b) contribution of the reduction of bicarbonate (c) evolution of CPL's thickness and (d) evolution of the corrosion rate (homogeneous CPL with fixed porosity). and
and 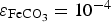 .
.
When the porosity of the CPL is very low ( ), the change in pH is weak, then the corrosion rate is limited solely by the porosity of the CPL at the metal surface. For
), the change in pH is weak, then the corrosion rate is limited solely by the porosity of the CPL at the metal surface. For  and
and 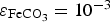 , the pH of the solution at the surface increases to basic values for t > 106 s. In these conditions, the corrosion rate is controlled by the porosity of the CPL only at short times (t < 106 s). Afterwards, the contribution of the reduction of
, the pH of the solution at the surface increases to basic values for t > 106 s. In these conditions, the corrosion rate is controlled by the porosity of the CPL only at short times (t < 106 s). Afterwards, the contribution of the reduction of 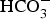 is decreasing (Figure 7(b) indicates that water reduction becomes the main cathodic contribution when the pH increases) causing the decrease of the corrosion rate.
is decreasing (Figure 7(b) indicates that water reduction becomes the main cathodic contribution when the pH increases) causing the decrease of the corrosion rate.
Results obtained using the second approach (varying porosity), by multiplying the precipitation rate constant of siderite ( (a) Evolution of the pH at the metal surface, (b) contribution of the reduction of bicarbonate, (c) evolution of porosityat the metal surface and (d) evolution of the corrosion rate (heterogeneous CPL with a varying porosity).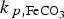 ) by 102 and 10−2, are summarised in Figure 8. The value of this rate constant influences the porosity in the CPL, more especially at the metal surface. The first inflexion corresponds to the beginning of the precipitation of siderite. The second one corresponds to the establishment of steady state: no more precipitation is observed.
) by 102 and 10−2, are summarised in Figure 8. The value of this rate constant influences the porosity in the CPL, more especially at the metal surface. The first inflexion corresponds to the beginning of the precipitation of siderite. The second one corresponds to the establishment of steady state: no more precipitation is observed.
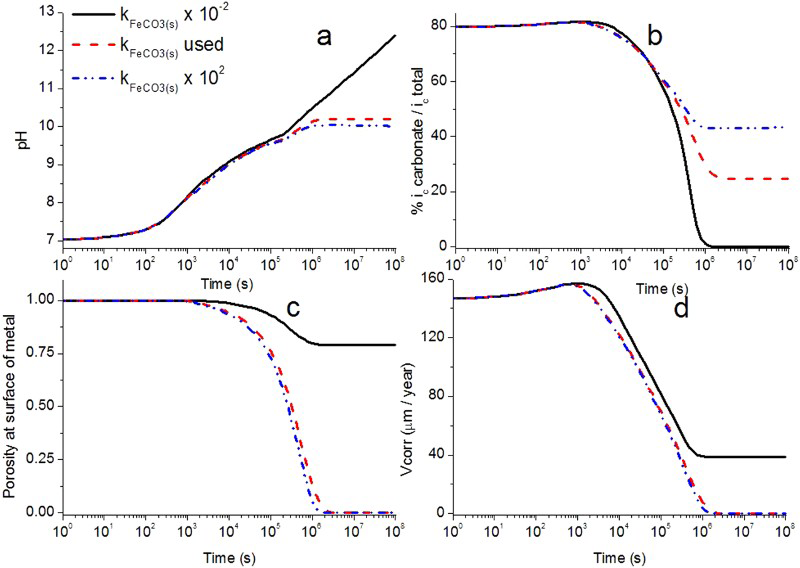
This effect can be seen in Figure 8(c), where the porosity at the metal surface is decreasing to very low values for higher precipitation rate constants. The evolution of the corrosion rate is controlled by the porosity at the metal surface (Figure 8(c,d)). When the precipitation rate is very high (102 × 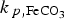 ), then a very thin (ca. 32 µm thick at t = 108 s) and dense CPL is formed. In contrast, when the precipitation rate is low (10−2 ×
), then a very thin (ca. 32 µm thick at t = 108 s) and dense CPL is formed. In contrast, when the precipitation rate is low (10−2 × 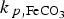 ) a porous and thick CPL is formed (ca. 295 µm thick at t = 108 s). In this latter condition, like in the previous case (Figure 7), a higher corrosion rate is obtained at long term due to a less protective CPL.
) a porous and thick CPL is formed (ca. 295 µm thick at t = 108 s). In this latter condition, like in the previous case (Figure 7), a higher corrosion rate is obtained at long term due to a less protective CPL.
Conclusion
The evolution of the corrosion rate of carbon steel in anoxic conditions has been simulated using two numerical models. These models take into account the growth of a porous CPL assuming either a constant and homogeneous porosity or a varying and heterogeneous porosity.
The numerical results have been compared to experimental data. The second approach (varying porosity) better reflects the experimental evolution of the corrosion rate. Assuming porosity is varying during the corrosion process and, during the CPL growth, allows the system to evolve in a larger extent leading to a significant decrease of the corrosion rate.
When the porosity is supposed fixed inside the CPL (regardless of the value of the porosity), the system reaches a steady state and hence a quite constant corrosion rate. However at long time, as shown by the parametric study, the pH controls the corrosion rate. Hence, the corrosion rate is controlled mainly by the surface porosity at short times and by the pH evolution at long time.
Finally, the parametric study allows establishing a relation between the precipitation rate of the siderite and the protectiveness of the CPL: for a high precipitation rate, a dense and thin CPL is observed leading to a low corrosion rate.
This approach is being adapted for other environments (aqueous medium, a room temperature and different chemical compositions) considering also the possibility for the CPL to be dissolved when under saturated.

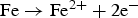
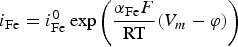
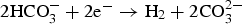
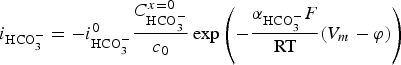
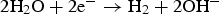
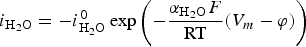
 at
at