Abstract
The increasing number of scrapped tyres constitutes a global problem. One way of recycling them is to mill the rubber waste to produce ground tyre rubber (GTR), which is utilised as a component of fresh rubber compounds for manufacturing new tyres. This paper is focused on the characterisation and application of five grades of GTR produced via three methods: ambient, cryogenic, and waterjet milling. Each GTR was separated using sieves into five size fractions. Each fraction was mixed into a fresh rubber compound based on styrene–butadiene rubber (SBR), natural rubber (NR), and butadiene rubber (BR), designed for agricultural tyre tread. The higher Mooney viscosities of GTR-containing compounds indicate different processability while the curing properties changed only moderately. Smaller GTR particles result in better mechanical properties than larger ones. The waterjet milling process seems to be the most perspective.
Keywords
Introduction
The increasing amount of waste tyre rubber constitutes a considerable problem worldwide. Among other methods of recycling, the physical way is of high interest, consisting of the grinding or milling of scrapped tyres and the utilisation of the produced ground tyre rubber (GTR) as a component of fresh rubber compounds for producing new tyres. Three main methods of rubber milling have been developed: ambient, cryogenic, and wet [1-18].
The ambient process involves fragmentation of the rubber in knife mills. The product is a rubber powder consisting of particles with an irregular shape and rough surface that are tenths of a millimetre or larger in size [1, 3, 4, 7, 9, 11, 14-16, 18]. The milling can also be conducted in twin-screw extruders, which is a continuous process. Berstorff's method uses a rolling mill line with ribbed rollers and a twin-screw extruder [1]. In the cryogenic process, the preliminary shredded tyres are cooled below the glass transition temperature of the rubber with liquid nitrogen and then milled in hammer mills, which is less energy-consuming than ambient grinding. The procedure creates a rubber powder consisting of very fine particles with smooth surfaces and sharp edges [1, 3, 4, 7-9, 11, 16-18]. In the waterjet process, water at high pressure (more than 200 MPa [7]) is the only milling element. Rubber strips are selectively separated from the steel cord that remains intact. Such a process produces rubber powder of high purity with fine particles having a convex–concave microstructure and highly specific surface [1, 4, 7, 14, 18, 19].
Ground rubber from scrapped tyres and other rubber waste is frequently blended with common rubbers such as NR, SBR, BR, EPDM, and NBR because of their good compatibility [1-3, 5, 6, 11, 17, 19-33]. The maximum concentration of GTR is considerably limited, however, due to the quality and strength requirements for such products [1-3, 11, 17]. Karger-Kocsis et al. [3] reported a previously deduced rule of thumb stating that each 1% of GTR results in a 1% deterioration of mechanical properties; therefore, the concentration of GTR in rubber compounds usually did not exceed 10% (w/w). Nevertheless, much higher concentrations are reported in other papers [5, 6, 11, 17, 19-33].
The type of milling process used to produce GTR influences the properties of the rubber composition containing GTR. Gibala et al. [21] reported that a compound containing ambient GTR has a higher viscosity than one with cryogenic GTR. This is attributed to occlusion of continuum rubber with sponge-like ambient GTR, whereas such occlusion is not possible with the smooth cryo-ground particles. Hassim et al. [24] analysed the influence of cryogenic and ambient GTR from truck tyres on the properties of NR and NR/BR compounds. Despite the fact that the cryogenic powder had much smaller particles, the tear strengths of the rubbers containing cryogenic GTR were inferior to the ones containing the ambient GTR. This was attributed to the less convoluted surface of cryogenic powder with sharp edges and hence lower interfacial adhesion between the GTR and rubber matrix. On the contrary, in BR-based compounds, the tear strength was higher with the cryogenic powder because of its reinforcing effect.
The particle size of GTR influences the properties of GTR-containing rubbers as well. As the particle size of GTR increases, the tensile strength, hardness, abrasion resistance, and crosslink density deteriorate whereas the swelling degree increases [5, 6, 25, 26]. The deterioration of properties is attributed to phase separation of the GTR and matrix, which is more pronounced with bigger GTR particles [6]. Conversely, the particle size hardly influences the viscosity [21] and curing properties [6] at all. Naskar et al. [5] reported that smaller GTR particles contain less rubber and carbon black and more silica and transition metals (except zinc), which results in poorer aging characteristics of GTR-containing rubber, probably because the metals catalyse its thermal degradation.
The abovementioned papers refer to experiments with various rubbers and formulations; this could lead to contradictory results in some cases. Therefore, our intent was to focus on the application of GTR in SBR/NR/BR-based compounds designed especially for agricultural tyre treads. The influence of the type of waste tyre milling process and of the GTR particle size on the properties of raw and cured rubber is investigated and discussed.
Experiment
Materials and chemicals
To investigate the influence of the type of ground tyre rubber (GTR), we used five market-available GTR samples. They were prepared via ambient milling (denoted as A), cryogenic milling (C), and waterjet process (W). Fresh rubber compounds containing GTR were mixed using a carbon black-filled rubber masterbatch based on styrene–butadiene rubber (SBR), natural rubber (NR), and butadiene rubber (BR), containing all the necessary additives except the curing system that we added later. The curing system consisted of sulphur, CBS accelerator (N-cyclohexylbenzothiazol sulphenamide), and PVI scorch inhibitor (N-(cyclohexylthio) phthalimide). Acetone, n-heptane, and deuterated chloroform of appropriate purity were used identify the polycyclic aromatic hydrocarbons in the oils contained in the GTRs (ISO 21461).
Characterisation of ground tyre rubbers
To sort the GTRs into individual fractions and investigate their particle size distributions, a sieve analysis was conducted. Based on the assumed particle sizes and availability of sieves, we used sieves of 100, 200, 315, and 400 μm. The GTRs were sieved in a sieving tower fixed in a shaking machine for 15 min with a maximum input of 350 g of GTR.
The results of the sieve analysis are depicted in Figure 1. Almost all the GTRs follow the Gaussian distribution of particle sizes. The cryogenic GTRs (C1, C2) contain large portions of smaller particles, as expected. The waterjet GTRs (W1, W2) have the distribution shifted to larger particle sizes, namely W1, which shows that more than 60% of the particles did not pass the 400 μm sieve, probably due to agglomeration of particles, which was observed during the sieving. This is attributed to the convoluted shape of the waterjet particles. Ambient GTR (A) is also expected to have convoluted particles; however, its distribution is similar to the cryogenic GTRs.
Histograms for each particle size of the ground tyre rubbers (GTR) prepared via ambient (A), cryogenic (C), and waterjet (W) milling based on a sieve analysis.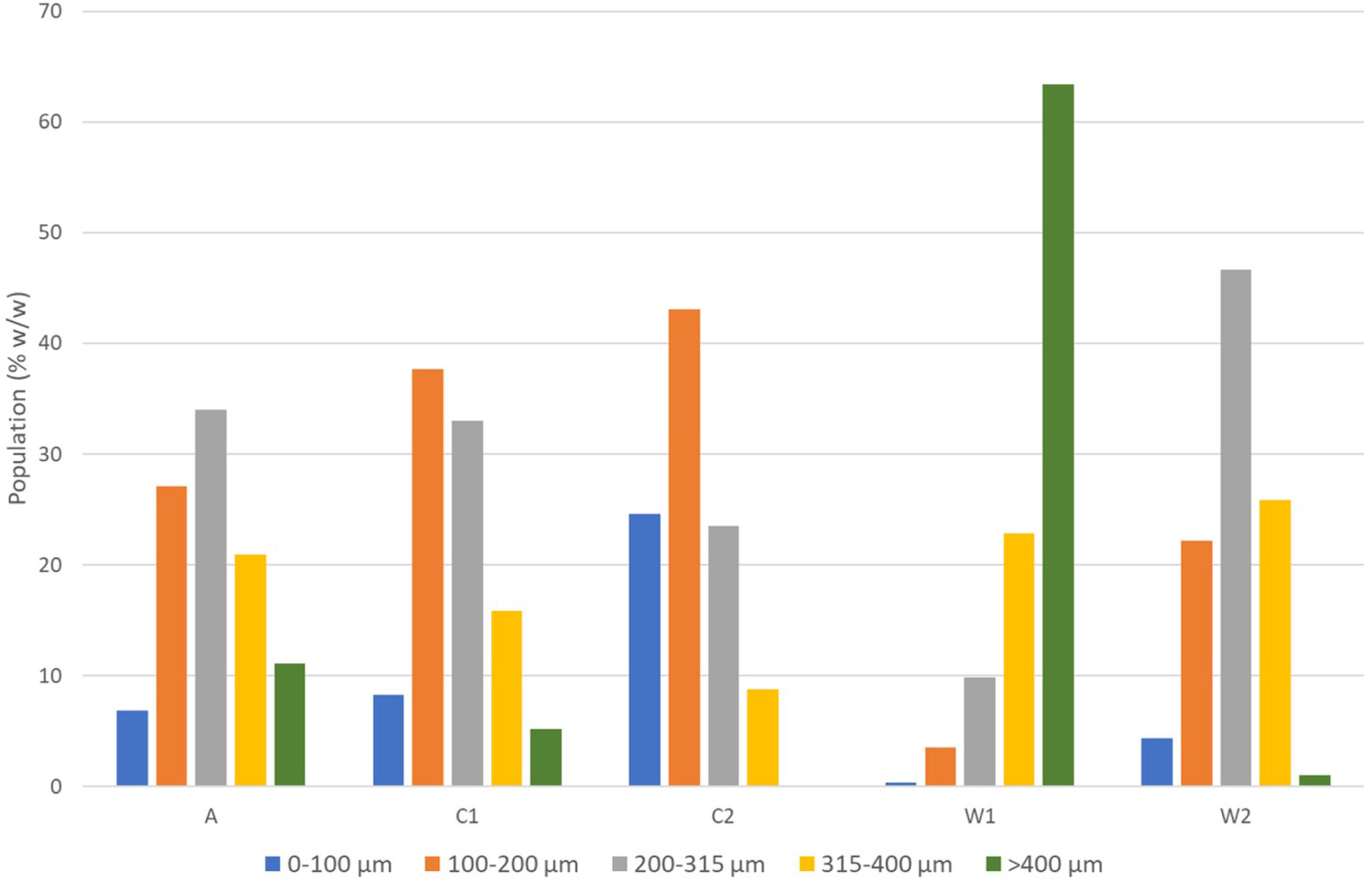
The thermogravimetric analysis (TGA) was conducted on a TGA Q500 (TA Instruments, USA). Each sample, with a mass of 10–20 mg, was heated at a rate of 10 °C min−1 in nitrogen from ambient temperature to 600°C and then in the air from 600 to 800°C. To disclose the GTR composition, we also measured the standards of the oil, butadiene rubber (BR), styrene–butadiene rubber (SBR), natural rubber (NR), and blends of these rubbers (40% NR, 30% SBR, and 30% BR).
Figure 2 shows the thermogravimetric curve of the ambient GTR (A). In the region from ambient temperature to 600°C in an inert atmosphere, a 62% decrease in weight was recorded, which is related to the thermal decomposition of the rubbers, oils, and some of the additives (e.g. antioxidants). Two peaks can be seen on the derivative curve (Figure 2), indicating that the decomposition takes place in two partially overlapping steps. The determination of the particular components (natural rubber, synthetic rubbers, oils) is somewhat complicated and will be reported below. After the change to the oxidative atmosphere, a 29% drop was observed, which was attributed to the decomposition of carbon black. A residue of 9% of the original weight was attributed to the content of incombustible matter that is mostly composed of silica (or other white fillers) and zinc compounds.
Thermogravimetric curve of the ambient GTR (A): Percent of the original weight and derivative weight change.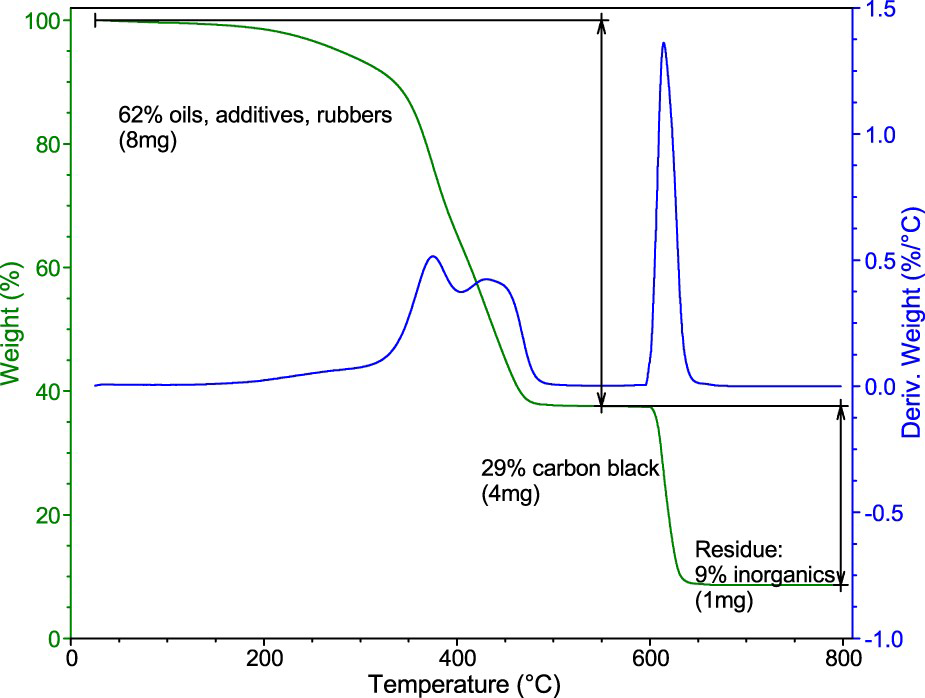
When comparing the curves in Figure 3(a), one can see that GTR W2 contains much more incombustible matter than the other GTRs. This may indicate that W2 was produced from passenger tyres while the other GTRs were milled from heavy tyres (truck, bus, agricultural vehicles, etc.) because, recently, more silica has been used in the production of the former. Furthermore, the ratio of the heights of the two partially overlapping peaks (Figure 3(b) in the region of 300–500°C) is the opposite for W2 than for the other GTRs. This may indicate differences in the composition of the rubbers and the other organic components in the GTRs. To further investigate this, we conducted a TGA of the most common neat components: that is, NR, SBR, BR, and oil. The results are depicted in Figure 4 together with the ambient GTR (A).
Thermogravimetric curves of the GTRs: Percent of the original weight (a) and derivative weight change (b). Derivative thermogravimetric curves of the neat rubbers and oil, together with the ambient GTR (A).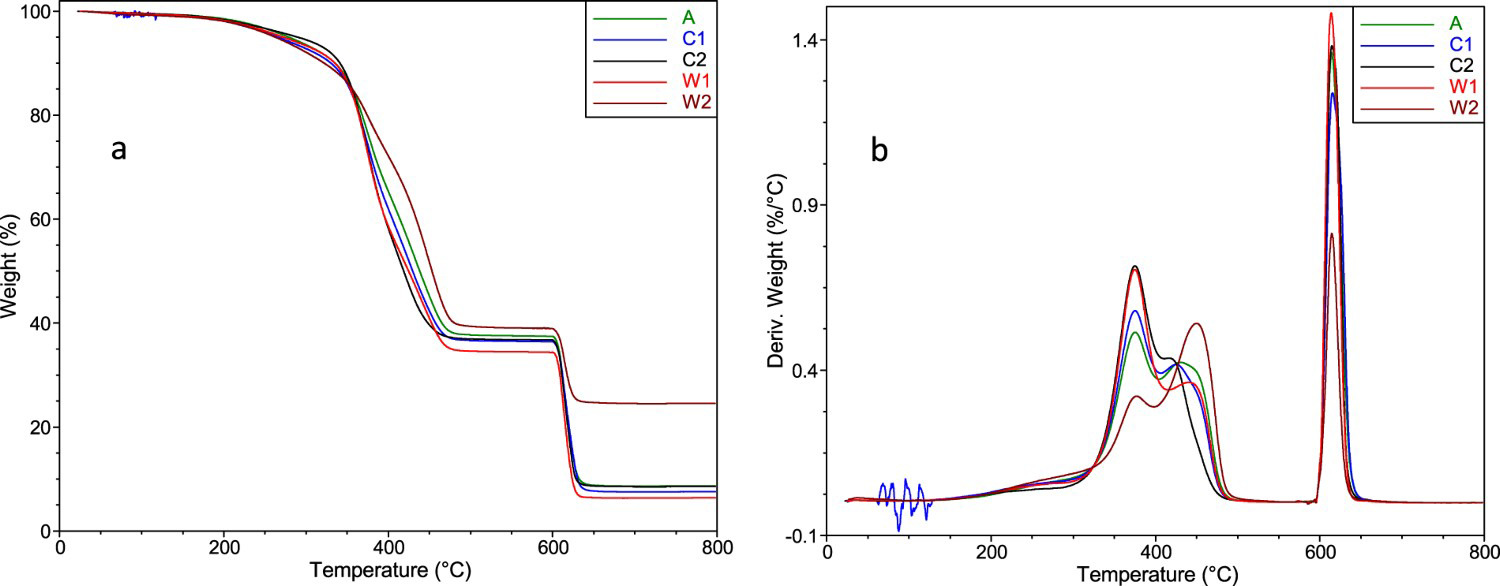
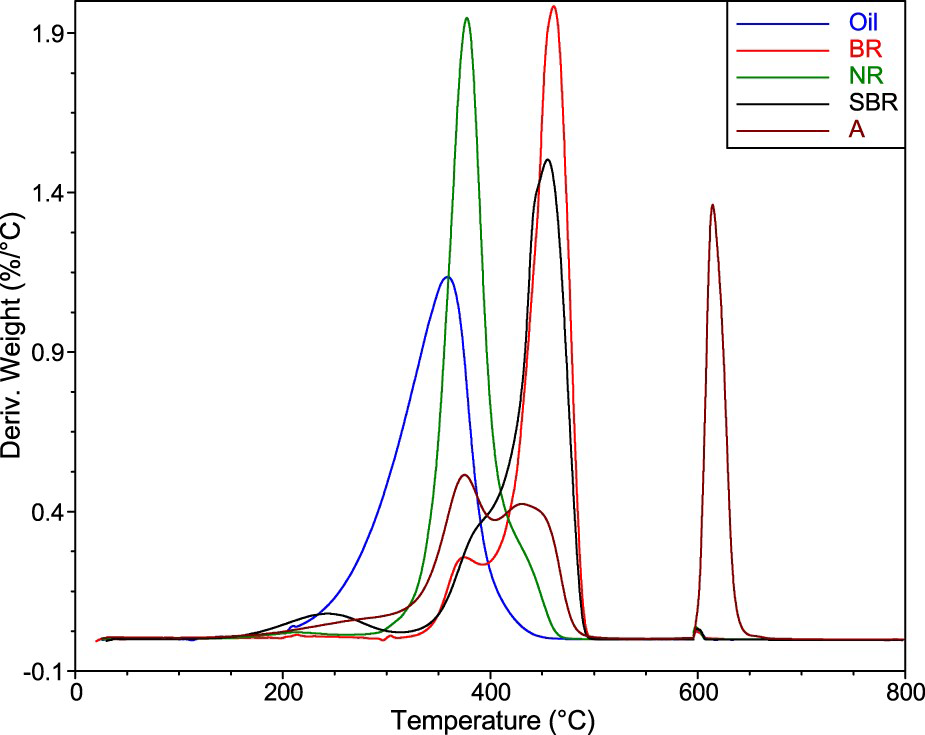
When comparing the TGA curves of the GTRs (Figure 3(b)) with those of the neat components (Figure 4), one could determine the estimated concentrations of the components other than fillers. For the example of the ambient GTR (A), as seen in Figure 4, we could assign the peaks occurring in the region of 200–500°C to either natural rubber (NR), synthetic rubbers (SBR, BR), or oil. It is obvious that all the peaks overlap with each other; therefore, it is not possible to reveal the exact composition of the GTRs from the TGA. Nevertheless, it is possible to distinguish the natural rubber from the synthetic rubbers, to some extent, as can be seen in Figure 5(a) for the model blend of rubbers (40% NR, 30% SBR, 30% BR). The relative drop of the sample mass corresponds to the formulation of the model blend. Figure 5(b) depicts an evaluation of the TGA curve and an estimation of the composition of GTR A. Note that this is only a rough estimate calculated with the help of another method: that is, the determination of the aromaticity of extender oils (described below). The procedure was as follows: The concentrations of inorganic residues and carbon black were derived from the TGA curves. The concentration of oil is the result of the determination of its aromaticity. Having omitted the components present in low concentrations (e.g. antioxidants, curatives), the rest is considered to be the sum of the rubbers. This sum was divided into natural and synthetic rubbers according to the height ratio of the respective peaks from the TGA (approximately in the region of 350–500°C). The results are reported in Table 1.
Thermogravimetric curves of the model blend of rubbers: 40% NR, 30% SBR, and 30% BR (left) and of GTR A with an assessment of the specific components (right). Estimated composition of the ground tyre rubbers (GTR, % w/w) and the aromaticity of the extender oils. from the TGA. from the determination of the oil's aromaticity according to ISO 21461. based on both methods, see the text for details.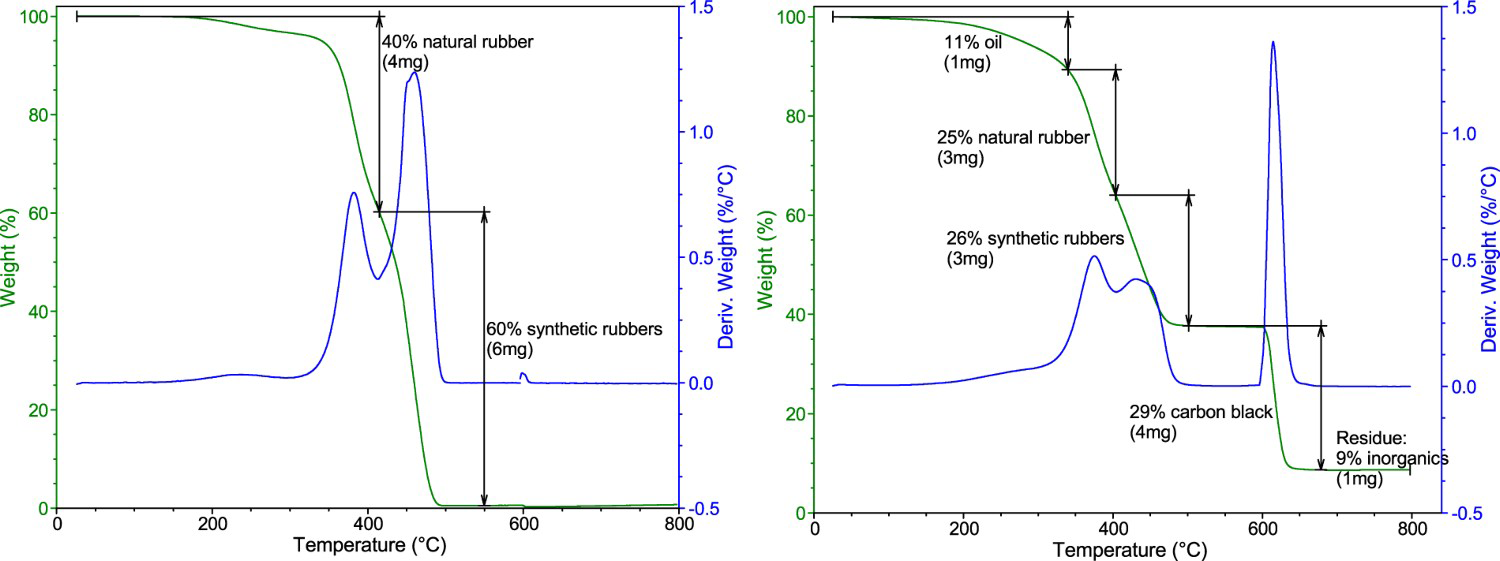

The concentrations of natural and synthetic rubbers in C1, C2, and W1 are very similar: approximately 30% and 23%, respectively. Sample A contains less natural rubber (25%). W2 is very different from the other GTRs: 15% natural rubber and 31% synthetic rubbers. This confirms the hypothesis written above that W2 was mainly made from passenger tyres while the other GTRs were made from heavy ones.
The aromaticity of the oils contained in the GTRs were determined according to ISO 21461. The data obtained from this analysis were:
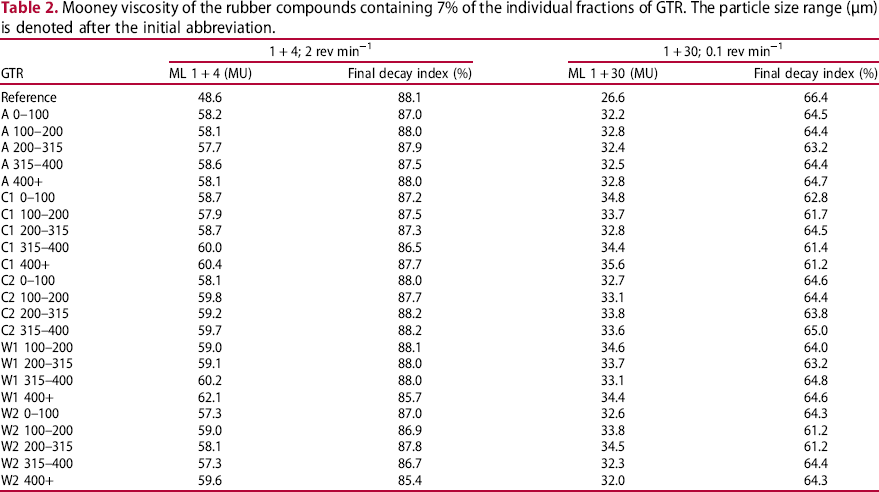
The total amount of oil in the GTR, as determined by acetone extraction. This was found to vary between 6% and 16% (Table 2). The aromaticity of the oil, as determined by 1H-NMR spectroscopy (Table 2). This was found to be independent of the method of GTR production. The results vary between 0.174 and 0.309, probably having been influenced by the type of oil used. Mooney viscosity of the rubber compounds containing 7% of the individual fractions of GTR. The particle size range (µm) is denoted after the initial abbreviation.
Compounding and testing
Each GTR fraction was mixed into a fresh carbon black-filled rubber compound with a composition adopted from a formulation for agricultural tyre treads. The compounds were mixed on a laboratory two-roll mill (diameter 150 mm, length 400 mm, friction 9: 11). The total mass of each mixed batch was 750 g, of which 91.29% was rubber masterbatch, 7.00% was GTR, and 1.71% was curatives. It was mixed as follows: The masterbatch was re-plasticised for 2 min. Then, the GTR was added and homogenised for 4 min. After that, the curatives PVI, CBS, and sulphur were added in that order, and the compound was cross-mixed for 2 min. Similarly, a reference compound without GTR was prepared as well.
The Mooney viscosity of the rubber compounds at 100°C was determined with a V-MV 3000 Variable Mooney Viscometer (MonTech) in two modes – Classic ML 1 + 4 at 2 rev min−1 and ML 1 + 30 at 0.1 rev min−1 – both of which were followed by a relaxation test after the rotor stop. The final viscosity and relaxation decay indexes were evaluated.
The course of the vulcanisation was measured with the rubber process analyser RPA 2000 (Alpha Technologies) at a temperature of 160°C, an oscillation amplitude of 0.5 degrees of arc, and a frequency of 100 cpm (cycles per minute) for a period of 30 min.
The specimens for mechanical testing were prepared by compression moulding using the laboratory hydraulic press LabEcon 300 (Fontijne Presses) as follows: For tensile tests, sheets (150 mm × 150 mm × 2.5 mm) were moulded in a flat mould for (
Ten dumb-bell specimens (type 2) were punched from each moulded sheet. Then, the specimens were subjected to a tensile test according to ISO 37 on an Instron 3365 tensile tester equipped with a long travel extensometer. The tensile strength, elongation at break, and 100% and 300% moduli of the cured compounds were evaluated. The Shore A hardness of the cured compounds was measured according to ISO 48-4 with a manual durometer (Mitutoyo) using residual parts of the sheets layered on top of each other to reach a 6 mm thickness. The hardness value was registered 15 s after the start of the indentation.
The abrasion resistance test was conducted on a drum tester (Polymertest Zlin). The test is based on ISO 4649. A cylindrical specimen was secured in a sample holder, which was pushed towards a rotating drum covered with emery paper. The sample was moved alongside the drum axis during the test. The total abrasion distance of the sample on the drum was 40 m. Apart from the standard, the abrasion resistance was calculated as the mass difference of the sample before and after the test, as the average from three measurements.
Results and discussion
Mooney viscosity
The results of the Mooney viscosity measurements are reported in Table 2. There is a considerable difference between the reference compound (without GTR) and those containing GTR: GTR increases the Mooney viscosity because it contains crosslinked gel, thus restricting the rubber chain mobility. This may indicate worse processability of GTR-containing compounds. This is in agreement with the literature [17, 21, 23, 25]. However, the processability can be improved by plasticisers, processing aids, or longer mixing. For GTR-containing compounds, the Mooney viscosity and the Mooney decay indexes fluctuate with minor changes and no clear trend.
Curing properties
Typical vulcanisation curves are depicted in Figure 6; the curing data are summarised in Table 3. Compared with the reference compound, the minimum torque ( Vulcanisation curves of the compounds containing size fractions of GTR C2 and of the reference compound. The particle size range (µm) is denoted close to the ‘C2’ abbreviation. Curing properties of the rubber compounds containing 7% of the size fractions of GTR prepared via ambient (A), cryogenic (C), and waterjet (W) milling. The particle size range (µm) is denoted close to the initial abbreviation. Note: 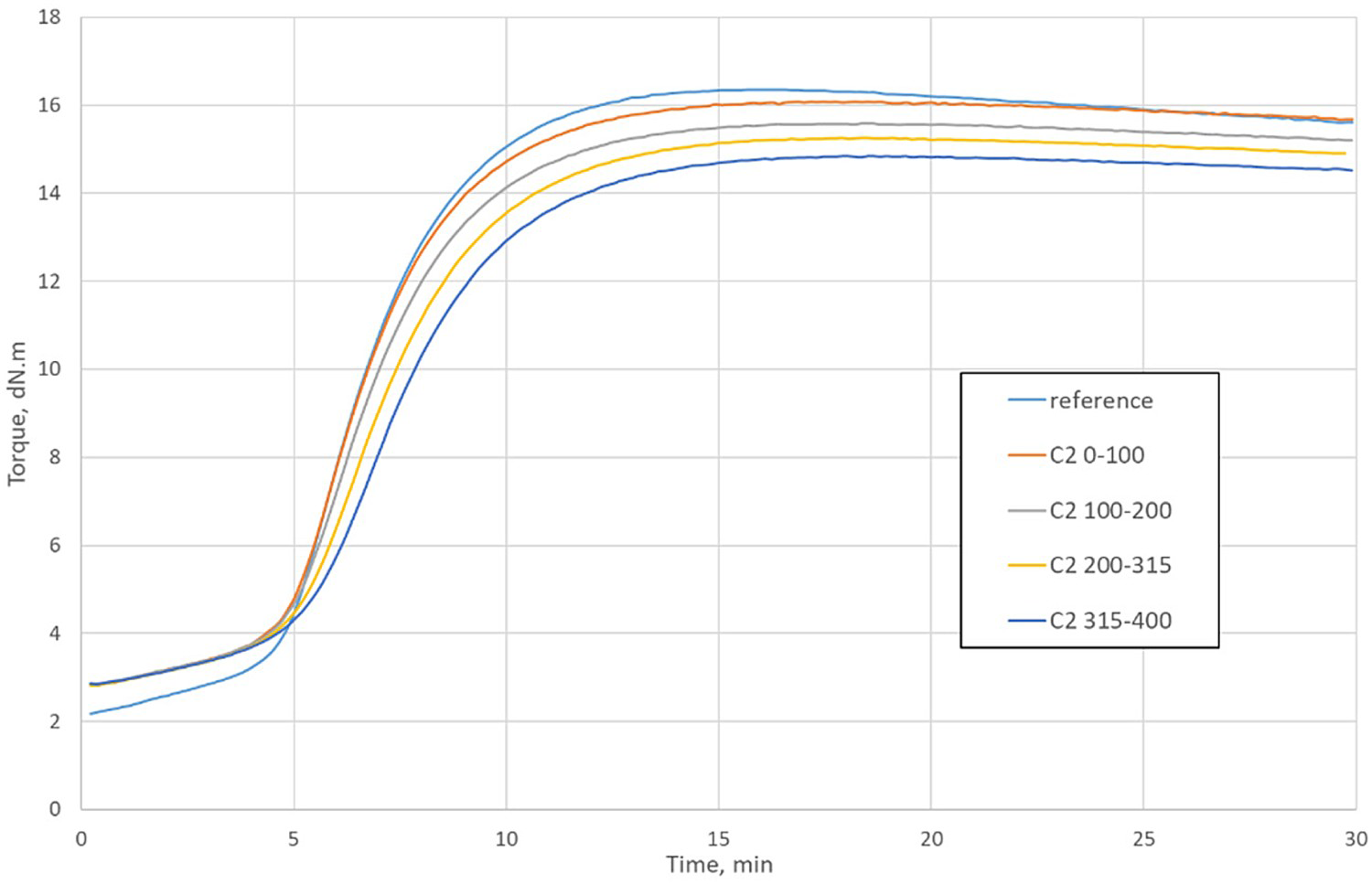
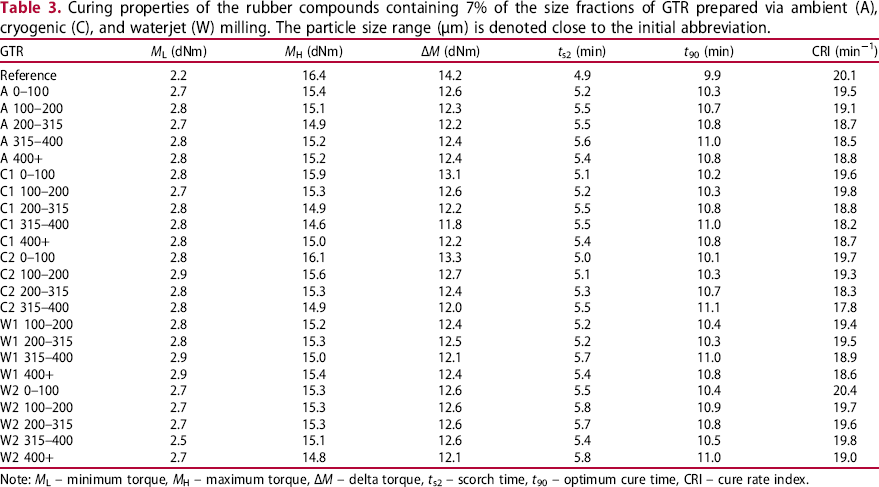
Furthermore, there is a considerable drop in the maximum torque (
Unlike in many other studies, [2, 6, 17, 20, 23-25, 27, 28], the scorch time (
Mechanical properties
The mechanical properties of the cured rubbers containing 7% of the size fractions of GTR prepared via ambient (A), cryogenic (C), and waterjet (W) milling. The particle size range (µm) is denoted close to the initial abbreviation. Each result is reported as an average ± standard deviation.
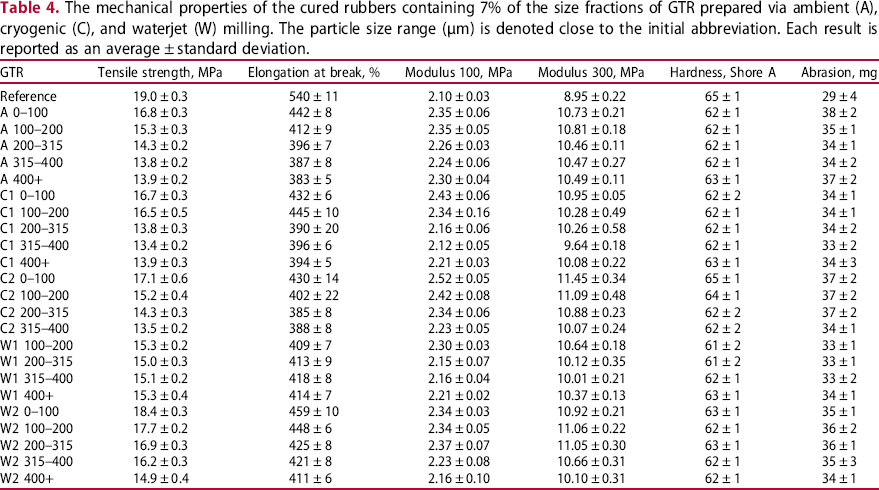
The 100 and 300 moduli increase after GTR is added. As the particle size increases, both moduli usually decrease with small differences. The highest hardness (65 ShA) was achieved for both the reference compound and C2 0-100 μm. The differences in the hardness data among the compounds are small (2–3 Shore units) and thus not relevant with respect to the size of the Shore indenter and the size of the GTR particles.
The abrasion resistance was evaluated as the mass difference of the original and abraded sample: The higher the number, the worse the abrasion resistance of the rubber. The best abrasion resistance, however, was achieved with the reference compound (29 mg), showing the highest data deviation. The GTR-containing compounds show much higher abrasion (33–38 mg), that is, lower abrasion resistance. This trend is in agreement with some papers [6, 23-25]. The variation of the abrasion data across the milling processes and particle size is small and does not show any clear trend with respect to data variation and somewhat limited interpretation of the method results.
Conclusions
Five grades of ground tyre rubber (GTR) produced via three different milling methods – that is, ambient, cryogenic, and waterjet – were characterised. A thermogravimetric analysis revealed that the GTRs were based on both natural and synthetic rubber and were filled with both carbon black and mineral fillers. The GTRs were sieved into five size fractions. The cryogenic (C1, C2) and ambient (A) GTRs contained high portions of small particles (100–200 µm) while the waterjet GTRs showed the distribution shifted to a larger size, probably due to particle agglomeration. Each GTR size fraction was added (7% w/w) to a rubber compound based on virgin SBR/NR/BR for production of agricultural tyre treads. GTR increases the Mooney viscosity by about 20%, showing no clear dependence on the particle size or type of GTR. GTR influences the curing properties with minor dependence on the particle size and the type of GTR: GTR increases the minimum torque (+26% on average), scorch (+10%), and optimum cure time (+8%) while it decreases the maximum torque (−7%) and delta torque (−12%). For cured rubbers containing GTR, the tensile strength and elongation at break are lower while the modulus is higher than those of the reference compound. The mechanical properties are more sensitive to the size of GTR particles: The tensile strength, elongation at break, and moduli mostly decrease as particle size increases. The best tensile strength (18.4 MPa) and elongation at break (459%) of the GTR-containing compounds was achieved using the waterjet GTR W2 0–100 µm (reference compound: 19.0 MPa and 540%). For most GTRs, the difference in tensile strength between the finest (0–100 μm) and coarsest (>400 μm) particles is about 3 MPa. The hardness of the GTR-containing rubbers is 1–4 ShA lower and the abrasion resistance deteriorated more than that of the reference, showing no clear dependence on particle size.
Footnotes
Acknowledgements
The financial support received from the Ministry of Education, Youth and Sports (Ministerstvo Školství, Mládeže a Tělovýchovy) via specific university research grant No. 21-SVV/2020 is gratefully acknowledged. The authors would also like to thank Dr Veronika Rippelová for providing the sieves and Prof. Richard Hrabal for the NMR analysis.
Disclosure statement
No potential conflict of interest was reported by the author(s).
