Abstract
A reprocessable elastomeric vulcanisate based on Epoxidised Natural Rubber with 50 mol-% epoxide content (ENR-50) was evloved by applying a self-assembled network based on thermochemically exchangeable ester crosslinks to the system. Hydrolysed Maleic Anhydride (HMA) as crosslinking substance in the presence of 1,2-DiMethylImidazole (DMI) as esterification accelerator and Zinc Acetate Dihydrate (ZAD) as transesterification catalyst was employed to generate the exchangeable ester crosslinking system. A sulphur-cured ENR-50 vulcanisate possessing a permanent sulphide crosslinking network was prepared as reference. Based on the results from cure characteristic and chemical structure analyses, the ENR-50 crosslinked with HMA behaves as a dynamic network because of a transesterification reaction catalysed by ZAD, promoting an exchangeable crosslinking network in the system. This dynamic network contributes to an intermolecular rearrangement of the ester crosslinking bonds at elevated temperatures, enabling interfacial self-adhesion and so reprocessability of the vulcanisates. The obtainable vulcanisates can be reprocessed, yielding relatively high retention of mechanical properties compared to their pristine counterpart. The interfacial self-adhesion and reprocessability of the vulcanisates have shown to significantly be improved with a higher loading of ZAD and elevated temperatures. This concept essentially shows a prospect towards developing e.g. novel recyclable and self-healing systems for elastomers.
Introduction
Thermoset polymers are commonly distinguished by the presence of chemical crosslinks between polymer molecules generated through a vulcanisation process that yields high mechanical properties, thermal stability and solvent resistance to the final materials. Crosslinked rubbers or vulcanisates are classified as thermoset polymers with excellent elastic properties; they are widely used in a variety of applications. However, a conventional vulcanisation reaction generates a permanent network in elastomers, making the obtainable vulcanisates impossible to be reprocessed or reshaped by a simple approach, unlike thermoplastics. This is one of the major challenges for waste management and the circular economy [1, 2].
Advancing materials towards intermolecular auto-reparation to obtain the so-called ‘self-healing materials’ enlighten polymer scientists to enhance their efforts in evolving innovative vulcanisates possessing a reprocessability characteristic [3]. To date, there is still limited in practically reprocessing/recycling the crosslinked rubbers, in particular in terms of investments and final properties of the materials. Thus, most of the rubber wastes have been ended up in landfills [4]. Therefore, acquiring a system that makes rubber vulcanisates reprocessable would be of paramount industrial and ecological benefits.
Epoxidised Natural Rubber (ENR) is one of the reactive polymers, as it possesses double bonds and epoxide groups which are reactive towards intermolecular crosslinking reactions. With the double bonds, the ENR can be vulcanised using sulphur, creating sulfidic crosslinks between the rubber molecules. While, peroxide can also be used as a crosslinking agent as it can vulcanise the hydrocarbon molecules, forming carbon–carbon crosslinks between the rubber molecules [5, 6]. Similar to an epoxy resin, the ENR can be crosslinked at the epoxide groups, called oxirane rings [7], most often by amine functional groups [8], carboxylic acids [9] and anhydrides [10, 11] in the presence of a suitable catalyst, for instance, bisphenol or phenol derivatives [12].
Regarding the modification of ENR by Maleic Anhydride (MA), there are only a few studies reported in the literature. ENR can be modified by MA in a latex state [13]. Results show that the particle size and zeta potential of the maleated ENR particles increase, due to the change in chemical composition on the ENR particle surface. However, the grafting percentage is decreased with increasing the MA content, attributed to side reactions taken place such as chain scission and chain transfer reactions. For the properties of ENR with the presence of maleic anhydride, tensile strength and thermal stability of the vulcanisate are improved, owing to a chemical crosslinking reaction between MA and epoxide groups; the ester crosslink was postulated for this mechanism [13]. In general, dicarboxylic acids can react with ENR to form
It has been reported that a catalyst significantly activates the reversibility of the esterification reaction, called ‘transesterification’. Zinc acetate was used to promote the transesterification kinetics at a high temperature, which is beneficial towards enabling the exchangeability of the ester crosslinks [9, 21-23]. With the presence of a proper transesterification catalyst, the ester crosslinking networks could provide a crosslinked ENR with vitrimer-like properties. The vitrimers are a new class of organic materials crosslinked through exchangeable covalent bonds; this concept was introduced in 2011 by Leiber et al. [22] This network is able to exchange its topology via a bond-interchange mechanism, giving a vulcanisate with unique intermolecular characteristics, including thermally malleability and self-repairability of the network at higher temperatures. Continuous exploration on this interesting subject would be of importance to elucidate, in more depth, the mechanisms involved. Moreover, it will provide further support on the performance and pragmatic applications of this system in thermosets, in this case, vulcanised elastomers.
In the present study, ENR was vulcanised using hydrolysed maleic anhydride, i.e. dicarboxylic acid, which can generate ester crosslinks and possibly yield a thermochemically exchangeable network with the presence of a transesterification catalyst. Reprocessability of the vulcanisates was investigated by remoulding the ground vulcanised ENRs to prepare new vulcanisate samples prior to their property determinations. The experiment was designed to investigate the effects of ZAD contents on the interfacial self-adhesion and reprocessing properties of the vulcanisates. The thermochemically exchangeable ester crosslinks which would influence the reprocessability of the vulcanisates were verified. The main intended objective of the present study is to validate the exchangeable ester network on promoting reprocessibility of ENR vulcanisates. The outcomes from this work might provide further inspiration for evolving a novel system for rubbers to promote good mechanical properties with a combination of recyclability and self-healability. This concept might be applicable for various applications such as wearable electronics, medical devices, soft robotics, engineering rubber goods, high-performance tyres, and so on.
Experimental
Materials
Epoxidised Natural Rubber containing 50 mol-% epoxide groups (ENR-50) was purchased from Muang Mai Guthrie PCL, Thailand. Kaolin clay was purchased from Siam Chemicals Co., Thailand. Maleic Anhydride or MA (99%, Sigma-Aldrich, China), 1,2-DiMethylImidazole or DMI (98%, Alfa Aesar, China) and Zinc Acetate Dihydrate or ZAD (Zn(OAc)2·2H2O as its empirical formulation, analytical reagent, Ajax Finechem, Australia) were used without further purification. Zinc Oxide or ZnO (Thai-Lysaght Co. Ltd., Thailand), stearic acid (Imperial Industrial Chemicals Co. Ltd., Thailand), DiPhenyl Guanidine (DPG) (Vessel Chemical Co. Ltd., Thailand), and N-Tert-Butyl-2-BenzothiazoleSulfenamide or TBBS and sulphur (all from Siam Chemicals Co. Ltd., Thailand) were employed as additives for rubber compounds.
Sample preparation
Preparation of dicarboxylic acid by hydrolysing MA
Maleic Anhydride (MA) was first hydrolysed using distilled water before a modification process in order to obtain the reactive dicarboxylic acid in a form of maleic acid. A commonly accepted mechanism proposed in the literature suggests that an anhydride ring is susceptible to be opened in excess water [24, 25], yielding a monoester and a carboxylic acid as depicted in Figure 1(a). 20 g of MA in briquette form and 30 ml of distilled water were introduced in a 100 ml capped laboratory flask. The flask was placed in a warm water bath at 70°C for 2 h until the anhydride melted and the formed globules disappeared. The anhydride was then reacted to form a diacid which is soluble in hot water. Then, the bottle was removed from the water bath and left to cool down to room temperature. After that, the bottle was immersed in an ice-water bath for 45 min until some crystals were formed because the maleic acid is insoluble in cold water. The dry crystals of Hydrolysed MA (HMA) were collected on a Hirsch funnel and kept in a tightly closed container for further purposes.
Proposed reaction mechanisms: (a) hydrolysis of maleic anhydride under a thermal treatment with a small amount of moisture; and (b) crosslinking of ENR by hydrolysed maleic anhydride.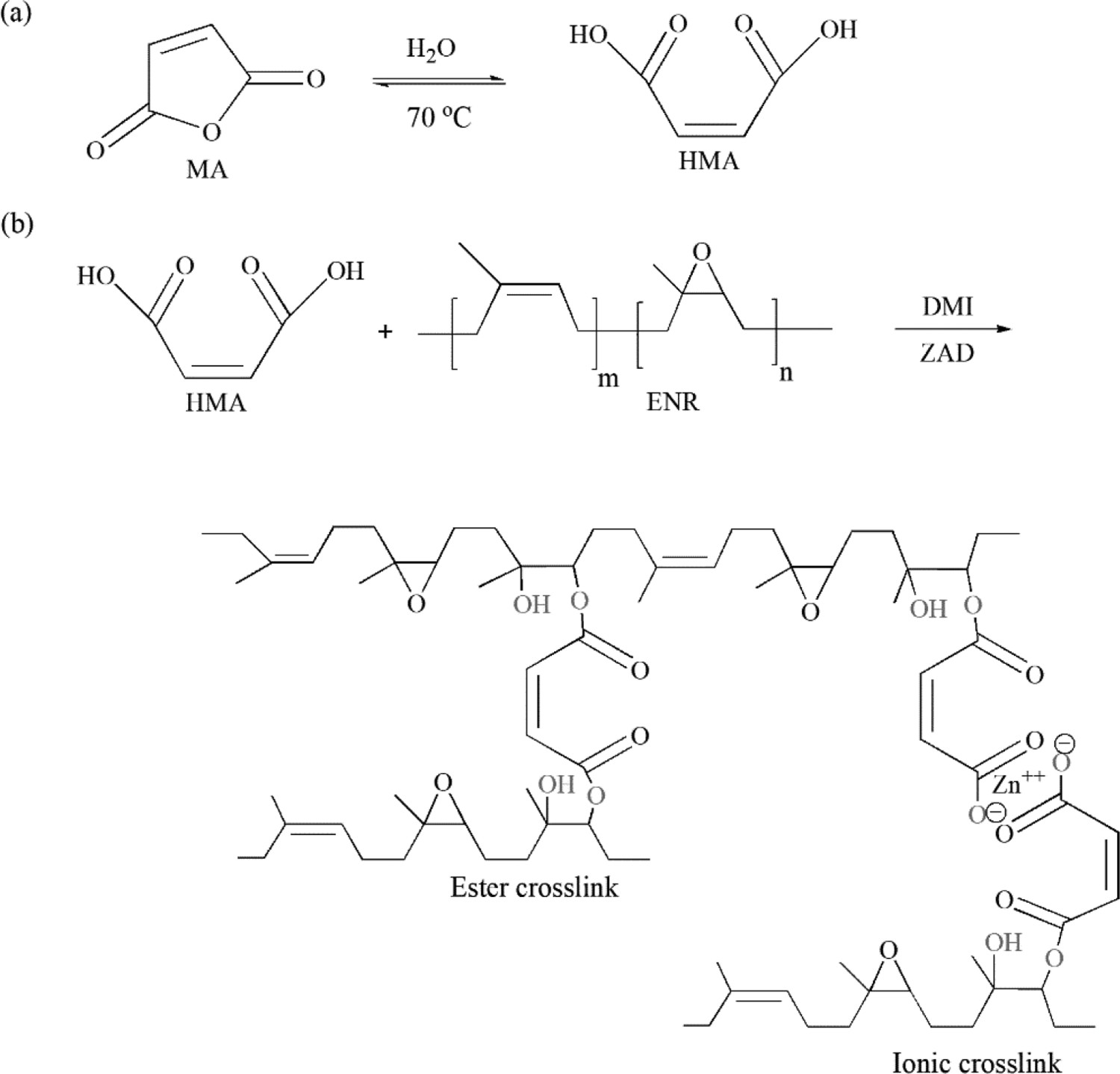
Compound preparation
Formulations of the mixes used in the present investigation.
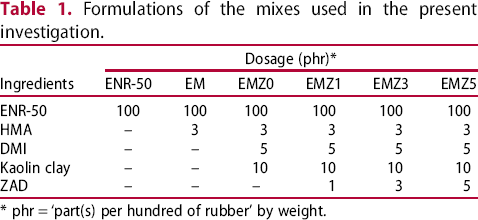
* phr = ‘part(s) per hundred of rubber’ by weight.
Property characterisations
Cure characteristics
Cure characteristics of the samples were determined using a Moving Die Rheometer (MDR2000, Alpha Technologies, USA) according to ASTM D5289 at a test temperature of 180°C and time of 60 min unless stated otherwise. Optimum cure time (
Chemical structures
The chemical structures of pure ENR-50 and HMA-crosslinked ENR-50s were analysed using a Fourier-Transform InfraRed spectroscopy or FTIR (Bruker Tensor 27 FT-IR, Bruker Co., UK). The spectra were recorded between 4000 and 400 cm−1 with 256 scans and a resolution of 4 cm−1. The spectrometer was equipped with an ATR accessory at room temperature with controlled humidity.
Tensile properties
The tensile test was carried out using a universal testing machine (Instron 3365, Instron Co., USA) with a crosshead speed of 100 mm min−1 according to ASTM D638 at room temperature with no humidity control. The specimens were prepared with a die-cut type V. Five specimens of each vulcanisate were tested, and the median values were reported.
Reprocessability analysis
Cured rubber sheets were ground into small grains using a mortar grinder (Fritsch Pulverisette 2, Fritsch GmbH, Germany). Liquid nitrogen was used to conduct the cryogenic grinding at a temperature below the glass transition temperature of a sample [26]. The obtained ground rubber of each vulcanisate was pulverised on a two-roll mill for 1 min [27] and finally heated at 180°C for 40 min in a compression moulding to obtain a reprocessed rubber sheet. The resulting sheets were determined for their property retention.
Interfacial self-adhesion assessment
A previously described setup and procedure for the interfacial self-adhesion measurement were adopted to prepare the adhered samples in this investigation [17]. Briefly, a reprocessed rubber sheet was cut into a rectangular shape of 100 × 40 × 1.5 mm. Two sheets of the same vulcanisate were attached together and tightened in a homemade device that has a compression zone to pressurise the half-length (i.e. 50 mm) of the two-layer sheet together. As a result, the obtainable sample has the adherent zone for half of the sample length and another half is a non-adherent or free zone. In the homemade system, the intensity of the compression was kept constant for all samples with an applied strain at around 30% by controlling the moving of the upper part. This system was then left in a vacuum oven hot-air at 180°C for 3 h. The final samples were cooled down to room temperature and rested for 1.5 h, and five samples from each vulcanisate with a size of 100 × 6 × 1.5 mm were prepared for a T-peel measurement.
For the measurement protocol, a T-peel sample was symmetrically peeled apart by pulling the free zone ends in the opposite direction at room temperature without a humidity control using a universal tensile testing machine (Instron 3365). The peel strength (load (N)/width (m)) was assessed in the average plateau region of a peel stress–strain curve. The detachment energy (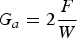
Where
In addition, the already adhered samples were re-adhered after their peeling test, and the interfacial self-adhesion of the re-adhered samples was assessed. This experiment was carried out for the compound EMZ5 to validate whether the vulcanisate can be reprocessed multiple times. The measurement was conducted for four cycles, and the detachment energy was reported as a function of the repeating cycle.
Results and discussion
Acceleration of dicarboxylic acid crosslinking by dimethylimidazole
Figure 2 and Table 2 show the cure characteristics of ENR-50 based compounds with and without DiMethylImidazole or DMI, as well as with different quantities of Zinc Acetate Dihydrate or ZAD. Comparing between the ones designed to reflect the impact of dimethylimidazole as an accelerator for HMA cure, i.e. EM versus EMZ0 compounds, the results clearly indicate that the optimum cure time ( Rheometer cure curves at 180°C of the investigated compounds based on HMA-crosslinked ENR-50 with different concentrations of zinc acetate dehydrate. Cure characteristics of the investigated compounds vulcanised at 180°C.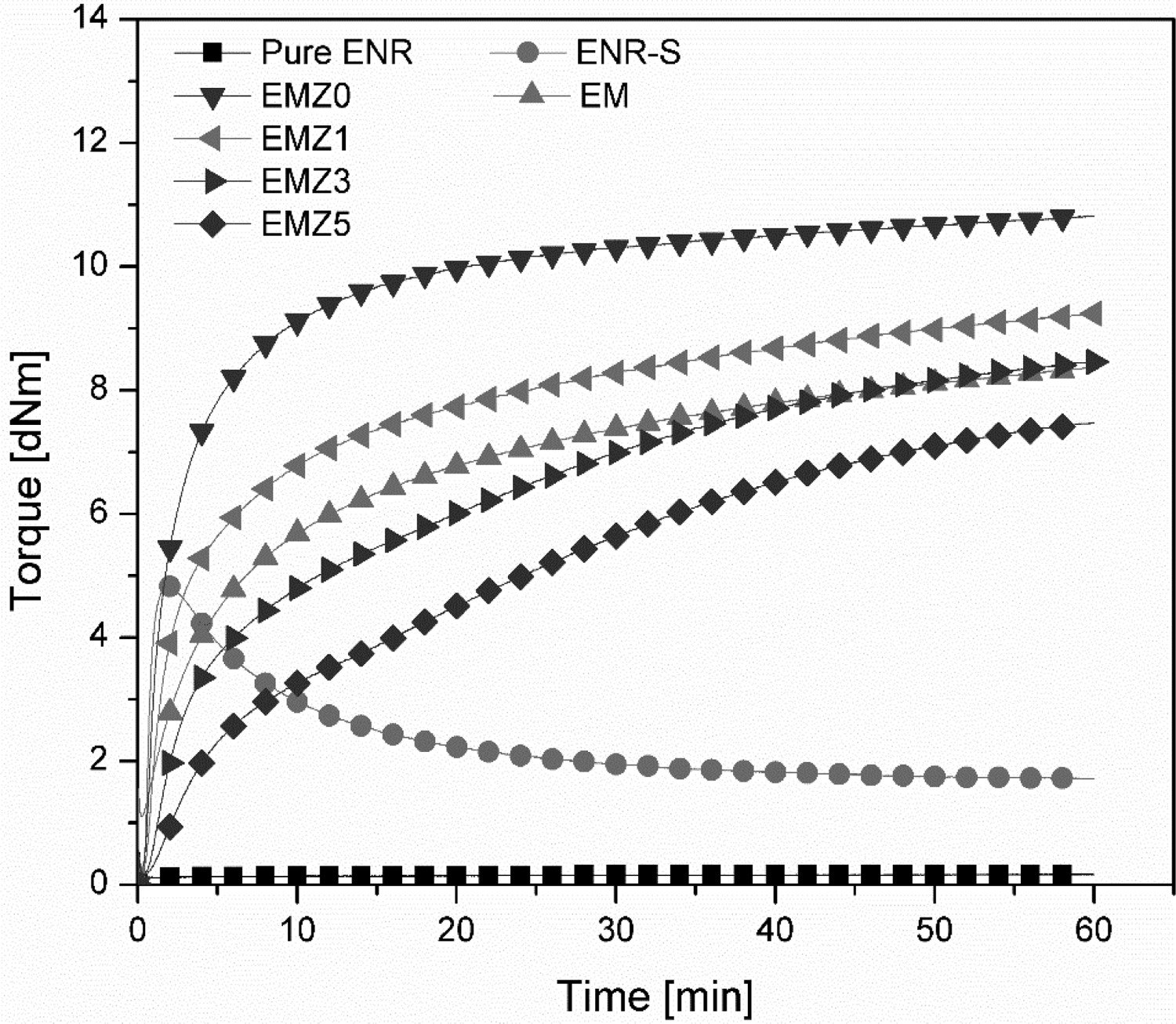

The cure kinetics of HMA-cured ENR-50 compounds are by far slower than that of the conventional sulphur-cured system: see
Overall, the cure characteristics shown in Figure 2 illustrate that HMA is clearly able to cure an epoxidised natural rubber, i.e. ENR-50, with the aid of DMI as cure accelerator. The crosslink created through this mechanism is a strong covalent bond which is undesired for the reprocessable system since the thermoreversible crosslinks are literally required. Interestingly, it has been reported that this ester bonding can thermochemically be modified to transform to a reversible linkage via a bond exchangeable process. An effective substance for this purpose to promote the transesterification reaction by shuffling the ester crosslinks to be exchangeable is ‘Zinc Acetate Dihydrate or ZAD’ [9, 17].
Enabling thermochemically exchangeable crosslinks by zinc acetate dihydrate
Considering the effect of ZAD with its empirical formulation Zn(CH3COO)2.2ּH2O (see Figure 2 and Table 2), the
In this system, as discussed in a previous paragraph, the initial rubber network is formed by an irreversible reaction between hydroxyl groups from the opened epoxide structure and carboxylic acid groups through an esterification reaction, i.e. an alcohol in reaction with an acid. In addition, based on the literature, zinc acetate dihydrate can function as a transesterification catalyst [31, 32]. Therefore, with the addition of zinc acetate dihydrate to an HMA-cured compound, the pendant hydroxyl groups and ester crosslinks undergo a rapid transesterification reaction at elevated temperatures, shuffling the bonds and enabling the network rearrangement. Hence, the desired exchangeable ester crosslinks are achievable. However, with this unique characteristic of ZAD, the crosslinking reaction of HMA is retarded and becomes more pronounced in marching modulus, as can be seen in Figure 2. Regarding the marching modulus, this mechanism will be discussed in a following paragraph.
To verify the creations of the aforementioned exchangeable crosslinks, a Fourier Transform Infrared Spectroscopy (FTIR) was performed to check the newly generated bonds/functionalities of the modified compounds. From Figure 3, the FTIR spectra provide an additional indication for the proposed crosslinking mechanism of ENR-50 molecules by HMA with the presence of ZAD. The spectrum of ENR-50 shows the presence of epoxide groups at 875 and 1251 cm−1 corresponding to the C–O–C stretching vibrations of oxirane rings [6, 33]. During the mixing and curing processes, epoxide groups can be opened transforming into hydroxyl groups due to the acidic and high-temperature conditions [6]. A broad peak in the region of 3250–3600 cm−1 on the spectrum of cured EMZ5 is observed, suggesting the presence of hydroxyl groups [34]. The peak at 1098 cm−1 assigned to the C–O–C stretching vibration of the ester [35, 36]. Whereas the peaks at 1008 and 1030 cm−1 have never been discussed before in the literature, but some researchers mentioned that several peaks appearing in the region of 1000–1200 cm−1 can be assigned to the moieties of i.e. –OH, C–O and ether groups generated from the ring-opening of epoxide groups [6, 36]. Note that, in this study, to minimise some errors presented due to the thickness variation of the samples, the FTIR spectra were normalised; the band due to the methyl functionalities vibrated at 1378 cm−1 was used as a normalising factor since no change is expected for the band of these groups [37].
FTIR spectra of pure ENR-50, uncured and cured EMZ5 compounds.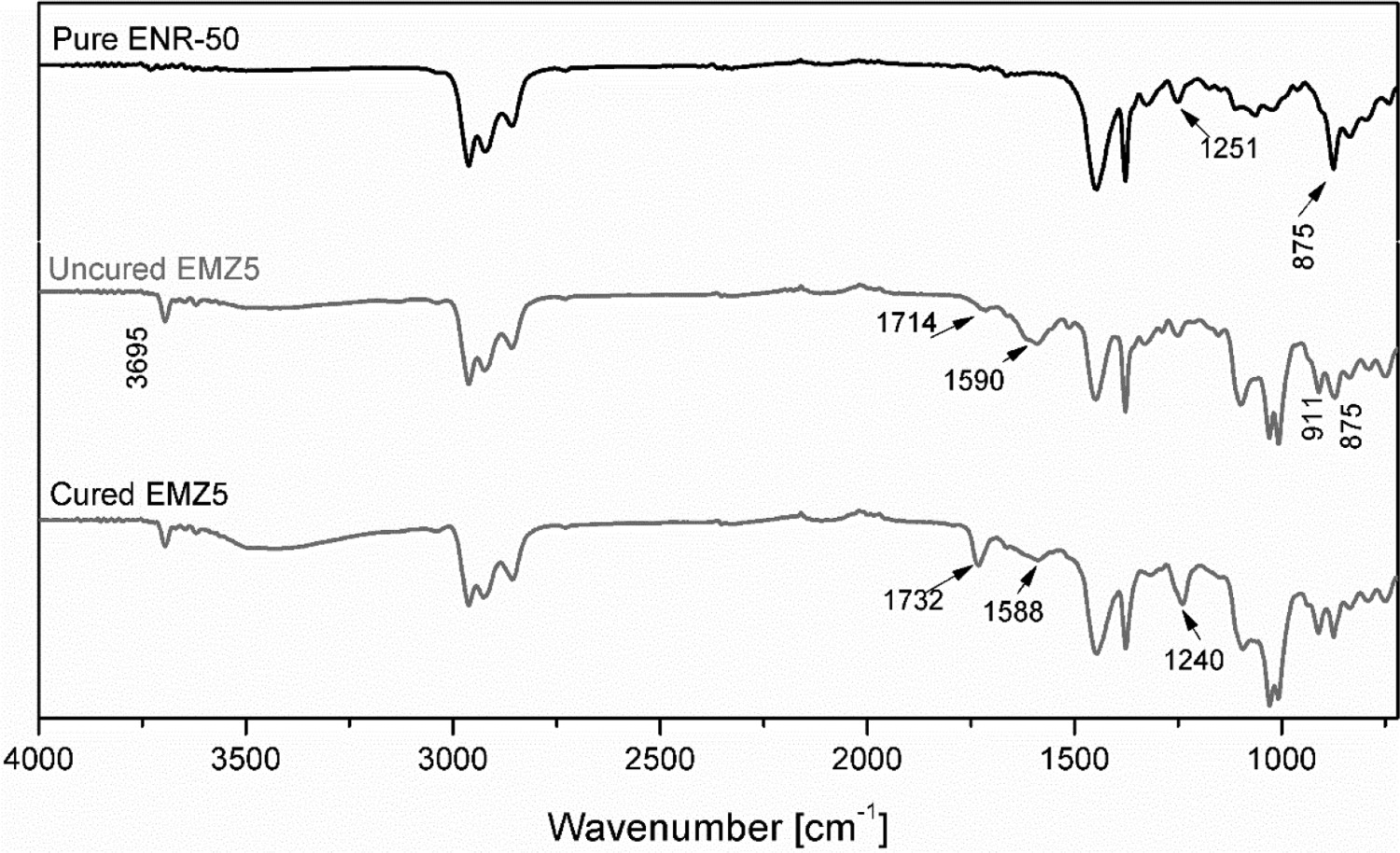
The hydroxyl groups from the ring-opened structure of epoxide groups further react with the carboxylic acid groups to generate the ester bonds via a transesterification reaction (Figure 1(b)). A new absorbance band at 1241 cm−1 emerged on the spectrum of cured EMZ5 is assigned to the C–C–O stretching vibrations due to the new ester bonds formed. The presence of carboxylic acid groups caused by the hydrolysis process of MA as evidenced by the appearance of the peak at 1714 cm−1 on the spectrum of uncured EMZ5 is due to the –C=O stretching vibrations of hydrolysed MA. This functionality can be formed by the reaction of the anhydride functional groups with a small concentration of moisture [38-40]. Besides, a new peak at 1732 cm−1 is associated with the C=O stretching vibrations of the ester functionality [41]. It is important noticing that the intensity of the peak of epoxide groups appears to be lessened (but not completely disappeared), on the spectrum of cured EMZ5. This indicates that the epoxide groups do not react completely with HMA. Moreover, an occurrence of the band at 1590 cm−1 on the spectrum of uncured EMZ5 is due to the absorbance of the associated carboxylate groups derived from HMA, suggesting the presence of neutralised carboxylic groups on the molecular structure [42, 43]. After curing, the peak intensity at 1590 cm−1 is significantly lessened due to the formation of ester bonds as discussed earlier, and it is slightly shifted to 1588 cm−1 that might be attributed to the hydrogen bonding interactions between the excess amount of blended components – the remaining free acid pending groups may form hydrogen bonds with unreacted epoxide groups [5] or with the excess DMI [30].
Nevertheless, the cure curves of the samples containing zinc acetate dihydrate or ZAD exhibit a continuous increase in the elastic modulus, i.e. marching modulus, which is more pronounced when increasing the ZAD content, as can be seen in Figure 2. This might indicate that these samples were not fully crosslinked after curing for 60 min. Therefore, in order to reach a plateau cure state, the cure analysing time of EMZ5 was extended to be 3 h instead of 1 h at 180°C. The obtained cure curves are compared in Figure 4. It is clear that adding ZAD into the compound substantially delays the vulcanisation rate, as the optimum cure time ( Cure curves of EMZ5 compound from a rheometer cure analysis at 180°C for 1 and 3 h.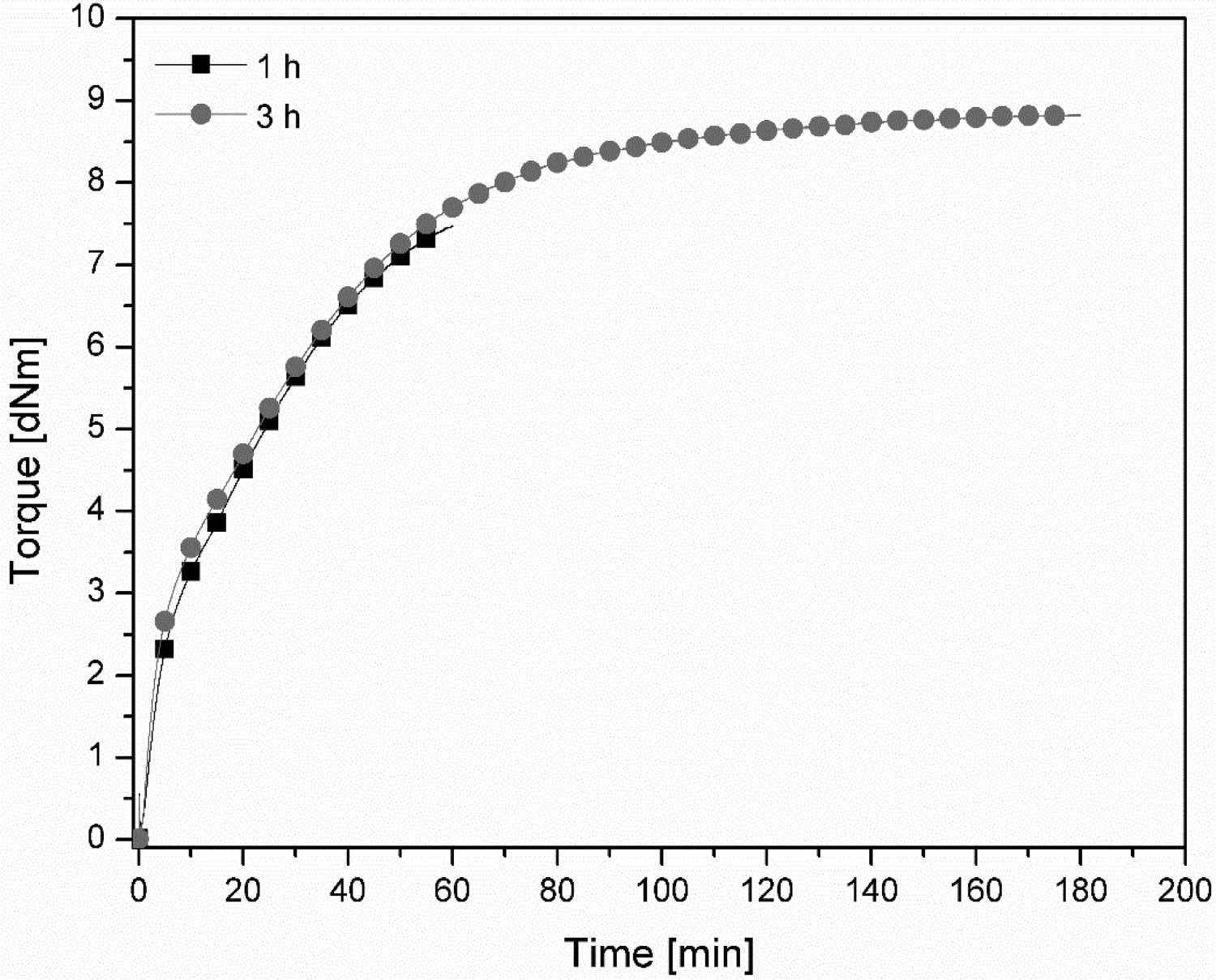
The degree of vulcanisation was also confirmed using an FTIR spectroscopy: see Figure 5. The band of epoxide groups at 875 cm−1 is significantly lessened, and the intensity of the –OH peak at 3450 cm−1 increases for the one cured with a longer cure time (i.e. 68 min). This is attributed to the ring-opening of epoxide groups on ENR molecules. The FTIR spectrum might imply that a certain extent of the hydroxyl groups originated from the ring-opening of epoxide groups still remain unreacted. Adding more amounts of dicarboxylic acid with the presence of dimethylimidazole should increase the cure rate of the compound. Further investigation on this issue would be suggested.
FTIR spectra of EMZ5 compound cured at 180°C for 43 and 68 min.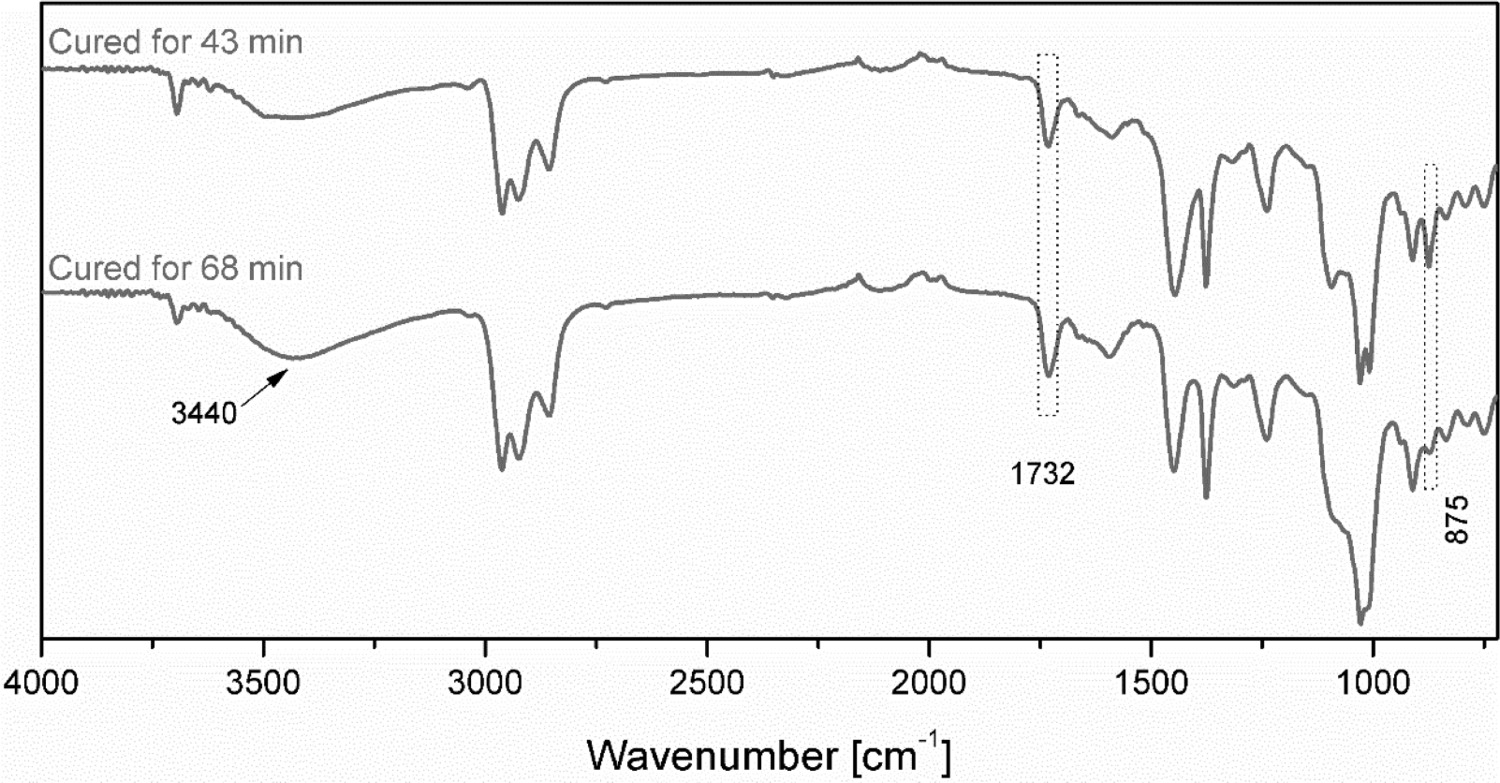
Tensile properties of ENR-50 compounds crosslinked by HMA with different contents of ZAD, as well as with and without DMI and kaolin clay.

Pristine samples were cured at 180°C to their optimum cure time obtained from a cure rheometer.
Reprocessed samples were remoulded at 180°C for 40 min.
Note that the compounds ENR-S, EM and EMZ0 could not be reprocessed as they were crumbled after remoulding.
Interfacial self-adhesion characteristics of the vulcanisates
Self-adhesion ability is a primarily required characteristic for self-healable and recyclable or reprocessable materials. As elaborated in the previous sections, the compounds prepared in the present work have the potential to exhibit as a self-adhesion material due to the presence of exchangeable ester crosslinks. A peeling test experiment was employed to assess the self-adhesion ability of the vulcanised rubbers; a T-peel test was performed to determine the adhesion energy. Based on the results from T-peel measurements, the calculated values of detachment energy Interfacial self-adhering ability: (a) detachment energy of cured EMZ5 samples containing various concentrations of the transesterification catalyst–zinc acetate dehydrate, tested at different temperatures; and (b) detachment energy of EMZ5 samples prepared by self-adhering the samples at 180 °C from one to four cycles.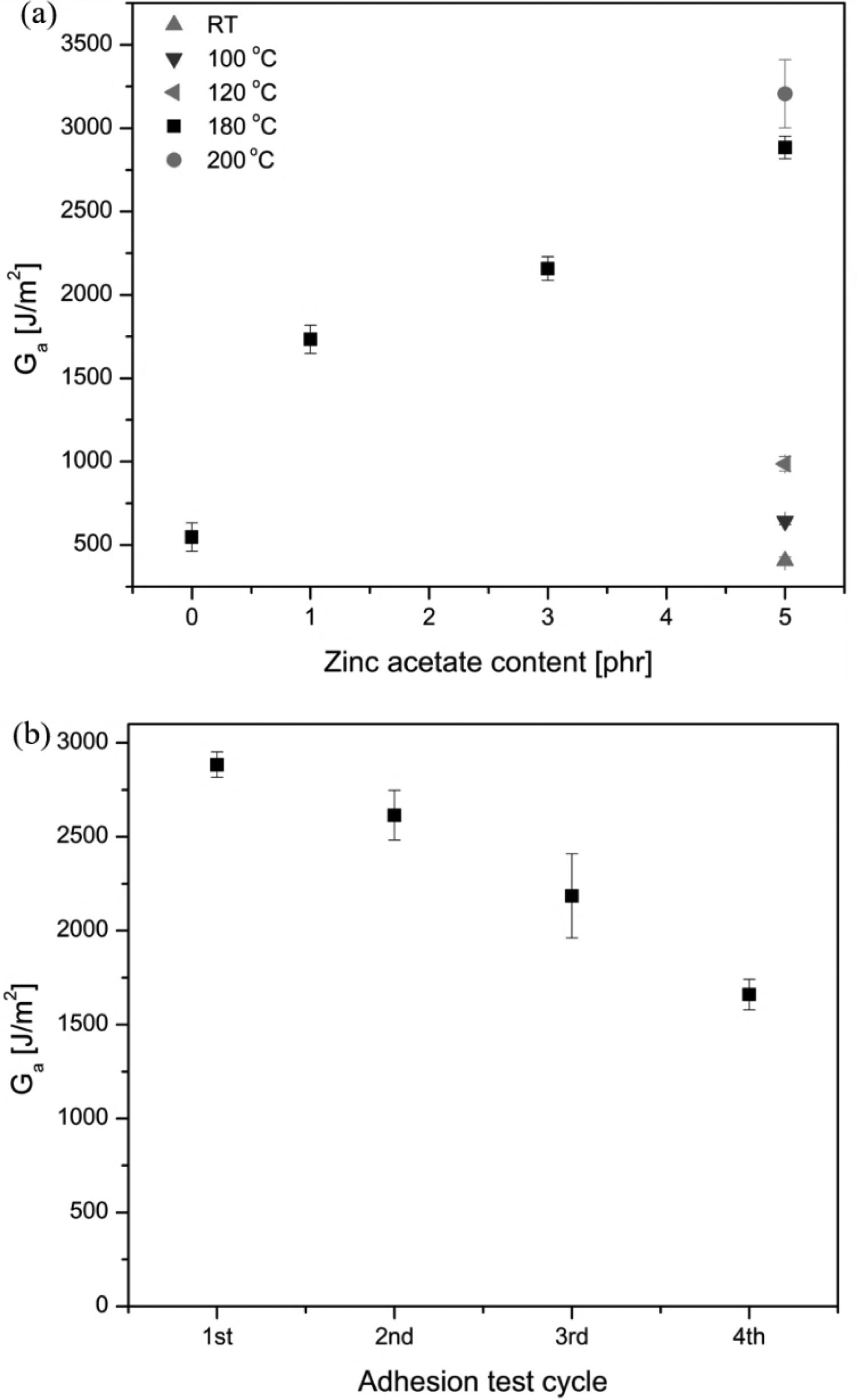
The influence of contact temperatures was investigated for the EMZ5 sample, by keeping the contact time constant for 3 h. The calculated
The same self-adhesion experiment was conducted for the reference sample, i.e. sulphur-crosslinked ENR-50 at 180°C for 3 h, the calculated detachment energy of the EMZ5 (2884 J m−2) is approximately 3 times higher than the detachment energy of the reference sample (973 J m−2). The low adhesion observed in the case of a permanent network-based vulcanisate (ENR-S) implies that no rearrangement reactions in the network are assumed. This can be attributed to the absence of interfacial interaction unlike in the case of the one with an exchangeable crosslink-based network, i.e. ENR with ester crosslinks from dicarboxylic acid curing precursor with zinc acetate dihydrate as bond-exchanging catalyst. However, the small detachment energy observed for ENR-S can only be attributed to the polymer chain diffusion as higher temperatures may facilitate the mobility of the chains – a physical mechanism [44, 45], leading to chain entanglements. Besides, some chemically interfacial interactions also involve in this system, owing to the polar elements in ENR. An ENR molecule has polar epoxide groups. Moreover, ENR is modified from NR as the base; it intrinsically consists of non-rubber constitutes, e.g. protein and phospholipid. These non-rubber-constitutes are polar and so contributing to the tack property or interfacial adhesion of NR and ENR. Note that, after modifying NR into an ENR, the concentration of these non-rubber constitutes is less compared to that of the unmodified NR. Whereas the high detachment energy of EMZ5 is attributed to the combination of three mechanisms: the physically intermolecular diffusion, chemically interfacial interactions as well as the exchangeable ester crosslink characteristic.
Moreover, the interfacial self-adhesion repeatability of the samples was conducted in order to evaluate the ability of the crosslinks to exchange multiple times at high temperatures. With increasing the number of interfacial self-adhesion cycle, the sample became more brittle accompanying with a darker colour. Figure 6(b) shows the detachment energy of the EMZ5 sample as a function of the interfacial self-adhesion test cycle. The apparent detachment energy of the compound is deteriorated with increasing the interfacial self-adhesion cycle. The detachment energy decreases from about 2900–2100 J m−2 for the first cycle. This small reduction in the detachment energy could be attributed to the influence of thermal annealing and oxidative modification. Natural rubber and epoxidised natural rubber are commonly known to be prone to thermal and oxidative degradation that could lead to molecular modifications on the sample interface. The exchangeable ester crosslinking mechanism might be intervened and disrupted by a degrading species. Oxygen radicals on the rubber interface generated during the transesterification reaction under the interfacial self-adhesion circumstance are most likely to be deactivated by the environmental oxygen, and this process exhibits more pronounced with a thermal annealing condition. Hence, at the sample interface, the exchangeable ester crosslinks could partially be transformed to permanent crosslinks [17, 46, 47], limiting the intermolecular rearrangements of the network, and thus lessened interfacial adhesion. In addition, the self-adhesion experiment was carried out at 180°C for 3 h; therefore, the high temperature for a prolonged time could lead to more molecular chain scission. This results in a decrease in the molecular weight, as a consequence in the reduced strength of the elastomer matrix at the interface and so deteriorated detachment energy.
To summarise, the obtainable results from the peel test reflect that a higher temperature facilitates the interdiffusion of the molecular chains. Whereas a higher content of zinc acetate dihydrate promotes a rapid transesterification exchange, leading to a greater extent of interfacial interactions, and so improved self-adhesion of adjoining surfaces of HMA-crosslinked ENR samples. Therefore, the results clearly illustrate an effective self-reformation of the network of this particular rubber vulcanisate, which further hints that the reprocessability of this material is highly promising to be achievable.
Reprocessability of the dicarboxylic acid-cured ENR vulcanisates
Since the ester crosslinks in HMA-vulcanised ENRs can thermochemically be exchangeable in the presence of a transesterification catalyst under a high-temperature stimulation. Additionally, the ring-opening reaction of the epoxide groups in ENR-50 will produce a certain amount of hydroxyl (–OH) groups, forming hydrogen bonding with the polar groups in the system. The hydrogen bonds and polar interactions, non-covalent bonds, are thermally reversible [27], and the deconstructed structures based on these interactions can be reformed during a dynamic rearrangement of the network [48]. Thus, the ruptured sample could recover to its original properties. In principle, this ester crosslink network could provide a crosslinked rubber with a potentiality to be reprocessable under elevated temperatures when the surfaces of the rubber particles are in contact under an appropriately stimulating condition.
In this study, all ENR-50 samples vulcanised to their optimum cure state using HMA system with ZAD as transesterification catalyst as well as the references EM and ENR-S were ground to be fine rubber pellets and then remoulded at 180°C for 40 min. After the treatment, the integrity of the EM, EMZ0 and ENR-S compounds was not achievable, as these rubber samples were still crumbled. This is due to that these samples were cured using dicarboxylic acid without a transesterification catalyst, i.e. ZAD. Hence, they are based mainly on the permanent ester crosslinks, constraint to the exchangeability of the crosslinking bonds, and so hindered chain mobility, intermolecular diffusion and, most importantly, the network reformation at the interface. By adding 1 phr of zinc acetate dihydrate (EMZ1), the uniformity of the sample was partially recovered after reprocessing, i.e. 66 and 76% relative to its tensile strength and elongation at break, respectively, as reported in Figure 7 and Table 3. One of the features of zinc acetate dihydrate is a straightforward control of the bond-interchange reaction kinetics through its catalysis as graphically illustrated in Figure 8.
Reprocessability: (a) images of a ground rubber and reprocessed rubber sheet from the vulcanisate based on EMZ5; and (b) stress-strain curves of the investigated samples before grinding (pristine) and after reprocessing. Pristine samples were cured at 180°C to their optimum cure time obtained from a cure rheometer, while reprocessed samples were remoulded at 180°C for 40 min. Postulated reformation mechanism at the interface of two separate rubber particles through the thermochemically exchangeable ester crosslinks in ENR-50 cured with HMA containing ZAD as transesterification reaction catalyst.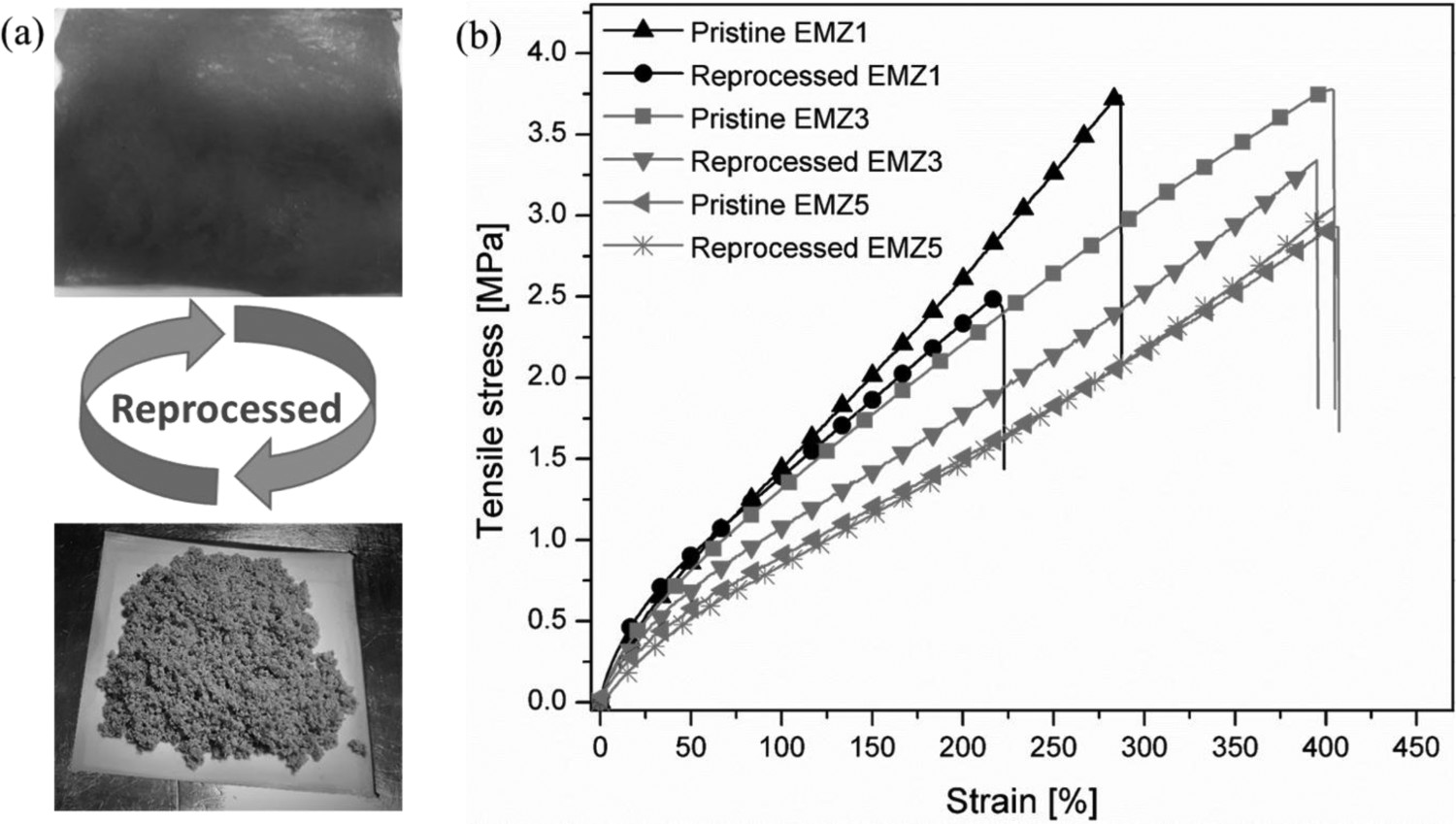
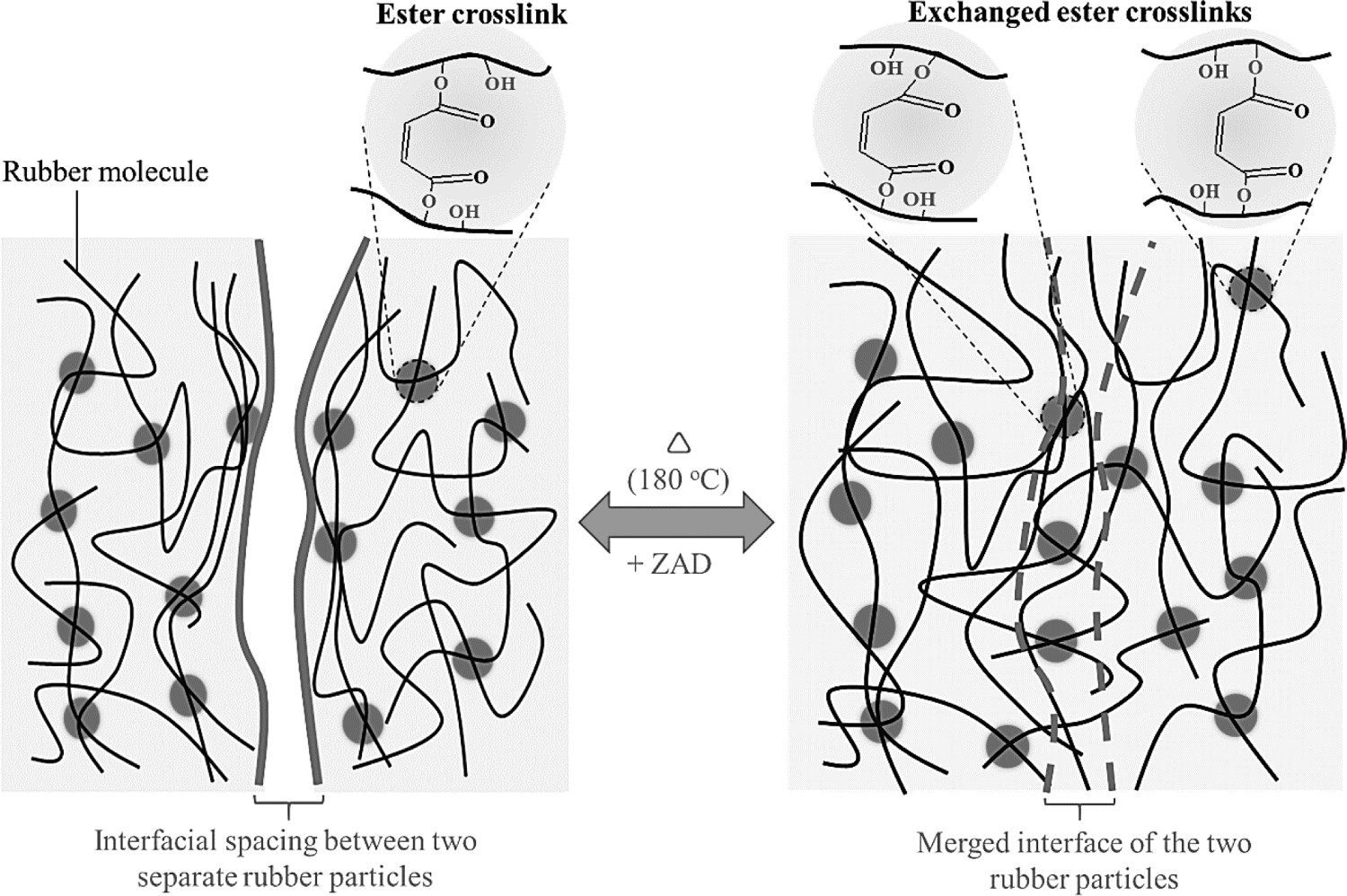
When the concentration of transesterification catalyst ZAD increases, the mechanical properties of the reprocessed rubber, in particular EMZ5, are significantly improved. With 3 phr of ZAD catalyst, the reprocessed rubber vulcanisate can maintain about 88% of its tensile strength and 98% of its elongation at break, which shows superior to the one with ZAD at 1 phr. Whereas, with 5 phr ZAD, the mechanical properties of the reprocessed vulcanisate are completely recovered after pulverisation due to a positive characteristic of the exchangeable ester bonds as elucidated earlier in this context. Figure 8 demonstrates that, in the initial stage under a high temperature, the ester crosslinks on the surface of rubber particles start to exchange their bonds through a transesterification reaction, generating new bonds/crosslinks at different positions. This bond-interchange mechanism can lead to intermolecular diffusion and the formation of bonding among the rubber grains, allowing an intimate self-adhesion of the crushed rubber particles. Therefore, the fully retained properties of the reprocessed vulcanisate were obtained.
Furthermore, the reprocessing of EMZ5 samples was evaluated. The stress–strain curves of the pristine and reprocessed EMZ5 compounds initially cured for different times, i.e. 43 and 68 min, are demonstrated in Figure 9. The EMZ5 compound cured to 68 min has a relatively excellent reprocessability as the reprocessed rubber has 97 and 92% of the retained tensile strength and strain at break, respectively. Generally, the intrinsic molecular structures of rubber vulcanisates, particularly molecular weight, are inevitably deteriorated after a pulverising process, leading to reduced mechanical strength and elongation at break of the materials even if the crosslinks can be reconstructed [49]. Interestingly, the tensile stiffness of the sample cured for 68 min is slightly higher than that of the pristine one. This could be due to the marching modulus when the compound undergoes a prolonged thermal treatment. For the compound cured for 43 min, the mechanical properties of the reprocessed vulcanisates are fully retained. All-in-all, the mechanical properties of the reprocessed EMZ5 vulcanisates show highly promising prospects towards recyclability and healability when compared to their pristine counterpart.
Stress–strain curves of non-reprocessed (pristine) and reprocessed EMZ5 compounds cured at 180°C for two different curing times.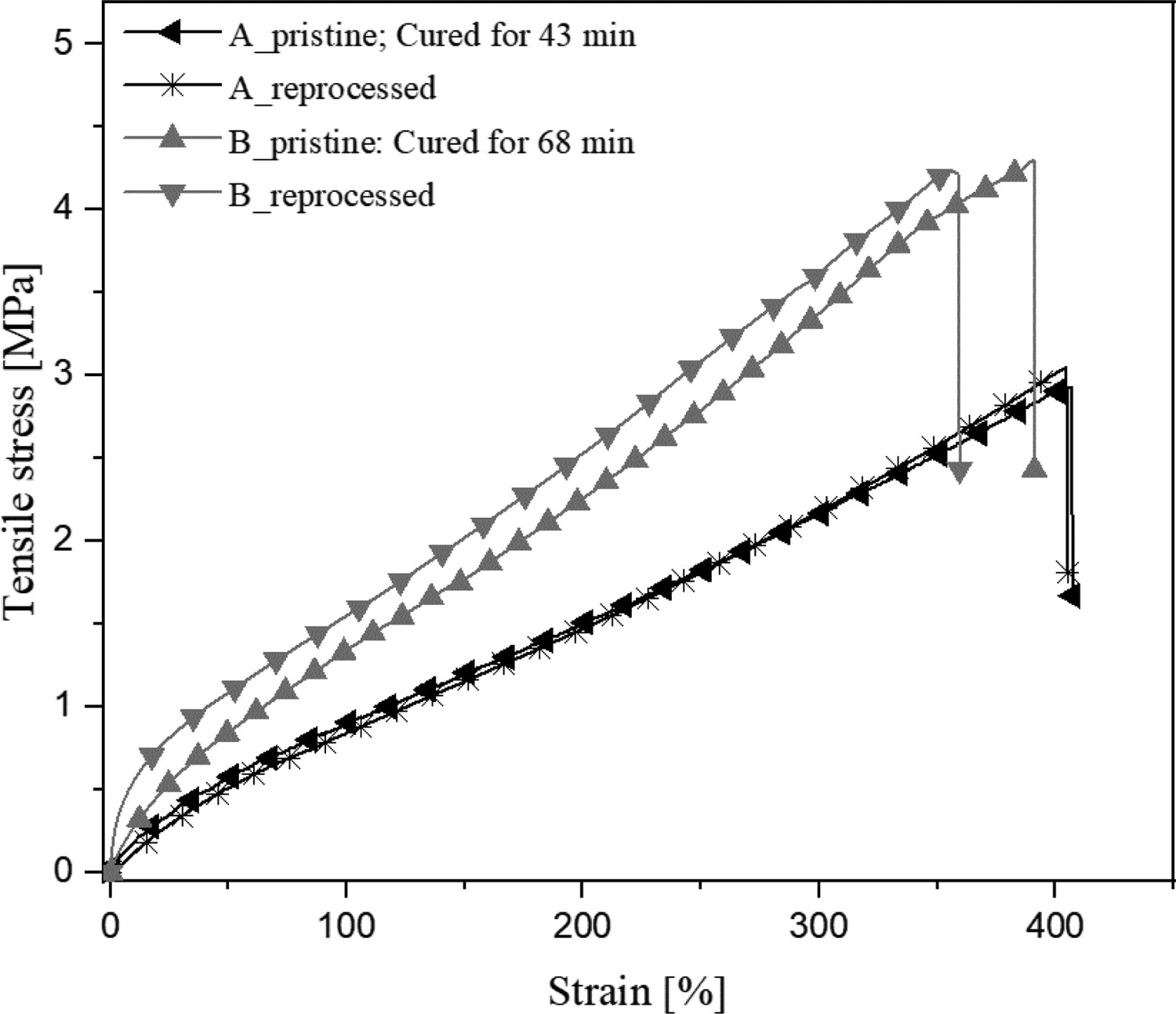
Conclusions
The results from cure characterisation confirm that Hydrolysed Maleic Anhydride (HMA) is able to vulcanise Epoxidised Natural Rubber (ENR). The vulcanisation rate can be accelerated by DiMethylImidazole (DMI). FTIR analysis gives useful information that the ester bonds are generated at the epoxide functionalities on ENR molecules. Dicarboxylic acid based on HMA reacts with the hydroxyl groups originated from the opened structure of oxirane rings. Zinc Acetate Dihydrate (ZAD) as transesterification catalyst has shown to retard the cure rate of HMA-cured ENR compounds and gives a higher degree of marching modulus with increasing its dosage. However, when prolonging cure time, the cure curve shows the optimum cure state with a plateau behaviour.
The assessment of interfacial self-adhesion of the compounds clearly demonstrates that the HMA-cured ENR-50 compounds with a higher concentration of ZAD have a stronger interfacial self-adhesion and show further substantial improvement under elevated treatment temperatures. However, the interfacial self-adhesion deteriorates over the number of re-adhering cycle. The reprocessability of the compounds with different quantities of ZAD was validated. The results of mechanical properties reflect that the higher ZAD loadings, the better reprocessability of the vulcanisates is obtainable, corresponding well to the results from the interfacial self-adhesion evaluation. Interestingly, the tensile properties of the reprocessed HMA-cured ENR-50 with 5 phr of ZAD are fully retained. This is because of the transesterification reaction of ester crosslinks in the rubber matrix, which is catalysed by ZAD and accelerated by a thermal treatment. Therefore, the overall results presented in the present study support that a thermochemically exchangeable crosslinking system based on dicarboxylic acid cure plus a transesterification catalyst can be enabled to yield a desired reprocessable elastomer vulcanisates. This concept shows a high potential to be extended for a variety of rubber applications, in particular with the better aspects in terms of durability and sustainability.
Footnotes
Acknowledgements
The authors would like to acknowledge the financial supports from the Higher Education Research Promotion and Thailand's Education Hub for Southern Region of ASEAN Countries Project Office of the Higher Education Commission, as well as from the Natural Rubber Innovation Research Institute (grant number SCI6201170s), Prince of Songkla University.
Disclosure statement
No potential conflict of interest was reported by the author(s).
