Abstract
This study investigated the validity of post-amputation application of removable rigid dressings (RRDs) for trans-tibial amputees, regarding preparation for prosthetic management and key rehabilitation timelines. It was hypothesised that the use of RRDs would result in faster preparation of the residual limb for prosthetic management and shorter rehabilitation times, compared with conventional soft dressings. A retrospective case-note audit was conducted, in which consecutive trans-tibial amputees who underwent amputation in the 2 years before RRD implementation (non-RRD group, n = 37) and in the 2 years after RRD implementation (RRD group, n = 28) were eligible for inclusion. There was a significant reduction in the geometric mean time-to-first-prosthetic-casting in the RRD group, compared with the soft-dressing group (36.4 days vs. 27.6 days, respectively, p < 0.05). A significant reduction in acute length of stay (LOS) for the RRD group was also identified (15.9 days vs. 8.7 days, respectively, p < 0.001). There were no significant differences in other rehabilitation timeframes, such as rehabilitation LOS, total LOS, outpatient rehabilitation days, and total rehabilitation days between the two groups. This study shows that the application of RRDs reduces acute LOS and time-to-first-prosthetic-casting, thereby providing substantial benefits in preparing the trans-tibial amputee for early rehabilitation and prosthetic intervention.
Introduction
The fundamental goals of trans-tibial post-operative dressings are to improve wound healing and to control pain, through the provision of a sterile wound-healing environment. (Smith et al. 2004). Wound-dressing techniques have evolved and are now expected to provide more than simply the provision of a sterile environment. Dressing strategies now attempt to incorporate a number of post-operative goals, including the prevention of knee flexion contractures, reduction of oedema, protection from external trauma, facilitation of weight bearing (Smith et al. 2004), and shaping for early prosthetic management (Wu et al. 1979).
There is currently no definitive evidence to support one dressing strategy over another. Adding to the uncertainty are the different methodologies, dressing types, and outcome measures that have been used in the various studies. Outcome measures that have been used include residuum healing, residuum volume, prosthetic fitting, and functional outcome (Nawijn et al. 2005). Furthermore, time-to-prosthetic-casting is often used as an indication of wound healing, due to the absence of a ‘gold standard’ for the prediction of residuum healing (Nawijn et al. 2005).
The literature indicates that non-removable rigid dressings result in significantly accelerated rehabilitation times and decreased oedema when compared with soft gauze dressings (Smith et al. 2003). Used widely in the 1970s and 1980s, non-removable rigid dressings were reported to reduce pain and healing time and increase weight-bearing tolerance (Baker et al. 1977; Barber et al. 1983), although not all these claims were supported in the literature. These dressings lost favour because of concerns about infection and revision rates, especially for dysvascular trans-tibial amputees, where wound inspection is paramount. (Cohen et al. 1974). The removable rigid dressing (RRD) was described in 1979 (Wu et al. 1979), and a recent systematic review suggests that use of this dressing may lead to improved residuum healing and oedema control, compared with conventional soft dressings (Nawijn et al. 2005).
It has been suggested that RRDs reduce the time from amputation to first prosthetic casting (Wu et al. 1979) and have been shown to result in earlier primary wound healing, compared with soft dressings (Deutsch et al. 2005). Another study investigating RRDs found other benefits, including rapid volume reduction, compared with no volume reduction with elastic bandages (Mueller 1982), which should allow early rehabilitation. Coupled with improved time-to-prosthetic-casting, there is also some evidence linking reduced inpatient stay with the introduction of RRDs, although no definitions of ‘definitive’ prosthesis, rehabilitation process, or controls were provided (Hughes et al. 1998).
The claims supported by controlled studies relate predominantly to non-removable rigid dressings and prefabricated post-operative prosthetic systems (Smith et al. 2004; American Academy of Orthotists Prosthetists, 2003). Although the five post-operative management strategies cited in a review by the American Academy of Orthotists Prosthetists are widely used, to date there is little evidence to support one method over another (American Academy of Orthotists Prosthetists, 2003). In trials where controls were used, inconclusive results and variations in definitions and techniques for measuring wound healing exist (American Academy of Orthotists Prosthetists, 2003), making interpretation and application of results in clinical settings difficult.
At the Repatriation General Hospital (RGH), RRDs fitted immediately after trans-tibial amputation became standard protocol in early 2000. It remains unclear, however, if the introduction of RRDs has led to any improvement in wound healing and patient management. With little evidence from the existing literature available to determine the effects of RRD use on wound-healing and rehabilitation times, further evaluation was required. While randomised, controlled trials are considered the gold standard of research, we decided that it was not ethical to withhold a treatment that was routine and considered superior to soft dressings. Therefore, we decided that a retrospective case-note audit was the best tool we had available to us to determine whether the introduction of RRDs into our clinical practice had a real and positive effect on our patients.
Therefore, the aim of the present study was to compare, using a retrospective case-note audit, RRDs to conventional soft dressings, with reference to preparation of the residual limb for early prosthetic management. Specifically, this study examined whether the introduction of RRDs reduced the number of days to initial prosthetic casting, determined RGH rehabilitation times and whether these were influenced by RRD implementation, and provided results on timelines and lengths of stay, enabling comparison to other facilities for benchmarking.
Methods
Study design
This study used a retrospective case-note audit design. All consecutive trans-tibial amputees who underwent amputation in the 2 years before RRD implementation (non-RRD group) and in the 2 years after RRD implementation (RRD group) were eligible for inclusion. A 2-year transition interval between the two periods of data collection was allowed. Data were collected from medical files.
The audit was granted ethical approval from the RGH Research and Ethics Committee.
Participants
Patients were considered for the audit if they had undergone trans-tibial amputation in the years 1998 or 1999 (non-RRD group), or if they had undergone one in 2002 or 2003 and received an RRD within 24 h of amputation (RRD group). Patients were excluded from the audit if they were identified as not for ambulatory rehabilitation, had been discharged prior to rehabilitation commencing, if rehabilitation or prosthetic casting had been delayed (not associated with wound healing), or if revision surgery occurred before the first prosthetic cast.
Treatment procedures
All hospital sites used in our study had a similar protocol regarding amputation surgery: most commonly, long posterior flap surgical technique with anterior suture line was used. The study included two different post-operative management techniques. In the non-RRD group, conventional residuum dressing techniques were used. This comprised the application of soft gauze dressing encased by traditional crepe bandaging, applied post-operatively by a member of the surgical team, followed by the elastic bandaging on advice from a rehabilitation specialist. Elastic bandaging usually began at the time of suture removal (typically, alternate sutures were removed at day 14, with remainder usually removed between days 21 and 28, as determined by the surgeon), and was changed regularly by the physiotherapist or another member of the rehabilitation team (typically every 4–6 h). Interim prosthetic management began when the wound was capable of withstanding weight-bearing forces with minimal oedema, as determined by the rehabilitation specialist in conjunction with the rehabilitation team. Interim prosthetic management involved casting and fitting with a thermo-set laminated socket. There was no use of pre-prosthetic aids or pneumatic air splints during this time.
The RRD Group protocol has been previously described by Deutsch et al. (2005). In brief, a synthetic cast RRD was manufactured by a member of the prosthetic team using a Shapemate casting sock (Seattle Limb Systems, Poulsbo, WA) over the residuum, typically while the patient was in recovery after their trans-tibial amputation. A 1-ply sock was applied under the RRD, which was suspended using a supracondylar cuff and stockinette (Figure 1). Daily inspection of the wound site was conducted by a member of the prosthetic team from day 1. As residuum volume decreased, extra socks were applied underneath, until a 4-sock fit was achieved, leading to RRD replacement. The beginning of interim prosthetic management followed the same process as with the non-RRD group.

Removable rigid dressing. a) RRD alone. b) RRD placed on a patient.
Outcomes
Residuum healing. The key outcome measure was residuum healing, which was determined by the number of days from amputation to first prosthetic cast. The number of days until elastic bandaging began in the non-RRD group, the number of RRDs, and the number of prosthetic sockets provided within 6 months of amputation was also recorded.
Length of stay.
The length of stay (LOS) in various settings during the hospital episode was recorded. Acute LOS was the number of days from amputation to admittance to the rehabilitation ward. Rehabilitation LOS was the period from rehabilitation-ward admission until discharge from hospital. Note that rehabilitation LOS does not indicate that prosthetic rehabilitation has been completed, as it may have continued on an outpatient basis. Total rehabilitation LOS is the total number of days required for the amputee to progress from the interim prosthetic program to the definitive program (i.e. the time from rehabilitation admittance until rehabilitation discharge). Total LOS was the time from amputation to discharge from the rehabilitation service.
Data analysis
Not all data were available for every participant, hence the variation in the patient numbers that are reported within each statistical analysis. Patient characteristics and outcome-measure results between the two groups were compared, using chi-square tests of association for categorical variables and independent-samples t-tests for continuous variables. As there was substantial skewness in the time-to-cast and LOS data, a logarithmic transformation was applied to these variables. Geometric means and interquartile ranges were used to summarise outcome measures. A significance level of p < 0.05 was used for the interpretation of results. SPSS® Version 12.0 for Windows was used in all analyses.
Results
In total, 37 patients were included in the non-RRD group and 28 patients were included in the RRD group. The RRD group consisted primarily of patients who had undergone their surgery at the RGH. The non-RRD group had a broader patient-catchment area and included amputations performed at other local hospitals. This difference is associated with the inclusion criterion that the RRD must have been applied within the first 24 h of surgery, which excluded many non-RGH amputations in the RRD group. No other significant differences were detected between the two groups (Table I).
Participant characteristics.

RGH, Repatriation General Hospital; RRD, removable rigid dressing.Note: ∗p < 0.04.
Residuum healing
The RRD group had significantly less time from amputation to first prosthetic cast (Table II). Time-to-cast was 36.4 days (IQR 24–50) with soft dressings, compared with 27.6 days (IQR 21.2–32.7) with RRDs (p < 0.05). The mean number of RRDs provided to achieve this result was 1.71 and the mean day of elastic-bandage application in the non-RRD group was 19 days post-amputation (Table III). There was a non-significant trend towards a higher number of sockets being provided in the RRD group (p = 0.08).
Geometric mean number of days (interquartile range) for time-to-first-prosthetic-casting and length of stay.

LOS, length of stay; RRD, removable rigid dressing.∗p < 0.05, # p<0.001.
Mean general residuum-healing results (SD) for each of the intervention groups.
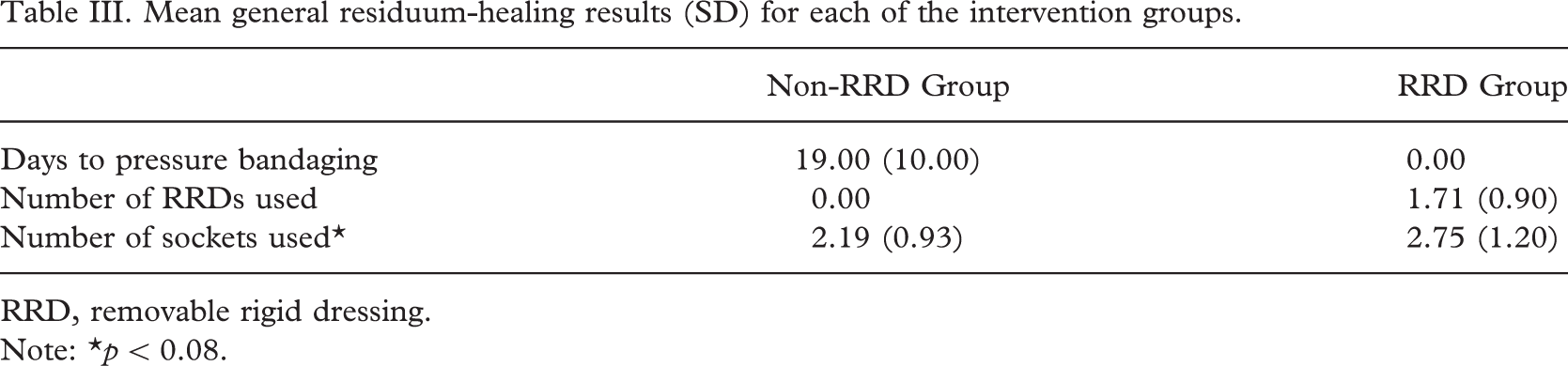
RRD, removable rigid dressing. Note: ∗p < 0.08.
Length of stay
Acute LOS was significantly shorter in the RDD group than in the non-RDD group: 8.7 days (IQR 6.2–12.5), compared with 15.9 days (IQR 9.0–26.7), respectively (p < 0.001, Table II). There were no significant differences in rehabilitation LOS, total LOS, outpatient rehabilitation days, and total rehabilitation days between the two groups.
Discussion
The results of this audit indicate a significant reduction in time-to-cast after the implementation of RRDs, with a geometric mean of 8.8 days in the RRD group and 27.6 days in the RRD group, post-amputation (p < 0.05). Our results can be considered clinically and financially significant, as delayed prosthesis fitting is regarded as the main cause of increased costs of rehabilitation after amputation (Cutson et al. 1994). The resource cost of providing 1.71 RRDs per patient is negligible.
The results of this audit show a geometric mean reduction of 7.2 days in the acute LOS, resulting in a geometric mean acute LOS of 8.7 days. The significant reduction in acute LOS does not flow on to a significant reduction in inpatient rehabilitation stay, outpatient rehabilitation, or overall rehabilitation days. This outcome may have partly been the result of the relatively small sample size, in that small, clinically meaningful differences on some variables may not have been detected as statistically significant. Some studies have demonstrated significantly shorter average hospital stays with thigh-length non-removable rigid dressings as compared was soft dressings (Baker et al. 1977), whilst a comparison of RRDs with soft dressings showed no significant difference in regards to acute LOS (Deutsch et al. 2005). Other studies however, have reported reduced overall rehabilitation times with the implementation of both non-removable rigid dressings (Baker et al. 1977; Kane and Pollak 1980; Nicholas and DeMuth 1976) and RRDs (Hughes et al. 1998).
Variation in the definitions of rehabilitation timeframes and hospital discharge and rehabilitation practices makes comparison of LOS and timeframes across facilities difficult. This significant reduction in acute LOS can be compared to national LOS data, which has shown that, for patients staying at least 1 night, average LOS over a 10-year period has remained relatively constant (Australian Institute of Health and Welfare 2006). This suggests that the changes in acute LOS we found are not simply be the result of a national trend to shorter LOS.
The time when the residuum is suitable for casting can be considered an indication of wound stability; however, it does not specifically define primary or secondary wound healing. Due to the continual nature of the healing process, defining these end-points is challenging (Smith et al. 2003). Time-to-cast is an important milestone in amputee rehabilitation, which, regardless of primary or secondary healing, indicates rehabilitation progression due to the wound being strong and stable enough to withstand weight-being forces.
The significant reduction in time-to-cast and acute LOS cannot be solely attributed to RRD post-operative management. The nature of a retrospective study presents difficulties for controlling important variables such as surgical skill, amputation-level selection, healing potential, co-morbidity, nutrition, immune status, and functional ability (Smith et al. 2003). Any differences attributed to soft dressings or RRDs are confounded with the year of dressing application, which is a limitation of our methodology. Rehabilitation-service differences between pre-RRD and post-RRD implementation may also have influenced the results.
Furthermore, rehabilitation services are unique to geographical and population bases; thus, careful consideration is required when comparing results across institutions. It is difficult to compare these results with those of other studies and centres, because of varying measures and definitions of wound healing and rehabilitation timeframes. The literature suggests that uniform wound-healing identification criteria are lacking. Development of a standardised wound-assessment tool would assist future research in defining this critical point and enable study comparisons.
Conclusions
This study has shown a reduced time-to-first-prosthetic-casting and acute LOS with the application of RRD within 24 h post-operatively, which may have positive benefits in preparing trans-tibial amputees for early prosthetic intervention. Further randomised, controlled trials comparing RRDs with dressing types other than soft gauze in regards to functional outcome, hospitalisation costs, and wound healing are warranted. The development of a wound-assessment tool that specifically assesses residuum healing and wound stability would assist researchers in measuring this variable. When used in conjunction with carefully selected controls, this tool may enable cross-institutional research and comparisons.
