Abstract
Sub-sensory electrical or mechanical stimulation can enhance the sensitivity of the human somatosensory system to improve the balance control capabilities of elderly. In addition, clinical studies suggest that visual-auditory biofeedback can improve sensory compensation for the elderly. This study hypothesizes that the static balance and gait performance of single leg quiet standing and treadmill walking could be improved for providing proprioceptive neuromuscular facilitation using sub-sensory stimulation and visual-auditory biofeedback in amputee subjects. To test this, a computerized foot pressure biofeedback sensory compensation system using sub-threshold low-level electrical stimulation combined with visual-auditory biofeedback was developed. Seven unilateral trans-tibial amputees who wore prostheses over 2 years were recruited. The subjects performed multiple single leg quiet standing trials with sub-sensory electrical stimulation applied at the quadriceps muscle during half of the trials. Static balance performance was characterized by using a Zebris motion analysis system to measure the sway distance and duration of the centre of mass on the second sacral (S2) of the subjects. In addition, multiple treadmill ambulatory trials with or without visual-auditory biofeedback was performed. Dynamic gait performance was characterized with a Zebris instrumented insole to measure the temporal responses of foot pressure sensors. Experimental results showed an improvement in three balance performance indices (Holding Time Index, HTI, Maximum Sway Distance Index, MSDI, and Average Sway Distance Index, ASDI) during single leg quiet standing by applying sub-sensory stimulation. The improvement ratio of these balance performance indices across subjects for single leg quiet standing tests resulted in 132.34% in HTI, 44.61% in MSDI, and 61.45% in ASDI. With visual-auditory biofeedback as a cue for heel contact and toe push-off condition during treadmill ambulation, the improvement of four dynamic gait performance measures (Double Support Period, DSP, Constant Time Cadence, CTC, Single Support Period, SSP, and Stance/Swing Ratio, SSR) in amputees was verified. This resulted in 7.89% in DSP (affected side), 5.09% in CTC, 16.67% in SSP (sound side), 45.30% in SSR (sound side), and 40.30% in SSR (affected side) respectively. These findings suggest that sub-threshold electrical stimulation and visual-auditory biofeedback rehabilitation strategies may be effective in compensating sensory loss and improving static balance and dynamic ambulation performance for amputees.
Introduction
Balance is a critical functional capability that greatly influences the ability to perform activities of daily living. For patients after amputation, the decrease in body weight accompanied by an alteration in the position of the centre of mass over the base of support will be inadequate in weight acceptance, single limb support and limb advancement (Yigiter et al. 2002). Inadequacies in weight acceptance, single limb support and limb advancement prevent the amputee in achieving adequate balance control and a normal walking pattern. Experimental evidence shows that an amputee has a decreased stance time on the prosthetic side, shortened stride length on the sound limb or lateral trunk bending over the amputated side. A well planned post-operative prosthetic training programme can improve balance control and walking pattern for the amputees (Yigiter et al. 202; Roll et al. 2002; Gill et al. 2001; Mouchnino et al. 1998; Fernie 1981; Gailey and Clark 1992; Olsson 1990).
Somatosensory feedback is an important component of the human balance control system. Recent studies proved that tactile stimulation in foot sole contributes to the coding and the spatial representation of body posture (Roll et al. 2002). Several clinical studies show that the sub-sensory low level noise stimulation (electrical or mechanical), the so-called stochastic resonance stimulation (SRS) can enhance the sensitivity of the human somatosensory system (Gravelle et al. 2002; Priplata et al. 2002, 2003). In addition, clinical studies suggest that visual-auditory biofeedback can improve sensory compensation for the elderly were reported (Gravelle et al. 2002). In the present study, the effects of sub-sensory low level input to the somatosensory system on postural control in amputees was examined. It was hypothesized that the static balance and dynamic gait performance of amputee individuals during single leg quiet standing and treadmill walking could be improved by applying sub-sensory electrical stimulation and visual-auditory biofeedback respectively. To test this hypothesis, a plantar pressure monitoring system integrated with a sub-threshold low-level electrical stimulation and visual-auditory biofeedback mechanism was developed and used for clinical study. To investigate the effect of the sub-threshold low-level electrical stimulation and visual-auditory biofeedback on human balance control, a series of tests on amputee subjects were conducted.
Methods
Sensory compensation biofeedback system design
A computerized foot pressure sensory compensation system with sub-threshold stimulation and visual-auditory biofeedback mechanisms was constructed as shown in Figure 1. The proposed system consists of a custom prosthesis with plantar pressure measurement sensors attached, a microcomputer-based control unit, sub-threshold low-level electrical stimulation unit and visual-auditory biofeedback unit. Foot pressure sensors were attached on the bottom of the prosthesis to monitor the subject's gait sequence and balance performance during tests. A microcomputer-based control unit was designed to receive the foot pressure signal and to generate the sub-threshold low-level electrical stimulation signal based on built-in control rule. The visual-auditory biofeedback units were integrated to provide a cue for heel contact and toe push-off condition during dynamic treadmill ambulation.
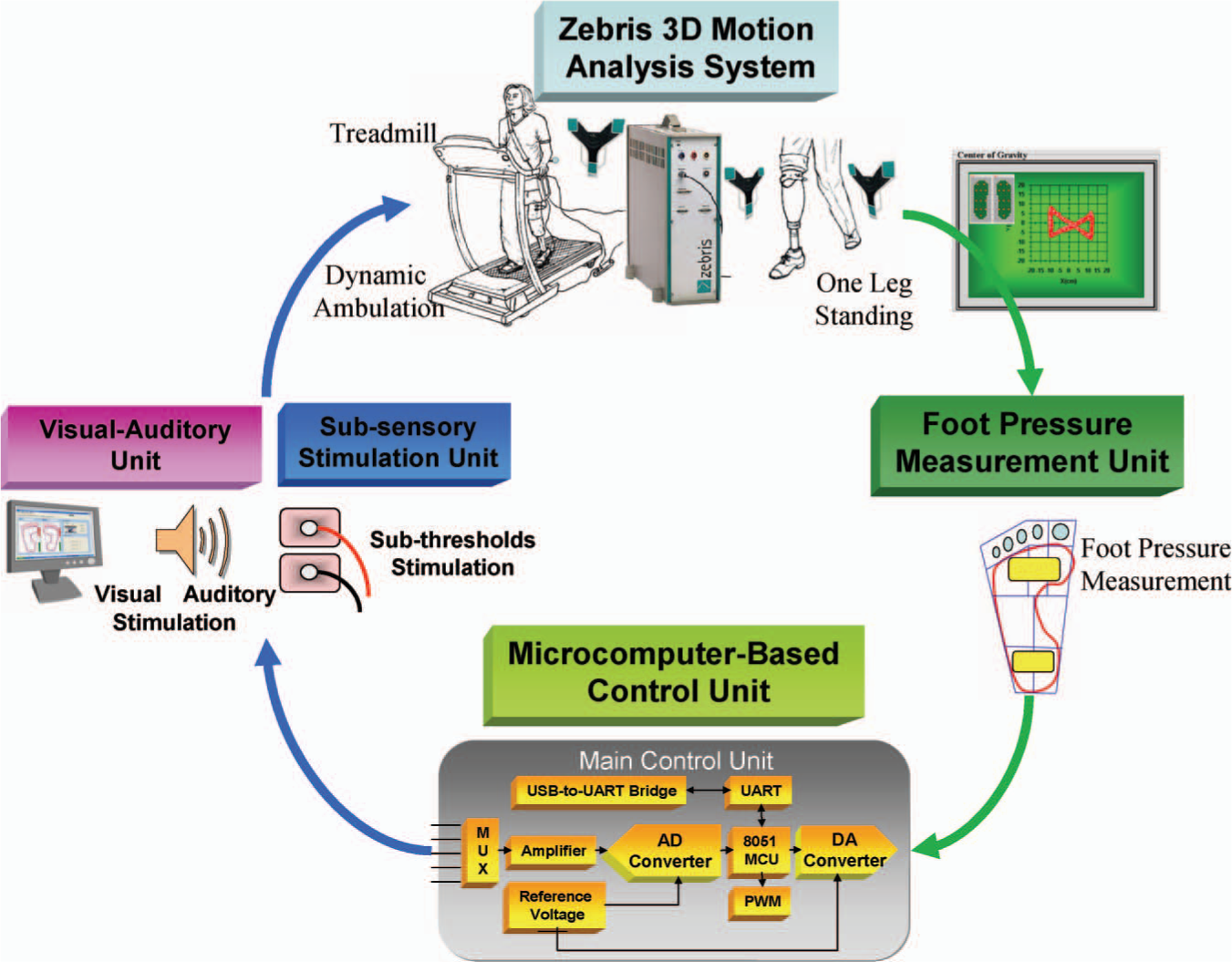
System architecture of the proposed sensory compensation biofeedback system.
Foot pressure measurement unit
In this study, two force-sensing resistor (FSR) sensors (18.3 mm×12.7 mm×0.46 mm, Interlink Electronics, CA, USA) were used to detect the heel contact and toe push-off conditions of the foot (or prosthetic foot). The foot pressure signal is sent to the main control unit to generate the visual-auditory biofeedback signal to the subject once the amount of ground reaction forces exceeds a certain specified threshold value; thus the visual-auditory biofeedback unit will be triggered according to the amputee's gait pattern for providing sensory compensation.
Microcomputer-based control unit
The microcomputer based control unit (Figure 2) comprises an 8051 single chip microcomputer (AT89C52, Atmel Corporation, USA), signal amplifier, A/D converter, RAM, 9V power supply and electrodes. The functions of the microcomputer-based unit included receiving a foot pressure signal from the foot pressure measurement unit, generating the stochastic resonance stimulation signal, facilitating functional electrical stimulation signals, uploading foot pressure information to visual-auditory stimulation unit, etc.
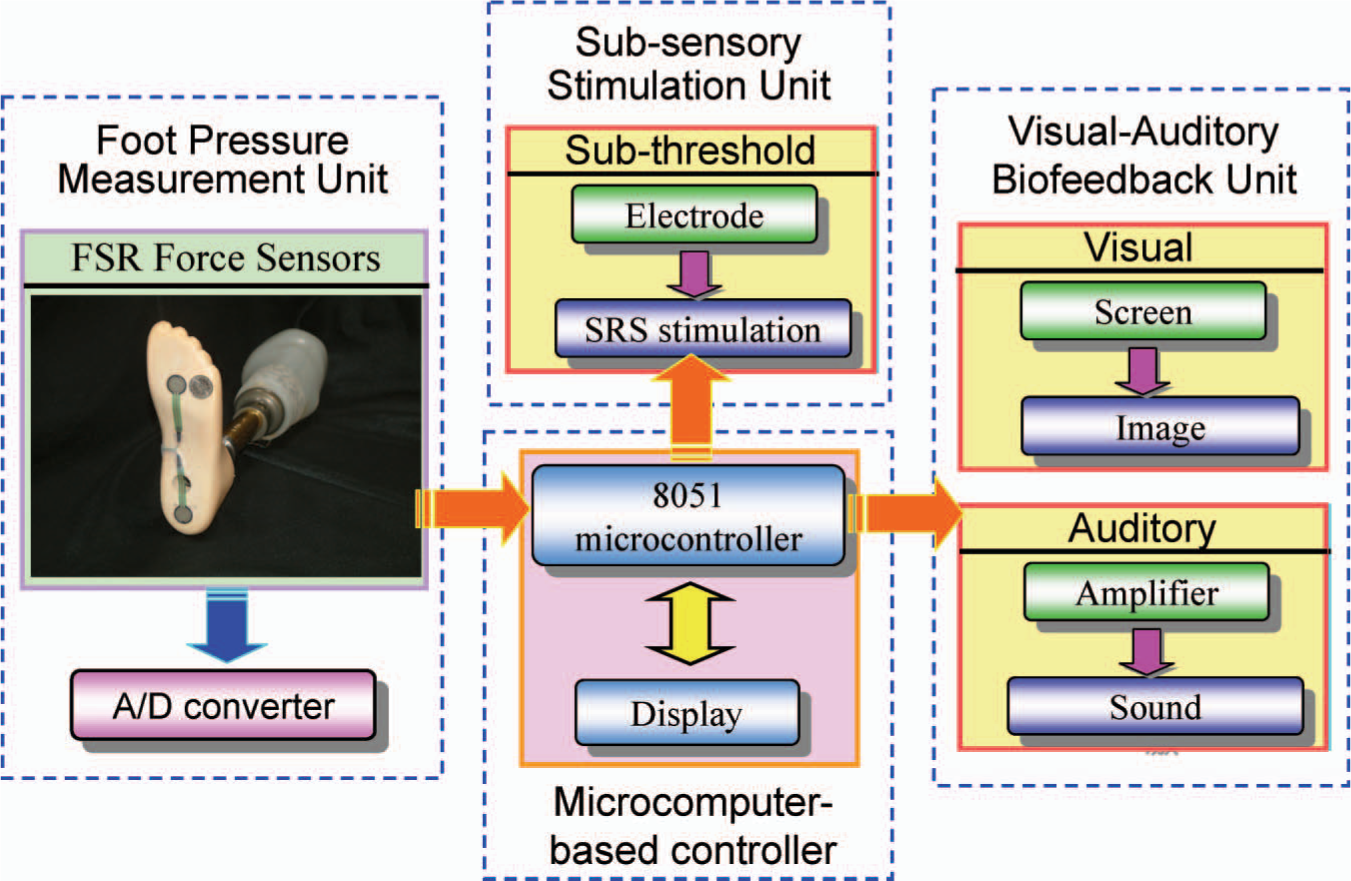
Control signal flow of proposed sensory compensation biofeedback system.
Visual-auditory biofeedback unit
The auditory biofeedback functioned as an alarm and was fed back to the subject through two loudspeakers connected to the computer. Auditory feedback was triggered when the local pressure under the six selected high pressure areas exceed a previously determined threshold, the critical peak pressure (CPP). Each channel had its own CPP, which was determined and adjusted by the physician according to the patient's plantar pressure pattern. A switch activated or deactivated the warning signal for each channel so that the auditory feedback of a single sensor could be individually reset. Each pressure sensor had a different tone in the auditory feedback. The lowest tone frequencies (500, 1000 Hz) corresponded to the sensors positioned under the heel. The highest frequency reached 10,500 Hz and was triggered by the sensor located under the hallux. The frequencies were determined in a way that allowed the subject to easily distinguish the overload foot pressure area.
The visual feedback, which was designed through a control screen placed in front of the subject whose plantar foot pressure distribution can be easily visualized. On the right part of the control screen is the real-time display of the plantar foot pressure distribution. To indicate the heel strike and toe off condition, two sensors were activated to detect the pressure underneath heel and metatarsal regions. The detected pressures were shown with colours in the corresponding region of the foot-shape picture. A range of colours, from white to yellow to red, indicated the intensity of the pressure exerted under each location. The higher the pressure, the more the colour is closer to red. The visual biofeedback provides a hint for the subject to control and modulate his/her gait pattern and weight shift.
Sub-sensory low level stimulation unit
The sub-sensory low level stimulation unit is an electrical stimulation device with controlled input below the cutaneous sensation threshold of amputees. In this study, the sub-sensory low level signal was used to assess its effects to the somatosentory system on balance control in amputees. At the outset of the testing session, each subject was asked to determine his or her threshold of tactile perception quadriceps muscle of the sound side. A potentiometer was used to adjust the amplitude of the low level signal output. Thus, the applied low level signals were sub-sensory, and subjects could not distinguish between low level and control trials. The low level signal used in this study is a triangular waveform with 0 – 60 mA in amplitude and 500/μs in period. In this study, a function generator to produce the triangular wave with appropriate frequency and amplitude was used. The sub-threshold triangular signals were applied through surface electrodes on quadriceps of the subject's lower limb. The electrodes were rectangular (4×4.8 cm) self-adhesive gel pads, aligned with the longitudinal axis along the joint axis line formed by the femoral and tibial condoyle. The low level stimulation signal was applied for the entire duration of the stimulation trial.
Zebris motion analysis system
In this study, the balance performance was characterized using a Zebris force measurement system (Model CMS-HS, Zebris Medizintechnik GmbH, Isny im Allgäu, Germany) for measuring the body's centre of mass (COM) sway pattern of the subjects. A passive marker was placed on the second sacral vertebra (S2) to represent the COM of the subject (Alexander 1996; Alexander et al. 1992). This passive marker consists of a small ultrasonic microphone and was connected to the control unit of Zebris system. Three sequential active ultrasonic transmitters in the measuring unit of Zebris system send continuous pluses during operation. The distance between transmitter and the passive marker was determined through the running time of the pulse. By triangulation the absolute 3D coordinates of the COM of the subject can be determined. To synchronize the gait phases, Zebris instrumented insoles were used. Each of the insoles contains four force sensors and a total of 100 capacitive force sensing elements. The analogue output signals of force sensors are directly proportional to the forces supplied to the measurement points.
In this study, the duration and body's COM sway pattern were recorded for postural stability evaluation during single leg quiet standing tests. For the dynamic ambulatory tests, the force distributions of the left and right instrumented soles were recorded.
Patient selection
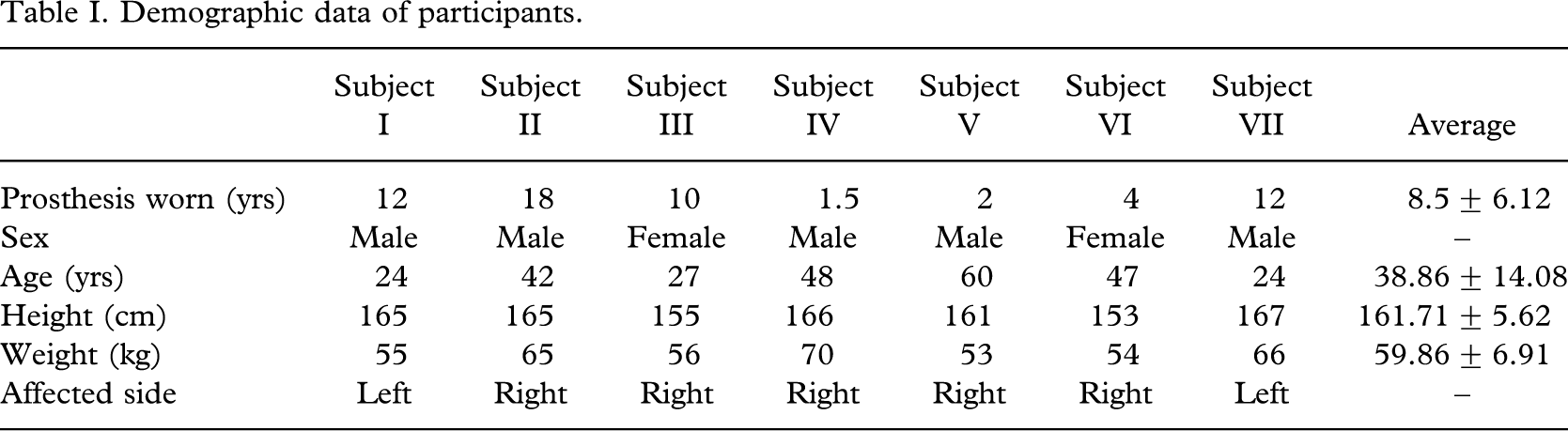
Seven lower limb amputee subjects (5 males and 2 female), age 24 – 60 years, mean age 38.86±14.08 years, height 161.71±5.62 cm, weight 59.86±6.91 kg (Table I), volunteered to participate in this study.
Demographic data of participants.
Potential subjects were randomly selected and contacted from a database of Department of Physical Medicine and Rehabilitation Hospital, Chung Shan Medical University, Taichung, Taiwan. A self-reported medical history screened potential participants for orthopaedic or neurological conditions such as Parkinson's disease, diabetes, peripheral neuropathy, stroke, disabling arthritis, uncorrected visual problems, dizziness or vertigo, use of assistive walking devices, joint injury, and joint implants. Subjects who reported these conditions were excluded from the study. Of the seven unilateral trans-tibial amputees who took part in the study one subject was excluded due to medical instability at the time of evaluation of single leg quiet standing. The study was approved by the ethics committee of cooperation hospital. All subjects signed an informed consent before entering the study.
Treatment protocol
Single leg quiet standing test
In this study, the experiment for evaluating the effects of sub-threshold low level stimulation to the balance performance during single leg quiet standing for amputee subjects was first performed. Subjects were asked to maintain balance without sub-threshold low level stimulation and with sub-threshold low level stimulation on the sound side leg for as long as they could, with arms across their chest and with a steady forward focus. Subjects were allowed to raise their arms out to the side if needed for balance. Subjects maintained a small amount of flexion in their stance knee joint during the balance task which was performed with the subject in single leg quiet standing position. Subjects performed two to three practice trials before data were recorded by the force measurement system. The single-leg balance task was performed 6 times, without sub-threshold low level stimulation applied in random order during 3 of the 6 trials. Sit-down rest for 15 min was provided every trial.
To provide the safety measure for preventing subject's falling during the experiments, a safety harness was used for all subjects. The harness was suspended from the ceiling and was adjusted so that it was not supporting the subject's weight but would catch the subject if they completely lost balance. During the three stimulation trials, electrical input was applied through surface electrodes on the lateral aspects of the subject's quadriceps muscle in the sound side leg. The electrical stimulation signal was applied for the entire duration of the stimulation trial. The stimulation amplitude was well below the cutaneous sensation threshold of all subjects, as confirmed by a threshold measurement prior to testing.
Dynamic treadmill ambulatory test
In this study, the second experiment for evaluating the effects of visual-auditory biofeedback to the gait performance during treadmill dynamic ambulatory test for lower limb amputee subjects was performed. To implement the visual-auditory biofeedback mechanism, two force sensors were attached underneath the prosthesis as foot touch switches. The sensors were placed at front and back positions of the foot. These two foot switches were used for detecting the heel contact and toe push-off stage during gait cycle. The visual-auditory biofeedback signals were provided to give subjects a clue to recognize the correct gait phase. If the two foot switches caused contact signals simultaneously this indicated an abnormal gait such as when flat-foot occurs. In this condition, the information from switches was ignored.
A commercially available treadmill (Model AG-7600, Aerogym Trading Company, Taichung, Taiwan) was used as a dynamic ambulatory test platform in this study. Subjects walked on the treadmill for 20 min in each test session. Each test session included a 5 min warm-up, 10 min training period and 5 min cool-down. Speed was increased each minute, as tolerated. No physical assistance was provided and heart rate of the subject was monitored. In order to provide visual-auditory biofeedback synchronized with subject's gait cycle, the Zebris instrumented insoles were used.
Outcome measures
Single leg quiet standing balance indices
Four different static balance performance indices were defined and used for assessment. One index is a time related measure and the other three indices were body sway related measures. The detailed definition of each balance index was elaborated as follows: (i) Holding Time Index (HTI): total duration in which the subject maintains balance on a single leg; (ii) Constant Time Sway Length Index (SLI): total distance between consecutive points on the COM trajectory over a constant time interval; (iii) Maximum Sway Distance Index (MSDI): the maximum sway distance is defined as the point that has the longest distance from the origin point on the COM trajectory; (iv) Average Sway Distance Index (ASDI): the average sway distance is defined as the summation of sway length between origin point to each sampling point on the COM trajectory divided by the total number of sampling.
Dynamic treadmill ambulatory gait performance indices
In addition, four different dynamic ambulatory gait performance indices were defined and used for assessment. All of the four indices are temporal (timing) domain related measures. The detailed definition of each balance index was elaborated as follows: (i) Double Support Period (DSP): the time between initial contact on one foot and toe off on the other foot. For right double support phase, right foot is forward having just landed on the ground and the left foot is backward, being just about to leave the ground; (ii) Constant Time Cadence (CTC): the number of steps of one foot per minute. As there are two steps in a single gait cycle, the cadence is a measure of half-cycles; (iii) Single Support Period (SSP): During the swing phase on one foot, only the other foot is on the ground, giving a period of single support, which ends with initial contact by the swing foot; (iv) Stance/Swing Ratio (SSR): the ratio between time fraction of stance phase and swing phase over a single gait cycle. In each gait cycle, there are thus two periods of double support and two periods of single support.
The stance phase usually lasts about 60% of the cycle, the swing phase about 40%, and each period of double support about 10% (Huitema et al. 2002). Therefore, the ratio of the time elapsed between stance phase and swing phase can normally be assumed as 60%:40% (i.e., 1.5).
Results
Single leg quiet standing tests
From the results of the quiet standing balance tests, the single leg stand holding time of six subjects increased with sub-threshold low level electrical stimulation, indicating an overall improvement in balance performance in the stimulation condition. Aslo, the SLI, MSDI and the ASDI were found decreased for six subjects. The improvement ratio of these balance performance indices across subjects for single leg quiet standing tests resulted in 132.34% in HTI, 44.61% in MSDI, and 61.45% in ASDI respectively as shown in Table II. No significant change was found in the SWI.
Comparison of static balance performance indices under single leg quiet standing with and without stimulation.
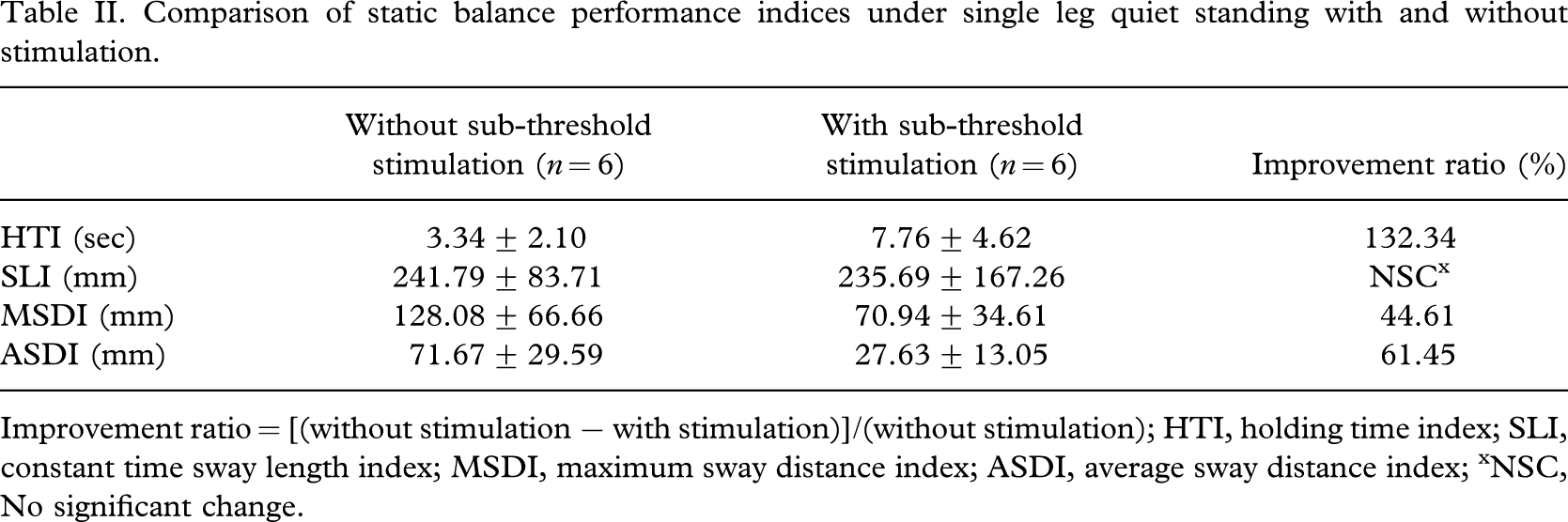
Improvement ratio = [(without stimulation − with stimulation)]/(without stimulation); HTI, holding time index; SLI, constant time sway length index; MSDI, maximum sway distance index; ASDI, average sway distance index; xNSC, No significant change.
Dynamic treadmill ambulatory tests
From the results of the dynamic treadmill ambulatory tests, both affected side and sound side, double support time periods were found to be increased. The improvement ratio of gait performance indices across subjects resulted in a 6.67% increase in DSP (affected side), 5.09% in CTC, 16.67% in SSP (sound side), 45.30% in SSR (sound side), 40.30% in SSR (affected side) respectively. For each of these measures, all of the seven subjects showed improvement with visual-auditory biofeedback (Table III).
Comparison of gait performance indices under treadmill dynamic ambulatory with and without visual-auditory clue.
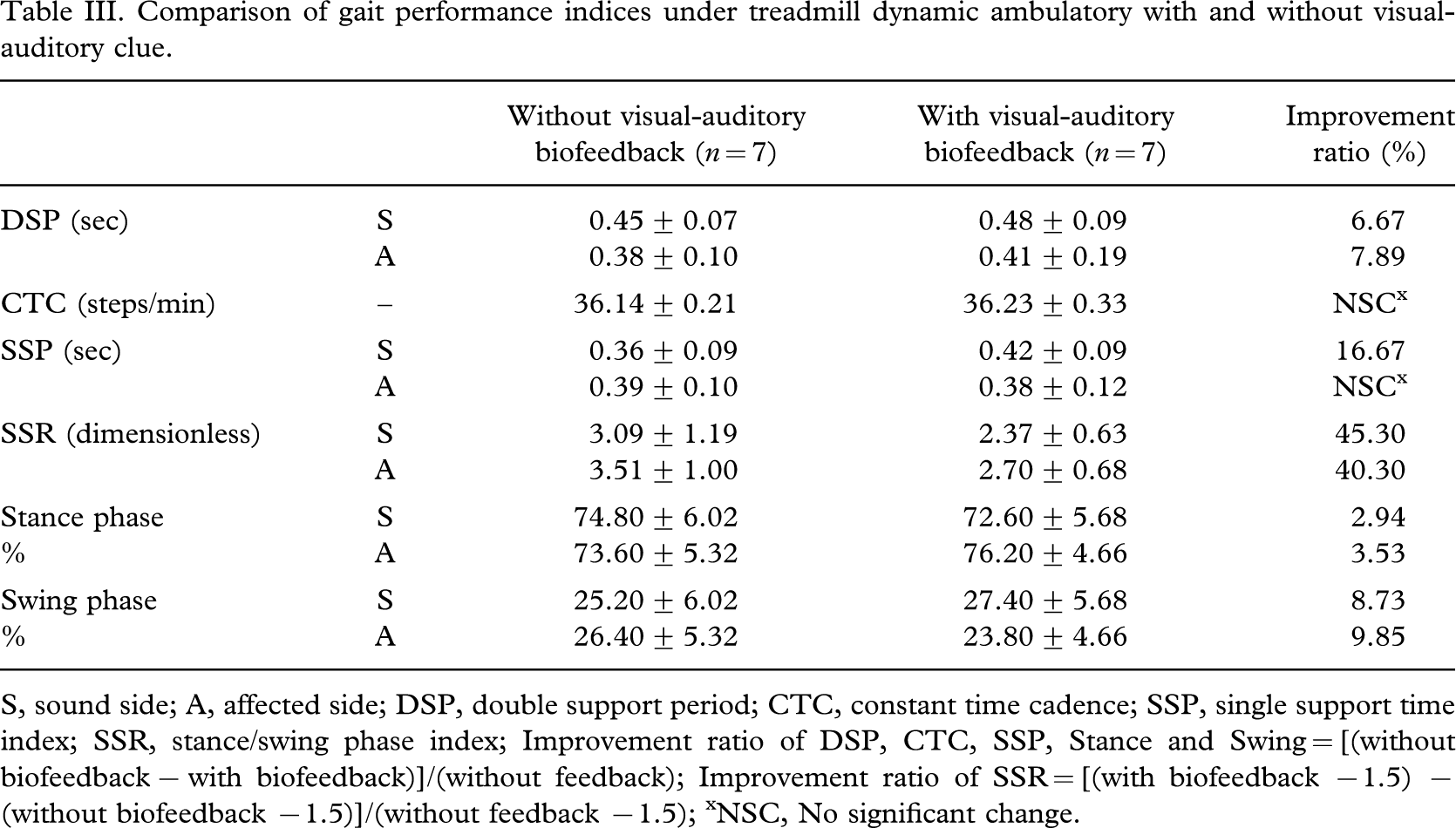
S, sound side; A, affected side; DSP, double support period; CTC, constant time cadence; SSP, single support time index; SSR, stance/swing phase index; Improvement ratio of DSP, CTC, SSP, Stance and Swing = [(without biofeedback − with biofeedback)]/(without feedback); Improvement ratio of SSR = [(with biofeedback −1.5) − (without biofeedback −1.5)]/(without feedback −1.5); xNSC, No significant change.
Discussion
Static balance performance
This study demonstrated that imperceptible low level electrical stimulation with triangular waveform, when applied to the quadriceps, could enhance the static balance performance of lower limb amputee patients. This improvement was demonstrated in single leg quiet standing test condition. The fact that balance performance improved when sub-threshold low level electrical stimulation was applied was demonstrated as an overall reduction in postural sway and increase in single leg support time, and suggests that balance perturbations in any task might be more easily overcome with the application of low-level electrical stimulation signals. From Table I, the improvement ratio of three static balance performance indices noted in this study is the range of 44.61 ∼ 132.34%. The most significant improvement was shown in single leg support time index (HTI).
A promising rehabilitation strategy using low-level noise stimulation (either electrical or mechanical) has recently been shown to improve the sensitivity of the human somatosensory system (Dhruv et al. 2002; Liu et al. 2002). In addition, noise-based sensory enhancement to improve human balance control for healthy elderly adults was reported (Gailey and Clark 1992; Olsson 1990). In this study, the authors extended the effect of low-level electrical inputs, instead of noise, applied to the quadriceps muscle on the amputee subjects. The result of this study suggests that sub-threshold low level electrical stimulation even with triangular waveform can enhance static balance performance. The comparison of sub-threshold low level electrical stimulation with different waveforms (i.e., noise, triangular etc.) on amputee's balance performance will be followed.
The sub-threshold low level electrical stimulation used in this study is likely effective in enhancing the function of the human sensor-motor system because of the electrical nature of information transfer in sensory neurons. Low-level electrical signals can cause small changes in receptor trans-membrane potentials. This depolarization in the local membrane potential brings the neuron closer to threshold, thus making it more likely to fire an action potential in the presence of a weak signal. The electrical depolarization when combined with graded potentials from mechanical stimuli could provide a mechanism by which normally sub-threshold mechanical stimuli become detectable in the presence of electrical low level signals.
Dynamic balance performance
This study also demonstrated that the visual-auditory proprioceptive biofeedback was more effective to improve dynamic postural control capabilities for lower limb amputee patients. Possible explanation for the increased double support period of the experimental group may be related to visual-auditory biofeedback providing subjects a clue to recognize the correct gait phase. It has also been suggested that the more intensive training provided by the treadmill may account for the greater improvement observed in several of the gait parameters of amputee subjects trained by treadmill ambulation. It has been shown, for instance, that gains in cadence are positively correlated to actual time spent practising gait or visual-auditory biofeedback (Davis et al. 2004). On the other hand, the ratio between time fraction of stance phase and swing phase of the amputees with visual-auditory biofeedback is more symmetrical to normal gait compared with the amputees without visual-auditory biofeedback. This experimental evidence indicates that the visual-auditory biofeedback may guide the subject in the right way of dynamic ambulation. These results implied that visual-auditory or proprioceptive biofeedback is an effective way of achieving dynamic balance, gait symmetry in lower limb amputee patients. The improvement of dynamic balance performance indices shows in Table III.
Several studies have shown that amputee gait is asymmetrical compared to normal gait (Inman et al. 1981; Boonstra et al. 1994). In addition, experimental results indicated that the asymmetry occurred in stance-phase (longer for the intact leg) and the swing-phase (shorter for the intact leg). The majority of the patients took a longer step with the prosthetic leg. Some studies indicated that the amputee could only vary gait-speed with the healthy leg, because of the inability of the artificial knee-joint to adjust actively (Boonstra et al. 1993). In this study, it is needed to pay more attention to the increase in cadence (CTC) and symmetry in gait (SSR) with visual-auditory proprioceptive biofeedback which can be achieved because all the subjects are trans-tibial amputees.
This study is unique in that, unlike most investigations in this field, visual-auditory biofeedback was not used during treadmill training of the subjects. This study demonstrates that the experimental group with very limited gait abilities (e.g., average cadence 36.23 steps/min and affected side single support period 0.38 sec), are able to tolerate treadmill training with visual-auditory biofeedback. The proposed system was accepted by all amputees in this study. All tested subjects rated it as comfortable and helpful in encouraging ambulatory training. In particular, auditory feedback was found useful by both amputees and therapists; amputees to monitor the adequacy of their gait pattern, therapists to monitor patient progress and achievements during therapy sessions. However, visual biofeedback seemed to be of less value. Most of the tested subjects did not make good use of the information presented on the monitor which may be mainly due to interference with auditory biofeedback. Most subjects easily adapted to auditory rather than visual biofeedback.
The outcome of this study suggested that both sub-threshold low level electrical stimulation and visual-auditory biofeedback were effective in the improvement of either static balance or dynamic gait performance respectively and the combined effect of these two proprioceptive rehabilitative strategies on balance performance remained at follow-up. However, further work involving a larger sample of subjects is needed to test the hypothesis. In addition, there is also a need for more extensive clinical assessment and classification of amputees to determine the extent to which different amputee populations can benefit from the proposed strategies. Future studies should include larger randomly controlled groups, which will further analyse and compare the changes induced by the different training protocols, and will provide more information concerning effect of visual-auditory biofeedback on various patient sub-groups. Further research should also investigate other important related issues such as long-term effect of training technique and relation between intensity of training and response. Furthermore, it is indicated that for some important gait parameters, which include functional walking ability, stride length, percentage of single stance period, and EMG muscular activity, treadmill training may induce changes not obtained by conventional gait training. Additional research with larger study groups and long-term information gathering is recommended to further evaluate the effectiveness of visual-auditory biofeedback in the early stages of amputee rehabilitation.
Conclusion
This pilot study constitutes the first steps toward assessing the clinical significance of using sub-threshold low level electrical stimulation and visual-auditory biofeedback to improve static balance and dynamic gait performance for amputees. This work indicated that the sub-threshold stimulation and visual-auditory biofeedback techniques may prove useful in overcoming sensory loss for unilateral trans-tibial amputees. The experimental results suggested that proposed proprioceptive biofeedback implementations can be effective in achieving static balance and symmetric gait performance for amputees respectively. The results have important implications on the design of rehabilitation training protocols for individuals with amputee. In the future, the proposed device may be redesigned as a wearable prosthetic which may potentially reduce the frequency and severity of falls for lower limb amputee patients.
Footnotes
Acknowledgement
The authors would like to thank physical therapist, Chun-Hou Wang, for his assistance with recruiting amputee subjects and guidance during the conducting of this study.
