Abstract
Creep resistant steels must be stable over long periods of time under severe operating conditions. This paper reviews precipitation in Cr–Mo steels and maraging steels destined for elevated temperature applications. It also covers aspects of structural evolution, strengthening and degradation, all of which are essential for alloy design. Two classes of steels are emphasised, those with origins in the power generation industries, and a novel approach based on intermetallic precipitates.
This review was submitted as part of the 2019 Materials Literature Review Prize of the Institute of Materials, Minerals and Mining run by the Editorial Board of MST. Sponsorship of the prize by TWI Ltd is gratefully acknowledged.
Keywords
Introduction
 to 450
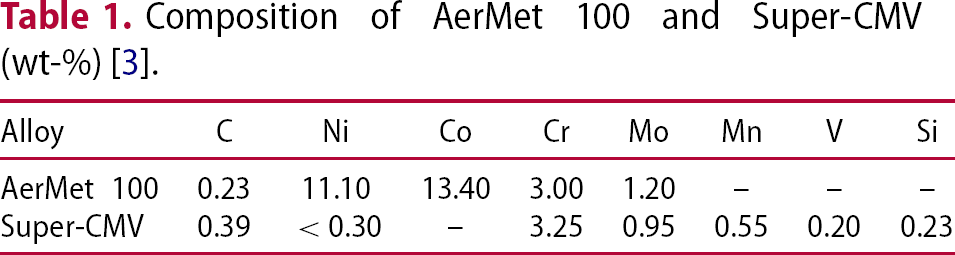
to 450 where creep resistance, toughness and microstructural stability become important [1]. An improved shaft would help reduce the engine core diameter, engine weight, with a high bypass ratio and enable high bypass ratio leading to greater propulsive efficiency [2]. Precipitates have an important role in the achievement of good creep properties. This review covers common precipitates and their effects in creep-resistant steels, in particular, Cr–Mo steels and maraging alloys (Table 1).
where creep resistance, toughness and microstructural stability become important [1]. An improved shaft would help reduce the engine core diameter, engine weight, with a high bypass ratio and enable high bypass ratio leading to greater propulsive efficiency [2]. Precipitates have an important role in the achievement of good creep properties. This review covers common precipitates and their effects in creep-resistant steels, in particular, Cr–Mo steels and maraging alloys (Table 1).
Composition of AerMet 100 and Super-CMV (wt-%) [3].
Current shaft materials
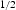 due to the nano-scale Mo-rich M
due to the nano-scale Mo-rich M C [3,8]. Although AerMet 100 is used as part of the shaft, it is not a creep resistant alloy and does not have microstructural stability if tempered above
C [3,8]. Although AerMet 100 is used as part of the shaft, it is not a creep resistant alloy and does not have microstructural stability if tempered above 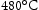 because of unstable reverted austenite formation and precipitation coarsening, resulting in loss in toughness and strength, respectively [9]. The nature of the inertia friction welding process generates a heat-affected zone with metastable martensite and retained austenite in Super-CMV and retained austenite in AerMet 100, which must be eliminated through heat treatment [3].
because of unstable reverted austenite formation and precipitation coarsening, resulting in loss in toughness and strength, respectively [9]. The nature of the inertia friction welding process generates a heat-affected zone with metastable martensite and retained austenite in Super-CMV and retained austenite in AerMet 100, which must be eliminated through heat treatment [3].
Mechanical property of Super-CMV and AerMet 100 (‘fatigue’ is the maximum stress at R = 0 (R= ) to give a fatigue life of 105 cycles) [7].

Design criteria
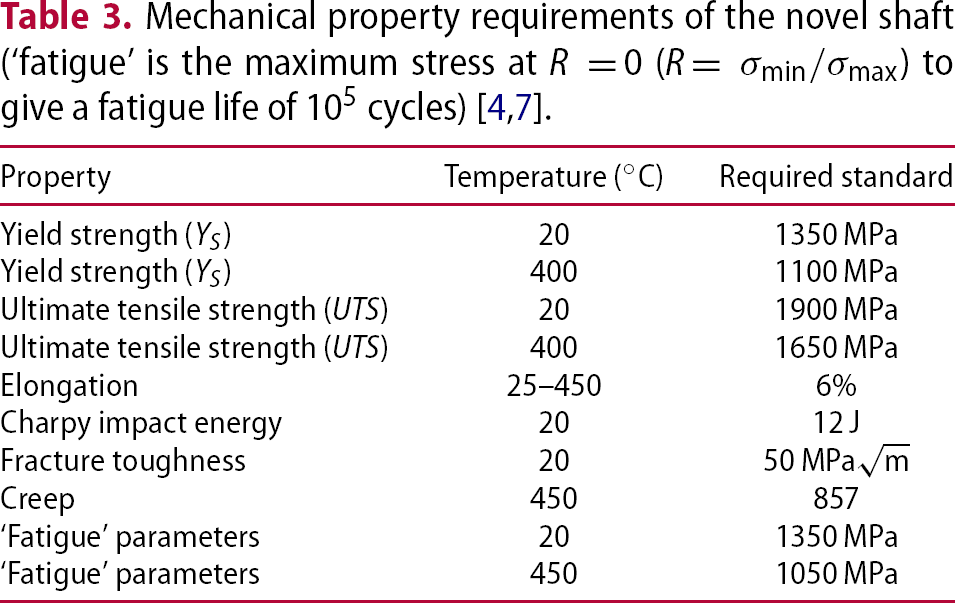
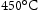 . Based on current shafts, the novel shaft material must combine the mechanical properties of Super-CMV and AerMet 100 in a single alloy, which requires an excellent combination of strength, ductility, toughness, thermal stability and creep resistance so that it can serve in the temperature gradients expected during service in an aircraft engine as listed in Table 3 [5]. The required ultimate tensile strength is approximately 1900 MPa at
. Based on current shafts, the novel shaft material must combine the mechanical properties of Super-CMV and AerMet 100 in a single alloy, which requires an excellent combination of strength, ductility, toughness, thermal stability and creep resistance so that it can serve in the temperature gradients expected during service in an aircraft engine as listed in Table 3 [5]. The required ultimate tensile strength is approximately 1900 MPa at 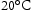 and 1150 MPa at
and 1150 MPa at 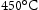 . The creep properties of Super-CMV are acceptable, however, the aim is to achieve strength similar to that of Super-CMV at
. The creep properties of Super-CMV are acceptable, however, the aim is to achieve strength similar to that of Super-CMV at 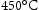 for the designed alloy at
for the designed alloy at 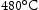 with 0.2% or 0.5% total plastic strain [10]. During service, high cycle fatigue from the torque transfer to the shaft and engine (over 105 cycles before failure) is required under conditions of R = 0 (R=
with 0.2% or 0.5% total plastic strain [10]. During service, high cycle fatigue from the torque transfer to the shaft and engine (over 105 cycles before failure) is required under conditions of R = 0 (R= 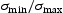 ). High purity and good surface finish are desirable for better fatigue properties. It also needs to be able to operate at
). High purity and good surface finish are desirable for better fatigue properties. It also needs to be able to operate at 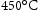 for more than 3000 h, without the occurrence of dislocation creep [7]. In addition, microstructural stability and mechanical stability over the service life are required.
for more than 3000 h, without the occurrence of dislocation creep [7]. In addition, microstructural stability and mechanical stability over the service life are required.
Cr–Mo steels
Chromium–molybdenum (CrMo) and chromium–molybdenum–vanadium (CrMoV) alloys are intended mostly for high temperature (350–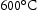 ) service. These alloys are expected to have a service life at least for 25,000 h [11]. Owing to their excellent combination of tensile strength, corrosion resistance and creep resistance at high temperatures and high pressure, the alloys have a good strength to weight ratio, allowing the use of smaller diameter shafts to reduce the overall weight. Higher creep strength can be achieved by adding solutes such as Mo, W, V, Nb [12], but to use the Cr–Mo alloy in aeroengine shafts, necessitate a high fracture toughness, low ductile-to-brittle transition temperature and significant creep resistance. Meanwhile, microstructural degradation needs to be minimised to achieve higher service temperature. Therefore, the carbide formed during tempering must be resistant to coarsening.
) service. These alloys are expected to have a service life at least for 25,000 h [11]. Owing to their excellent combination of tensile strength, corrosion resistance and creep resistance at high temperatures and high pressure, the alloys have a good strength to weight ratio, allowing the use of smaller diameter shafts to reduce the overall weight. Higher creep strength can be achieved by adding solutes such as Mo, W, V, Nb [12], but to use the Cr–Mo alloy in aeroengine shafts, necessitate a high fracture toughness, low ductile-to-brittle transition temperature and significant creep resistance. Meanwhile, microstructural degradation needs to be minimised to achieve higher service temperature. Therefore, the carbide formed during tempering must be resistant to coarsening.
Microstructure
 C
C and minor phases as spherical NbC, fine needle-like V(C,N), M
and minor phases as spherical NbC, fine needle-like V(C,N), M C (M = metal) etc. [12]. Its microstructure can be changed by thermal or mechanical processes [16], therefore gradual microstructural degradation may take place during service in creep conditions, resulting in reduced properties. Furthermore, the nucleation and coarsening of precipitates during service are detrimental to the microstructure, resulting in the final creep failure.
C (M = metal) etc. [12]. Its microstructure can be changed by thermal or mechanical processes [16], therefore gradual microstructural degradation may take place during service in creep conditions, resulting in reduced properties. Furthermore, the nucleation and coarsening of precipitates during service are detrimental to the microstructure, resulting in the final creep failure.
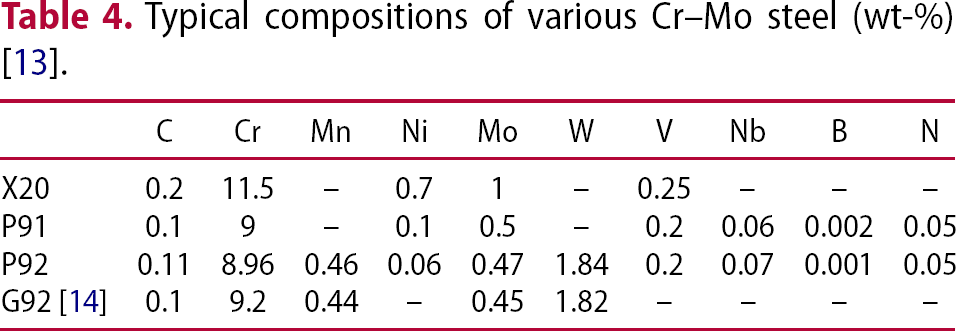
Typical compositions of various Cr–Mo steel (wt-%) [13].
MX precipitates
‘MX’ represents a compound of metal atoms with X = C or N. In the presence of strong carbide or nitride forming elements (Nb, V, Ti, Zr, Ta, etc.), the formation of MX is helpful in enhancing long-term creep resistance. A typical heat treatment involves austenitisation between 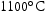 and
and 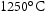 for 30–60 min conducted normally before service to dissolve as much as MX possible [17]. Subsequent fine precipitation is beneficial in retarding creep. There are four types of MX here, coarse NbX that remains after normalising, fine spherical NbX and platelike VX that form during tempering, and VX ‘wings’ formed on fine NbX particles during tempering [18].
for 30–60 min conducted normally before service to dissolve as much as MX possible [17]. Subsequent fine precipitation is beneficial in retarding creep. There are four types of MX here, coarse NbX that remains after normalising, fine spherical NbX and platelike VX that form during tempering, and VX ‘wings’ formed on fine NbX particles during tempering [18].
The MX carbonitride has a NaCl face-centred cubic lattice, and precipitates mainly in the matrix within laths and some along boundaries (Figure 1). The creep property of steel is highly dependent on the solubility of MX and proportion of each element that add. Kikuchi et al. [20] proposed that excessive additions of M and X lead to coarse MX and rapid coarsening of later precipitated MX. However, this does not apply before the solubility limit is reached.
Illustration of precipitation in high Cr ferritic steel after Maruyama [19].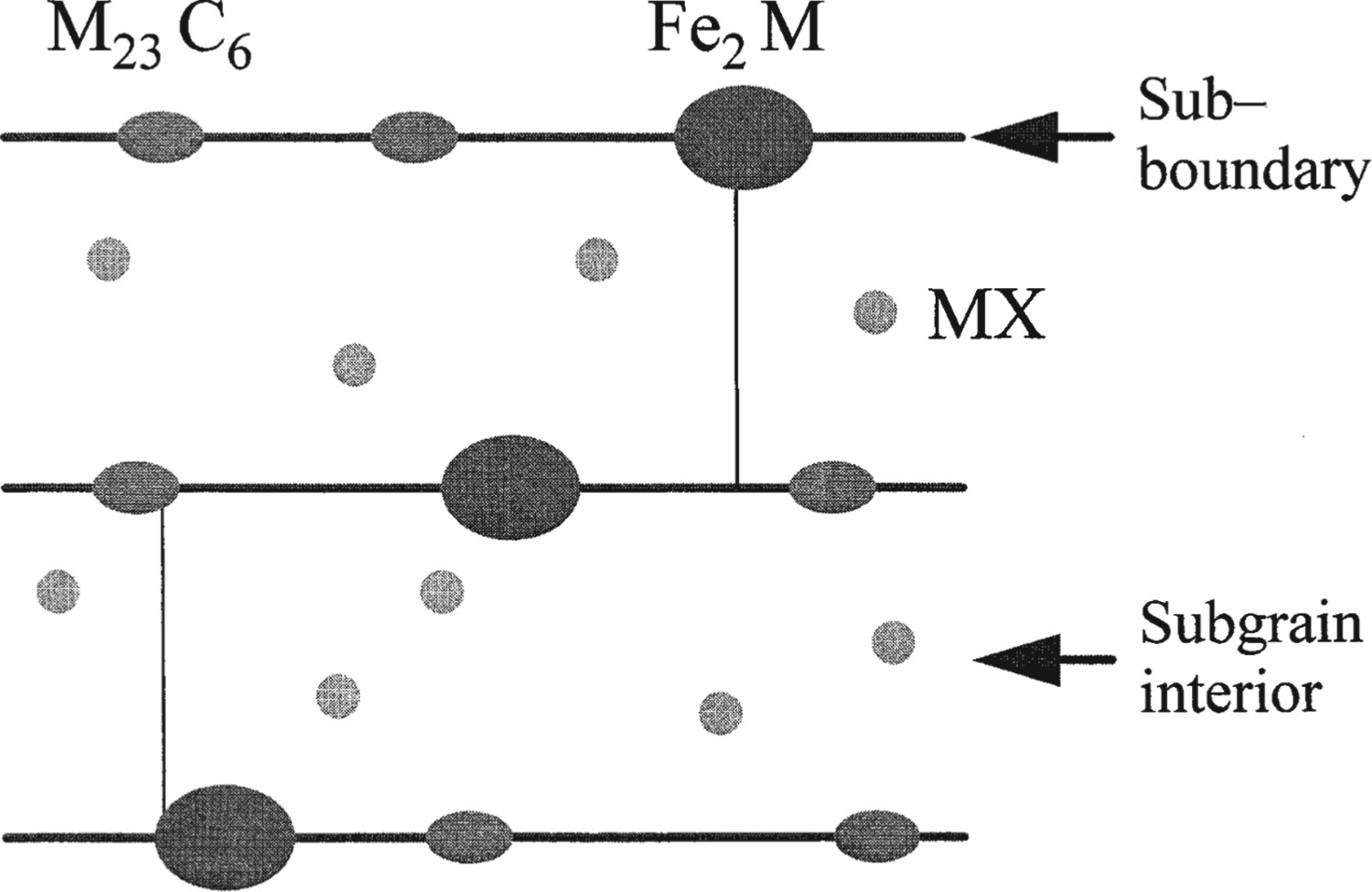
M C
C
‘M C
C ’ is a general notation for Cr
’ is a general notation for Cr C
C where the Cr is partially substituted by Ni, Mo and Fe. It has a fcc lattice, and precipitates in the vicinity of grain boundaries, sub-grain boundaries and also within austenite grain. It can form in 30 min at
where the Cr is partially substituted by Ni, Mo and Fe. It has a fcc lattice, and precipitates in the vicinity of grain boundaries, sub-grain boundaries and also within austenite grain. It can form in 30 min at 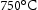 in high chromium steel [21]. These precipitates are usually coarse (∼ 200 nm in size) with a comparatively high coarsening rate and detrimental to mechanical properties, especially creep strength [17]. With the addition of small amounts of boron, the coarsening rate of M
in high chromium steel [21]. These precipitates are usually coarse (∼ 200 nm in size) with a comparatively high coarsening rate and detrimental to mechanical properties, especially creep strength [17]. With the addition of small amounts of boron, the coarsening rate of M C
C is reduced, thus the number density of M
is reduced, thus the number density of M C
C along the grain boundaries increases, impeding grain boundary sliding [19].
along the grain boundaries increases, impeding grain boundary sliding [19].
Beckitt and Clarck [22] have found that M C
C also grow on dislocations, and form continuous chains and clusters. M
also grow on dislocations, and form continuous chains and clusters. M C
C can stimulate dislocation emission, while providing the nucleation sites for further precipitation. Salsmal [23] have also found M
can stimulate dislocation emission, while providing the nucleation sites for further precipitation. Salsmal [23] have also found M C
C plates form around undissolved Nb(C,N) in 16Cr–16Ni–0.8Nb steel, perhaps due to the strain around the undissolved particles.
plates form around undissolved Nb(C,N) in 16Cr–16Ni–0.8Nb steel, perhaps due to the strain around the undissolved particles.
M C
C
M C has a wide composition range, and forms at long aging times. The composition of M
C has a wide composition range, and forms at long aging times. The composition of M C in steel can be Fe
C in steel can be Fe Nb
Nb C or Fe
C or Fe Mo
Mo C. Honeycombe [24] has proposed that prolonged tempering at
C. Honeycombe [24] has proposed that prolonged tempering at 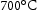 leads to the massive formation of M
leads to the massive formation of M C particles on the grain boundaries which replaces M
C particles on the grain boundaries which replaces M C. This results in softening of the steel, which is not desirable for creep resistant steel. However, the addition of vanadium is able to stabilise Mo
C. This results in softening of the steel, which is not desirable for creep resistant steel. However, the addition of vanadium is able to stabilise Mo C [25].
C [25].
Laves phase
Laves phase is an intermetallic compound, with the general formula A B (A = Fe, Cr, B = Mo, W, Nb) [26]. It normally is found in steel containing 2 Mo wt-% and 9–12 Cr wt-%, where the introduction of Cr increases its temperature stability [27]. In common alloys, Laves phase is thermodynamically stable between 600
B (A = Fe, Cr, B = Mo, W, Nb) [26]. It normally is found in steel containing 2 Mo wt-% and 9–12 Cr wt-%, where the introduction of Cr increases its temperature stability [27]. In common alloys, Laves phase is thermodynamically stable between 600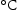 and
and 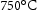 [7], which is higher than the service temperature of aerospace shafts (
[7], which is higher than the service temperature of aerospace shafts (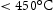 ).
).
There is debate on the effect of Laves phase on the long-term creep strength; its precipitation depletes the Mo and W concentrations in the matrix, which can lead to a reduction on the creep strength if the Laves phase precipitates are coarse with a low number density [28]. However, Laves phase precipitates have been found to contribute to creep strength by precipitation hardening under certain circumstances recently [28].
Lee et al. [14] have reported the size of Laves phase of ∼80 nm in diameter after exposure at 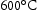 for more than 300 h in Grade 92 steel (Table 4). He also found creep strength dropped when Laves phase reached an average diameter of 130 nm where the formation of cavity at Laves phase triggers the brittle intergranular fracture. Exposure at
for more than 300 h in Grade 92 steel (Table 4). He also found creep strength dropped when Laves phase reached an average diameter of 130 nm where the formation of cavity at Laves phase triggers the brittle intergranular fracture. Exposure at 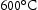 for 5000–10,000 h also leaves a particle size of 280 nm in P92 steel [29].
for 5000–10,000 h also leaves a particle size of 280 nm in P92 steel [29].
Strengthening mechanisms
There are three main strengthening mechanisms in the heat resistant Cr–Mo steel that are active under the circumstances of shaft applications:
Solution strengthening
W and Mo in solid solution improve the creep strength of martensite [30]. As shown in Figure 2 [31], the creep rate of α-Fe is reduced by two orders of magnitude by the addition of 2.3 wt-% W in a binary Fe–W alloy. The α-Fe is free of particles, subgrain structures and free dislocations so these data represent a controlled experiment. The magnitude of the benefit is, however, small when there is an overwhelming constitution.
Minimum creep rate of α-Fe and ferritic steels at 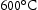
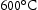 (mass%) [31].
(mass%) [31].
Precipitate strengthening
The addition of alloying elements enhances the creep strength either by being dissolved in the matrix and precipitates as fine carbides (NbC, Mo C, M
C, M C
C ) or intermetallic (β-NiAl, Laves phase) [32]. NbC and VC particles remain the most stable in terms of their resistance to coarsening [33]. As shown in Figure 2, they reduce the creep rate tremendously in Fe-VNbC (mass%) compared with α-Fe [31] by hindering dislocation movement and retarding dislocation recovery [34]. Fine M
) or intermetallic (β-NiAl, Laves phase) [32]. NbC and VC particles remain the most stable in terms of their resistance to coarsening [33]. As shown in Figure 2, they reduce the creep rate tremendously in Fe-VNbC (mass%) compared with α-Fe [31] by hindering dislocation movement and retarding dislocation recovery [34]. Fine M C
C is also effective in this respect, by slowing down the recovery of dislocation structure while its coarsening during creep results in acceleration of creep deformation [35]. This is also consistent with Mikami [36] who reported a condensation and coarsening of precipitates accelerating the degradation. The increase in creep resistance due to fine Fe
is also effective in this respect, by slowing down the recovery of dislocation structure while its coarsening during creep results in acceleration of creep deformation [35]. This is also consistent with Mikami [36] who reported a condensation and coarsening of precipitates accelerating the degradation. The increase in creep resistance due to fine Fe M has been reported by Igarashi [37]. The thermal stability of precipitates at elevated temperature is important in resisting grain growth and slowing down dislocation recovery; thus enhancing long-term creep strength.
M has been reported by Igarashi [37]. The thermal stability of precipitates at elevated temperature is important in resisting grain growth and slowing down dislocation recovery; thus enhancing long-term creep strength.
Dislocation strengthening
A comparison between the creep rate of ferrite (F) without dislocation substructure and banite (B) with dislocation substructure is shown in Figure 3 [38]. Th bainite has a similar dislocation substructure to tempered martensite [19]. The contribution of particles, solute atoms and dislocations lower the creep rate. However, the dislocation substructure is not effective without the introduction of particles and solute atoms.
Minimum creep rate of ferritic (F) and banitic (B) steels at 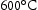
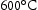 . +P and +S represent the addition of MX particles and solute atoms (0.6 W + 0.15 Mo in wt-%) to the steels [38].
. +P and +S represent the addition of MX particles and solute atoms (0.6 W + 0.15 Mo in wt-%) to the steels [38].
Steel with a smaller subgrain size has better creep than one with a coarser [19]. In the same study, 11 Cr steel tempered at 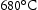 has a much higher dislocation density than tempered at
has a much higher dislocation density than tempered at 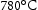 , suggests the dislocation density increases with decreasing tempering temperature. Notably, the former one reaches 1% strain at 70% rupture life, and latter at 10% of rupture life, even with the same subgrain size. It can be concluded that the large free dislocation density as well as fine subgrain size both contribute to better creep resistance [39]. Moreover, Endo et al. also have reported that the reduced dislocation density and lath boundaries movement are responsible for about 65% loss of hardness in 9Cr–1Mo wt-% steel during creep in the range of stress and temperature from 71 to 167 MPa and 600 to
, suggests the dislocation density increases with decreasing tempering temperature. Notably, the former one reaches 1% strain at 70% rupture life, and latter at 10% of rupture life, even with the same subgrain size. It can be concluded that the large free dislocation density as well as fine subgrain size both contribute to better creep resistance [39]. Moreover, Endo et al. also have reported that the reduced dislocation density and lath boundaries movement are responsible for about 65% loss of hardness in 9Cr–1Mo wt-% steel during creep in the range of stress and temperature from 71 to 167 MPa and 600 to 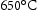 [40].
[40].
Effect of alloying elements
The effects of alloying elements have been summarised in Table 5.
Carbon and nitrogen: For steels with up to 0.2 wt-% carbon, approximately 90% of carbon segregates to the dislocations and lath boundaries during quenching. During tempering, the carbon precipitates, contributing to the overall strength. However, it can reduce toughness, weldability, corrosion resistance and by making carbides more stable can make it difficult to dissolve them during austenitisation [42]. Nitrogen also strengthens by precipitation as nitride of Ti or Nb. However, nitrogen in solid solution is much more effective at strengthening than carbon [17]. It retards the nucleation and coarsening of M Alloying elements: Figure 4 shows that solutes such as Mn, Mo, W delay the transformation to ferrite and pearlite so that martensite can be generated at relatively low cooling rates. They also reduce the martensite-start temperature, giving a fine subgrain structure. Besides enhanced hardenability, wear resistance, corrosion resistance, impact resistance and machinability, Cr and Mo improve the creep properties by precipitation strengthening [19]. They also facilitate carbide precipitation and promote the formation of Laves phase on long-term aging. As shown in Figure 5, the addition of W delays the coarsening of M Time–temperature–transformation diagrams of austenite to ferrite isothermal transformation (a) carbon steel and steel alloyed with weak or non-carbide forming elements; (b) carbon steel and steel alloyed with strong carbide forming elements, after Maalekian [46]. Effect of W on the growth of M
 C
C due to its low solubility in this carbide [43–45].
due to its low solubility in this carbide [43–45]. C
C [47] and the addition of a very small amount of boron has a similar effect [48–50]. Both of these solutes do not influence MX coarsening [51,52]. As strong carbide formers, Nb, Ti and V improve the creep strength by precipitating carbides, nitrides or carbo-nitrides which are stable and do not coarsen during service [17].
[47] and the addition of a very small amount of boron has a similar effect [48–50]. Both of these solutes do not influence MX coarsening [51,52]. As strong carbide formers, Nb, Ti and V improve the creep strength by precipitating carbides, nitrides or carbo-nitrides which are stable and do not coarsen during service [17].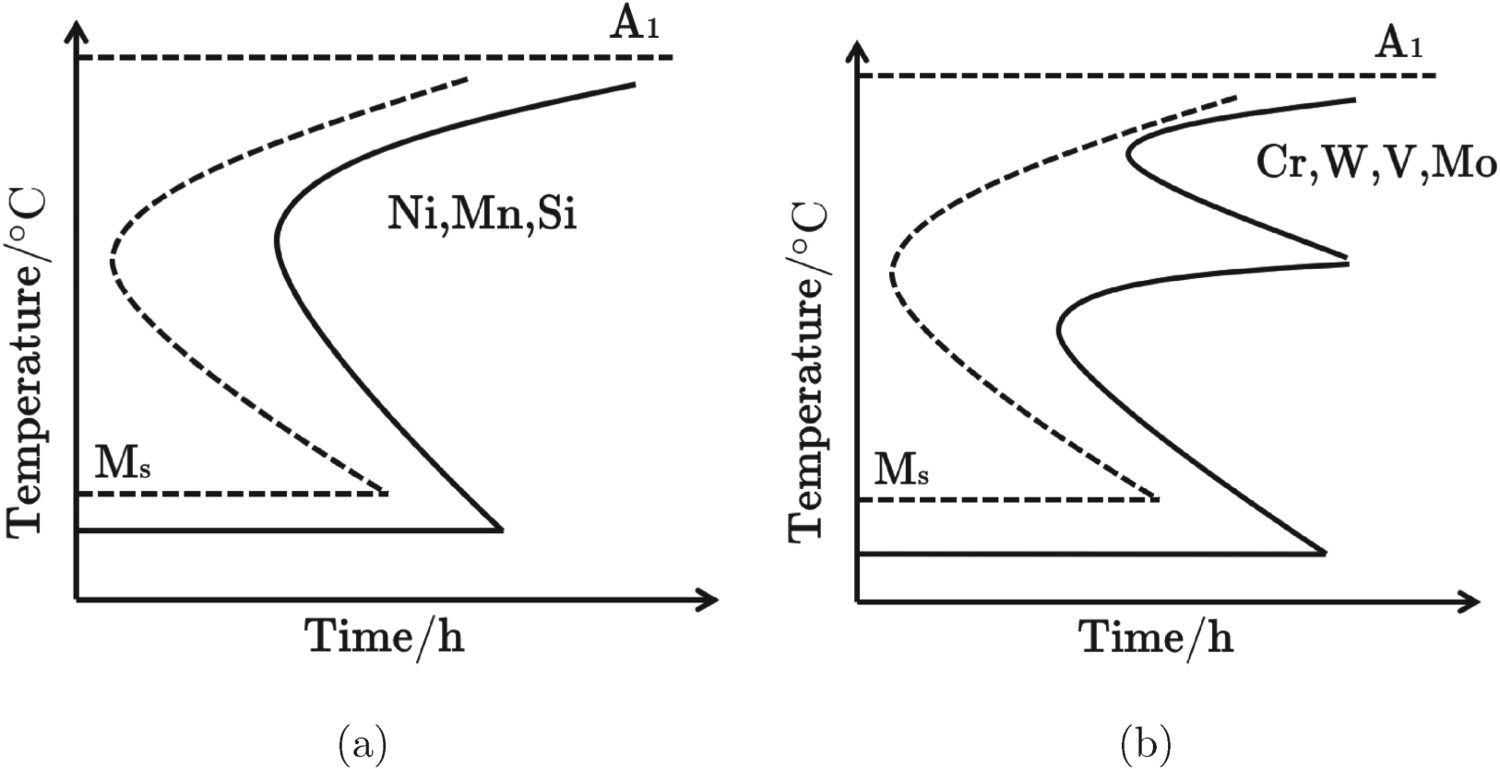

 C
C particles during creep of 9Cr–1W wt-% steel at
particles during creep of 9Cr–1W wt-% steel at 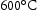 [19].
[19].
Secondary hardening
Secondary hardening in a series of steels with increasing molybdenum content at around The effect of molybdenum on the tempering of quenched steel Fe–0.35C–2Cr wt-% [53]. Comparison of WC and MoC dispersion at peak hardness (tempering at ) [54].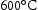 is illustrated in Figure 6 [53]. Needle-shaped Mo
is illustrated in Figure 6 [53]. Needle-shaped Mo C about 100–200 Å long and 10–20 Å in diameter at peak hardness in Fe–2Mo–1C (at-%) [54], growing along <100> direction in ferrite. Tungsten produces similar secondary hardening carbides (Table 6), but the formation of W
C about 100–200 Å long and 10–20 Å in diameter at peak hardness in Fe–2Mo–1C (at-%) [54], growing along <100> direction in ferrite. Tungsten produces similar secondary hardening carbides (Table 6), but the formation of W C is slower because of the smaller diffusivity of tungsten in ferrite relative to that of molybdenum.
C is slower because of the smaller diffusivity of tungsten in ferrite relative to that of molybdenum.
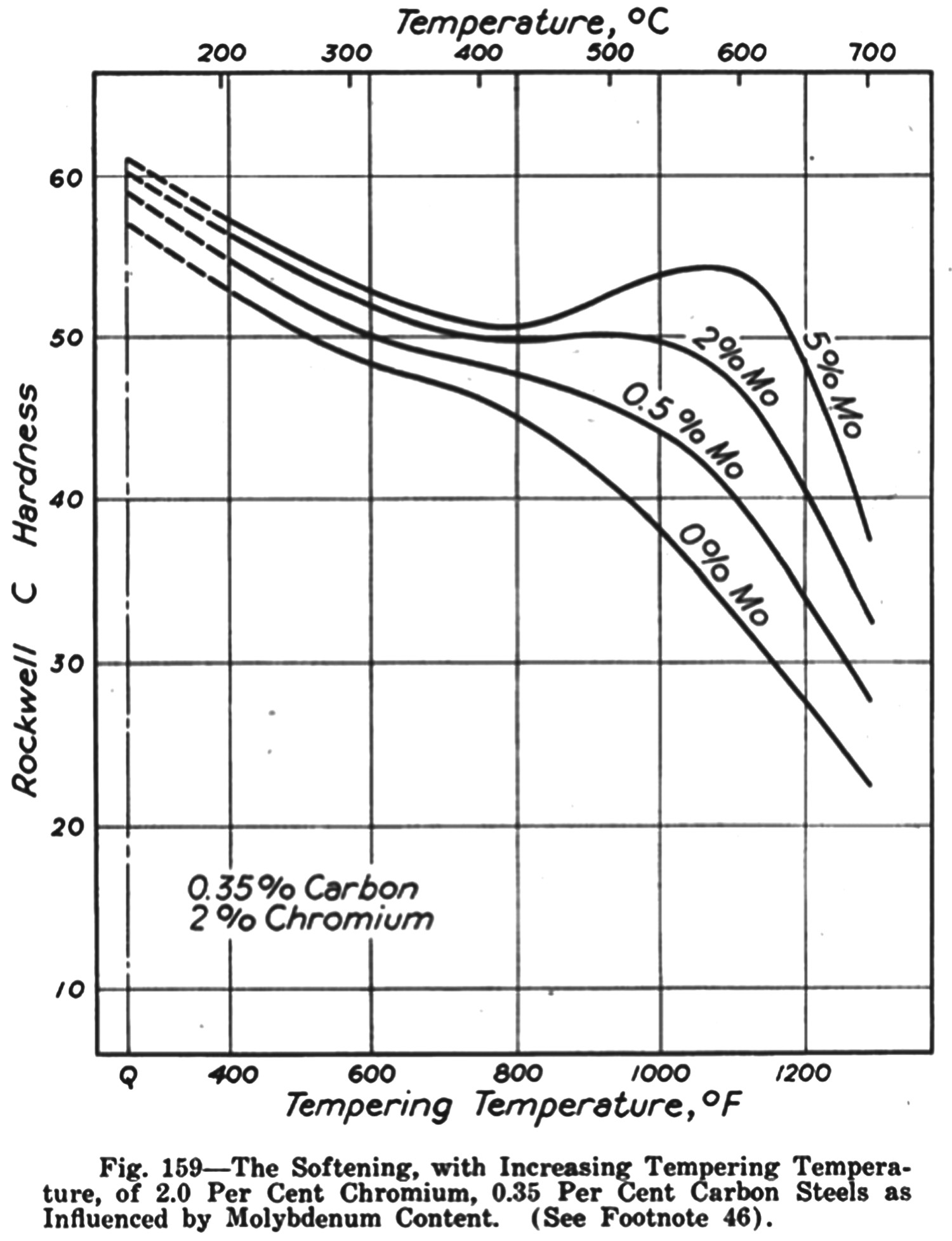

Secondary hardening is more effective with strong carbide formers such as Nb, Ti, V, which enhances the peak hardness and resist overaging. Raynor et al. [25] found that 0.06 Nb at-% or Ti to Fe–3.6 Mo–0.2C wt-% increases the peak hardness by approximately 20 HV at 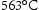 ,and retards overaging. The introduction of 0.5 V wt-% to Fe–2.5 Mo–0.2C wt-% is more effective in this context because of the fine precipitation of a large amount of V
,and retards overaging. The introduction of 0.5 V wt-% to Fe–2.5 Mo–0.2C wt-% is more effective in this context because of the fine precipitation of a large amount of V C
C and its influence on the coarsening of Mo
and its influence on the coarsening of Mo C.
C.
Without forming carbide, cobalt it has a similar effect by limiting dislocation recovery during tempering, which provides more nucleation sites for precipitates [55,56]. It also increases carbon diffusivity, promoting fine precipitate formation [57].
The replacement of coarse cementite by fine (Mo, Cr)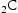 enhances toughness during tempering [55] of 10Ni–8Co–2Cr–1Mo–0.2C wt-% steels.
enhances toughness during tempering [55] of 10Ni–8Co–2Cr–1Mo–0.2C wt-% steels.
Maraging steels
Maraging steels are a class of low-carbon steels with Ni, Cr and other substitutional solute that may precipitate during aging [58]. On cooling, the austenite transforms into relatively soft martensite albeit with a high dislocation density, and intermetallic compounds are then induced by tempering in the range 450–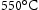 . These steels are strong and tough in the absence of carbon, which makes for a wide range of applications including aerospace, military industry, tooling and machinery [59]. Typical requirements include 0.2% proof strength of 1250–1350 MPa, the tensile strength of 1500–1600 MPa, fracture toughness of 50 MPa
. These steels are strong and tough in the absence of carbon, which makes for a wide range of applications including aerospace, military industry, tooling and machinery [59]. Typical requirements include 0.2% proof strength of 1250–1350 MPa, the tensile strength of 1500–1600 MPa, fracture toughness of 50 MPa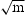 and elongation of 6% as listed in Table 3 [4].
and elongation of 6% as listed in Table 3 [4].
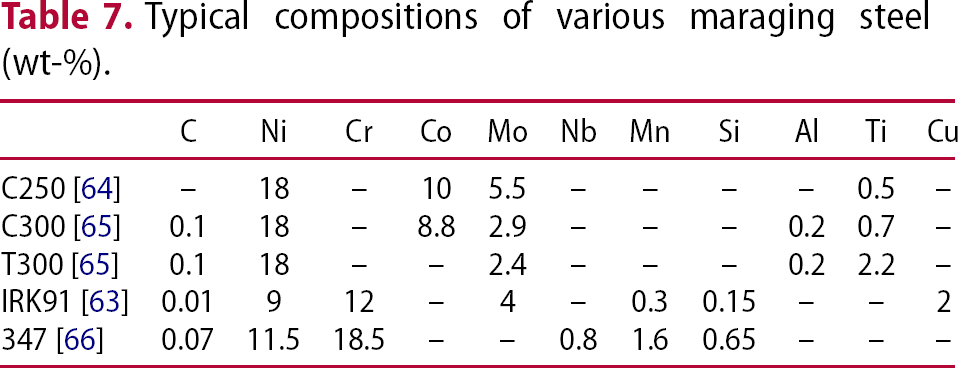
Typical compositions of various maraging steel (wt-%).
Microstructure
The matrix following cooling from the austenite phase field is martensite with some austenite retained. The large concentrations of Ni, Cr and other solutes allow martensitic transformation during oil cooling. A cryogenic treatment may be applied to reduce the retained austenite content [61].
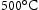 , accelerated by the high dislocation density and the diffusion of substitutional solutes [62]. The precipitated intermetallic compounds in maraging steel are listed in Table 8. The critical precipitates depend on the detailed chemical composition. For applications at approximately 450–
, accelerated by the high dislocation density and the diffusion of substitutional solutes [62]. The precipitated intermetallic compounds in maraging steel are listed in Table 8. The critical precipitates depend on the detailed chemical composition. For applications at approximately 450–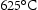 , Laves phase and β-NiAl are desirable due to their excellent high temperature stability.
, Laves phase and β-NiAl are desirable due to their excellent high temperature stability.
Precipitate phases in maraging steels [7].

Laves phase
Fine Laves phase (50 nm) has been reported [63] to impede the movement of subgrains and dislocations in IRK91 (Table 7), these continuing to precipitate strengthening, even though solid solution strengthening described as Mo, W or Cr are depleted [67,68]. W tends to accelerate the coarsening of Laves phase [69–71].
In niobium stabilised 347 steels (Table 7), Fe Nb forms after long aging, as a transient phase which disappears to give way to Fe
Nb forms after long aging, as a transient phase which disappears to give way to Fe Nb
Nb C. Laves phase also appears as a transition precipitates, dissolving as Fe
C. Laves phase also appears as a transition precipitates, dissolving as Fe Nb
Nb C or NbC form [66].
C or NbC form [66].
Similarly, unstable Ni Mo formed at the initial stages of aging gives way to the more stable Fe
Mo formed at the initial stages of aging gives way to the more stable Fe Mo during prolonged aging in C250 maraging steel (Table 7) [64].
Mo during prolonged aging in C250 maraging steel (Table 7) [64].
β-NiAl
The β-NiAl is the main strengthening phase in Al-containing alloys with ordered B2 (CsCl) structure [72]. It typically is stable up to 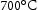 but only precipitates in the absence of Ti; this has been attributed to its tendency to form η-Ni
but only precipitates in the absence of Ti; this has been attributed to its tendency to form η-Ni Ti. β-NiAl precipitates from solute-rich clusters, and grow to be spherical at first [73], become more cuboidal as it grows, and overtime revert to be almost spherical again [74]. β-NiAl uniformly distributed in the matrix with coherency up to 150 nm in radius [62], and maintain its coherency at the aging temperature [75].
Ti. β-NiAl precipitates from solute-rich clusters, and grow to be spherical at first [73], become more cuboidal as it grows, and overtime revert to be almost spherical again [74]. β-NiAl uniformly distributed in the matrix with coherency up to 150 nm in radius [62], and maintain its coherency at the aging temperature [75].
Austenite
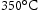 through aging by a diffusion controlled reaction [76,77]. The precipitated austenite is in the form of thin films at the martensite boundaries, and is able to resist coarsening and enhance fracture at the expense of yield strength [78,79]. As shown in Table 9, Co-free T300 tends to form Ni
through aging by a diffusion controlled reaction [76,77]. The precipitated austenite is in the form of thin films at the martensite boundaries, and is able to resist coarsening and enhance fracture at the expense of yield strength [78,79]. As shown in Table 9, Co-free T300 tends to form Ni Ti which results in a lower matrix Ni composition [65]. A low Ni concentration in the matrix reduces the tendency to form austenite. Also, the reversion to austenite can be retarded to reducing the Ni concentration [80].
Ti which results in a lower matrix Ni composition [65]. A low Ni concentration in the matrix reduces the tendency to form austenite. Also, the reversion to austenite can be retarded to reducing the Ni concentration [80].
Matrix composition of C300 and T300 after tempering at for 8 h (wt-%).

Strengthening mechanisms
The dominant strengthening factor is precipitate hardening by the intermetallic compounds [81], for example, fine Laves phase and β-NiAl in the novel maraging steel studied here [82–85].
Apart from precipitation, strong elements such as nickel have meltable sites, for example in enhancing hardenability. If nickel is formed to result in too much retained austenite, then some of it can be replaced with Cr to maintain hardenability [7]. Chromium also provides oxidation and corrosion resistance to the alloy and is effective in solid solution strengthening. Excessive chromium can promote the precipitation of δ-ferrite at higher temperatures [86].
Cobalt increases the M temperature while maintaining the hardenability [59,87]. Tungsten and molybdenum improve the resistance to creep by solution strengthening and forming M
temperature while maintaining the hardenability [59,87]. Tungsten and molybdenum improve the resistance to creep by solution strengthening and forming M C and Laves phase particles [30], although the latter represents a minor contribution due to the tendency for the precipitate to be coarse [88]. Aluminium is necessary for β-NiAl, 1.75 Al wt-% produces 5.0 wt-% β-NiAl at
C and Laves phase particles [30], although the latter represents a minor contribution due to the tendency for the precipitate to be coarse [88]. Aluminium is necessary for β-NiAl, 1.75 Al wt-% produces 5.0 wt-% β-NiAl at 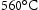 [59]. However, it raises the M
[59]. However, it raises the M temperature, so its concentration needs to be controlled.
temperature, so its concentration needs to be controlled.
Nitrogen promotes coarse Ti(C,N) and AlN particles that form at very high temperatures, which initiate fatigue. Mn and P raise the hardenability but their segregation at grain boundaries can lead to embrittlement in the aged condition [89]. Therefore, N, Mn and P concentrations should be as low as possible.
Grain size and mechanical properties
The relationship between grain size and strength [90,91], ductile to brittle transition [92,93], creep [94,95] and fatigue [96,97] have been measured and modelled for steels. An austenite grain after transformation will contain packets, blocks and laths. A packet has a series of parallel laths, slightly misorientated, whereas a block consists of laths with similar orientations. Based on previous researches on the steel [98], the relationship between the block size (d) and austenite grain size 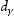 is:
is:
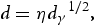
Grain refinement is a traditional way to improve strength according to Hall–Petch Equation [99,100]:
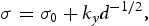
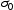 is friction stress, (d) is grain sizes,
is friction stress, (d) is grain sizes, 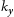 is 2190 MPa μm
is 2190 MPa μm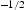 for
for 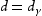 [91,101] and ∼684 HV μm
[91,101] and ∼684 HV μm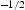 for block size [85].
for block size [85].
The ductile to brittle transition temperature (T ) is highly dependent on the stress required to move dislocations; fine structure is generally associated with a low T
) is highly dependent on the stress required to move dislocations; fine structure is generally associated with a low T , thus longer elongation for crack propagation [102].
, thus longer elongation for crack propagation [102].
A large grain size can benefit creep resistance if the dominant mechanism is grain boundary diffusion [103], but not otherwise. It has often been found that smaller grain size tends to have a lower creep rate and a longer creep life at low to intermediate temperatures (below half of the melting temperature) [94,95,104–106]. Grain boundaries would act as barriers and dislocation sources, which would impede dislocation movement [94]. This suggests additional grain refinement would be promising for improved creep performance.
Methods of grain size refinement
Grain refinement can be achieved through dispersed fine carbides, nitrides or carbo-nitrides that hinder growth at the austenitising temperature by pinning grain boundaries during hot deformation [107]. For 0.10 Nb wt-% or Ti and 0.06 C wt-%, NbC and TiC are stable up to 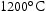 [46]. The vanadium nitride remains undissolved until the temperature exceeds
[46]. The vanadium nitride remains undissolved until the temperature exceeds 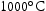 in steel containing 0.10 V wt-% and 0.10 N wt-% [108]. The addition of 0.005 Nb wt-% and 0.06 V wt-% has been found to reduce the prior austenite grain size (PAGS) from approximately 25 μm to 18 μm in 0.01 C–1.25Mn wt-% steel after austenitisation at
in steel containing 0.10 V wt-% and 0.10 N wt-% [108]. The addition of 0.005 Nb wt-% and 0.06 V wt-% has been found to reduce the prior austenite grain size (PAGS) from approximately 25 μm to 18 μm in 0.01 C–1.25Mn wt-% steel after austenitisation at 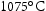 for 1 h by precipitation of fine (Nb,V)C with an average size of around 25±16 nm [109]. For Nb, a fine dispersion of nanosized NbC ( 2–10 nm diameter) is more effective in retarding recrystallization than dissolved Nb in the matrix. An atomic ratio of Nb/C >1 or Nb/(C N) >1 is found to be preferred due to the combination of precipitation strengthening and solid solution strengthening in refining prior austenite grain size[110]. This is also consistent with Kikuchi et al. [111], Adamson [112] and Keown and Pickering's study [113].
for 1 h by precipitation of fine (Nb,V)C with an average size of around 25±16 nm [109]. For Nb, a fine dispersion of nanosized NbC ( 2–10 nm diameter) is more effective in retarding recrystallization than dissolved Nb in the matrix. An atomic ratio of Nb/C >1 or Nb/(C N) >1 is found to be preferred due to the combination of precipitation strengthening and solid solution strengthening in refining prior austenite grain size[110]. This is also consistent with Kikuchi et al. [111], Adamson [112] and Keown and Pickering's study [113].
The combination addition of Nb, Ti with B is found to be more effective in retarding recrystallisation than single addition of B, Ti or Nb [114]. Tamehiro et al.[115] found that the addition of Nb to B steel not only slow down the recrystallisation but also suppress the M (B,C)
(B,C) precipitation, which gives a fine structure.
precipitation, which gives a fine structure.
Elements that are not deliberate additions, like Al, V and Ti, favour forming nitrides in austenite which may be effective in inhibiting grain growth but can be detrimental to the overall mechanical properties [116].
Concluding remarks
The application of the material is based on the application for aerospace shaft, will only tolerance for up to 0.5% creep strain. Its particular required strength level determines the particular level of precipitate strengthening needed. A uniform distribution of intermetallic compounds and stable carbides would contribute to improved creep properties. However, coarse M C
C and Laves phase present at prior austenite grain boundaries, or even junctions, would result in creep voids to link together, coalescing and be detrimental to creep life. Therefore, the designed alloy need to have a compromise on the size and number of intermetallic compounds. The heat treatment must happen at a temperature above the service temperature in order to ensure some level of thermal stability.
and Laves phase present at prior austenite grain boundaries, or even junctions, would result in creep voids to link together, coalescing and be detrimental to creep life. Therefore, the designed alloy need to have a compromise on the size and number of intermetallic compounds. The heat treatment must happen at a temperature above the service temperature in order to ensure some level of thermal stability.
The Super-CMV is well established, so it is not discussed further. But research on steels based on intermetallic compounds for shaft application as lead to a novel ultra-high strength maraging steel developed in Cambridge [82]. Owing to its unique combination of nanoscale Laves phase and β-NiAl, it has a balanced ductility and creep resistance. It has a yield strength above 1800 MPa, the ultimate tensile strength of approximately 2000 MPa, tensile ductility up to approximately 8% at room temperature. This alloy ticks all the right boxes with respect to the properties required exclude creep rupture life which is below 3000 h under 700 MPa at 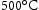 . Based on the literature, grain size refinement would be promising to enhance its creep properties.
. Based on the literature, grain size refinement would be promising to enhance its creep properties.
Futuer aspects that need to be considered and researched, particularly in the context of intermetallic compounds strengthened alloys are listed below:
Thermal stability of any material developed is an extreme requirement for aeroengine shaft applications. So not enough is understood for controlling factors for thermal stability over service conditions of the shaft. These service conditions are much more onerous than those in the power generation industries because of far greater stresses and longer service lifetime, therefore more research needs to be done. Moreover, changes in toughness over the duration of the service life needs to be explored. Recording systems for any new alloys are necessary for development. Component level testing is required for designed alloy for the aeroengine applications.
Footnotes
Disclosure statement
No potential conflict of interest was reported by the author.
References
 C
C in austenitic steels
in austenitic steels c
c plates around undissolved nbc particles in a stabilized austenitic stainless steel
plates around undissolved nbc particles in a stabilized austenitic stainless steel
 )
)