Abstract
Repeated administration of theophylline, a phosphodiesterase inhibitor, induces the enlargement of the salivary glands in rats. Time-course changes after a single administration of theophylline were examined in the salivary glands, including phosphodiesterase enzyme activity, and the expression of aquaporin 5 (AQP5), a water channel. We also examined the contribution of β-adrenergic receptors to theophylline-induced salivary changes. Male F344 rats were given 50 mg/kg of theophylline intraperitoneally either alone or concurrently with a 10 mg/kg subcutaneous injection of propranolol. After treatment with theophylline alone, the weight and histology of the submaxillary and parotid glands were examined. Phosphodiesterase activity and AQP5 were detected by enzyme- and immuno-histochemistry, respectively. At 4 hours, 8 hours, or both, organ weights were decreased with depletion of secretory vesicles in the acinar cells. In the submaxillary glands, reduced activity of phosphodiesterase and increased expression of AQP5 in the intercalated ducts were observed at 4 hours. When co-administered, propranolol partially abolished theophylline-induced glandular reduction. These results suggest that the theophylline-induced transient reduction in size of the salivary glands is attributable not only to phosphodiesterase inhibition but also to β-adrenergic receptor activation and that the intercalated ducts in submaxillary glands play a role in the production of saliva.
Keywords
Introduction
Although the reduction and enlargement of the salivary glands are common in toxicity studies, there are few studies on salivary toxicity. When toxic doses of phosphodiesterase inhibitors, including theophylline (1,3-dimethylxanthine), are given repeatedly to rats, salivation as well as enlargement of the salivary glands are often observed (Wells, 1967; Wells and Humphreys-Beher, 1985; Westwood et al., 1991; Larson et al., 1996; Kajikawa et al., 2003). Since phosphodiesterase inhibitors have been shown to induce saliva secretion as well as to increase cyclic AMP in the acinar cells in in vitro studies (Afari, Tenenhouse, and Vachan, 1982; Fuller and Gallacher, 1984; Takuma, 1990), the cause of salivary toxicity was thought to be related to their pharmacological properties. The measurement of intracellular cyclic AMP, however, Were unsuccessful in the in vivo studies, and it required the detection of phosphodiesterase activity in the tissues.
The β-agonists are known to exhibit tropism towards the salivary glands and induce salivary gland enlargement when administered repeatedly. β-adrenergic receptors elevate intracellular cyclic AMP via activation of adenylate cyclase. Although the elevation of cyclic AMP by phosphodiesterase inhibitors or β-agonists is mediated by independent mechanisms, co-administration of theophylline and isoproterenol, a β-agonist, failed to yield a synergistic effect (Wells, 1967). This suggests that theophylline may have some effects on β-adrenergic receptors in salivary glands.
Membrane water channel aquaporins are known to regulate the movement of water across the plasma membrane of secretory and absorptive cells. A significant amount of the aquaporin 5 (AQP5) subtype has been found to be present in the salivary gland (Funaki et al., 1998). AQP5-deficient mice have shown depressed rates of salivary secretion and reduced amounts of saliva (Ma et al., 1999; Krane et al., 2001). A number of secretagogues have been noted to increase the amount of AQP5 in the plasma membranes of salivary acinar cells (Ishikawa et al., 1999; Matsuzaki et al., 1999).
The purpose of this study is to elucidate the time-course changes after a single administration of theophylline in the weight and histopathology of salivary glands including phosphodiesterase activity using enzyme histochemistry and AQP5 expression using immunohistochemistry. The contribution of β-adrenergic receptors to theophylline-induced changes in the salivary glands will also be examined by coadministration with propranolol, a β-antagonist.
Materials and Methods
Animals
Male F344 rats were supplied by Charles River Japan (Kanagawa, Japan) and acclimated until used. They were housed in stainless steel cages in an animal room under controlled conditions (23 ± 3°C with a relative humidity of 55 ± 5%, ventilation 10 times/hour and a 13-hour light/11-hour dark cycle). At the commencement of dosing, the animals were 9-11-weeks old. They were fed a commercially available diet (CRF-1, Oriental Yeast, Tokyo, Japan) and tap water ad libitum. The experiments were conducted in compliance with the institutional guidelines for the care and use of laboratory animals.
Experiment 1
The first experiment was designed to focus on the time-course theophylline-induced changes in the salivary glands. Twenty rats each were given either saline or theophylline (Wako Pure Chemical, Osaka, Japan) at 50 mg/kg (theophylline dissolved in physiological saline at 5 mg/ml). A total of 10 ml/kg was given intraperitoneally. Five rats each were sacrificed under ether anesthesia at 1, 4, 8, and 24 hours after administration. Immediately after exsanguination via the caudal vena cava, the major salivary glands (the parotid gland and submaxillary gland with the sublingual gland) were taken. All rats were weighed on the day before administration and at the time of necropsy. Additionally, 6 rats each were given either saline or theophylline and sacrificed, 3 as control subjects and 3 as the treated group, at 1 and 4 hours each in the same manner. The salivary glands taken were used for enzyme histochemistry.
Organ Weight and Histopathology: The salivary glands were fixed overnight in 10% neutral-buffered formalin. After removal of fat and connective tissues, they were weighed, processed routinely, embedded in paraffin, and stained with hematoxylin and eosin (H&E).
Enzyme Histochemistry: The principle of the phosphodiesterase cytochemical procedure is based on the hydrolysis of the cyclic adenosine triphosphate into 5'-adenosine monophosphate. The procedure was carried out as described elsewhere (Ueno et al., 1984), with minor modifications. Briefly, the submaxillary glands and parotid glands were embedded in OCT compound and frozen at -80°C. Then, 6-μm sections were cut in a cryostat and fixed in 1% glutaraldehyde and 2% paraformaldehyde in 0.1 M sodium cacodylate buffer, pH 7.4, for 15 minutes at 4 C. The sections were first incubated for 30 minutes at room temperature in a solution containing 60 mM tris maleate buffer, pH 7.4,
2 mM MgCl2, and 250 mM sucrose (TMS buffer) containing 5 mg/ml 5'-nucleotidase (lyophilized Crotalus atrox snake venom, Sigma, St. Louis, MO) for degradation of intrinsic cyclic AMP. The sections were then incubated for 60 minutes at 37°C in TMS buffer containing 2 mM lead nitrate, 3 mg/ml 5'-nucleotidase, and 3 mM cyclic AMP as substrates, in either the absence or presence of 50 mM theophylline. After incubation the sections were washed briefly in TMS buffer and rinsed in distilled water. They were treated with 1% ammonium sulfite for 2 minutes.
Immunohistochemistry: Salivary gland sections were stained for AQP5 by the routine labeled streptavidin biotin technique. Briefly, 4-μm paraffin sections were prepared. AQP5 antigen was retrieved using a microwave oven in 0.01 M citrate buffer, pH 6.0, for 20 minutes at 95°C. Nonspecific binding sites were blocked with 1% bovine serum albumin. Antibody to AQP5 (Chemicon International, Temecula, CA) was applied at a concentration of 4 jug/ml, for overnight at 4°C, followed by biotin-labeled anti-rabbit IgG (Vector, Burlingame, CA), diluted 1:100, for 30 minutes. For visualization, streptavidin-conjugated Alexa Fluor 594 (Molecular Probes, Eugene, OR) was applied at a dilution of 1:1,000 for 30 minutes, followed by 0.2 μg/ml of YO-PRO-1 iodide (Molecular Probes) for nuclear staining for several minutes. The localization of AQP5 was examined with a confocal microscope (TCS SP2, Leica Microsystems, Wetzlar, Germany). All sections were examined under the same operation procedure; single images were projected from 8 consecutive optical-sections every 0.49 μm (3.42 μm in total thickness).
Experiment 2
In the second experiment, designed to investigate potential involvement of β-adrenergic stimulation in theophylline-induced salivary changes, DL-propranolol hydrochloride (Sigma) was used to block β-adienergic receptors. Fifteen rats were assigned randomly to 3 groups of 5 rats each. Animals in groups 1 and 3 were subsequently injected with 10 mg/kg propranolol 30 minutes before intraperitoneal administration of saline and 50 mg/kg theophylline, respectively. The dosage of propranolol was thought to be sufficient to produce the β-antagonistic effect based on the results from a previous study (Laemont et al., 1999). Animals in group 2 received saline subcutaneously 30 minutes before intraperitoneal administration of 50 mg/kg theophylline. All rats were sacrificed at 4 hours after intraperitoneal administration of theophylline or saline, and organ weights and histopathology of the submaxillary and parotid glands were examined with the same procedures as those in Experiment 1.
Statistics: Differences in submaxillary and parotid glands to body weight ratios between the control and treated groups were tested using the Student's t-test in the first experiment and Dunnett's test in the second one, after 1-way analysis of the variance using SAS (version 8.2). A p-value of <0.01 indicated a statistical significance of differences.
Results
Experiment 1
Organ Weight: In control rats, the weight of both the submaxillary and parotid glands increased slightly at hours 1 through 8 (during light phase) and decreased at hours 8 through 24 (during dark phase). This variation was attributed to the circadian rhythm. In theophylline-treated rats, submaxillary gland weight decreased transiently as the post-dose hours passed, and then increased. Compared to the control rats at each time point, the submaxillary gland to body weight ratio decreased significantly by 12% and 16% at 4 and 8 spectively and then increased significantly by 19% at 24 hours (Table 1A). Similar trends were noted in the parotid gland. The parotid gland to body weight ratio decreased (p = 0.04) by 13% at 4 hours and then increased significantly by 18% at 24 hours, compared to the control group at each time point (Table 1B).
Theophylline induces the transient reduction of the submaxillary and parotid glands to body weight ratio in male F344 rats.

For each group, values are means ± standard deviations.
p-value <0.01 (Student's t-test), compared to the control, for each time point.
Histopathology: There was an association between histology and organ weight changes. In control rats, abundant secretory vesicles and eosinophilic zymogen granules were present in the acinar cytoplasm of the submaxillary and parotid glands, respectively (Figures 1A, B). By 1 hour after administration, a slight decrease in cellular size and the amount of secretory materials was observed in both salivary glands. Four hours after administration, marked depletion of the secretory materials in the acinar cells as well as large vesicular nuclei were observed in both glands (Figures 1C, D). Salivation or frequent preening, which was exhibited by the rats immediately after administration, disappeared in most rats by this time. At 8 hours, the size of acinus was still smaller than that of the time-matched control. The secretory materials in both glands were slightly overaccumulated at 24 hours.
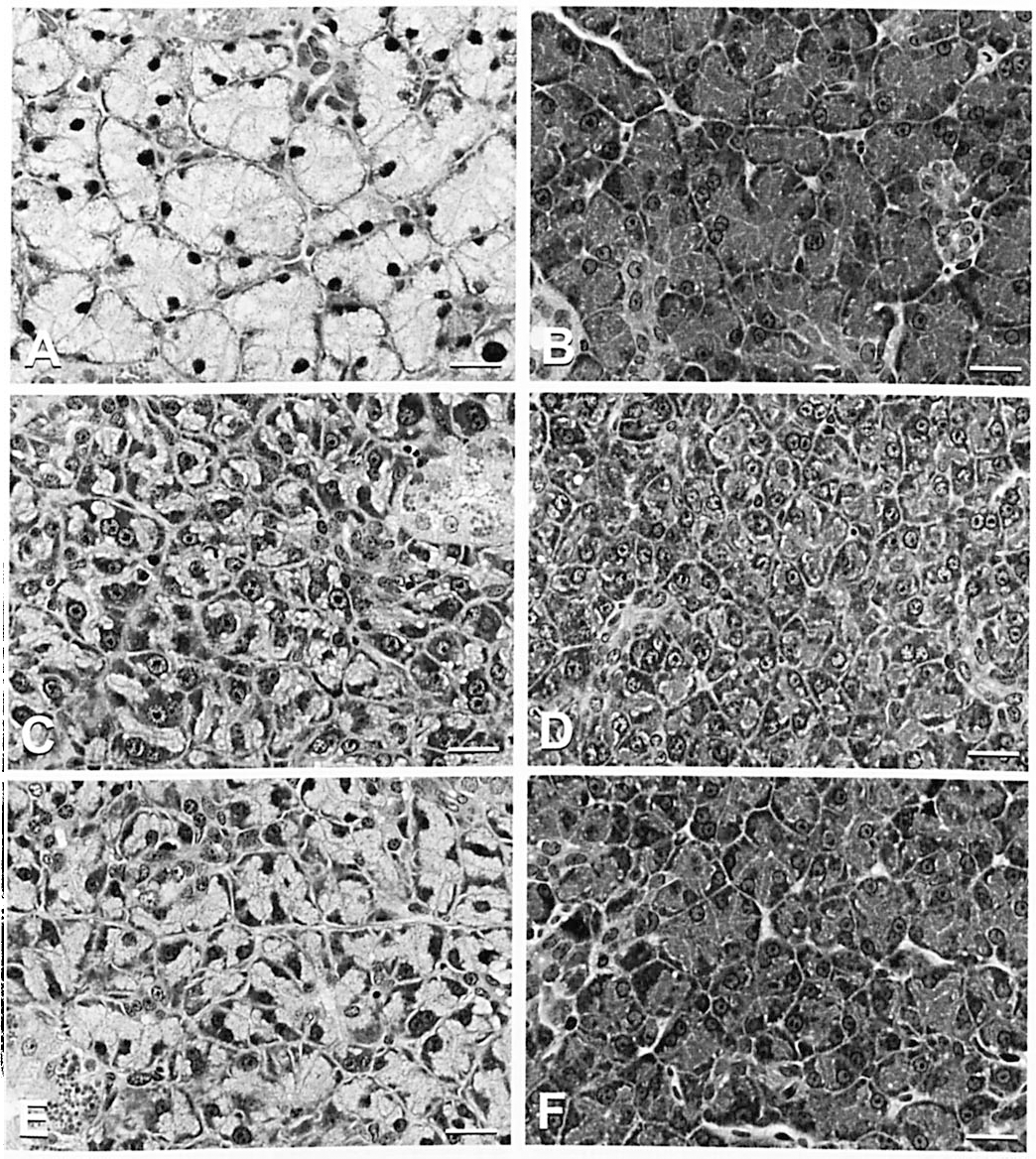
Light microscopy of theophylline-induced reduction in the size of acinus and the antagonistic effect of a β-blocker. H&E stain. Bar = 20 μm. (A) Submaxillary gland, 4 hours after saline administration: abundant secretory vesicles are present in the cytoplasm. (B) Parotid gland, 4 hours after saline administration: abundant zymogen granules are present in the cytoplasm. (C) Submaxillary gland, 4 hours after theophylline administration: the size of the acinar cells is smaller than the control due to the depletion of the secretory vesicles, showing large vesicular nuclei with prominent nucleoli and a basophilic cytoplasm. (D) Parotid gland, 4 hours after theophylline administration: cellular size of acinar epithelium is decreased by the depletion of zymogen granules. (E) Submaxillary gland, 4 hours after theophylline administration following pretreatment with propranolol: a moderate amount of secretory vesicles is stored in the cytoplasm, and the nuclear changes shown in Figure 1C are less evident. (F) Parotid gland, 4 hours after theophylline administration following pretreatment with propranolol: a moderate amount of zymogen granules is stored in the cytoplasm.
Enzyme Histochemistry: The reaction product demonstrating phosphodiesterase activity in control submaxillary glands was located in the acini and intercalated ducts (Figure 2A). The intense reaction was present on the basal side of the intercalated ducts. In addition, the reaction product was found on the arterial walls and the outer layer of the excretory ducts. No reaction product was observed in the granular or striated ducts. In treated submaxillary glands, although no alteration was apparent at 1 hour after administration, the intensity of the precipitate at 4 hours after administration was weaker than that of control submaxillary glands (Figure 2C).
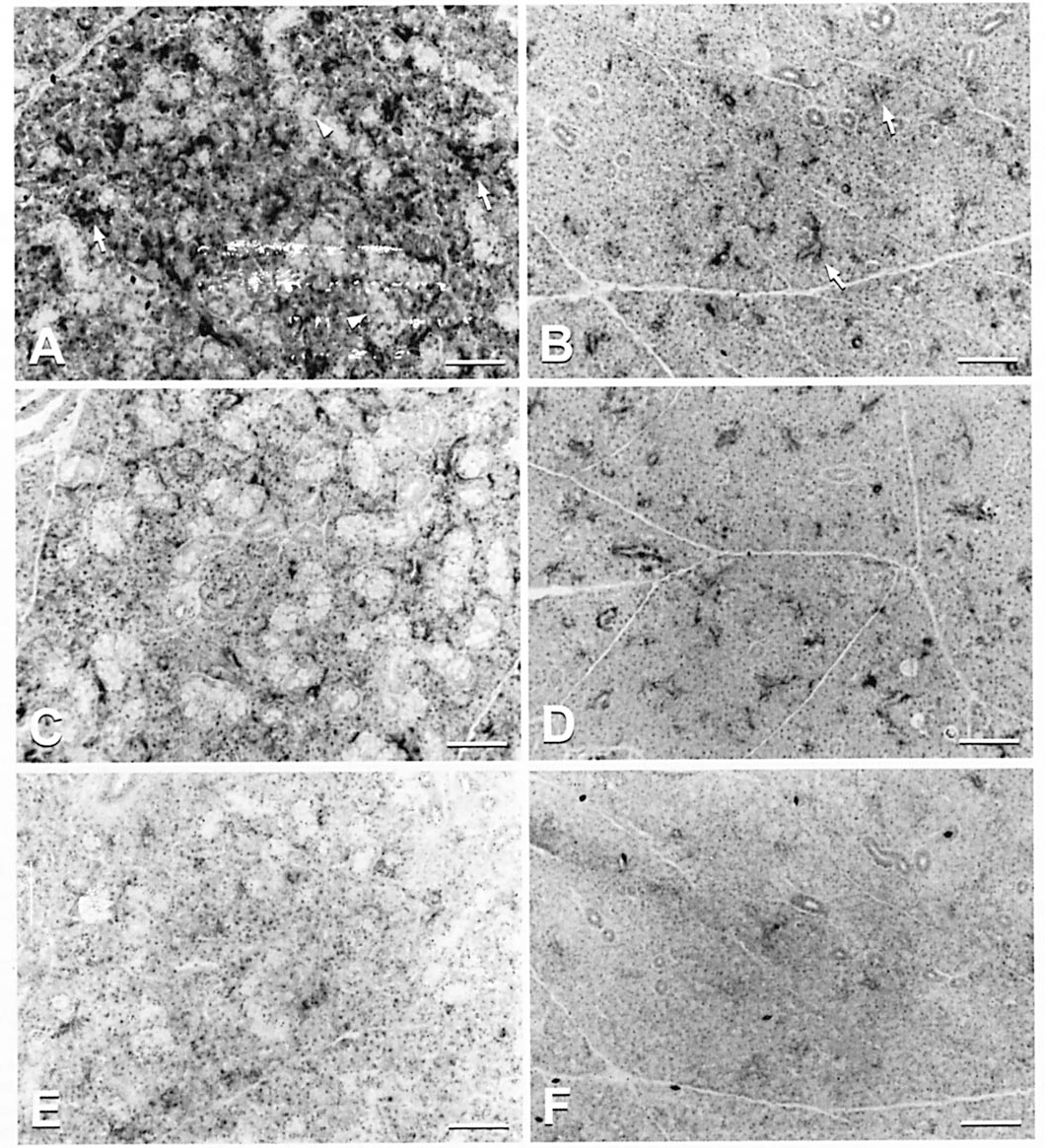
Enzyme histochemistry for cyclic AMP phosphodiesterase activity. Bar = 100 μm. (A) Submaxillary gland, 4 hours after saline administration: the reaction products were in the acini and the intercalated ducts (arrows), whereas granular and striated ducts were devoid of the products (arrowheads). (B) Parotid gland, 4 hours after saline administration: the reaction products were in the intercalated ducts (arrows), whereas no products were seen in the acini and excretory ducts. (C) Submaxillary gland, 4 hours after theophylline administration: the intensity of the precipitate representative of phosphodiesterase activity is weaker than that of the saline control. (D) Parotid gland, 4 hours after theophylline administration: the intensity of the precipitate is similar to that of the saline control. (E) Submaxillary gland, the section adjacent to that of Figure 2A incubated in a medium containing theophylline: the reaction is nearly abolished. (F) Parotid gland, the section adjacent to that of Figure 2B incubated in a medium containing theophylline: the reaction is completely abolished.
The reaction product in the control parotid glands was located exclusively in the intercalated ducts. The intense reaction tended to be present on the basal side of the cells. No reaction product was observed in other components including the acini and the excretory ducts (Figure 2B). The intensity of the precipitate in the treated parotid glands was unchanged at both 1 and 4 hours after administration (Figure 2D).
To confirm the enzyme reaction, 50 mM theophylline was added exogenously to the incubation mixture. The result showed little or no reaction product, indicating inhibition of cytochemically demonstrable phosphodiesterase activity (Figures 2E, F).
Immunohistochemistry: Immunohistochemical reaction to AQP5 protein in the submaxillary glands was detected in the apical plasma membrane and the secretory canaliculi of the acinus epithelial cells and in the luminal membrane of the intercalated ducts. The labeling intensity was stronger in the intercalated ducts than in the acinar cells. The staining characteristics in the acinus units were almost identical among time points. In the intercalated ducts, the increased staining density was noted by laser confocal microscopy at 4 hours after administration (Figure 3).
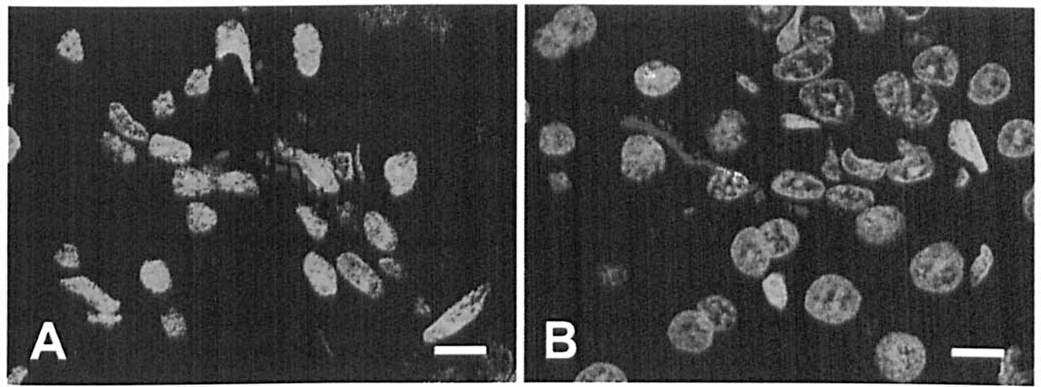
Fluorescence immunohistochemistry for AQP5 (red) in the intercalated ducts of the submaxillary gland. Nuceli were counterstained with YO-PRO-1 (green). Bar = 8 μm. (A) 4 hours after saline administration: dispersed or dotty distribution of the protein is seen on the luminal membrane. (B) 4 hours after theophylline administration: increased labeling intensity of AQP5 is evident along the luminal membrane of the intercalated duct.
In contrast, immunoreactive AQP5 in the parotid glands was found in the apical plasma membrane of the acinus epithelial cells, but not in the ductal segment, including the intercalated ducts. The AQP5-staining characteristics in the parotid acini of the control and theophylline-treated rats did not differ from each other.
Experiment 2
The effects of propranolol on the theophylline-induced reduction in the size of the salivary glands are shown in Table 2. Compared to the control in the first experiment, it was confirmed that propranolol administration did not affect the organ weight or histopathology in either the submaxillary or parotid glands. The submaxillary and parotid glands to body weight ratios decreased by 17% and 13% in the theophylline-treated rats, respectively. These results reproduced the first experiment. When propranolol was given concomitantly with theophylline, the weight ratio in both glands decreased by 5% as compared to the group given propranolol alone. The depletion of secretory vesicles or zymogen granules was attenuated in both glands, in accordance with the results of organ weight (Figures 1E, F).
β-adrenergic receptors contribute to the reduced weight of salivary glands induced by theophylline in male F344 rats.

Subcutaneous injection was given 30 min before intraperitoneal administration. Salivary glands were taken 4 hours after the intraperitoneal administration.
For each group, values are means ± standard deviations.
p-value < 0.01 (Dunnett's test), compared to the propranolol (sc) + saline (ip) group.
Discussion
Our studies demonstrated that secretory vesicles in the salivary glands decreased during salivation after administration of theophylline in coincidence with organ weight changes and that phosphodiesterase in situ also was decreased in its activity. The reduced size of the acinus was partially abolished by blocking β-adrenergic receptors. In the submaxillary gland, the expression of AQP5 on the luminal membrane of the intercalated duct increased during the salivation phase.
Reduction of secretory granules during the secretory cycle has been reported. Amsterdam et al., (1969) observed depletion of zymogen granules in the parotid gland from 1 to 3 hours after isoprenaline, a catecholamine, was given intraperitoneally to rats. Our study showed similar secretory cycles with theophylline. These results indicate that salivary acini change their morphology according to the secretory cycle. In toxicity studies, it is not rare to observe both hypertrophy and atrophy in salivary tissues within the same dosage group. These contradictory findings may simply reflect a different phase of the secretory cycle at necropsy.
In previous studies of salivary gland enlargement induced by multiple doses of phosphodiesterase inhibitors, organ weight was examined on the next day of the terminal administration (Wells, 1967; Wells and Humphreys-Beher, 1985; Westwood et al., 1991; Larson et al., 1996). Interestingly, we observed a slight increase in organ weight at 24 hours after a single administration. This may suggest that repeated secretory stimulation lead to salivary enlargement and this enlargement induced by the phosphodiesterase inhibitor is of compensatory nature. In support of this, diffuse acinar cell hypertrophy recorded in a 6-month study of a repeatedly dosed phosphodiesterase inhibitor completely disappeared following a 1-month withdrawal period (Westwood et al., 1991).
The results of our enzyme histochemical analysis indicated that the phosphodiesterase activity in the submaxillary gland was decreased by treatment with theophylline. This technique was developed primarily to determine the subcellular distribution of cyclic AMP-dependent phosphodiesterase (Shanta et al., 1966; Florendo et al., 1971; Okruhlicova et al., 1996). Since the precipitated products originate from phosphates hydrolyzed by phosphodiesterase in the tissue section, the amount of precipitation is related to the phosphodiesterase activity (Ueno et al., 1984). We did not observe any apparent reduction in activity in the parotid gland. The reason was thought to be that although phosphodiesterases have previously been demonstrated (Imai et al., 1995), the sensitivity of the enzyme histochemical technique was not sufficient to detect the difference where the activity was undetectable in the first place. Unexpectedly, the phosphodiesterase activity in the intercalated duct was stronger than that in the acinar unit. The biological significance of this finding remains unclear.
Our study of the pretreatment with propranolol demonstrated that the β-blockade abolished theophylline's influence on the reduction in the size of the salivary gland to some extent. This result suggests that β-adrenergic receptors in the salivary tissue are stimulated by a toxic dose of theophylline. A similar result was obtained in a study using parotid acinar cells, in which amylase release and cyclic AMP accumulation stimulated by 3-isobutyl-l-methylxanthine, a phosphodiesterase inhibitor, were decreased dose-dependently by propranolol (Takuma, 1990). Our results, however, do not rule out the possibility that the theophylline produces autonomic stimulations. In the salivary gland, receptors for a variety of neurohumoral agonists including acetylcholine, norepinephrine, and vasoactive intestinal peptide have been found to be present as well as second messengers including cyclic AMP, cyclic GMP, and calcium. This implies that the regulation of protein and fluid secretion involves complex cellular mechanisms (Cook et al., 1994). More extensive studies are needed in order to fully understand theophylline-induced salivary toxicities.
Immunohistochemistry for AQP5, a water channel protein, showed that the expression of AQP5 in the intercalated ducts of the submaxillary glands increased after treatment with theophylline. Cyclic AMP regulates the trafficking of AQP5, where the proteins are rapidly translocated to the plasma membrane from cellular reservoirs in response to stimuli (Yang et al., 2003). Theophylline increases the saliva flow rate from the submaxillary glands (Abdollahi and Simaiee, 2003). Accordingly, it is suggested that the increased level of cyclic AMP elicited by theophylline leads to the membranous relocation of AQP5 in the intercalated ducts and thereby to salivation. To date, the process of the secretion of fluid by the salivary glands is thought to consist of 2 stages in which the secretory endopieces, or acini, generate an isotonic, plasmalike fluid that is modified as it flows through the ductal system (Cook et al., 1994). As previous studies reported, we observed the abundant expression of AQP5 in the submaxillary intercalated duct cells (Nielsen et al., 1997). Not only acinus units but also the intercalated ducts, therefore, participate in saliva secretion.
In conclusion, theophylline induces a reduction in the size of salivary glands. This change is caused by phosphodiesterase inhibition as well as by steps involved in β-adrenergic receptor activation. The fact that theophylline alters the expression of AQP5 in the intercalated ducts suggests that the intercalated ducts play a significant role in the production of saliva fluid in the submaxillary gland.
Footnotes
Acknowledgements
We acknowledge Kyoko Ito for helpful discussion and advice. We also thank Satoru Sasaki and Yoko Kaneko for the administration of test articles, Shuji Ishikawa and Eiichi Nukui for the preparation of numerous specimens, and Kenji Nakano for histochemistry and confocal scanning laser microscopy.
