Abstract
As part of regulatory safety testing program, a 13-week oral toxicity study with a new antipsychotic drug candidate was performed in beagle dogs. During this study, dark red/brown feces were recorded in treated dogs and increases in liver parameters (alanine aminotransferase, alkaline phosphatase, bilirubin) were measured biochemically. At the end of the study, livers of high-dose (50 mg/kg) animals were (mottled) dark brown, sometimes with pale foci. Histopathological examination of these livers showed dark globular pigment deposits in the hepatocellular cytoplasm and within the bile canaliculi. Varying numbers of inflammatory cell infiltrates were additionally present in association with the deposits. These pigment deposits showed birefringency with characteristic “Maltese Cross”-like structures under polarized light. Electronmicroscopy revealed the typical, so-called “sunburst” pattern with radiating double-lined crystalline structures. These morphologic characteristics strongly indicated at the presence of porphyrins, which was definitely confirmed biochemically. Published reports of drug-induced hepatic porphyria in dogs are rare. The possible underlying mechanism in the dog and man is discussed.
Introduction
Porphyria is a disorder caused by a disturbance of the heme biosynthetic pathway (Frank and Christiano, 1998). The synthesis of heme occurs in mammalian erythropoietic cells and in the hepatocytes of the liver. It is a multistep process of enzymatic conversion of succinyl CoA via series of heme-precursors (porphyrins) into heme. When the heme synthesis is hampered, the precursor porphyrins accumulate and additional injury can develop as a consequence of the toxic properties of the accumulating pigment (Bloomer, 1998; McCullough et al., 1988). The different precursor porphyrins are excreted via different routes depending on their solubility. One of them, protoporphyrin, is water-insoluble and therefore excreted solely in the bile and feces (Schwartz et al., 1960). In contrast, the precursors uro- and coproporphyrin are better water soluble and are excreted via bile and feces as well as kidney and urine.
In man, porphyria has been reported as a congenital and inherited disease but can also be induced and/or aggravated by a number of drugs and environmental chemicals (Bickers, 1982; Smith et al., 1997; Gross et al., 2000; Sassa and Kappas, 2000). Domestic animals mainly develop congenital and hereditary porphyria (With, 1980) whereas in laboratory animals (rats and mice) drug-induced porphyria have been described in literature (Matilla and Molland, 1974; Gralla et al., 1977; Cantoni et al., 1983; Frater et al., 1993; Smith, 1997). For dogs, information about experimentally induced porphyria is scarce and not of recent date (Stokvis, 1895; Zaki et al., 1973; Stejskal et al., 1975).
In a 4-week toxicity study performed in our laboratory, dogs were treated with a new drug candidate under development for the treatment of psychosis. In this regular study the increased values of several biochemical liver parameters (alanine aminotransferase or ALT, alkaline phosphatase or ALP and bilirubin) as well as the histopathology of the liver changes draw our attention. Especially the microscopic changes in the liver suggested a possible disturbance in the heme pathway. To investigate this hypothesis, a subchronic (13-week) dog study was performed and special attention was paid to parameters specifically indicative of porphyria. For this purpose many hematologic and biochemical parameters were determined in blood, urine and feces and an extensive histopathologic examination was performed. A parallel 4-week rat study with the same drug did not show any abnormalities.
Material and Methods
Study design
This study design has been approved by the Animal Experimentation Committee and complies with the Dutch Act on Animal Experimentation (Stb.1977, 67). Beagle dogs, Hsd/Hfr: DoBe, pure bred were supplied by Harlan S.A.R.L., Gannat, France. All dogs had been vaccinated against rabies, canine distemper, canine hepatitis, leptospirosis and canine viral enteritis and had received one or more courses of antiparasitics. At arrival the dogs were 5 to 6 months old and the dosing period of the study started when they were 6 to 7 months old.
The dogs (4 males and 4 females per group) were dosed orally for 13 weeks with 0, 5, 15 or 50 mg/kg of the new antipsychotic drug. The dose levels were based on the earlier 4-week toxicity study.
Housing conditions
Dogs were housed in pairs in centrally heated kennels with an outdoor run (temperature: 19–29°C). A fixed proportion (200 g/dog/day) of Canex Brocks standard expanded food (supplied by Hope Farms BV, Woerden, the Netherlands) was given from the predosing period onwards and free access to tap water from an automatic water supply was available.
Clinical examinations
Routine examinations included clinical signs, electrocardiography, physical examination, ophthalmoscopy, body weight, and food consumption.
Laboratory investigations
Blood was collected with K2-EDTA, citrate or heparin as anticoagulant depending on the subsequent analysis. Hematology (with Cell-Dyn 3500 hematology analyzer), blood clotting (APTT: Activated Partial Thromboplastin Time (s) with a Sysmex CA-5000 coagulation analyzer, optical method) and blood biochemistry (with Hitachi-911, Boehringer-Mannheim) was performed following standard procedures. Urinalysis: conjugated bilirubin (μmol/l) was determined with a DPD method (37°C, Wahlefeld et al., 1972), glucose (mmol/l) with a Hexokinase method (37°C), hemoglobin with a SANGUR test (Boehringer Mannheim), osmolality (mOsm)/kg by freezing point depression method, protein (g/l) with the benzethonium chloride method (37°C), volume (ml) in a graduated flask and the pH (−logH+) by pH measurement (pH-meter, Knick 764 Hilkomy, Rijswijk, The Netherlands). Protoporphyrin in feces was measured by HPLC-UV analysis after liquid-liquid extraction as is described by Christensen and Romslo (1979) and Gray et al. (1977) with some modifications. In short, calibration was done by spiking blank dog feces with different protoporphyrin concentrations (7.5, 10, 20, 30, 40, 50, and 62.5 μg/gram wet feces). Three quality samples were prepared similarly (concentrations 20.0, 35.0, and 50.0 μg/gram wet feces). Samples were extracted by liquid-liquid extraction with ethylacetate and purified by solid phase extraction with C18 cartridges. After evaporation residues were redissolved in 0.2 ml acetonitrile/milli Q water 1:1 (v/v) and an aliquot of 5 μl was measured. HPLC-UV analysis was performed using a column Symmetry C18 3.9 × 150 mm 5 μM (Waters) with a flow of 1.0 mL/min (Temp 40°C) and step gradients of 30-10-30% (solvent A: 0.1% TFA-solution) and 70-90-70% (solvent B: Acetonitrile) at 0, 4.01 and 8.01 minutes. Detection was performed with a Diode Array Detector at 400 nm. In the graph the mean values are given with the lowest and highest values. The Student’s t-test has been used to evaluate significancy.
Necropsy/histopathology
Dogs were sacrificed at the end of the dosing period by exsanguination from the brachial plexus under Nesdonal and Rapifen anesthesia. Major organs were weighed and a full gross examination was performed. Tissues were fixed in 4% neutral-buffered formalin and processed using routine methods for histopathologic examination (paraffin embedding, sectioning, and Hematoxylin & Eosin staining). Additionally, liver and kidney samples were kept frozen and extra liver tissue was fixed in glutaraldehyde and processed for electron microscopy. This processing comprised postfixation of the tissue in 1% osmium tetroxide, embedding in Epon 812 via propylene oxide and the examination of ultrathin 80 nm sections with a JEOL 1010 electron microscope.
Compound
The compound used was a heterocyclic drug (4-[(4-fluorophenyl)-4-chlorothienyl-2-methylene]-methylpiperid-inebutane-1,4-dioate). It antagonized several subtypes of dopamine, serotonin, and muscarine receptors and had expected antipsychotic activities. Structural formula:
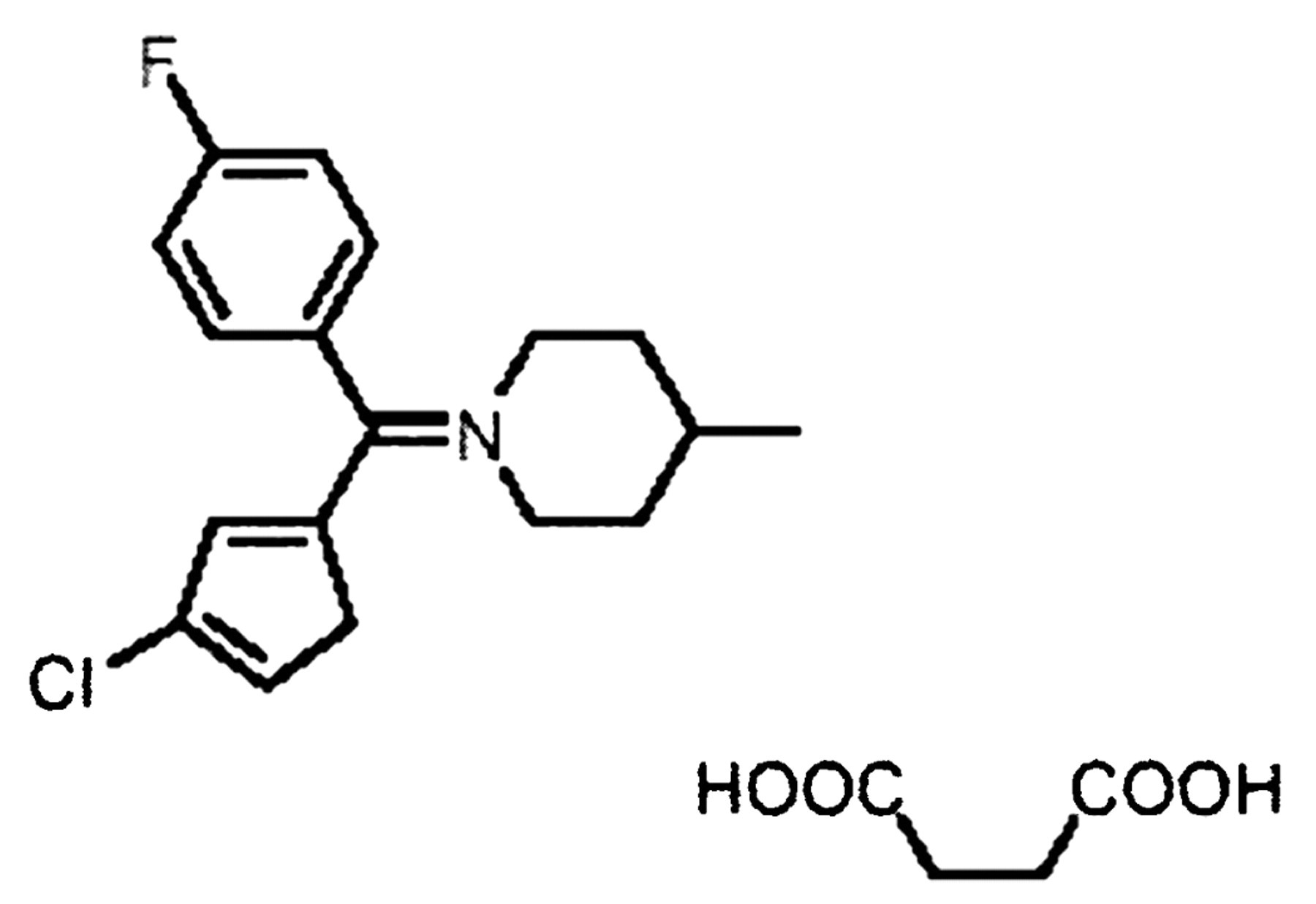
Results
Clinical signs
Mild behavioral changes were observed in all dogs treated with 50 mg/kg and mild body tremors were recorded in few dogs. A dose-related increase in heart rate was observed in the treated dogs. Feces of dogs treated with 50 mg/kg of the compound were reddish/brown or sometimes dark brown.
Laboratory results
In the feces of the males treated with 15 mg/kg, total protoporphyrin was slightly increased (from undetectable in all control animals to 12.6 μmol/L in males, and 8.9 μmol/L in 1 female) after 23/24 days whereas the males and females treated with 50 mg/kg showed markedly increased total protoporphyrin (males 94.3 μmol/L, females 62.7 μmol/L). This remained steady after 84/85 days. In addition, in dogs treated with 50 mg/kg mean values of the following parameters were increased after 86 days: plasma alanine aminotransferase (in males from 37 to 188 U/L, in females from 60 to 144 U/L), alkaline phosphatase (in males from 61 to 319 U/L, in females from 76 to 405 U/L), and bilirubin (in males from 1.6 to 3.1 μmol/L, in females 1.6 to 4.3 μmol/L) (Table 1). Hematology showed a mild decrease in activated partial prothromplastin time in most of the dogs treated with 50 mg/kg at the end of the dosing period.
Pathology
Grossly, the livers of all dogs treated with 50 mg/kg were dark brown, sometimes mottled and/or with pale foci. The livers of the dogs of the other groups did not show abnormalities. The thymus showed atrophy with corresponding decreased weights in almost half of the animals treated with 50 mg/kg. Changes in other organs were unremarkable.
Upon histopathology, in routine H&E stained liver slides, dark brown pigment deposits were present in all dogs treated with 50 mg/kg (Figure 1A). The deposits were localized intracellularly in hepatocytes, in macrophages and between hepatocytes within the bile canaliculi, often in a string-wise manner (Figure 1C). Under polarized light, this pigment exhibited birefringence characteristics with the typical, so-called “Maltese cross”-like appearance (Figure 1B). The severity of the pigment accumulation varied among the different animals. In addition to the pigment deposits some affected dogs showed mild to severe inflammatory changes (Figure 1D) and 1 dog bile plugs (as was demonstrated by a Fouchet’s staining). In some cases these changes were accompanied by hepatocellular necrosis and cholangiolar “oval” cell proliferation (Figure 1E). In one dog a hyperplastic (regenerative) nodule was present (Figure 2A). Electron-microscopic evaluation revealed that the pigment deposits were closely packed with short curved or straight bars and rods in a so-called “sunburst” pattern (Figure 2B).
Discussion
In the present study, beagle dogs treated subchronically with a new antipsychotic drug candidate showed a variety of clinical abnormalities, gross- and histopathological liver changes, a mild decrease in one of the hematology clotting parameters and a considerable increase in the values of some biochemical liver parameters. The reddish/brown feces observed in treated dogs during the study indicated a disturbance in normal excretion and/or liver function. This was confirmed by obvious changes in the liver of these animals during autopsy (mottled livers, sometimes with pale foci). Histopathologic examination revealed hepatocellular necrosis, aggregates of inflammatory cells and cholangiolar cell proliferation, the presence of bile plugs and a hyperplastic regenerative nodule in 1 dog. The most prominent feature in these affected livers was the presence of dark-brown pigment deposits. The pigment deposits were of variable size and were found at different intra- and extracellular locations (in the cytoplasm of hepatocytes and macrophages and within the bile caniculi).
The characteristic combination of the liver changes and clinical observations in treated dogs fitted well with those described for porphyria (Ghadially, 1988; Komatsu et al., 2000). Pigment deposits are however quite common in the liver. Well-known pigments are for instance hemosiderin, bilirubin, and lipofuscin. Next to differences in staining properties, important additional characteristics to discriminate porphyrins from other types of liver pigment deposits are the microscopical properties when examined under polarized light, and the electron-microscopic properties. The deposits in the affected livers of the treated dogs in this study were birefringent under polarized light, showing the typical “Maltese-cross”-like appearance. Using electron microscopy, a “sunburst” pattern with straight bars and rods was clearly evident. Both microscopic characteristics strongly indicated at protoporphyria.
Besides the histopathologic lesions, changes in several biochemical parameters in blood and urine indicated liver injury as well. The increased activity of alanine aminotransaminase supported the observed hepatocellular necrosis at sites of inflammation, as a sign of diminishing integrity of the hepatocytes. Marked increased alkaline phosphatase activity suggested an impaired hepatobiliary function, and also the high total bilirubin in blood together with the dark color of the feces of treated animals strongly indicated at disturbed bile-transport and -excretion. This was in accordance with the bile plugs (bile stasis) observed in 1 dog on histopathologic examination. It is likely that the pigment globules within the bile canaliculi caused obstruction of the bile flow by obliterating the canalicular lumina. The combination of reddish/dark brown feces, the microscopic properties of the pigment deposits and the fact that protoporphyrin (in contrast to other porphyrins) is the most important water insoluble heme precursor, strongly favoured protoporphyrin as being the most important factor involved in this drug-induced pathologic process. Finally, the dose-dependent increase in fecal protoporphyrins (in dogs treated with 15 and 50 mg/kg) proved the involvement of protoporphyrins.
In the literature, porphyria in man is described as a disease of abnormal porphyrin biochemistry in the body. The usual classification is based on deficiency of enzymes in the heme biosynthetic pathway (Frank and Christiano, 1998; Canadian Porphyria Foundation, 2004; American Porphyria Foundation, 2005) and divides the disease in at least eight separate types (Acute Intermittent Porphyria; AIP, Congenital Erythropoietic Porphyria; CEP, Porphyria Cutanea Tarda; PCT, Aminolevulinic acid Dehydrogenase Porphyria; ADP, Hepatoerythropoietic Porphyria; HEP, Hereditory CoproPorphyria; HCP, Variegate Porphyria; VP, Erythropoietic Protoporphyria; EPP). As synthesis of heme occurs in mammalian erythropoietic cells and in the liver, either bone marrow (blood cells) or the liver (hepatocytes) is the main site of defect, dependent of the porphyria type. Except for PCT, the human porphyria types are in general genetically determined. PCT is an acquired disease although in some individuals a genetic deficiency of the enzyme uroporphyrinogen decarboxylase contributes to the disease severity and development (Sassa and Kappa, 2000; American Porphyria Foundation, 2005). The main porphyrin precursor that accumulates in the human porphyria type EPP is protoporphyrin (Kansky and Glavaè, 2004). In EPP patients a markedly decreased activity of the enzyme ferrochelatase is found (Sarkani and Cox, 1995; Straka et al., 1991). This enzyme catalyzes the insertion of ferrous iron into protoporphyrin IX to form heme in the last step of the heme-biosynthetic pathway (Brenner et al., 1992; Ferrreira, 1999). Several studies point to a molecular defect in the coding region of the ferrochelatase gene in EPP (Brenner et al., 1992; Sarkany and Cox, 1995).
Since, in our study, the accumulation of protoporhyrins in the feces of the treated dogs also pointed to a deficiency in ferrochelatase, this mechanism was investigated at the Erasmus University Hospital in Rotterdam (the Netherlands). Measurements of enzyme activity in the livers of the 50 mg/kg treated dogs demonstrated a clear decrease in ferrochelatase activity. Furthermore, a slight increase in the activity of a second enzyme earlier in the heme-pathway, porphobilinogen-deaminase, was measured (Van den Dobbelsteen, D. J., personal communication). The involvement of ferrochelatase in both the inherited EPP in man as well as in the drug-induced protoporphyria in the 50 mg/kg treated dogs in our study, suggests a strong resemblance in pathogenesis. In the dog study no histopathological evidence for disturbance of the heme synthesis is demonstrated in the bone marrow. Therefore, in contrast to the situation in EPP in man, in this dog study the liver seems to be the primary affected site. However, despite the major erythropoietic involvement in most of the human patients with EPP, the liver can be heavily involved as well (Bruguera et al., 1976; Mac-Donald et al., 1981; Pimstone et al., 1976; Sarkani and Cox, 1995; Wolff et al., 1975). This liver involvement is supposed to be secundary to the disease. When the hepatic excretory capacities of the liver fail to cope with high quantities of protoporphyrins, accumulation of this product occurs in the liver (Pimstone et al., 1976).
More recent studies indicate that patients with liver complications share a common genetic feature (“null allele mutation”) that results in the formation of a truncated protein (Kansky and Glavac, 1994). The striking difference between the dog and man might be due to the fact that impairment of ferrochelatase activity occurs by a different mechanism, i.e. at the level of the gene in human and at the level of interaction between enzyme and drug (or metabolite) in the dog. Furthermore, it is not surprising that when a drug is given orally (as was the treatment route in the present dog study) the liver is the firstly affected organ.
Liver changes as described in our study and/or changes in biochemical parameters in blood and feces are not restricted to dogs (Gralla et al., 1977). In other laboratory species such as mice, rats and genetically engineered (ferrochelatase deficient) mice, such changes have been described as well after drug-treatment (Zaki et al., 1973; Matilla and Molland, 1974; Cantoni et al., 1983; Tutois et al., 1991; Frater et al., 1993; Smith et al., 1997). However, in a rat study carried out in our laboratory with the same drug candidate as used in the dog study, no lesions indicative for porphyria were observed. The most logical explanation would be a difference in metabolism of the compound between rats and dogs as different metabolites were detected for both species (Van den Dobbelsteen, D. J., personal communication).
In conclusion, the new drug candidate tested in a toxicity study induced protoporphyria in dogs. This resembled one of the porphyria-types in human (Erythropoietic Protoporphyria) since the activity of the same enzyme, ferrochelatase, is impaired. Differences in the primary site of defect and the genetic involvement in man strongly suggest a distinct underlying mechanism, however.
Footnotes
Acknowledgments
The authors would like to acknowledge Dr. F. de Rooij and Mrs. A. Edixhoven from Internal Medicine Department of the Erasmus University Hospital in Rotterdam (The Netherlands) for the excellent analysis of the enzyme activities. We would also wish to thank the biotechnical staff for providing animal treatment support and the histotechnological staff, the clinical chemical staff and the chemical analytical staff for their technical assistance, Dr. P. Jap for his help in the interpretation of the electron microscopy, Mrs. W. Benneker for coordinating all studies with the antipsychotic drug and Dr. H. Joosten and Prof. Dr. Ir. W. H. van der Putten for their critical reading of this manuscript.
