Abstract
The aim of the present paper was to describe the mechanisms and management of antipsychotic-induced weight gain in schizophrenia patients. A comprehensive literature review of all available articles on the mechanisms and management of antipsychotic-induced weight gain was done by searching databases PsychINFO and PubMed. A summary of the available guidelines for monitoring of antipsychotic-induced weight gain and metabolic syndrome is also provided. There has been a substantial increase in the number of studies investigating the mechanisms and management of antipsychotic-induced weight gain after 2002. These include advances in the understanding of pharmacogenomics of weight gain and several randomized controlled trials (RCTs) evaluating pharmacological and psychological treatments to promote weight loss. The most effective strategy for prevention of weight gain is the choice of antipsychotic medication with low weight gain potential. In individuals with established weight gain and metabolic issues, switching to an antipsychotic agent with lower weight gain potential and/or lifestyle modifications with physical activity are most effective in promoting weight loss. Pharmacological agents such as orlistat and sibutramine are effective in general obesity but have not been sufficiently evaluated in antipsychotic-induced weight gain. The case to prescribe routine pharmacological treatment to promote weight loss is weak. Long-term, pragmatic studies are required to inform clinical practice. Weight gain in schizophrenia is associated with significant physical and psychological morbidity. Achieving an optimal trade-off between effectiveness and side-effects of antipsychotic agents, although difficult, is achievable. This should be based on three main principles: (i) a shared decision-making model between the patient, clinician and carer(s) when choosing an antipsychotic; (ii) a commitment to baseline and follow-up monitoring with explicit identification of the responsible individual or team; and (iii) the adoption of clear structured protocols for clinicians to follow in case of clinically significant weight gain and metabolic issues, which should incorporate greater collaboration between various health professionals from psychiatric and medical specialist services.
Keywords
Obesity has reached epidemic proportions throughout the world. The report of a World Health Organization (WHO) consultation on obesity in Geneva in 1998 endorsed the body mass index (BMI) as a measure of obesity. It is expressed as weight in kilograms divided by the square of height in metres (kg m−2). BMI < 18.5 kg m−2 is defined as underweight, 18.5–24.9 kg m−2 as normal range, >25 kg m−2 as overweight and >30 kg m−2 as obese.
Obesity has become a particular concern in individuals with psychiatric illness and the recent debate has focused on patients with schizophrenia. Much of the weight gain is attributed to antipsychotic medications. Weight gain causes significant morbidity and mortality notwithstanding the societal costs associated with it.
This article focuses on the mechanisms of weight gain in schizophrenia due to antipsychotic drugs and the various evidence-based management strategies to tackle this issue. A literature search was performed in PsychINFO and PubMed to identify relevant articles.
Weight gain in schizophrenia
Weight gain is a common issue in schizophrenia and the prevalence of obesity exceeds general population estimates [1]. Individuals with schizophrenia have threefold as much intra-abdominal fat as controls matched for age, gender and lifestyle [2]. Therefore patients with schizophrenia are at an intrinsically increased risk of complications of obesity. In addition they have several life style factors (e.g. poor diet, smoking and a sedentary lifestyle) that further increase this risk [3]. After the advent of atypical antipsychotics prevalence of obesity has increased significantly, at estimates of approximately 10% [4]. There is general consensus that atypical antipsychotic medications are most associated with weight gain and metabolic disturbances. Henceforth in the present article the term ‘antipsychotics’, ‘atypicals’ or ‘atypical antipsychotics’ are used to indicate second-generation antipsychotics unless otherwise specified.
The atypicals differ in their potential to cause weight gain, with olanzapine and clozapine causing the most weight gain [5]. This difference in weight gain potential has been borne out in various studies including the recent Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) trial, which showed that olanzapine was associated with greatest weight gain, at an average of 0.9 kg per month compared to other antipsychotics [6]. A meta-analysis of 81 studies of antipsychotic weight gain showed an average of 4.5 kg and 4.0 kg of weight gain with clozapine and olanzapine, respectively over 10 weeks [7].
Considering that olanzapine has a high weight gain potential and is the second most widely prescribed atypical in the UK and the most widely prescribed in Australia, it has significant implications in the management of patients with schizophrenia [8]. Interestingly though, there are suggestions that the extent of weight gain is associated with improvement in psychopathology [9], [10].
Morbidity of antipsychotic-induced weight gain
Obesity is associated with significant psychological and physical morbidity. Obesity increases the risk of metabolic syndrome (Table 1) [11], cardiovascular disease (CVD) and stroke. Increased abdominal obesity, particularly visceral abdominal fat, is strongly associated with decreased insulin sensitivity, which impairs glucose control and increases triglycerides and highly oxidized low-density lipoprotein particles, increases blood pressure, and increases markers of blood clotting, all of which are associated with an increased risk of CVD [12]. A Spanish study showed that coronary heart disease risk and metabolic syndrome prevalent among patients treated with antipsychotics, was in the same range as that of the Spanish population 10–15 years older [13]. There is emerging literature linking obesity with depression [14], [15], low self esteem, shame and guilt [16]. Weight gain is associated with stigma, which in turn contributes to impaired quality of life and increases the risk of depression [17]. It is also associated with non-compliance of medication [18]. In the CATIE trial olanzapine had the lowest rate of discontinuation due to any cause, but in the rates of treatment discontinuation due to intolerable side-effects olanzapine had the highest rate, mainly due to weight gain and metabolic effects [6]. Weight gain also reduces the quality of life [19]. Weight gain has significant clinical and public health implications. The study by Fontaine et al. showed that the anti-suicidal effect of clozapine could be reduced by the deaths due to clozapine-induced weight gain. The study estimated that the use of clozapine for schizophrenia over a 10 year period would prevent 492 deaths from suicide per 100 000 patients. This would, however, be offset by an estimated 416 deaths due to a presumed clozapine-induced weight gain of 10 kg [20].
New International Diabetes Federation criteria for metabolic syndrome (2006)

BP, blood pressure; HDL, high-density lipoprotein; †South Asia and South-East Asia: men ≥90 cm, women ≥80 cm; Japan: men ≥85 cm, women ≥90 cm.
Mechanisms of antipsychotic weight gain
Antipsychotic-induced weight gain is a complex side-effect with multiple causal mechanisms.
Effect on energy expenditure and energy intake
Reduced rate of energy expenditure causing obesity was first proposed in 1988 [21]. Sharpe et al. found a lower than predicted resting energy expenditure (REE) in people taking antipsychotic medications [22]. A recent Australian study showed that the total energy expenditure rates in patients taking clozapine were 20% lower than the WHO recommendations. This indicates that weight management strategies should focus on enhancement of energy expenditure (e.g. exercise) rather than strategies to reduce energy intake (e.g. diet or food intake) [23]. But olanzapine treatment did not affect REE rates [24]. Moreover, weight gain with antipsychotics is associated with increased food intake and low habitual activity [25].
Effects on receptors and neurotransmitters
The two receptors that are most robustly associated with weight gain are the Serotonin 2C (5-HT2C) and histamine 1 (H1) receptors. Facilitation of serotonin transmission reduces food intake and decreases feeding behaviour [26]. Antagonism of serotonin receptors, on the other hand, induces food intake despite satiety leading to weight gain in the long term [27]. It is postulated that the antagonism of 5-HT2C receptors centrally by antipsychotics increases food intake despite the sensation of satiety, leading to weight gain [28]. Histamine receptor antagonism stimulates energy intake centrally by increasing appetite [29]. The H1 receptor is involved in the orexigenic actions of antipsychotics whereby the antipsychotics potently and selectively bind to the paraventricular and arcuate nucleus of the hypothalamus, increasing appetite and reversing the anorexigenic actions of leptin [30]. Two other receptors that have generated recent interest are α−2 adrenergic receptor [31] and the sterol regulatory element binding protein-1 (SREBP-1). SREBP-1 is postulated to regulate peripheral adipogenesis and is influenced by olanzapine [32]. The differential receptor binding profiles possibly account for the differential weight gain potentials of the antipsychotic agents. Thus clozapine and olanzapine, which have the greatest affinity for 5-HT2C and H1 receptor, have the greatest weight gain potential. Risperidone, in contrast, has lesser affinity for the 5-HT2C and H1 receptors, causing lesser weight gain than olanzapine or clozapine. Ziprasidone, which is considered weight neutral, is a potent 5-HT1a receptor agonist and a potent 5-HT2C antagonist. Its weight neutral profile may be explained by its synaptic re-uptake inhibition of serotonin and norepinephrine [33]. Aripiprazole, in contrast, has a unique mechanism of action with partial D2 and 5-HT1a agonism and 5-HT2a antagonism. Its low weight gain potential is attributed to its moderate affinity to H1 receptors and lack of 5-HT2C antagonism [34], [35].
Pharmacogenetics and weight gain
An important finding is that patients with lower BMI are at highest risk of weight gain and that early and rapid weight gain in the first few weeks of treatment appears to be a predictor of long-term weight gain. In some patients this weight gain is irreversible and discontinuation or switching antipsychotics does not reduce weight [36]. Thus there must be certain fundamental biological factors that make some individuals more susceptible to weight gain. This has prompted a search for susceptibility genes.
A common 5-HT2C receptor promoter region polymorphism demonstrates strong associations with weight gain and increased risk of metabolic syndrome in people taking antipsychotics [37–39]. More specifically a 5-HT2C-receptor promoter (−759c/t) is identified as a true genetic susceptibility factor for antipsychotic weight gain [36], [40]. It is hypothesized that the 5-HT2C polymorphism appears to determine levels of circulating leptin, providing a potential mechanism underlying the genetic association of the 5-HT2C receptor with weight gain [41].
Investigations of serotonin transporter (SERT) gene polymorphisms and the relationship of long allele/short allele (L/S) promoter (SERTPR) regions with olanzapine-related weight gain found that the SS (short allele) genotype was associated with significantly higher weight gain in those who were non-obese at time of admission [42]. Incidentally it is this genotype that is associated with poorer response to antidepressant treatment.
Another gene polymorphism that has raised interest is the 25 kDa synaptosomal associated protein (SNAP) polymorphism. The SNA protein encoded by the gene is involved in the regulation of hunger and satiety and release of neurotransmitters [43]. There are several other candidate genes for antipsychotic-induced weight gain and current pharmacogenomic approaches have estimated the number of candidate genes to be around 300 [44]. Results from genome-wide association and linkage studies point to several chromosomal regions (e.g. 12q24) and some specific genes (e.g. promelanin concentrating hormone (PMCH), polycystic kidney and hepatic disease 1 (PKHD1), peptidylglycine α-amidating monooxygenase (PAM)). Others include adrenergic α2a (ADRα2a) receptor, leptin, ghrelin, guanine nucleotide binding protein, α−1, β−2 and β−3 adrenergic receptor, tumour necrosis factor-α, adiponectin and dopamine D2 receptor genes [36], [45]
Role of leptin
Leptin is a hormonal product synthesized by fat cells (ob gene) and was initially thought to be one of the most important signals for the long-term regulation of weight gain. Leptin binds to the ventral medial nucleus (VMN) of the hypothalamus, known as the ‘satiety centre’. Binding of leptin to this nucleus signals to the brain that the body has had enough to eat: a sensation of satiety. This action occurs due to the specific inhibition of neuropeptide Y (NPY), which influences feeding behaviour and has an effect in regulating leptin levels itself. Several early studies postulated that leptin is related to weight gain through impairments in leptin secretion, leptin clearance and central leptin resistance, that is, loss of negative feedback inhibition from the brain [46–49]. All these studies, however, did not control for adiposity, which is a potent signal for leptin secretion. Three studies controlling for gender and adiposity showed that leptin, gender (female sex) and adiposity positively correlated with each other [50–52]. Additionally leptin also positively correlated with insulin, glucose and insulin resistance, which suggested leptin's role in insulin sensitivity regulation. Thus it is likely that leptin plays a perpetuating role in antipsychotic-induced weight gain and is a consequence of metabolic abnormalities rather than a primary promoter of weight gain [53].
The rise in leptin is clinically significant irrespective of the cause because it increases fat deposition in non-adipose tissues (e.g. blood vessels, liver, heart etc.), increasing the risk of metabolic syndrome. This occurs due to the disturbance in the normal regulatory function of leptin on peripheral subcutaneous fat [54–56]. Summarizing the evidence, the role of leptin in antipsychotic-induced weight gain is unclear. Further complicating the situation is the genetic variability in the leptin receptor, which may predispose some individuals to gain weight on antipsychotics [57], [58].
Ghrelin
Ghrelin is a newly discovered appetite-stimulating peptide that has a role in the regulation of feeding behaviour. Ghrelin is synthesized principally in the stomach, and the concentration of circulating ghrelin is negatively correlated with leptin and body fat mass. Atypical antipsychotics promote the orexigenic effects of ghrelin, that is, effects to increase appetite during therapy, which at the same time renders anorexigenic pathways ineffective. This may lead to weight gain, and further studies with a ghrelin antagonist may provide support for this hypothesis [59]. So far conflicting results have been obtained. Some studies have shown no increase in ghrelin on atypical antipsychotic treatment [60], [61]. Others have suggested that olanzapine may directly act on the secretion of ghrelin and induce appetite, resulting in weight gain [62]. The link between atypical antipsychotic treatment and elevated serum ghrelin levels is not clear so far, but a dysregulation of the central feedback mechanism can be hypothesized [63].
Orexins
A small number of lateral hypothalamic/perifornical area neurons release the orexins, peptides that are critically involved in body weight regulation and arousal. Antipsychotic-induced weight is associated with activation of distinct orexin neurons. This is a relatively new area and there has been only one study evaluating the effect of antipsychotics and orexins [64].
Neuroendocrinal mechanisms
Other less well-studied mechanisms include the effect of antipsychotics in increasing prolactin. Hyperprolactinemia can impair insulin sensitivity and affect circulating levels of gonadal hormones, leading to increased adiposity [65], [66].
Weight gain through indirect effect on glucose and lipids
Hyperglycaemia and hyperlipidaemia are often considered to be consequences of weight gain. Although this is well substantiated [12], there also exist independent associations of antipsychotic medications with hyperglycaemia and dyslipidaemia independent of adiposity. Olanzapine and clozapine have the greatest impact in increasing blood glucose and lipids independent of adiposity [6], [67–69]. The evidence for the association between atypical antipsychotic drugs and diabetes remains confusing [70] but the following is known. Schizophrenia and antipsychotics are both independently associated with an increased rate of diabetes and worsening blood sugar control. Atypical antipsychotics are more likely to produce this effect than the first-generation agents, but both contribute to impaired blood glucose mainly through insulin resistance [71]. Dyslipidaemia, on the other hand, also causes insulin resistance, which increases adipose tissue deposition [72]. Thus there exists a complex relationship between adiposity, blood glucose and serum lipids in relation to antipsychotics. This is outside the scope of this article and I refer readers to the following references for further reading [12], [70].
What does the evidence tell us?
It is clear that there are a number of biological and non-biological factors that contribute to antipsychotic-induced weight gain. The main non-biological factors include age, gender, response to drug treatment, weight before initiation of drug treatment, smoking, diet, physical activity and socioeconomic status. The most robust biological factors include the 5-HT2C and H1 receptors. The role of other neuroendocrine factors such as ghrelin, leptin, orexins and prolactin is unclear and further research is required to elucidate their mechanisms. Furthermore certain individuals are more prone to weight gain than others, which has prompted the search for susceptibility genes. Although several genes have been identified, the 5-HT2C receptor polymorphisms have shown the strongest association with antipsychotic-induced weight gain. Further research is needed to clarify the complex relationship between antipsychotics, glucose, lipids and adiposity. There exists a differential weight gain potential among atypical antipsychotics, with clozapine and olanzapine causing the greatest weight gain and ziprasidone and aripiprazole the least, although they have not been used as extensively as the other agents [73].
Management
There is no current international consensus on the management of weight gain associated with antipsychotics. Management strategies investigated can broadly be divided into choice of antipsychotic, pharmacological and psychological treatments for promoting weight loss and switching strategies. There has been one systematic review evaluating 18 trials of pharmacological treatments and five trials of cognitive behavioural treatments [74]. Overall, there have been approximately 50 trials evaluating pharmacological and psychological strategies to reduce or reverse antipsychotic-induced weight gain.
Choice of antipsychotic
Choice of an antipsychotic is the most important consideration in the prevention of weight gain but there is a consensus that optimal treatment of psychosis is a priority [71], [73]. There are a number of factors that should be considered when choosing an antipsychotic, which include the nature and severity of the illness; the past response of the patient; patient and family preference; psychiatric and medical comorbidity; effectiveness of the medication; and potential for extrapyramidal and metabolic side-effects. Thus although ziprasidone has the least weight gain potential among second-generation antipsychotics, there may be other factors that may militate against its prescription. It is difficult to generalize and each case should be assessed individually. A risk–benefit analysis is crucial because even with a high risk of worsening the metabolic profile, the benefits of the medication may outweigh the risks.
Pharmacological management
The main pharmacological agents investigated for antipsychotic-induced weight gain are H2 (histamine) antagonists (nizatidine, famotidine and ranitidine), sibutramine, topiramate, amantadine, metformin, antidepressants (fluoxetine, fluvoxamine, bupropion and reboxetine), fenfluramine and phenylpropanolamine. Orlistat and rimonabant are two other agents that are beneficial in general obesity but have not been evaluated in antipsychotic-induced weight gain.
Orlistat at 120 mg thrice daily is considered to be the best choice for treatment of obesity in people with chronic mental illness [3]. It may offer a pharmacological treatment option to support dietary efforts in obese and overweight psychiatric patients [75]. Orlistat is a lipase inhibitor, which acts by reducing the absorption of dietary fat by approximately 30% [76]; but there have been no RCTs in weight gain due to antipsychotics.
Sibutramine is effective in promoting weight loss in general obesity with improvements in cardiovascular and metabolic risk factors [77]. Two RCTs in antipsychotic-induced weight gain have produced contradictory results [78], [79]. Its use in psychiatric populations may be limited because it has been known to cause panic, psychosis and mania in case reports, and further studies are required to establish efficacy and safety in individuals with mental illness [80–82].
Rimonabant is a selective cannabinoid receptor antagonist that affects the endocannabinoid system in the brain that modulates the intake of palatable sweet or fatty foods. Acomplia (rimonabant; Sanofi-Aventis, France) has been available in Europe since the middle of 2006, and is indicated as an adjunct to diet and exercise for obese or overweight patients with associated risk factors, such as type 2 diabetes or dyslipidaemia. It is not available in Australia and does not have Food and Drug Administration approval in the USA. The manufacturers do not recommend its use in uncontrolled psychiatric illness [83]. Treatment with rimonabant at 20 mg day−1 is associated with better glycaemic control in type 2 diabetes mellitus, improved lipid profile, reduced blood pressure and an overall decrease in the prevalence of the metabolic syndrome, thus modifying cardiovascular risk. There is evidence that rimonabant can significantly reduce human visceral fat, and future studies may examine its role in individuals with mental illness [84].
Topiramate is a second-generation anti-epileptic agent and has a mixed GABAergic and anti-glutamergic action through its action at the AMPA receptors. Topiramate reduces NPY Y1 and Y5 receptors centrally (receptors that increase appetite). Peripherally topiramate reduces leptin mRNA transcription in adipose tissue, which increases energy expenditure [1]. There is also speculation that topiramate reduces appetite by altering taste due to its inhibition of carbonic anhydrase [85]. Topiramate induced weight loss in two RCTs of antipsychotic-related weight gain [86], [87].
Metformin is an oral hypoglycaemic agent that reduces hepatic glucose production and increases peripheral utilization of glucose. It also improves insulin sensitivity and thus indirectly decreases bodyweight. Metformin has shown some benefit in attenuating weight gain in antipsychotic-induced weight gain in children and adolescents [88]. Other studies have produced inconclusive results [89–91].
Amantadine increases the release of dopamine by blocking the re-uptake of dopamine and blocking N-methyl-
Psychological strategies
The main psychological strategies in antipsychotic-induced weight gain are cognitive behavioural therapy; behavioural therapy; nutritional interventions; and others incorporating all of these in various combinations. There have been seven RCTs. One has evaluated cognitive behavioural therapy [94], two have evaluated behavioural therapy [95], [96] and four others evaluated lifestyle modifications and education. [97–100]. There have been several other open-label studies evaluating all the aforementioned interventions. Overall psychological interventions may result in weight loss as shown in these studies. The most promising are behavioural interventions incorporating lifestyle modifications and physical activity. They are, however, resource intensive and require long-term intervention to produce clinically significant results. In the UK at the Cromwell clinic, a weight management clinic that included discussion, education and weight measurement within a group-based programme showed reductions in mean weight (6.2 kg) and BMI and this correlated with the number of sessions attended [101]. An Australian RCT investigating a nutritional intervention approach in patients commenced on olanzapine found weight gain in both groups but significantly lesser weight gain in the intervention group. The control group BMI increased significantly (2 kg m–2 vs 0.7 kg m–2). The intervention group also reported significantly greater improvements in moderate exercise levels, quality of life, health and body image compared to controls at 3 and 6 months [102].
Antipsychotic switching
Because for many patients with schizophrenia treatment is usually long term, one viable option to reverse weight gain is by switching antipsychotics. A review of the evidence for switching antipsychotics concluded that switching to an antipsychotic with a lower propensity to cause weight gain or dyslipidaemia can be effective for reversing the weight gain and dyslipidaemia caused by previous antipsychotic treatment [103]. Three separate open-label 6 week multicentric studies showed that switching to ziprasidone was an effective strategy in individuals on olanzapine and produced reductions in lipids and weight [104]. Quetiapine has shown benefit in case reports and aripiprazole has been evaluated in RCTs with positive effects on weight loss [105–109]. One Danish study showed a striking mean weight loss of 10.5 kg in six patients switching from olanzapine or risperidone to low-dose perphenazine plus buspirone. No deterioration of mental state was observed [110]. Amisulpiride, due to is selective affinity for dopamine receptor, is particularly likely to benefit patients who have gained weight on previous therapy, or who have negative symptoms of schizophrenia and depressive symptoms [111].
Summary of evidence
There has been a great increase in the number of studies conducted to manage antipsychotic-induced weight gain. The most effective strategy for prevention of weight gain is the choice of antipsychotic medication with low weight gain potential. In individuals with established weight gain and metabolic issues, switching to an antipsychotic agent with lower weight gain potential and/or lifestyle modifications (Table 2) with physical activity are most effective in promoting weight loss. Pharmacological agents such as orlistat and sibutramine are effective in general obesity but have not been sufficiently evaluated in antipsychotic-induced weight gain. The case to prescribe routine pharmacological treatment to promote weight loss is weak. It is difficult to collate the results of the studies into a meta-analysis to give a more robust pooled estimate of effect size due to heterogeneity in studies. Long-term effectiveness of studies cannot be extrapolated from the data because most studies are short term, with the longest being 12 months. Long-term, prospective, naturalistic studies are required to inform clinical practice.
Lifestyle modifications for obesity†

†Adapted and modified from [73].
Difficulties in implementation of evidence-based strategies
There is a lack of resources and reluctance to fund evaluations of preventive strategies, which would be the first step in any intervention. The development and evaluation of evidence-based investigation to manage obesity by diet and exercise in primary care costs approximately $AU12.5m [112]. This brings the principle of utilitarianism into question, because health managers could argue for funding to be channelled into already existing evidence-based resources.
There are also uncertainties around who should take the primary responsibility for monitoring of weight, lipids and blood sugar and for managing it in case of abnormalities. Some suggest that the primary responsibility lies with the psychiatrists and that the task should be clearly delegated to specific members of the community team if the primary treating psychiatrist does not monitor these [113].
Guidelines
There have been some attempts to develop a consensus for monitoring and management of patients with weight gain due to antipsychotic drugs. These, however, do not incorporate specific psychological or pharmacological strategies. Current practice of monitoring weight and other metabolic parameters falls short of recommendations. Despite the higher prevalence of CVD and metabolic syndrome and published recommendations, monitoring rates are poor and the physical health of patients with schizophrenia often deteriorates in the long term [114].
The various attempts at forming a consensus are as follows: (i) Mount Sinai guidelines [115]; (ii) Australian position statement [71]; (iii) American Diabetes Association, American Psychiatric Association, American Association of Endocrinologists, North American Association for the Study of Obesity, Consensus guidelines conference on antipsychotic drugs and obesity and diabetes [73]; (iv) expert group ‘Schizophrenia and diabetes 2003’ Expert Consensus Meeting (UK) [116]; (v) Canadian Diabetes Association position paper: antipsychotic medications and associated risks of weight gain and diabetes [117]; and (vi) Belgian consensus on metabolic problems associated with atypical antipsychotics [118].
A summary of the main points based on the American and Australian consensus statements is provided in the form of a flowchart (Figure 1).
Recommendations for monitoring and interventions (based on American Diabetes Association (ADA) and Australian consensus statement on Diabetes, Psychotic disorders and Antipsychotic therapy (ACS)). BMI, body mass index; CVD, cardiovascular disease; DM, diabetes mellitus; GP, general practitioner; HDL, high-density lipoprotein; LDL, low-density lipoprotein; SGA, second-generation antipsychotic.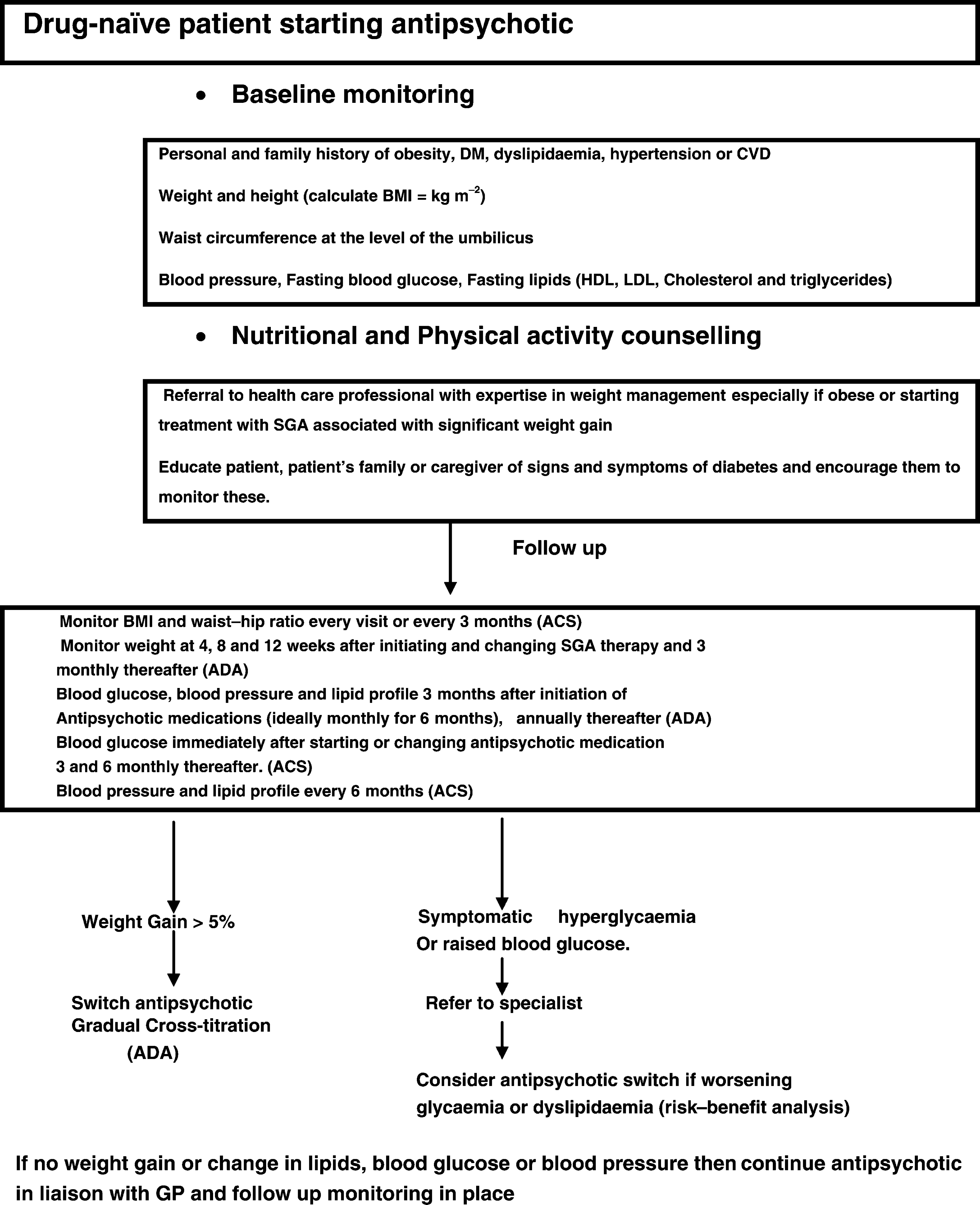
Future research
There are a number of treatments being evaluated for management of antipsychotic-induced weight gain. These can be found on the website http://clinicaltrials.gov/. Newer studies are focusing on elucidating the fundamental mechanisms of weight gain through pharmacogenomic approaches and evaluating changes in energy homeostasis, metabolism, energy intake and expenditure during antipsychotic treatment. New drugs being evaluated for antipsychotic-induced weight gain include atomoxetine (norepinephrine re-uptake inhibitor), zonisamide (anticonvulsant), β-histine (H1 receptor agonist), and mifepristone (glucocorticoid antagonist). Studies are also testing if sublingual olanzapine causes less weight gain than standard oral olanzapine. One study found that orally disintegrating olanzapine was associated with lower weight gain than standard tablets in first-episode never-treated psychotic patients. Theoretically this is due to partial sublingual absorption, leading to a difference between parent compound and metabolite [119]. Other switching strategies to agents with lower weight gain potential are also being evaluated.
Conclusion
Weight gain due to antipsychotic medication is associated with significant physical and psychological morbidity. Individuals with schizophrenia treated with antipsychotics have concerns about weight gain and weight management, like the general population, and express a desire to be more active [120], [121]. Achieving an optimal trade-off between effectiveness and side-effects of antipsychotic medications, although difficult, is achievable. It calls for a shared decision-making model, when prescribing an antipsychotic, involving the patient, clinician and carer(s) that takes into account several factors such as patient values and preference, response and tolerance. This should accompany a commitment to careful baseline screening and follow-up monitoring with explicit identification of the responsible individual or team. There is a need for services to adopt clear structured protocols for clinicians to follow in case of clinically significant weight gain and metabolic issues that should incorporate greater collaboration between mental health professionals, endocrinologists, general practitioners, dieticians, nurses and occupational therapists to achieve holistic care for individuals with schizophrenia.
Conflict of interest
The author has been sponsored by Janssen-Cilag to attend a conference.
Footnotes
Acknowledgements
I wish to thank Professor Chris Tennant for his valuable advice in preparing this manuscript.
