Abstract
In the treatment of major depressive disorders, combination strategies have a modest evidence base akin to alternative therapies [1], which is reflected in their low hierarchical staging in treatment guidelines [2]. Nevertheless, antidepressant combinations are often prescribed by psychiatrists as second-line treatment, with perceived benefits of synergistic efficacy and faster onset of action. [3]. A number of combination antidepressant strategies have been studied, with accumulating evidence in support of the strategy [4–6]. Among these, an increasingly popular strategy is the combination of venlafaxine and mirtazapine, an option that is mooted as being especially efficacious in clinical practice [7]. Dubbed ‘California rocket fuel’ by Stahl [8], the theoretical advantage of this combination lies in the amplified serotonin and noradrenaline release, and subsequent neurotransmission, and a putative rapid onset of action via α2-adrenergic antagonism. Although, generally, combinations of antidepressants are likely to increase side-effects [9], the combination of venlafaxine and mirtazapine is thought to be mitigating in this regard [3].
Pharmacologically, venlafaxine is a dual serotonin and noradrenaline re-uptake inhibitor, and although mirtazapine also acts on both neurotransmitter systems, its actions include α2-adrenergic, 5-HT2 and 5-HT3 serotonergic receptor antagonism [10]. In combination, venlafaxine and mirtazapine are thought to exert broader pharmacodynamic effects, which may include a more selective transmission of the augmented levels of synaptic serotonin through the 5-HT1A receptor [8], [11]. The notion of broadening the receptor profile of antidepressant therapy is indirectly supported by data suggesting that antidepressants with multiple mechanisms of action may possibly have an edge in efficacy over those with a single mechanism of action [12]. Indeed, early data on the combination of venlafaxine and mirtazapine support its utility in treatment-resistant depression.
The aim of the present paper was to add to the evidence base by reporting a prospective case series of patients with depression treated with a combination of venlafaxine and mirtazapine.
Methods
Patients
Cases in which combination venlafaxine and mirtazapine was naturalistically implemented in the treatment of major depressive episode, as defined by the DSM-IV-TR [13], were prospectively collected by the authors at several sites that consisted of both public and private psychiatric hospital settings and included both inpatients and outpatients. Case selection was independent of treatment outcomes and all individuals had failed monotherapy prior to combination therapy. Dosing of medications was at the discretion of the treating clinicians.
Clinical assessment
The following demographic and clinical patient data were gathered: age and gender, diagnostic types (unipolar or bipolar II major depressive episode, melancholic or non-melancholic subtype), comorbid psychiatric diagnoses, chronicity of the mood disorder (duration of condition, number of previous episodes and duration of index episode), treatment history of the index episode including antidepressants trialled and the use of augmentation or combination strategies, and the dose of the established agent (venlafaxine or mirtazapine) on commencement of combination therapy. Baseline scores on the 17-item Hamilton Depression Rating Scale (HAM-D17), Montgomery–Åsberg Depression Rating Scale (MADRS) and the Clinical Global Impression–Severity Scale (CGI-S) were recorded. These were repeated at end-point, which was determined by the naturalistic termination of the depressive treatment episode or the discontinuation of the combination treatment due to adverse effects. The length of treatment until end-point was documented for each patient. Dosages used for both medications were naturalistically determined. Descriptive statistics were used on the collated data.
Results
A total of 22 patients were included in this case series, with a mean age of 38.5 years (SD = 13.3, range = 22–75 years) and a gender distribution of seven men (31.8%) and 15 women (68.2%). The majority (20 patients) were diagnosed with unipolar major depression, while the remaining two had a longitudinal diagnosis of bipolar II disorder. Ten patients met criteria for a melancholic subtype. More than half of the patients (n = 12) had at least one lifetime psychiatric comorbidity, with anxiety disorders being the most common diagnoses (Table 1). For the parameters of illness chronicity, the mean overall duration of illness was 10.7 years (SD = 6.5, range = 2–26 years) and the mean number of previous illness episodes was 5.5 (SD = 3.8, range = 1–14).
Baseline and end-point measures for patients on combination venlafaxine and mirtazapine
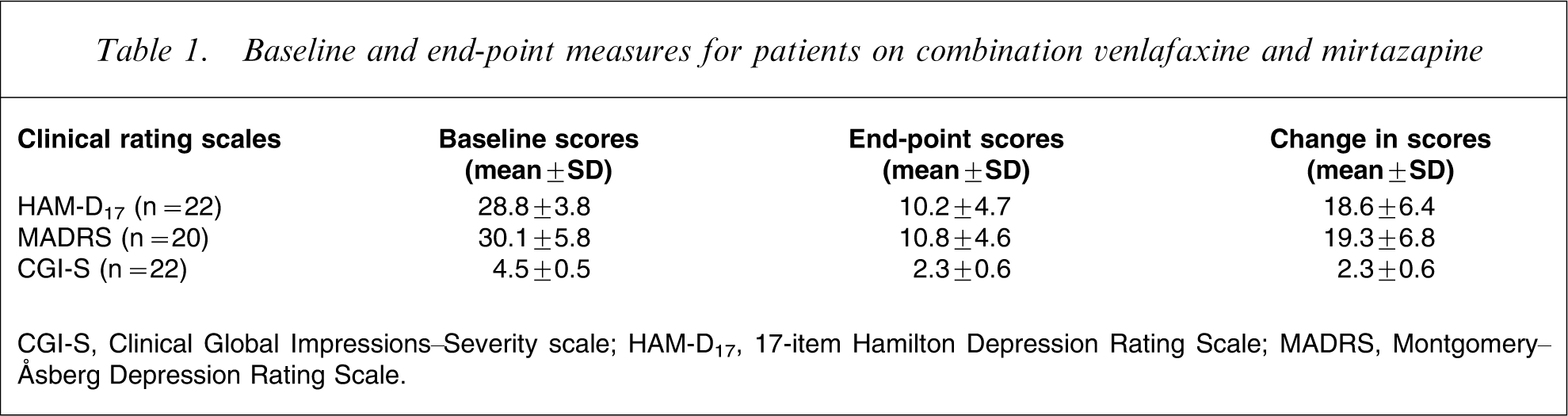
CGI-S, Clinical Global Impressions–Severity scale; HAM-D17, 17-item Hamilton Depression Rating Scale; MADRS, Montgomery–Åsberg Depression Rating Scale.
The mean duration of the index episode was 9.1 weeks (SD = 4.5, range = 2–16 weeks). Most patients had undergone unsuccessful trials of one (nine patients) or two (nine patients) antidepressants for the index major depressive episode, with selective serotonin re-uptake inhibitors and venlafaxine being the most commonly selected agents. Three patients had received lithium augmentation during the index episode, and none had been on combination antidepressants. On commencement of the combination, 18 patients had been established on venlafaxine and four on mirtazapine, with respective mean doses of 283.3 mg (SD = 70.7, range = 150–375 mg) and 56.3 mg (range = 45–60 mg) per day. Patients on combination therapy were not taking any additional psychotropic medications such as atypical antipsychotics or mood stabilizers.
At baseline, mean scores were 28.8 (SD = 3.8) for HAM-D17, 30.1 (SD = 5.8) for MADRS, and 4.5 (SD = 0.5) for CGI-S, reflecting a cohort at the moderate to severe end of the spectrum. At end-point, mean absolute scores were 10.2 (SD = 4.7) for HAM-D17, 10.8 (SD = 4.6) for MADRS, and 2.3 (SD = 0.6) for CGI-S. Mean change from baseline was 18.6 (SD = 6.4) for HAM-D17, 19.3 (SD = 6.8) for MADRS, and 2.3 (SD = 0.6) for CGI-S (Table 1). Response, as defined by change >50% in HAM-D17 score, was reached by 18 of the 22 patients (81.8%). Remission, defined as a final score of ≤7 on HAM-D17, was achieved by six patients (27.3%). The mean naturalistic duration of treatment follow up was 7.7 weeks (SD = 2.8, range = 2–10 weeks). Of those who responded, response as previously defined occurred after a mean duration of 4.6 weeks (SD = 1.9, range = 2–10 weeks). Mean daily doses of venlafaxine and mirtazapine, respectively, were slightly lower at end-point, and were 276.1 mg (SD = 67.0, range = 150–375 mg) and 48.4 mg (SD = 12.2, range = 30–60 mg). One patient discontinued treatment due to unremitting and intolerable nausea. There were no cases of serotonin syndrome and although in the cohort as a whole, 10 patients experienced side-effects, namely sedation (n = 8 patients), weight gain (n = 3 patients) and nausea (n = 3 patients), none of these was deemed to be severe enough to stop treatment.
Discussion
This case series lends support for the efficacy of venlafaxine and mirtazapine in combination as a post-first-line treatment in major depression in a naturalistic treatment setting. The outcomes appear favourable in comparison with the two published studies. Specifically, the current case series demonstrates a response rate of approximately 81.8% after a mean duration of under 8 weeks, compared with response rates of 44% after 8 weeks and 50% after 12 weeks in another case series [14], and a remission rate of 27.3% after approximately 6 weeks compared with 13.7% after 12 weeks in the STAR∗D study [15]. Clearly, these three studies are not directly comparable due to differences in sample size, population characteristics and methodologies. In particular, patients in the present case series had variable levels of treatment resistance and, as a whole, are likely to have been less resistant to treatment than patients in the other studies, as inferred from the number of unsuccessful medication trials prior to the combination under investigation. This may be a relevant factor in accounting for the relatively high response and remission rates, although the significance of these absolute values is indeterminate in view of the limitations pertaining to case series. The high response rate may also reflect a number of additional factors such as enhanced treatment compliance because of undergoing assessment and because of a heightened placebo effect by virtue of greater clinician involvement.
It is pertinent to observe that the mean venlafaxine and mirtazapine doses at naturalistic end-point, of 276.1 mg and 48.4 mg, respectively, fall within common dose ranges in psychiatric settings. This is consistent with the synergistic principle of combination treatment, which usually entails the use of lower medication doses, in comparison with an alternative strategy of extending antidepressant monotherapy to supratherapeutic doses, for which few supporting data are available [16].
The mean time to response of 4.6 weeks that was obtained in the present case series is notable, in spite of the limitations that undermine the strength of this finding, because it is shorter than response times typically reported in other major depression treatment trials, and the venlafaxine and mirtazapine combination has specific theoretical mechanisms for a more rapid onset of anti-depressant effect. From the STAR∗D trials, response times have ranged from 5.5 to 11.4 weeks [15], [17–19], and those for antidepressant combinations were reported as 6.3 weeks for citalopram plus sustained-release bupropion, 6.8 weeks for citalopram and buspirone [19], and 8.6 weeks for venlafaxine and mirtazapine [15]. One potential mediating factor for the discrepant response times between the STAR∗D venlafaxine and mirtazapine trial and the present study, besides the obvious methodological and sample differences, is the implementation strategy of the combination therapy. Whereas both medications were commenced simultaneously and their doses titrated in parallel in the STAR∗D trial, in the present case series a second medication was added to an established base medication, thus possibly accounting for extra lag time for the trial participants. In the present case series, there is also the possibility that the decision to augment may reflect the presence of a partial response rather than a non-response to the baseline treatment. Indeed, in pursuing algorithmic antidepressant management of patients with depression, it is arguably preferable to continue a medication that has produced a partial response as opposed to switching to an alternative.
Limitations
The lack of a control group is a significant limitation of this preliminary study and this clearly constrains the strength with which inferences can be made. But the use of a placebo design, although ideal, is difficult to implement in practice in patients already failing to respond to antidepressant treatment.
Conclusion
Data from randomized, controlled trials are required to confirm these preliminary but promising efficacy results for combination venlafaxine and mirtazapine, which seems to be an effective combination based on the pharmacodynamic synergism and increasingly common usage in clinical psychiatric practice. It may be particularly useful in patients with certain symptomatic profiles, such as melancholic or agitated depression, and a higher place in treatment algorithms may be appropriate, pending further data from clinical trials.
Footnotes
Acknowledgements
There was no pharmaceutical industry funding for this study and no company had any role in the data collection, analysis and interpretation or submission for publication. Professors Malhi and Berk have served on a number of pharmaceutical industry advisory boards in the past 3 years and have received honoraria for talks and presentations and grants for research. The companies include Wyeth, Astra Zeneca, Eli Lilly, Pfizer, Lundbeck, Organon and Jansen Cilag.
