Abstract
Keywords
There has been an increasing number of reports of hyponatraemia related to treatment with antidepressants since the first description with amitriptyline in 1974 [1]. This is probably due to the move towards early, aggressive treatment of depression in all age groups, and has coincided with the introduction of the selective serotonin re-uptake inhibitors (SSRIs) and subsequent generations of antidepressants.
The exact incidence of this phenomenon, however, is uncertain because most reports are of single cases and most studies of incidence have been of a retrospective nature, except for three studies [2–4]. Furthermore, hyponatraemia is commonly present in the elderly, with a reported incidence varying between 11 and 18% depending on the source of patients [5, 6]. In the case of hyponatraemia attributed to SSRIs and venlafaxine, some 11 studies have been published in which the criteria and method of compilation were clearly stated in the publication (Table 1). The lowest incidence of zero% was in a veteran's hospital where the average age of affected subjects was 51.4 years, but a low incidence (0.5%) was also recorded in an epidemiological study of patients ≥65 years [7]. In contrast, a high incidence of hyponatraemia of 40% was found in a prospective study in patients with a mean age of 75 years [2], while the highest incidence of 71% was in a group of patients treated with venlafaxine [12]. SSRIs are four times more likely to cause hyponatraemia than the older antidepressants [15], which parallels the increasing publications concerning this disorder since the introduction of the newer classes of antidepressant drugs.
Reported incidence of hyponatraemia with SSRI and venlafaxine
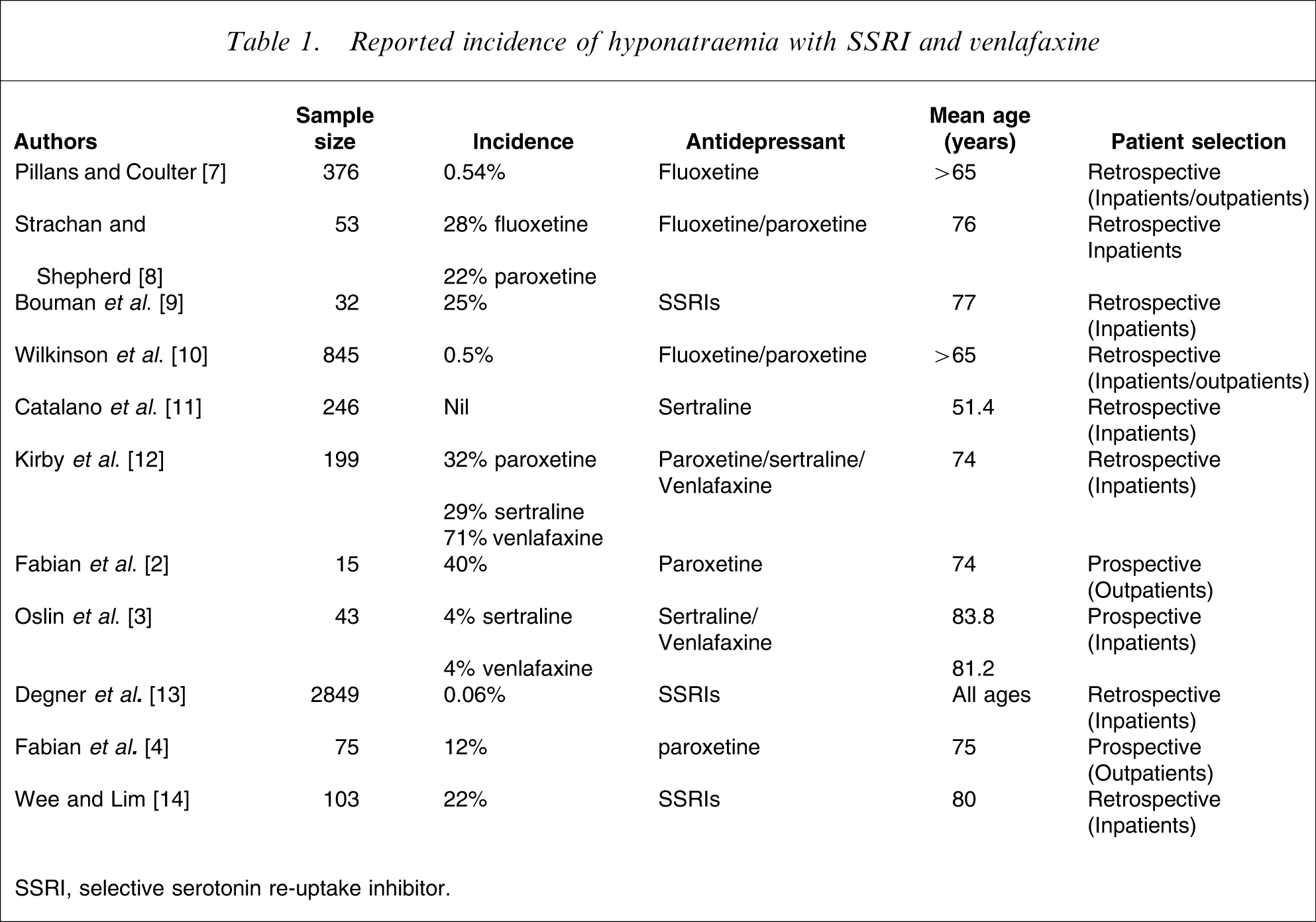
SSRI, selective serotonin re-uptake inhibitor.
The literature provides some indication of the possible risk factors for the development of hyponatraemia. Old age is a common finding in most [12, 16–19], but not all studies [20]. The elderly are often on many medications and may have impaired renal function, both of which predispose to hyponatraemia. Women are disproportionately represented [7, 17], low bodyweight [10], summer [17], and increased fluid intake [17] are other factors. Other drugs, especially diuretics, anticonvulsants and hypoglycaemic agents, have their own propensity to cause or contribute to hyponatraemia [18, 19, 21] and are commonly prescribed in the elderly.
It has generally been assumed that the mechanism of hyponatraemia secondary to antidepressants involves inappropriately high release (or non-suppression) of antidiuretic hormone (ADH, or vasopressin) from the posterior pituitary, although this hormone is frequently not measured because of the difficulty in doing so, or when measured it is reported as not being elevated. However, an adequate interpretation of the level of ADH can be made only when it is compared with the simultaneously measured serum osmolality, its secretion being normally stopped when serum osmolality falls below 280 mosmol L−1[22]. Clinical experience shows that almost all antidepressants have been implicated in the production of the syndrome of inappropriate antidiuretic hormone (SIADH) [17], including the newer mirtazapine [23], reboxetine [24], and duloxetine [25].
The usual management of hyponatraemia due to drug-induced SIADH is to stop the offending antidepressant and wait, or to institute fluid restriction as well. Rarely, patients may need i.v. hypertonic saline [21], demeclocycline, fludrocortisone and even lithium [17] to reverse the condition. However, seriously depressed patients need treatment, and the authors therefore decided to proceed with a prospective trial of subjects starting treatment with venlafaxine in which those developing hyponatraemia were continued on the drug with concurrent application of fluid restriction in a closely supervised protocol.
Methods
Setting
Patients were recruited from one author's (MR) practice; they were referred from private practice, hospital outpatients, private and teaching hospital inpatients.
Subjects
Altogether 58 patients aged ≥65 years, were recruited in a consecutive manner. They satisfied DSM-IV criteria for depression and, if there was no concomitant need for sedation, were routinely started on therapy with venlafaxine XR at a dose of 75 mg day−1 and titrated up to 150 mg day−1 as necessary to achieve an appropriate therapeutic response. In accordance with the treating psychiatrist's usual practice, patients were advised that the medication could potentially upset the body's salt balance, and that this would be monitored by blood tests taken before and after starting the antidepressant, with more intensive testing being required if this side-effect developed.
Because this was a practice-based study, patients were not excluded if they had comorbid illnesses or were taking other medications, as long as the baseline plasma sodium level was normal. Comorbidities and other medications are recorded in Table 2, 3.
Hyponatraemic patients completing study protocol
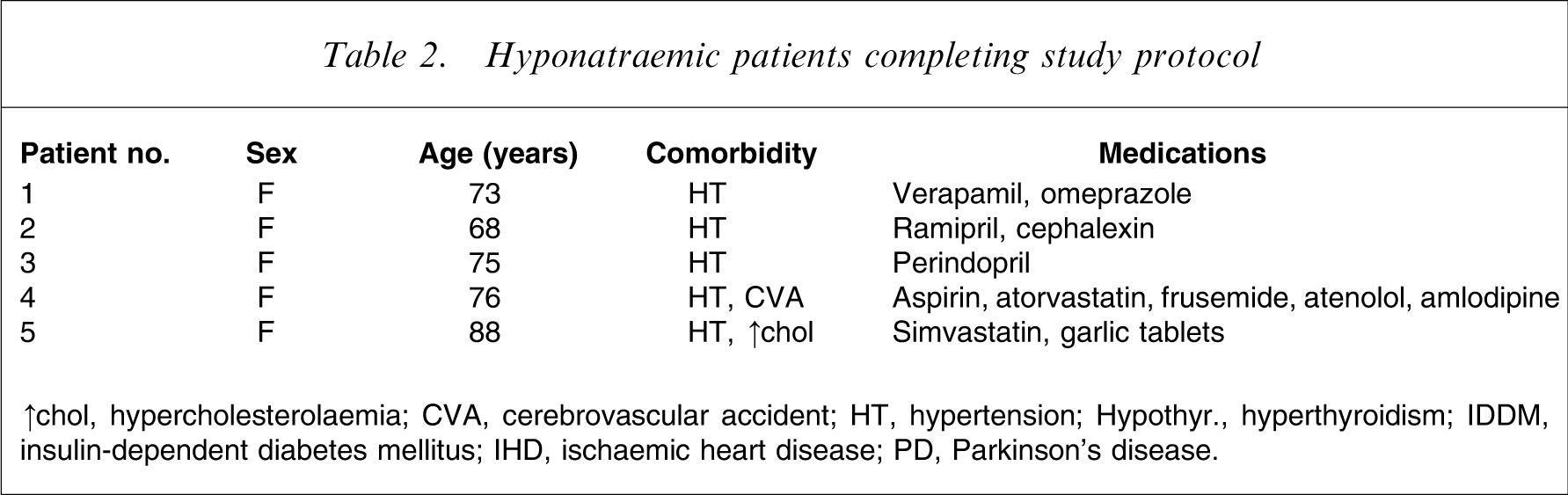
↑chol, hypercholesterolaemia; CVA, cerebrovascular accident; HT, hypertension; Hypothyr., hyperthyroidism; IDDM, insulin-dependent diabetes mellitus; IHD, ischaemic heart disease; PD, Parkinson's disease.
Assessments
Prior to starting venlafaxine, patients had baseline measurements of plasma electrolytes, urea, creatinine, uric acid and osmolality. These measurements were repeated 3–5 days after starting treatment, and if significant hyponatraemia was found (plasma sodium <130 mmol L−1), the patient was seen again within 1 week and examined clinically for volume status (blood pressure lying and standing, oedema, mucous membranes, skin turgor, jugular venous pressure, weight), and a careful neurological assessment was made. At this time, the aforementioned tests were repeated with the addition of assays of plasma ADH, thyroid-stimulating hormone, random cortisol, and a single urine specimen was obtained for urinary osmolality and spot urinary sodium concentration. The patient was advised to restrict total fluid intake to 800 mL day−1, and tests were repeated every 2 days until plasma sodium started to rise or it was deemed necessary to stop the therapy.
When plasma sodium remained at ≥130 mmol L−1 for 7 days, fluid intake was liberalized to 1200 mL day−1, and after a few days, if the sodium level continued to be within the normal range, fluid intake was unrestricted. Other clinical observations included mental state, response to treatment, and any other newly reported symptoms, and checks were regularly made on medication compliance.
Patients were followed on a monthly basis for 6 months, and tests of plasma electrolytes, urea, creatinine, osmolality, ADH, urine osmolality and spot urinary sodium were made at each visit.
Laboratory analysis
The serum sodium was measured by an ion-selective electrode using the Hitachi-Modular 1 unit (Roche Diagnostics, Diagnostics Division Switzerland, Basel, Switzerland), while the spot urinary sodium was measured with an ion-selective electrode in a Cobas-Integra 700 (Roche Diagnostics). The serum and urine osmolalities were measured by the freezing point depression method using a Micro-Osmometer (Advanced Instruments, Norwood, MA, USA).
Plasma ADH-vasopressin was measured by drawing blood into a 4 mL BD-Vacutainer K2E 7.2 mg (Becton-Dickinson, Franklin Lakes, NJ, USA) and the blood mixed by inversion several times; the tube was then centrifuged for 10 minutes at approximately 1450 g and then the plasma removed, frozen and transported on dry ice. The ADH was measured by a double antibody radioimmunoassay method (Buhlmann Laboratories vasopressin-RIA (RK-AR1) kit (Allschwil, Switzerland).
Results
A total of 58 consecutive patients ≥65 years who were started on venlafaxine were investigated and 10 patients developed hyponatraemia, giving an incidence of 17.2%. Clinical details of these patients are given in Table 2, 3. The hyponatraemia was evident in all cases within 3–5 days. Five of these patients were excluded from detailed further study: three who responded to fluid restriction could not be followed for the 6 month follow-up period because of moving out of area or changing antidepressant medication due to other side-effects, while two did not respond to fluid restriction and were severely ill with multiple medical problems (Table 3).
Hyponatraemic patients withdrawn from further study
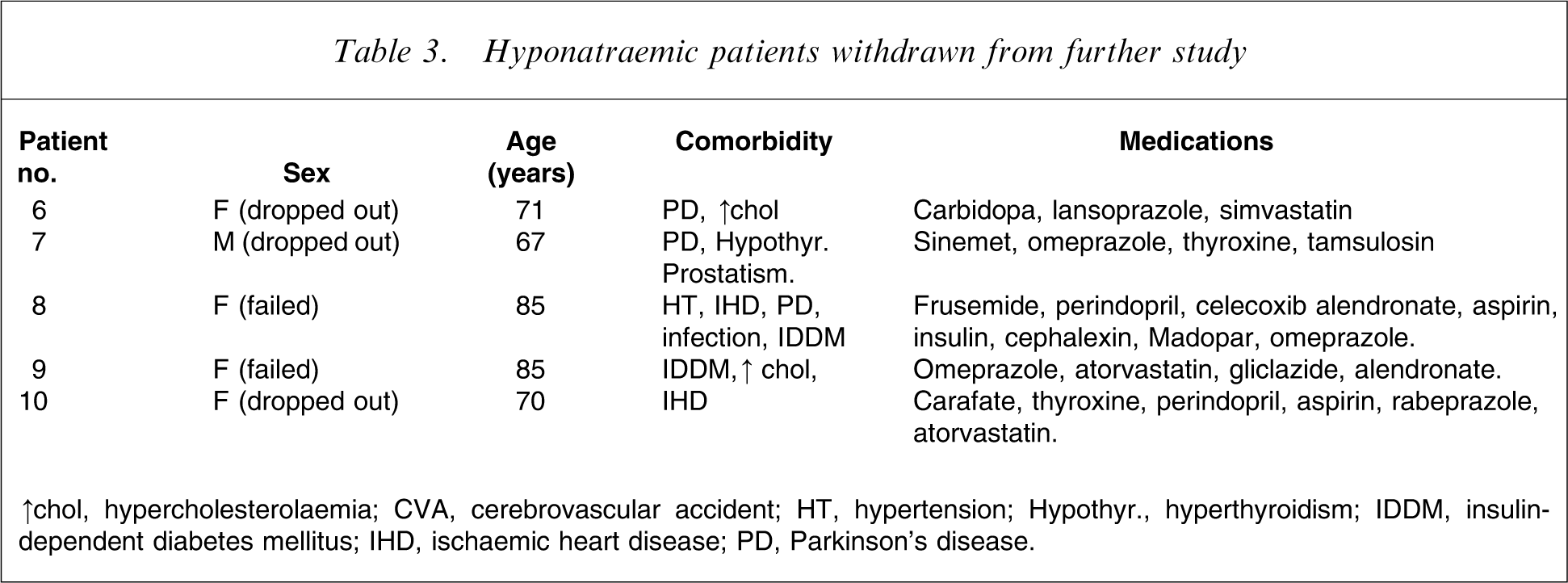
↑chol, hypercholesterolaemia; CVA, cerebrovascular accident; HT, hypertension; Hypothyr., hyperthyroidism; IDDM, insulin-dependent diabetes mellitus; IHD, ischaemic heart disease; PD, Parkinson's disease.
The results from 17 randomly selected normonatraemic patients from this study (nine male, eight female) were compared with the five remaining hyponatraemic patients using a non-parametric two-tailed Mann–Whitney U-test for the group comparisons (Table 4).
Clinical and laboratory data [mean (range)]
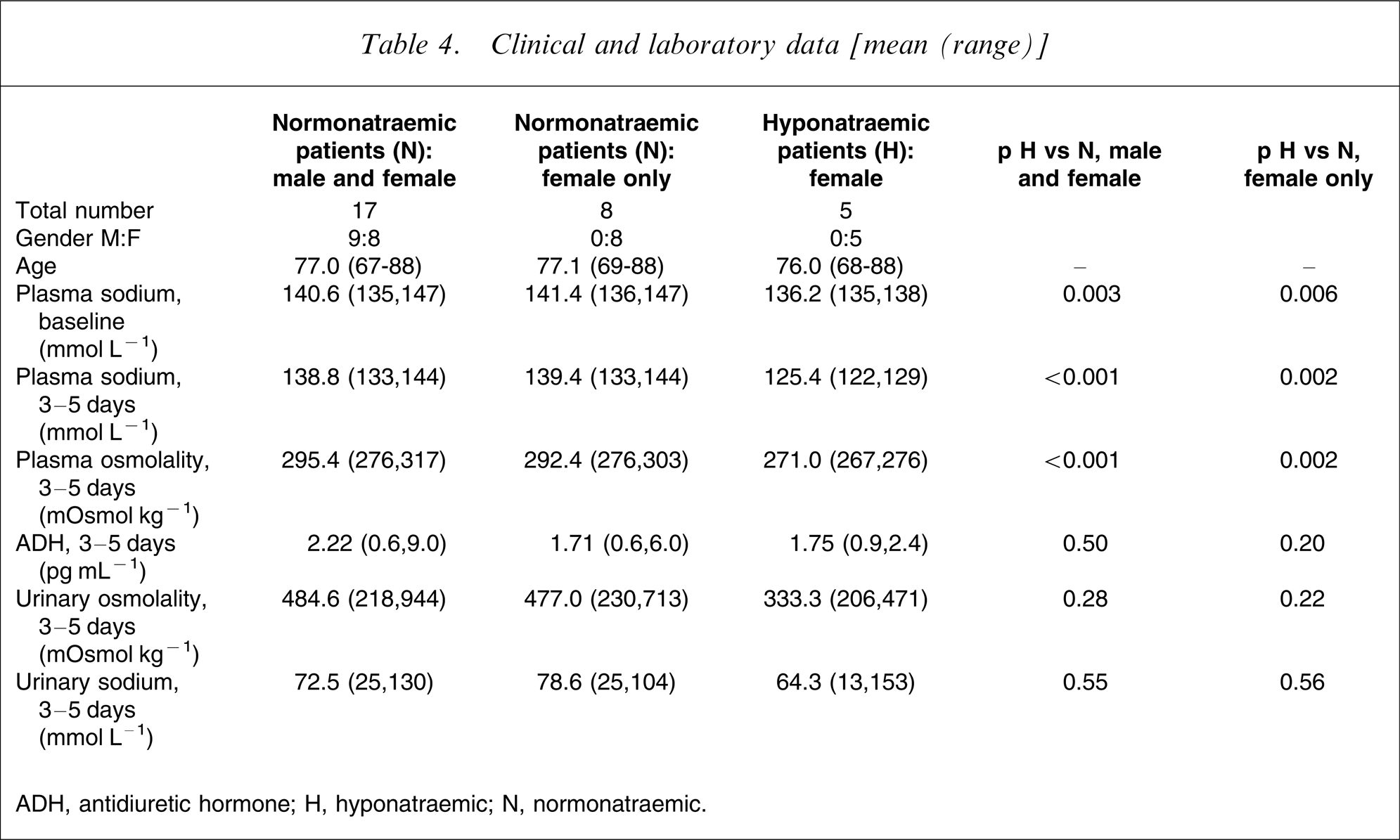
ADH, antidiuretic hormone; H, hyponatraemic; N, normonatraemic.
There was a statistically significant difference in baseline sodium level between the normonatraemic patients (both the combined male and female normonatraemic and the female-only normonatraemic groups) and the group of patients who later developed hyponatraemia, being some 4–5 mmol L−1 lower in the latter group. After 3–5 days, the plasma sodium was 125.4 mmol L−1 in the hyponatraemic group, being significantly lower by some 13–14 mmol L−1 than the two normonatraemic groups. Plasma osmolality at 3–5 days was likewise significantly lower in the hyponatraemic patients. There was no statistical difference in ADH level at 3–5 days between the hyponatraemic and the normonatraemic patients, which indicated non-suppression of ADH secretion at levels of plasma osmolality <280 mosmol L−1 (Table 4).
Figure 1 shows the time course of the plasma sodium concentration (mean values and SD) before and after fluid restriction during the 6 month follow up of the five patients who developed hyponatraemia and continued on venlafaxine throughout the full length of the study.
Time course of plasma sodium concentration in hyponatraemic patients maintained on venlafaxine therapy.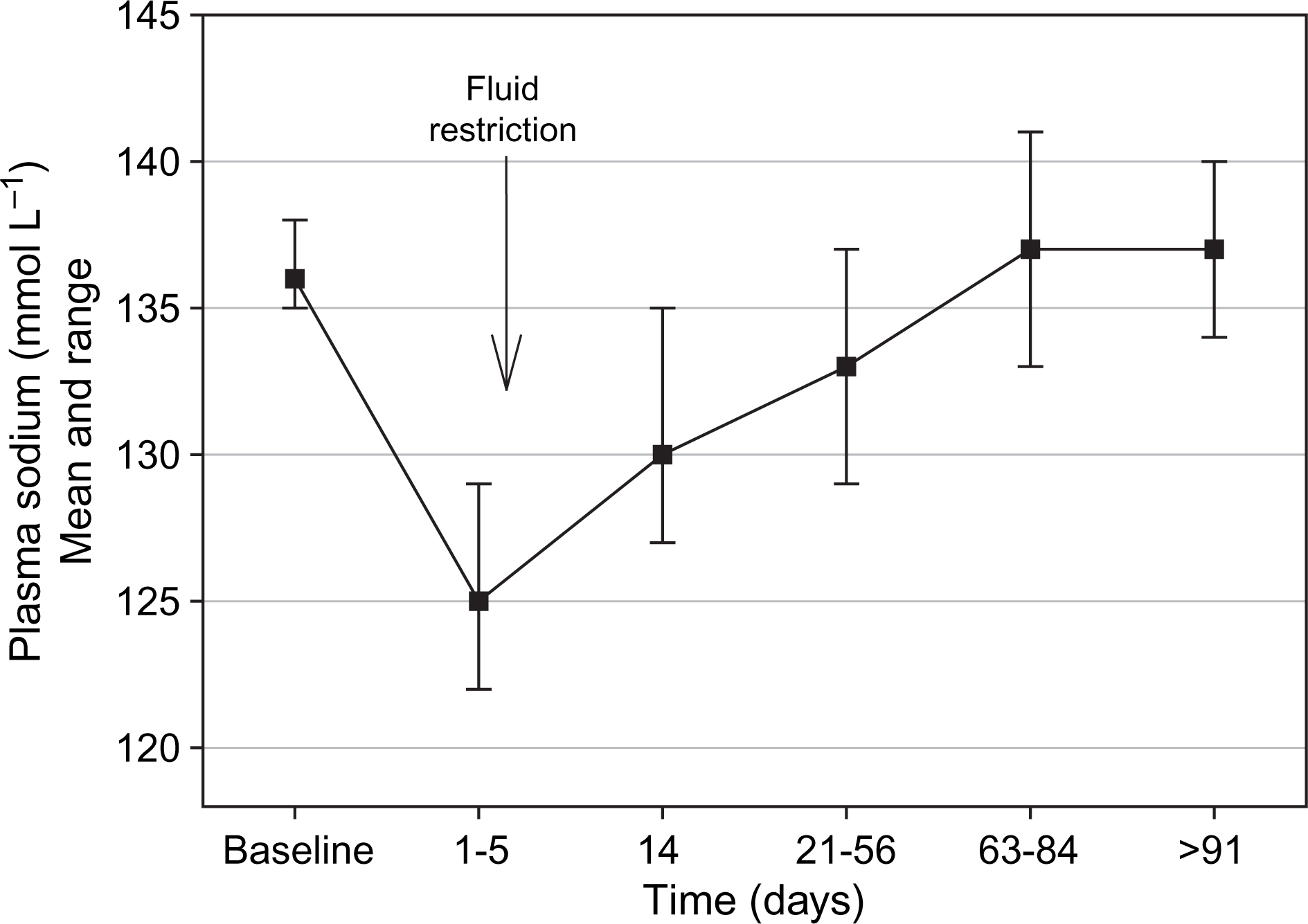
Figure 2 shows the results of plasma ADH assays in individual patients, plotted against their simultaneous plasma osmolality. It is clear that in patients with drug-induced hyponatraemia (black squares) ADH levels remain inappropriately raised in relation to the low plasma osmolality associated with their hyponatraemic state.
Plasma antidiuretic hormone (ADH) concentration against plasma osmolality for the different patient groups. All measurements taken 3-5 days after commencement of venlafaxine. The line shows the upper limit of the 95% confidence interval for the normal range.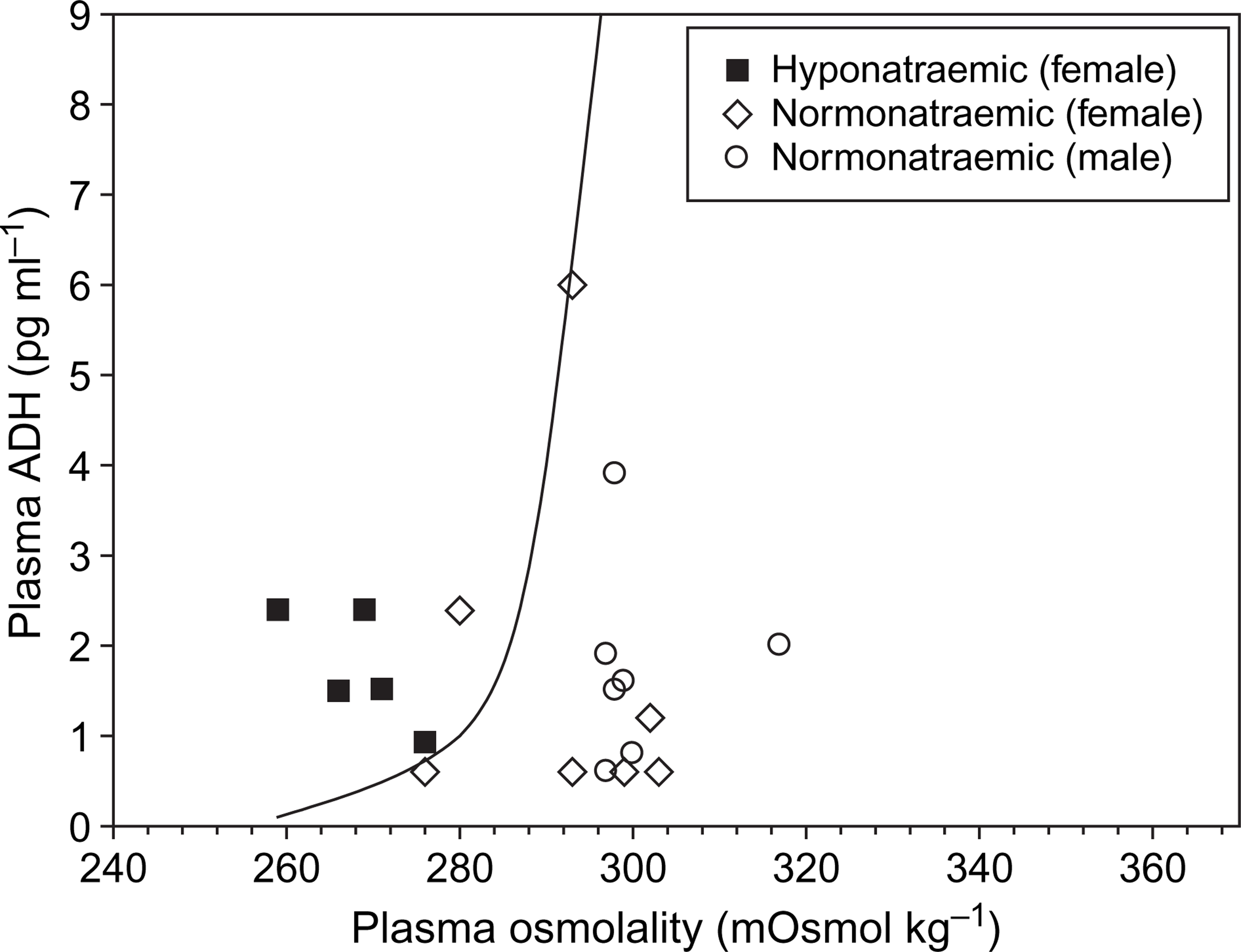
Discussion
This is, to our knowledge, the first prospective study to examine the incidence and mechanism of hyponatraemia in a sample of patients >65 years of age treated with venlafaxine. The 17.2% incidence of this complication found in the present study is much lower than the 71% found in a retrospective series [12] and is closer to that of Fabian et al., who found that 12% of patients on paroxetine became hyponatraemic [4]. However, patients in the Fabian et al. study were instructed to restrict their fluids on commencement of treatment, and hence their incidence may be an underestimate. One reason for the present relatively low incidence may be the wider background of the present patients, because many came from the community as well as from tertiary referral centres. The mean age of the present patients was 76 years, and nine out of 10 were female, in agreement with most other studies [7, 8, 12, 16, 17, 19]. The finding in the present study that patients who were to go on to develop hyponatraemia after starting venlafaxine treatment had a significantly lower plasma sodium at baseline was also observed by Fabian et al.[4], and may suggest that such patients may be predisposed to this complication, possibly because of a higher habitual water intake or other factors.
Following the pre-treatment baseline assessment, the plasma sodium level was measured again 3–5 days after starting venlafaxine, and the drop in plasma sodium was evident by that time in all patients who were to develop hyponatraemia during the course of treatment. Venlafaxine can induce hyponatraemia within 2–21 days of starting the antidepressant [26, 27], and this may be sooner than with SSRIs, whose range in the literature varies between 5 and 30 days [7, 28]. The prospective study by Fabian et al. involving paroxetine found a mean time of 9.3 days for SIADH to develop [4]. The review of Spigset and Hedenmalm found that 51.3% of patients who develop hyponatraemia on various antidepressants will do so within 2 weeks [17], and a similar period was found in another epidemiologic study of Movig et al. [15]. Reports of late diagnosis of hyponatraemia come from retrospective series or single case studies, suggesting that measurements were not made regularly after starting the antidepressant to detect this earlier [9, 29–32].
Strachan and Shepherd first described the normalization of plasma sodium in spite of continuing SSRI therapy in nine out 13 patients [8]. Bouman et al. described one patient who was able to continue treatment with an SSRI after starting fluid restriction while another of their patients failed this approach [9]. Fabian et al. pointed out the transient nature of hyponatraemia when three of six patients spontaneously reverted to normal in spite of continuing on paroxetine [2]. In the present study, eight of the 10 hyponatraemic patients responded in the short term to fluid restriction of 800 mL day−1, although only five were followed up over 6 months. Two of the present patients failed the treatment but they were seriously ill with multi-organ failure. As shown in Figure 1, hyponatraemia in the five patients followed over 6 months had a generally benign course. Their plasma sodium returned on average to >130 mmol L−1 within 14 days on fluid restriction while continuing on venlafaxine, and fluids were then progressively liberalized. The treatment was accepted by both hospitalized patients and outpatients.
The current study provides some evidence that the mechanism of hyponatraemia in the present patients involves non-osmotic stimulation of ADH release. ADH release is physiologically controlled by peripheral and central osmoreceptors, which trigger addition of ADH to the circulation when plasma osmolality rises above the normal range of 290–300 mosmol kg−1. ADH acts on receptors in the cortical and medullary collecting ducts in the kidney, causing activation of water channels (aquaporins), leading to enhanced water reabsorption and hence restoration of normal plasma osmolality. Conversely, when plasma osmolality falls below 285 mosmol kg−1, ADH release is suppressed and plasma ADH falls to undetectable levels [33]. Euvolaemic hyponatraemia is the result of unsuppressed release of ADH in the presence of a low plasma osmolality (SIADH). A key finding of the present study is that ADH release is not inhibited during development of hyponatraemia in patients taking venlafaxine (extended release formulation) at a dose of 75–150 mg day−1. Venlafaxine acts primarily as an SSRI but also has some inhibitory effect on noradrenaline re-uptake [34]. These actions may be involved in the inappropriate secretion of ADH triggered by the drug, because stimulation of central serotonin receptors (5HT1C and 5HT2) and α-1-adrenergic receptors can release ADH in experimental models [35–37].
Adaptation of the osmoregulatory feedback mechanism must occur in some patients with time because hyponatraemia corrects spontaneously in some [2, 8], and by fluid restriction in others (Figure 1) while continuing on various antidepressants (fluoxetine, paroxetine, venlafaxine). The present data further show that once the plasma sodium corrected itself, this was maintained for the duration of the 6 month observation period. Elderly people have a slightly increased ADH secretion [38] but the underlying mechanism of this change and its relevance to adaptation during varying water intake is currently unknown. It may be that differences in spontaneous fluid intake between subjects taking venlafaxine are important determinants of susceptibility to hyponatraemia. The experiments of Marar and Amico show that in rats, both vasopressin excess and liberal fluids are required to produce hyponatraemia [39], and it could be that hyponatraemic patients drank more than the others, but we are unable to comment on this because we did not attempt to measure the amount of fluid drunk by the present patients. Stress as part of illness may be another factor because interleukin-6, which is released under such conditions, has been shown to stimulate ADH secretion [40].
The interpretation of the present study must be qualified by the fact that the observations were made on small numbers of patients, some having comorbidities and taking other medications that could have contributed to or interacted with venlafaxine to produce hyponatraemia. Also, we did not measure venlafaxine levels, which may be a variable influencing the release of ADH. However, we do not consider that these reservations detract substantively from the main conclusions of the study.
From the findings of the present study on venlafaxine, we support the recommendation of other authors (4,8,21], that elderly patients should have their electrolytes measured soon (within 3–5 days) after starting an antidepressant drug, to detect the development of hyponatraemia. Once hyponatraemia is identified, modest fluid restriction should be commenced and further monitoring performed while drug treatment is continued. Most patients will be able to continue safely on the drug with return of plasma sodium to the normal range, and in the longer term fluid intake can generally be liberalized.
Footnotes
Acknowledgements
The authors wish to sincerely thank Dr Margaret Janu for advice with the biochemical testing, and Associate Professor Jenny Peat whose willing and enthusiastic statistical help and graph drawing made writing this article easier, and Mrs Jen Moore for graphic and typing assistance. MR received remuneration from Eli Lilly and Wyeth for speaking at educational meetings, and grants for conference attendance.
