Abstract
The purpose of the present review is to describe the neuropsychological correlates of long-term substance abuse and to discuss the findings within the context of premorbid vulnerabilities, comorbidity and adolescent neurodevelopment. The authors critically review key findings from the neuropsychological literature related to the long-term sequelae of alcohol, cannabis, inhalant, opiates, psychostimulants and ecstasy use. Leading electronic databases such as PubMed were searched to identify relevant studies published in the past 20 years. References identified from bibliographies of pertinent articles and books in the field were also collected and selectively reviewed. Across substances, individuals with long-term abuse consistently demonstrate neuropsychological impairments of executive (inhibitory) control, working memory and decision making, together with neurobiological abnormalities involving frontotemporal and basal ganglia circuits. In some instances these deficits are dose dependent, implying that they are a direct consequence of prolonged drug exposure. However, comorbid behavioural, personality and mental health problems are common among drug-using populations and are associated with similar neuropsychological deficits. Presented herein is a neuropsychological model of addictive behaviour that highlights the complex interplay between cognition, brain maturation, psychopathology and drug exposure.
‘Addiction’, derived from the Latin verb addicere (meaning ‘to enslave’), is characterized by an apparent loss of control or autonomy over one's behaviour. Indeed, the continued abuse of substances by addicted individuals, despite an apparent awareness of associated adverse consequences, suggests that addictive behaviour may involve deficits in inhibitory control, decision-making and the regulation of affect [1–7]. In line with this, recent neuroimaging studies across a variety of substance-using populations have implicated impairments in frontal cortical networks [6–10]. However, an question is whether these frontally mediated executive control deficits relate to the use of specific substances or are common across the major classes of drugs.
In the present paper we critically review the neuropsychological literature related to the long-term sequelae of alcohol, cannabis, inhalants, opiates, psychostimulants and ecstasy in humans. Leading electronic databases such as PubMed were searched to identify relevant studies written in English and published in the past 20 years. References identified from bibliographies of pertinent articles and books in the field were also collected and reviewed. Studies examining the acute effects of these substances were not included in this review because the focus is on the long-term sequelae. It was not our intention to comprehensively review all studies identified in our searches, but to selectively focus on key exemplary research to highlight methodological issues and determine the primary and most consistent findings across this broad literature. We discuss these findings in the context of premorbid neuropsychological vulnerabilities and comorbid personality traits and disorders (e.g. medical, neurological or psychiatric). We also discuss the neural and functional changes that occur in the adolescent brain and their relevance to the neuropsychological outcomes observed.
Neuropsychological sequelae of specific drugs
Alcohol
The most consistent findings of neuropsychological impairment in heavy and long-term drinkers are in the domains of attention, short-term memory, visuospatial abilities, postural stability and executive functions (such as problem-solving, mental flexibility, judgement, working memory, response inhibition and decision-making), with a relative sparing of declarative memory, language skills and primary motor and perceptual abilities [11–14]. The link between lifetime exposure and the development of cognitive problems is unclear. While some research findings suggest that cognitive performance worsens in direct proportion to the frequency and duration of drinking [15, 16], other studies suggest that cognitive deficits may be detectable only in those who have been drinking regularly for at least 10 years [17, 18]. Some researchers even suggest that neuropsychological impairments are not related to alcohol at all, but rather are a direct consequence of head trauma associated with drinking [19]. These contrasting findings highlight the need for further research to determine how patterns of alcohol use are related to cognitive impairment, especially in light of some evidence to suggest that long-term, light-to-moderate social drinkers may also have cognitive deficits [15].
The nature of the neuropsychological deficits observed in long-term drinkers is consistent with disruption to frontotemporal, frontoparietal and cerebellar brain systems. Indeed, structural magnetic resonance imaging (MRI) indicates a consistent association between heavy drinking and structural neuronal injury and volume loss that is more extensive in the frontal lobe, temporal lobe, and cerebellum [20, 21]. Results of autopsy studies show that individuals with a history of chronic alcohol consumption have smaller, lighter and more shrunken brains than non-alcoholic adults of the same age and gender [22, 23].
Perhaps the strongest evidence for a direct relationship between chronic substance abuse and neuropsychological/neurobiological impairments comes from observations of a dose–response relationship and longitudinal studies demonstrating progressive changes in such measures with ongoing abstinence or continued use. To this end, some alcohol-related cognitive impairment and structural brain deficits can be reversed with abstinence over a period of several months to years [14]. Abstinence-associated improvements have been documented in neuropsychological functions such as working memory, visuospatial functioning, and attention, accompanied by significant increases in brain volume, compared with alcoholics who subsequently relapsed [24–26].
Cannabis
Despite inconsistent findings from early studies and a small meta-analysis [27], recent well-controlled neuropsychological studies of chronic cannabis users in the non-intoxicated state have now demonstrated impaired performance on a variety of attention, memory, and executive function tasks [28–33]. Deficits have variously been attributed to duration of cannabis use [33], frequency of cannabis use [31] or cumulative dosage effects [32]. Performance on executive tests, such as the Stroop task [34], is not consistently impaired in cannabis users, but performance decrements have nevertheless been shown to be related to duration of use [33], or dose interacting with lower IQ [32]. Few studies have sought to specifically tease out differential impairments associated with varying patterns of cannabis use (e.g. heavy daily use for short periods vs light weekly use for long periods). Solowij showed that the ability to focus attention and filter out irrelevant information was progressively impaired with increasing years of cannabis use, while speed of information processing was impaired with increasing frequency of use (days per month) [35].
Recent neuroimaging studies of cannabis users demonstrate impaired performance in attention, verbal memory [36], working memory [37], response inhibition [34, 38, 39] and decision-making tasks [40], with concomitant alterations in blood flow, activation or brain tissue density primarily in prefrontal cortical, anterior cingulate, basal ganglia, cerebellar and hippocampal regions. For example, Matochik et al. found grey and white matter density changes in 28 day abstinent cannabis users in the medial temporal regions, and some of the density changes were associated with duration of cannabis use [41]. Our own study used high-resolution MRI to examine a well-characterized sample of long-term (>10 years of regular use) and very heavy cannabis users (5–7 joints per day over many years). We found robust reductions of 12.0% in the hippocampus and 7.1% in the amygdala of users relative to well-matched healthy control participants. Additionally, left hippocampal volume was negatively associated with cumulative dose of exposure to cannabis. While some studies found no structural brain alterations in cannabis users [36, 42], our findings corroborate the animal literature in human subjects suggesting that long-term heavy cannabis use is associated with significant and localized medial temporal reductions that are related to cumulative cannabis dosage [Yucel M, Solowij N, Respondek C et al: unpublished data, 2007]. Moreover, altered frontal cortical activation is apparent in cannabis users despite normal Stroop task performance [34, 39], suggesting that there may be disturbances in brain physiology that are not as yet apparent behaviourally.
The extent of persistence of effects or recovery of function following abstinence is also uncertain, with some studies suggesting no recovery after 25–28 days abstinence [32, 34, 40], while others report full recovery after 28 days abstinence [31] and partial early recovery after 2 years abstinence [35].
Inhalants
While the toxic effects of chronic inhalant abuse are well described [43], there is only a small research literature examining the neurobiological and neuropsychological effects of voluntary inhalant exposure [44, 45]. One early study demonstrated that inhalant abusers (mainly those abusing metallic paints) had deficits in motor coordination, learning, memory, executive functioning and overall verbal intelligence [46]. One of the largest investigations examining the effects of inhalant use on neuropsychological functioning in adolescents was conducted by Chadwick et al. [47]. Those who had used volatile substances performed significantly worse on tests of vocabulary and impulsivity, and had significantly lower verbal and full-scale IQ. However, these differences were no longer significant when background social disadvantage was taken into account, although it is important to note that the inhalant group consisted of primarily experimental or recreational users. Other studies suggest that solvent-abusing groups tend to perform worse on a range of neuropsychological tasks, especially those involving memory and executive functioning [48]. However, these individuals typically lack the motivation to perform well on such tasks and invariably present with complex problems, such as psychosocial disadvantages (e.g. unstable and dysfunctional families, state-based care, school absenteeism, trauma and abuse, forensic issues), and histories of comorbid drug use and mental health problems – issues that play a major role in neuropsychological outcomes [48].
From a neurobiological perspective there is more conclusive evidence that inhalant exposure is associated with adverse consequences. A recent study comparing 55 inhalant abusers (mean age 30 years) and 61 cocaine abusers (mean age 29 years) found substantial brain abnormalities (especially in subcortical and white matter regions) and cognitive impairment within both groups [45]. However, the structural brain abnormalities were more common (44% vs 25%) and more extensive in the inhalant-using group. In addition, the inhalant group performed significantly worse on tests of working memory and tests requiring focused attention, planning, and problem solving. Similar findings have been observed in other neurobiological and neuroimaging studies of chronic solvent exposure in occupational settings [49, 50].
Very few studies have specifically investigated recovery of function after prolonged exposure to inhalants. Cairney et al. found significant improvements in previously identified neurobehavioural impairments following 2 years abstinence from petrol sniffing [51, 52]. In fact, in many cases these deficits normalized completely. However, although those with the greatest levels of impairment showed the greatest degree of improvement with abstinence, they were less likely to recover completely. A handful of studies in solvent-abusing individuals have also reassessed their cases with further neuropsychological and/or neuroimaging investigations and found either greater impairments with continued solvent use [53, 54] or significant improvements with long-term abstinence [55]. Other studies have found associations between the parameters of solvent use (e.g. duration and extent) and MRI abnormalities [56, 57]. While these studies support the notion that solvent abuse directly leads to adverse outcomes, and that recovery of function is possible through abstinence, one study failed to find any significant improvement in MRI or neuropsychological measures following 5 months of abstinence [58]. Worryingly, another study found no improvements in MRI measures of brain pathology following an extended period (18 months) of abstinence [59].
Opiates
Research on the long-term neuropsychological effects of chronic opiate abuse has been relatively limited. Davis et al. reported that 60% of individuals currently abusing opiates had impairments of at least two standard deviations below published norms on two or more neuropsychological tests, a significantly higher incidence than found in matched controls with no history of drug abuse [60]. In particular, deficits were identified in impulse control in those with a history of ≥5 years of heroin use. Similarly, Pau et al. examined the impact of heroin on frontal executive functioning in three cognitive domains, namely attention, impulse control and mental flexibility [61]. They found that heroin abuse has adverse effects on impulse control but not attention or mental flexibility. Other studies have reported more diffuse deficits across the domains of attention, working memory, memory and executive function in chronic opiate abusers [61–65]. Ornstein et al. found that heroin addicts were impaired on performance of cognitive tasks (e.g. learning, spatial working memory, strategic thinking) known to be sensitive to cortical damage (including selective lesions of the temporal and frontal lobes) [63]. Darke et al. reported that methadone-maintained heroin addicts performed more poorly on all neuropsychological domains tested, including attention (information processing speed, attentional capacity), memory (visual and verbal learning and memory) and executive functioning (problem solving) compared with matched controls [66]. However, Darke et al. noted that the methadone group had high rates of polysubstance use, overdose, head injury and comorbid psychopathology [66]. They found that the neuropsychological deficits identified were more characteristic of those with associated comorbidities, further raising issues regarding the specificity of findings reported.
Chronic opiate users have been shown to have disturbances in prefrontal cortical activity when performing a reward-based decision task [67]. Moreover, this disturbance was observed in a group of drug users who had been abstinent for at least 1 year. Further support for prefrontal dysfunction comes from the large structural imaging study of Lyoo et al., which investigated grey matter density in 63 opiate-dependent subjects and 46 matched controls [68]. They found that relative to controls, the opiate-dependent group exhibited significantly decreased grey-matter in the prefrontal, as well as superior temporal cortex, insula and fusiform gyrus. Another study by Kivisaari et al. in long-term opiate users found that the sylvan fissures and ventricles were wider in opiate-dependent subjects than in controls, which may be related to brain atrophy within frontal and temporal lobes [69].
Our own multimodal neuroimaging study of long-term opiate users also found abnormalities in the neural substrates of inhibitory control [9, 10]. Specifically, opiate addicts (i) exhibited significantly reduced concentrations of the neuronal marker N-acetylaspartate (NAA) and glutamate/glutamine within the dorsal anterior cingulate cortex (d-ACC); (ii) failed to show the normal association between d-ACC activity and behavioural performance demonstrated by healthy controls; and (iii) had relatively increased task-related activation of frontoparietal and cerebellar regions. These findings suggest that neuronal abnormalities and a breakdown of normal brain–behaviour relationships within the d-ACC of chronic opiate users may result in the recruitment of a compensatory and inadequate network of brain regions when required to exercise inhibitory control.
Studies evaluating the persistence of cognitive deficits among abstinent opiate addicts remain mixed. A number of studies have found that abstinent groups of recovering addicts have no significant cognitive deficits [60, 62]. Guerra et al. reported that individuals with current heroin abuse demonstrated deficits in attention, working memory, episodic memory and verbal fluency, which normalized 7–14 days following rapid detoxification [62]. However, two other studies of abstinent heroin users (8 and 14 months, respectively) reported ongoing deficits in executive function [61, 65]. Using a more sophisticated battery of experimental neuropsychological tests, Ersche et al. reported that opiate-dependent individuals demonstrated marked impairments in spatial planning, paired associate learning and visual pattern recognition compared with matched controls [70]. Performance of former opiate users (abstinent for 8 years on average) was not statistically different from current users on any measure, suggesting that the deficits observed did not simply reflect the current effects of drug use. These findings are in keeping with other studies describing prefrontal dysfunction in chronic drug users [1, 3, 5–7].
Psychostimulants (cocaine, amphetamine/methamphetamine)
Few studies have comprehensively examined cognitive functioning among methamphetamine users. Some researchers point to studies that suggest memory and executive problems, while others maintain that no firm evidence for a link exists [71]. Recent studies of chronic amphetamine/methamphetamine abusers have shown that they perform poorly on decision-making tasks that involve regions of the frontal cortex (specifically the ventromedial prefrontal cortex), such that they make disadvantageous decisions that reflect valuing short-term gain over longer term losses [64, 72]. Methamphetamine users also appear to be more distractible and are unable to suppress processing task-irrelevant information [72], which is consistent with their clinical presentation. Other work has also shown cognitive deficits related to processing speed, learning, delayed recall and inhibitory control and working memory [73–75]. Another recent study found methamphetamine abuse to be associated with deficient strategic (i.e. executive) control of verbal encoding and retrieval, which is consistent with proposed prefrontostriatal circuit neurotoxicity [76]. Interestingly, comorbid cannabis does not appear to exacerbate methamphetamine neurotoxicity, but rather has been suggested to have neuroprotective effects [75].
Neuropsychological studies of chronic cocaine users, as in chronic amphetamine users, also demonstrate higher order cognitive impairments (e.g. inhibitory dysregulation) that is consistent with abnormal blood flow in frontal brain regions [77]. Several studies have reported that cocaine abuse is associated with decrements on neurobehavioural tests measuring executive control, visuoperception, psychomotor speed, manual dexterity, verbal learning, and memory [78, 79]. Ardila et al. found that neuropsychological test scores were correlated with lifetime amount of cocaine used, suggesting a direct relationship between cocaine abuse and cognitive impairment [80].
Neuroimaging studies of methamphetamine users have shown abnormalities of brain structure and function relative to healthy controls including alterations of frontal, temporal and subcortical metabolism [81–86]. Changes in neuronal biochemistry suggestive of neuronal injury have also been found in the frontal cortex and basal ganglia structures [87]. Using proton magnetic resonance spectroscopy (1H-MRS), Ernst et al. reported abnormally low levels of the neuronal marker NAA in the basal ganglia of abstinent methamphetamine-dependent subjects [87]. They also observed an inverse association between prefrontal white-matter NAA values and years of use, implying direct effects of this drug on the neuronal integrity of prefrontal tissue. Similar findings have been reported in the anterior cingulate cortex [88, 89]. Recent neuroimaging studies have shown that frontal and temporal lobe white matter continues to increase into the fifth decade of life [90]. However, Bartzokis et al. found that cocaine-dependent subjects (aged 19–47) do not demonstrate the normal pattern of age-related increases in white matter within these brain regions [90], suggesting that continued cocaine use may arrest normal white matter maturation.
Examination of cognitive function in abstinent cocaine-dependent individuals after both 6 weeks and 6 months abstinence shows persistent cognitive impairment across a wide range of functions compared to controls at both time-points. Further, a close relationship between the degree of neuropsychological impairments and dosage (i.e. quantity and dosage of peak usage) has also been reported [91]. Consistent with these neuropsychological findings, Fein et al. also found that cocaine-induced brain volumetric reduction in the prefrontal cortex persisted after 6 weeks abstinence [92].
3, 4 methylendioxymethamphetamine (MDMA, ecstasy)
A number of persisting cognitive problems have been attributed to regular N-methyl-3,4-methylenedioxy-amphetamine (MDMA) use and suggest underlying serotonergic dysfunction [93–95]. For example, impairments in memory (both visual and verbal) have been shown to correlate with in vivo measurements of brain serotonin function and levels of 5-hydroxyindoleacetic acid (5-HIAA) in cerebrospinal fluid, as well as relating to the level of previous MDMA use (i.e. are dose related) [96]. Other neuropsychological deficits that have been reported in regular ecstasy users include impairments of executive function and self-control (i.e. decreased inhibitory control and increased impulsivity) [97, 98]. McCardle et al. found that MDMA users exhibit difficulties in coding information into long-term memory, have impaired verbal learning, are more easily distracted, and are less efficient at focussing attention on complex tasks [97]. Interestingly, Spatt et al. have described a case of profound amnesia, associated with bilateral brain changes on MRI, following a single exposure to MDMA [99].
Although cognitive deficits among MDMA users have been well documented, little is known of the neurobiological sequelae of MDMA use. In one study, MDMA use in adolescence was associated with difficulties in the ability to focus and divide attention, as well as abnormal hippocampal activity during performance of a working memory task [100]. Other functional MRI studies have found abnormal frontotemporal, parietal and subcortical activity during performance of working memory tasks [101–104]. Additionally, Daumann et al. found that relative to a group of controls and currently abstinent but previously moderate users, the currently abstinent but previously heavy users showed more prominent frontal and temporal lobe activation abnormalities during performance of a working memory task [104]. Structural MRI findings among MDMA polydrug users includes evidence of diffuse grey matter reductions across the cortex, cerebellum and brainstem structures [105].
Little is known about the possible persistent neuropsychological effects of extensive MDMA use. However, there is tentative evidence that these cognitive deficits persist for at least 6 months after abstinence, whereas anxiety and hostility remit after 1 year of abstinence [93, 106]. Morgan et al. compared four groups of participants: current regular recreational MDMA users; ex-regular MDMA users who had abstained from using the drug for an average of 2 years; polydrug users who had never taken MDMA; and drug-naive controls [106]. They found that both current and ex-MDMA users exhibited elevated psychopathology and behavioural impulsivity compared with polydrug users and drug-naive controls, but current MDMA users exhibited a broader range of psychopathology than ex-users. Both groups of MDMA users also exhibited impaired working memory and verbal recall performance compared with drug-naive controls. These findings suggest that selective impairments of neuropsychological performance associated with regular MDMA use are not reversed by prolonged abstinence. This is consistent with evidence that MDMA may affect brain serotonergic systems in human users. Other studies have also found altered neural activations suggestive of prefrontal neuronal injury in abstinent MDMA users during performance of working memory tasks [102].
Summary
This selective review of the available literature suggests that chronic abuse of a wide range of addictive substances can adversely affect neuropsychological functioning. However, it is also clear that there is marked inter-individual variability in the patterns of substance use (e.g. duration, frequency, dosage, type) and, with the exception of a few studies, most researchers were not able to definitively isolate the effects of a specific drug due to a history of polysubstance use. An under-investigated issue is whether concurrent use of different substances (e.g. cannabis and alcohol) may potentiate the long-term adverse effects of each drug. While synergistic or additive effects are plausible, there is also speculation that use of some substances may mask or protect against the neurocognitive sequelae of other substances. For example, it has been hypothesized that the neuroprotective properties of cannabinoids may fortify against neurotoxic effects of MDMA or amphetamines in general to preserve cognition in Ecstasy users [93] or amphetamine users [75]. Working memory-related neural activation in cannabis users was found to be deficient only during nicotine withdrawal (but not in tobacco users during nicotine withdrawal), suggesting that nicotine may mask neuropsychological deficits associated with cannabis [107]. Nonetheless, there is consistent evidence that almost all substances affect the domains of attention, learning and memory, visuospatial abilities and executive functioning. However, the more robust findings relate to impairment in inhibitory control (variously referred to as response inhibition, inhibitory regulation, self-control or impulsivity), working memory and decision-making. Accordingly, the neurobiological findings from structural, functional and spectroscopic MRI studies suggest dysfunction in neural systems that subserve these functions, particularly frontotemporal and basal ganglia circuitry.
Nevertheless, the pathways into addiction are invariably complex, making it difficult to disentangle the neuropsychological effects of substance exposure from associated risk factors. While in some instances, reported deficits have been found to be dose dependent (implying direct effects of drug exposure), the fact that most studies are cross-sectional in nature means that it is not possible to categorically determine whether the identified deficits are a consequence of the drugs specifically, relate to pre-existing vulnerabilities, or are a combination of both. Specifically, it is not clear how broader individual characteristics (e.g. developmental stage, psychopathology, genetic polymorphisms) may affect neuropsychological functioning and contribute to the observed deficits.
Executive control deficits: broader individual considerations
Young people who have behavioural problems early (e.g. a difficult temperament in infancy or childhood oppositional, aggressive or impulsive behaviours) are at increased risk of developing substance use disorders [108]. Indeed, childhood diagnoses of oppositional defiant disorder, conduct disorder and attention deficit hyperactivity disorder are well-established risk factors [109–112]. In addition, a number of mental health disorders are associated with high rates of comorbid substance use, such as depression, anxiety, schizophrenia, bipolar disorder and obsessive–compulsive disorder [113–116]. Given that deficits in inhibitory control and affect regulation are found across many of these disorders, together with disruption to brain regions subserving these functions (e.g. prefrontal and temporal areas), it is possible that they form a key component of liability to not only behavioural and mental health disorders, but also to addictive behaviour.
Certain personality characteristics may also influence an individual's decision to use drugs, as well as their liability to develop problematic use. Indeed, inhibitory-control and affect-regulation difficulties may also be components of a premorbid personality style rather than the result of state-related cognitive–affective processes. To this end, there is a growing literature on temperament and personality as risk factors for substance use disorders and addiction. These studies highlight a relationship between measures of impulsivity and related constructs (such as risk-taking, sensation-seeking) in childhood and the development of later substance use disorders in adulthood [117]. In fact, studies of both adolescents and adults consistently report an association between impulsivity (e.g. acting in a sudden and unplanned manner, acting without having all the necessary information or failing to think through the pros and cons of a decision) and substance-related problems [118–120]. Other personality traits such as negative affectivity/neuroticism have also been identified as risk factors for substance use disorders. Negative affect/neuroticism is characterized by a general tendency to experience life as more negative and a difficulty in controlling one's mood (i.e. difficulties with affect regulation) and/or being less tolerant to stressful life events. There is growing evidence to suggest that adolescent substance use disorders may not only result in disinhibited behaviour or impulsivity (reviewed here), but may in fact reflect an attempt to reduce negative affectivity [121, 122]. From a neurobiological and neuropsychological perspective, this inability to internally regulate moods and the over-reliance upon external agents, such as psychoactive substances, suggests inappropriate neural (particularly prefrontal) and cognitive modulation of emotions [123].
Another issue is that of premorbid neuropsychological vulnerabilities. To this end, Tarter et al. and Kirisci et al. recently conducted a cross-sectional and longitudinal analysis of children at low and high risk of substance use (on the basis of parental substance use history) and found that deficits in behavioural regulation (referred to as ‘neurobehavioural inhibition’) at age 16 in high-risk children predicted a substance use disorder at age 19 with 85% accuracy [117, 124]. Their measure of behavioural regulation was derived using primarily prefrontal tests of cognition (e.g. Stroop interference task, Porteus mazes, motor restraint), affect (a temperament survey) and behaviour (number of behavioural disorder symptoms). The indices derived from these areas converge with other evidence to suggest that behavioural dysregulation is a key component of liability to drug addiction.
The initiation of substance use typically occurs during adolescence, a critical period of neural, cognitive, emotional, and social development. Notably, adolescence is a period during which there is increased affective reactivity together with significant but more protracted neural maturation [125–129] in areas associated with core executive and self-regulatory skills, including inhibitory control and affect regulation [129]. Given that the frontotemporal regions undergo dramatic developmental changes from adolescence to adulthood, drug exposure during adolescence may disrupt the functional maturation of these regions (i.e. inhibitory control and affect regulation). Indeed, there is growing evidence that adolescent rodents appear to be more vulnerable than adults to the adverse neuropsychological and neurobiological effects of addictive substances [130, 131], although studies in human subjects have been limited to date [132–136].
Finally, genetic factors appear to account for 30–60% of the overall variance in risk for developing a drug addiction [137]. While the precise mechanisms underlying this relationship remain unclear, recent work implicates the role of specific genetic polymorphisms. For example, the methionine polymorphism of the catechol O-methyltransferase (COMT) gene results in a slower breakdown of prefrontal dopamine, and is associated with better prefrontal cortical function (including working memory and inhibitory control) in both children [138] and adults [139, 140]. This polymorphism is less common in drug-addicted populations, thereby potentially increasing vulnerability to prefrontally mediated cognitive deficits with chronic drug use [141, 142].
Pathways into drug addiction: implications for neuropsychological interrelations
Long-term substance abuse is associated with significant and persistent neuropsychological impairments and neurobiological abnormalities, involving frontotemporal and basal ganglia circuits. However, it is not clear how broader individual characteristics that are common to addicted populations (e.g. psychopathology, genetic polymorphisms, head injury), and that also affect neuropsychological functioning, contribute to the reported findings. Age of onset of substance use is also clearly important given the plasticity of the developing brain, and may further accentuate the magnitude of neuropsychological and neurobiological impairments. We suggest that future neuropsychological evaluations of long-term substance abusing populations consider the potential influence of such factors.
As illustrated in Figure 1, the pathways leading to drug addiction are likely to be both multifaceted and complex. There is likely to be an intricate relationship between pre-existing neuropsychological vulnerabilities, the age of initiation of substance use (or neurodevelopmental maturity), patterns of substance use (type, dosage, duration, frequency) and associated adverse events (head injury, overdose, suicide). This latter issue is exemplified by Darke et al., who found that these associated adverse events increased the likelihood of neuropsychological impairment in drug-using groups [66]. Similarly, Solomon and Malloy critically reviewed how a head injury can modulate the relationship between substance use and neuropsychological functioning [19]. Clearly, each of these areas can confound the association between substance use and neuropsychological impairment, as well as impact upon the development and functional integrity of prefrontal circuitry, which is postulated to play an important role in the initiation and maintenance of drug-use behaviours. These adverse effects are likely to render the individual at increased risk for making decisions that are impulsive, focused on short-term gains, and lack inhibitory control. Such impairments may underpin difficulties regulating drug-seeking/taking behaviour, as well as increase the risk for the development of comorbid disorders. However, the integrity of prefrontal circuitry is also likely to depend on a number of individual factors, ranging from genetic polymorphisms through to psychosocial variables. Such relationships need further investigation, especially as few prospective, longitudinal neuropsychological studies have been conducted to date, and most previous studies have sought to specifically exclude such confounds rather than examine potential interactions.
Neuropsychological model illustrating how substance-associated neuropsychological sequelae may interact with adolescent neurodevelopment and pre-existing neuropsychological vulnerabilities to increase risk for addictive disorders. PFC, Prefrontal Cortex.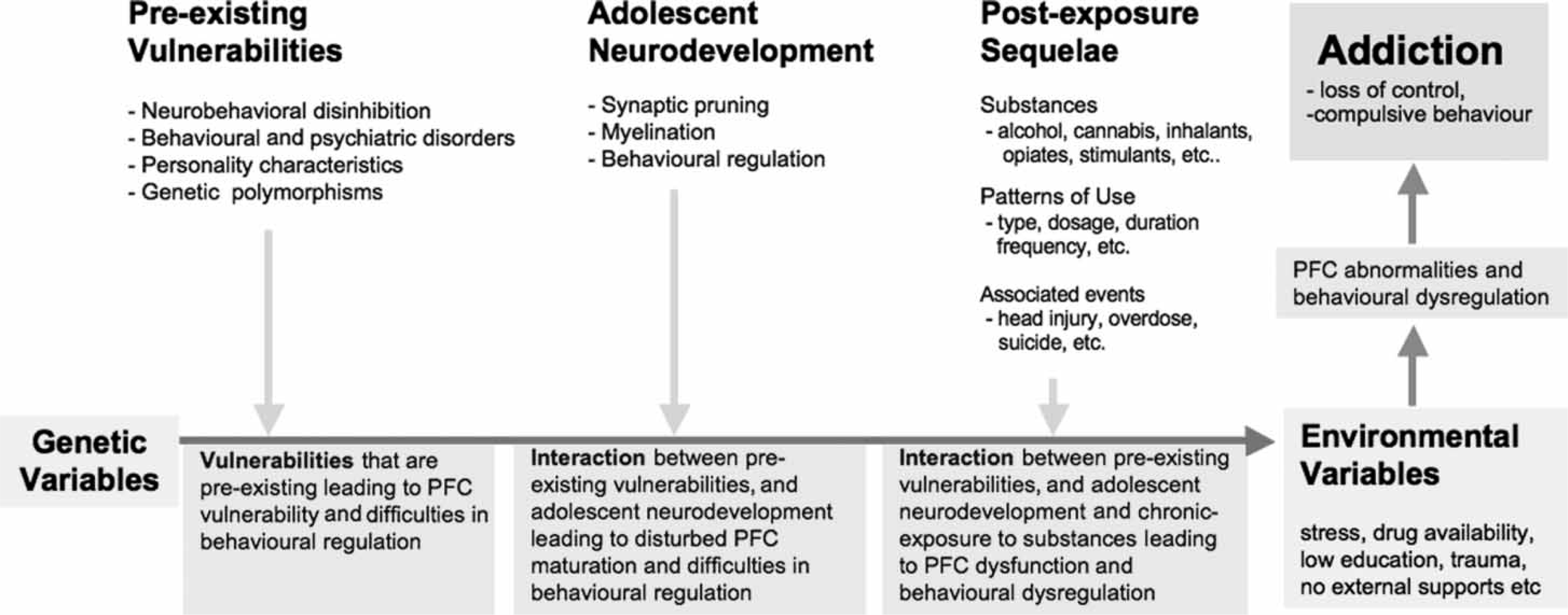
In closing, it should be noted that the selective review of studies highlighted in the present paper may have biased reporting toward positive findings with regard to neuropsychological sequelae of the major substances of abuse. Nevertheless, we endeavoured to maintain a balance by also acknowledging negative findings. A systematic and comprehensive review that covers all of the published literature on neuropsychological sequelae of substance use in humans was beyond the scope of this paper but would be a welcome addition to the literature and would enable a more rigorous exploration of the hypotheses we pose here.
Footnotes
Acknowledgements
Dr Murat Yücel is supported by an NHMRC Program Grant (ID 350241). Melbourne Neuropsychiatry Centre is supported by the Department of Psychiatry, University of Melbourne and Melbourne Health. Dr Dan I. Lubman and Assoc. Professor Warrick J. Brewer are supported by the Colonial Foundation. Assoc. Professor Warrick J. Brewer is supported by an NHMRC Clinical Career Development Award.
