Abstract
Keywords
Bipolar disorder (BD) is an illness of extremes of emotion and mixed mood states characterized by cognitive and social disability [1, 2]. Structural and functional imaging studies have identified a number of key brain regions that have been implicated in its pathophysiology [3, 4]. Among them the anterior cingulate cortex (ACC) has been shown to be particularly important by virtue of its role in decision-making processes [5]. The ACC is part of the limbic system and is located in the medial part of the frontal lobes. It partly encircles the rostrum of the corpus callosum, and is bordered superiorly and inferiorly by the cingulate and callosal sulci, respectively. It contributes along with other cortical and subcortical neural structures (e.g. amygdala, prefrontal cortex and ventral striatum) to the processing of complex emotional behaviours and responses, problem-solving capacity and mood regulation [6–8]. Individuals with BD have been found to have a variety of ACC neuropathological abnormalities including higher levels of white matter glial fibrillary acidic protein [9], decreased density of non-pyramidal neurons [10], and decreased synaptic density [11].
Functional neuroimaging studies have shown decreased ACC activity in unipolar depression [12, 13] but increased activity in mania [14, 15], and magnetic resonance spectroscopy (MRS) studies have identified anterior cingulate changes in both unipolar and bipolar patients [16, 17], strongly implicating this brain region in the pathophysiology of BD. Neuroimaging ACC volumetric studies, however, have been less consistent with reports of an overall reduction in ACC volume in both adults and children [18, 19], while investigations that have partitioned grey matter have in addition reported an increase in segments of the anterior cingulate [20–24]. Notably, many magnetic resonance imaging (MRI) volumetric studies have failed to detect any significant changes in the ACC of bipolar patients [25–27]. However, these studies have used a range of techniques and methodologies in examining the ACC and the definition of BD has varied. For example, some early studies that used manual region-of-interest (ROI) techniques found significant and sizeable reductions in the subgenual grey matter of the left ACC in familial bipolar patients [28]. More recent studies have been less impressive, with either generalized reductions across the entire ACC [29, 30] or no significant change whatsoever [31, 32]. Similarly, studies employing voxel-based approaches have found lateralized changes, reductions [33, 34], bilateral changes [35] or no significant differences at all [36]. Hence, the aim of the present study was to investigate the existence of volumetric abnormalities in the ACC of individuals with rigorously defined BD as compared to closely matched controls. Given the pivotal role of this structure in affect regulation and mood generation we anticipated differences in ACC volumes that would perhaps be related to illness variables and treatment factors.
Methods
Subjects
Eighteen female and six male patients aged 19–59 years (mean = 38.2±11.0 years) with DSM-IV BD I were recruited from a specialized mood disorders clinic. This is a secondary and tertiary referral service in which research psychiatrists make diagnoses using the Structured Clinical Interview for DSM-IV–Patient Edition (SCID-P) [37] supplemented by case note review. Patients who had at least one first-degree relative with an affective disorder were defined as having a positive family history. Subjects were defined as having BD with a history of psychosis if hallucinations and/or delusions had occurred during at least one affective episode. Subjects were excluded if they had a history of ongoing substance misuse (past 12 months), neurological disease or closed head injury, or had an additional Axis-I or Axis-II DSM-IV diagnosis, or a medical disorder currently necessitating treatment.
At the time of MRI, 12 subjects were taking lithium (daily dose 975.1±213.2 mg) with a mean plasma level of 0.77±0.1 mmol L–1, while four of them also received sodium valproate. Eight patients were taking valproate, carbamazepine or a combination of the two, and the remaining four patients were unmedicated. Fourteen patients had at least one first-degree relative with an affective disorder and 10 patients had no family history of mood disorder.
Patients were matched pairwise on age (within 2 years), sex, handedness (all right-handed), and education (14.8±2.1 years) with volunteers that were recruited by local advertisement. Comparison subjects were screened for a history of neurological or psychiatric disorder (with SCID version) or a family history of the same. They underwent the same clinical assessments and self-report questionnaires as patients immediately prior to scanning. The Prince of Wales Hospital and University of New South Wales research ethics committees approved the study and all participants provided written informed consent.
Magnetic resonance imaging
Imaging was conducted with a 1.5 T GE scanner (GE Medical Systems, Milwaukee, WI, USA) for T1-weighted 3-D structural MRI. A 2-D scout mid-sagittal cut for AC-PC plane alignment was first acquired. Then 3-D structural MRI was acquired in coronal orientation using a T1-weighted FSPGR sequence (TR/TE = 12.2/5.3; flip angle = 25°; matrix size = 256×256; field of view = 250×250 mm; no gap between), yielding coronal slices 1.6 mm thick with an in-plane spatial resolution of 0.977×0.977 mm pixel−1.
Volume measurement
All measurements of grey and white matter were performed by a single trained rater (AJ), blind to subject identity. The periphery of the ROI was manually outlined using 6.1 Analyse Software (BIR, Mayo Clinic Rochester, Rochester, MN, USA) on coronal T1-weighted slices. The boundary of the ROI was then outlined using a cordless infrared light-driven cursor and the number of voxels within the regions was calculated to produce a total ROI volume. Structural delineation was standardized from posterior to anterior. The tracing procedure was based on the anatomic atlas of Duvernoy [38], and morphologic criteria explained by Yucel et al. [39] in accordance with the method published by Takahashi et al. [40]. Interrater reliability of ACC measurement was determined on five scans that were randomly selected and measured independently by two raters (AJ, XC). The intraclass correlation coefficient for the left ACC was 0.83 (p = 0.001) and for the right ACC it was 0.81 (p = 0.026). Each volume was then measured after at least four weeks by the principal rater (AJ). The intra-rater intraclass correlation coefficient for the right ACC was >0.97 (p = 0.000) and for the left was >0.95 (p = 0.002).
Statistical analysis
Left and right anterior cingulate volumes were normalized to the total intracranial volume (TIV) of the control group using the following formula [41, 42]: normalized (or corrected) volume = R×raw volume, where R is the mean TIV of the controls divided by the patient's TIV. TIV was calculated by summing volumes of gray matter, white matter and cerebral spinal fluid (CSF), all of which were segmented from raw structural images using template in SPM5 software (Wellcome Department of Cognitive Neurology, Queen Square, London, UK). Raw and normalized volumes were compared between patients and comparison subjects using pairwise t test. Exploratory analyses were performed to determine the effects of five variables on anterior cingulate volumes: family history of affective disorder; history of psychosis; duration of illness; frequency of episodes; and lithium treatment. ACC asymmetry was investigated on pairwise t-tests comparing ratios of right to left ACC volume. Effects of clinical features of BD on ACC volumes were determined using a two-step approach. First, pairwise t-tests were applied to examine the effects of psychosis history, family history, use of lithium, episodes of depression and duration of illness on the normalized ACC volumes across patients and controls. The variables total number of affective episodes, and the duration of illness (defined from the age of onset) were dichotomized into subgroups that were comparable in sample size. Patients were divided into two groups according to the duration of illness (≤10 years, n = 10; >10 years, n = 14) and the number of episodes of mood disorder including both manic and depressive episodes (≤10 episodes, n = 11; >10 episodes, n = 13). Second, volumetric effects of these clinical variables were examined in bipolar patients using independent t-tests for continuous variables and Spearman correlation analysis for categorical variables. The effects of variables showing significance in univariate analyses were examined further using ANCOVA analyses to control for effects of age and sex. All statistical analyses were performed using SPSS 14.0 for Windows (SPSS, Chicago, IL, USA). A significance level of p < 0.05 was used in the exploratory comparisons of the effects of the five clinical variables on ACC volume. A significance level of p < 0.01 (using a Bonferroni adjustment) was used for analyses involving multiple comparisons.
Results
Clinical and demographic characteristics
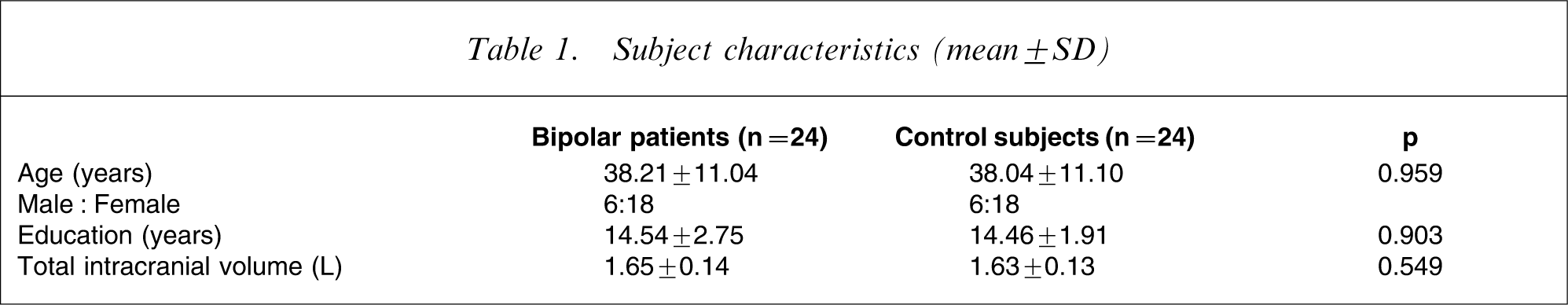
The demographic and clinical characteristics of the 24 bipolar patients and 24 control subjects are presented in Table 1. There were no significant differences between the two groups with respect to age, sex and years of education.
Subject characteristics (mean±SD)
Anterior cingulate cortex volumes
In pairwise analyses, bipolar subjects had significantly larger right ACC volume (by 26%). This difference remained significant when the volumes were normalized. Table 2 shows the mean values for both measured and normalized cingulate volumes. Between-group and within-group comparisons of normalized right ACC volumes in relation to clinical features are summarized in Table 3.
Anterior cingulate cortex volumes (mean±SD)
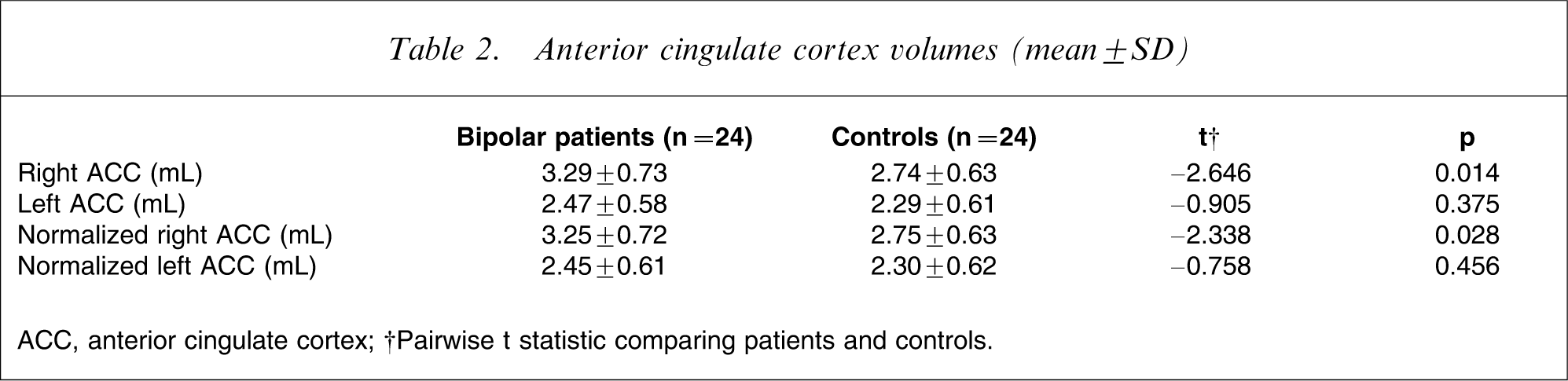
ACC, anterior cingulate cortex.
†Pairwise t statistic comparing patients and controls.
Right ACC volume and clinical features (mean±SD)
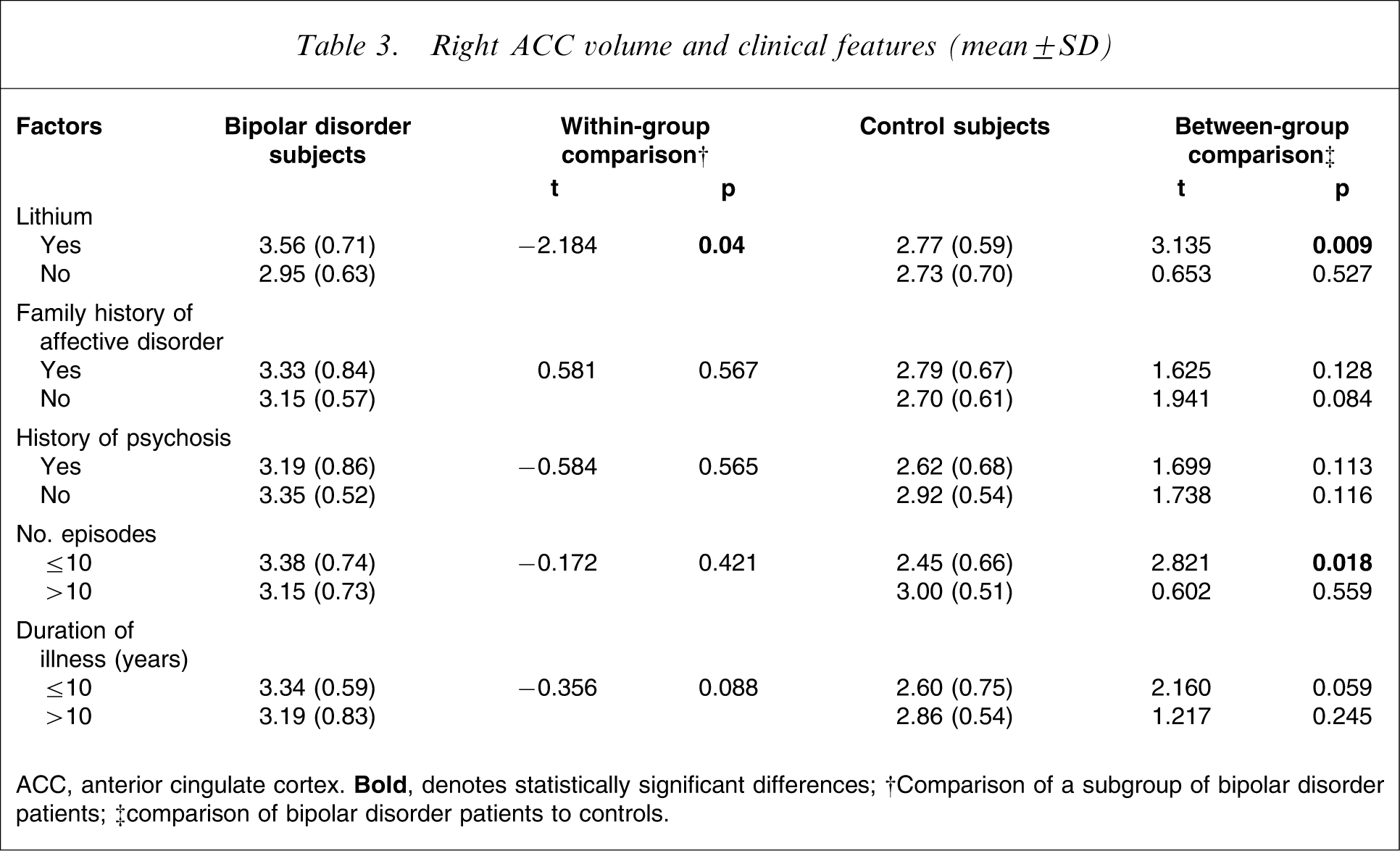
ACC, anterior cingulate cortex.
†Comparison of a subgroup of bipolar disorder patients; ‡comparison of bipolar disorder patients to controls.
ACC asymmetry
Analysis for right–left differences was not significant and right-to-left volume ratios did not differ significantly between the bipolar and control groups (t = − 0.965; p = 0.345).
History of psychosis
At the time of study 14 patients had a history of psychosis. Pairwise analysis did not show significant differences in ACC volume between patients with (t = 1.699, p = 0.113) or without psychotic features (t = 1.738, p = 0.116) as compared to controls. Furthermore, univariate analysis did not show any significant difference within patient groups (t = − 0.584, p = 0.565).
Family history
Of the bipolar patients, 14 had a family history of mood disorder (unipolar or bipolar). Pairwise analysis on the right ACC volumes did not indicate any significant difference between patients and controls with respect to family history of mood disorder (t = 1.625, p = 0.128). Univariate analysis also indicated no significant differences between patients with and without a family history of mood disorder (t = 0.581, p = 0.567).
Use of lithium
Patients treated with lithium (n = 12) were compared with those who were not (n = 12). Pairwise analysis found larger right ACC volumes in the lithium-treated group compared to controls (t = 3.135, p = 0.009), which was not significant for non-lithium treated patients (t = 0.653, p = 0.527). In univariate analysis on bipolar subgroups there was a significant difference between lithium-treated and lithium-naive BD patients with larger ACC volume favouring lithium treatment (t = − 2.184, p = 0.04). However, ANCOVA analysis that corrected the results for age and sex did not show a significant difference within BD patients according to lithium treatment status (p = 0.094).
Duration of illness and number of episodes
Patients had 20.92±18.56 episodes of mania and depression during 14.17±10.26 years of illness. Pairwise t-test determined an association between right ACC volume and number of episodes. Bipolar patients with ≤10 episodes had larger right ACC compared to healthy control subjects (t = 2.821, p = 0.018), which was not observed between patients with >10 episodes and control subjects. Additional analyses within patients group did not detect any correlation between the number of affective episodes and ACC volume (r = − 0.172, p = 0.421). Likewise, there was no correlation between length of illness and right ACC volume (r = − 0.356, p = 0.088). However, a trend of increased ACC volumes in patients with shorter illness duration was found (t = 2.16, p = 0.059).
Discussion
The key finding in the present study is that bipolar patients have a significantly larger right ACC as compared to an age-, sex- and education-matched comparison group, the difference being 26%. Findings from structural neuroimaging studies on BD report both decreases and increases in cingulate volume as compared to controls, with some failing to demonstrate any differences [18, 20–27]. This diversity of findings in the literature is possibly related to the large number of factors that may influence ACC volume. These include frequency of episodes [27], genetic susceptibility/familiality [21, 28, 43–45], duration of illness [11], life stressors [46], illness subtype [14, 15, 25] and treatment [18, 47, 48].
Drug treatment, in particular with lithium, has been considered to be an important factor. Lithium is the most widely used mood stabilizer in the treatment of BD [49], and chronic administration increases the neuronal levels of putative neuroprotective molecules such as N-acetyl-aspartate (NAA) and b-cell lymphoma protein-2 (bcl2) [50, 51]. Lithium-induced regional volume changes may therefore be the consequence of increased neuronal and glial cell size and density [47, 48, 51]. In keeping with this, right ACC volume was significantly increased in lithium-treated patients in the present study as compared with lithium-naive patients and controls. This relationship was, however, not significant after age had been taken into consideration. Because lithium had a positive correlation with ACC volume, and age had a negative correlation, the effect of lithium cannot be accounted for by an age-related effect. In the absence of treatment duration information, inferences from this association must be tempered [18, 47, 48], and we cannot discount the possibility that lithium played a role in this difference.
The number of episodes and duration of illness have been associated with structural abnormalities in patients with mood disorders [16, 20, 25, 52], but studies of first-episode patients also report structural abnormalities, suggesting emergence early in the course of illness [20, 21, 53]. Not all studies echo this association [27], suggesting instead that affective valence may be an important determinant. In the present study bipolar patients with fewer episodes had larger cingulate volumes as compared to controls, suggesting that ACC volume may be enhanced before or during the initial episodes of BD. Unfortunately, additional analyses did not correlate the number of affective episodes or length of illness with ACC volume. Similarly, although previous studies that have partitioned BD with respect to psychosis have identified trends toward volumetric differences [54] with a decrement in left subgenual volume favouring psychosis [21], the present study that examined total ACC volume did not corroborate this.
Genetic predisposition is another postulated precursor of structural abnormalities in BD, and volumetric analyses on patients with mood disorders have generally found smaller left cingulate volumes in patients with a positive family history [21, 45]. Abnormalities in both grey and white matter right ACC have also been associated with genetic liability in BD [55]. Histological and functional cingulate cortex abnormalities have also been associated with familiality [21, 28, 43–45], with decreased subgenual cortex metabolic activity in familial bipolar depression [28] and a reduction (24%) in glial cell density in familial major depressive disorder [43]. Once again, however, findings are not consistent, with one study finding a greater reduction in synaptic density in the ACC of bipolar subjects without a family history of BD [11]. The present findings add to the heterogeneity by finding no association between ACC volume and family history.
An increase in the size of the ACC may have important implications for the neurobiology of BD [56]. The ACC is strongly implicated in conflict resolution, perhaps signalling its involvement in exerting cognitive control. This is thought to occur so as to amplify target features as opposed to inhibiting responses to task-irrelevant information [57]. Symptoms in BD, namely those of affective instability, typically emerge in late adolescence and early adulthood and it is possible that attempts to regulate emotion necessitates greater ACC mediation that is reflected in increased size early in the course of the illness. The effects of medication may be similar and work in concert with psychological processes, such that it promotes the proliferation of the physiologically active regions. However, the lack of sufficient information on duration of lithium treatment in the present study and small sample size constrain further speculation in this regard. Interestingly, right ACC size also correlates with alexithymia especially in men, with a closer association in women to harm avoidance [58].
We acknowledge a number of limitations of the present study. First, there is inevitable heterogeneity and an inherent bias in sampling from hospital subjects. However, matching the controls for demographic features, in particular age and sex, and selecting only those patients with clearly defined bipolar I disorder perhaps compensates to some degree. Second, the sample size was small and therefore the present examination of the subgroups of bipolar patients was exploratory. Third, there is a growing literature on the variability of the surface morphology of the ACC that is perhaps unique to a number of psychiatric illnesses and the impact this has on volumetric measurements [59, 60].
Conclusion
In the present study the principal finding is that of an increase in the size of the ACC in patients with BD as compared to healthy subjects. The pivotal role of the ACC in the regulation of affect is no doubt fundamental to the psychopathology of BD and changes in the function of this neural structure may hold the key to unravelling its neurobiology. It is likely that the structural changes identified in the present study are associated with the aetiopathogenesis and phenomenology of BD. However, the present study could not fully accommodate a number of additional factors and some of the volumetric differences may indeed be better accounted for by changes in surface morphology, or at least better understood in this context. Future structural imaging studies that investigate BD or attempt to replicate these findings need to take methodological issues into consideration and should ideally be longitudinal in design and involve large samples of subjects that have been carefully controlled for medication.
