Abstract
General intellectual decline is thought to be common in patients with established schizophrenia (SZ) [1], as evidenced by a decline in IQ [2, 3] and poor performance on a wide variety of cognitive tests [4]. However, the available evidence suggests that executive [5–7] and memory functions [8] are impaired to a greater degree than the overall level of intellectual impairment. Severe memory deficits in SZ have been demonstrated using composite psychometric measures, which combine varieties of tasks [9–12], and some have even postulated the existence of a “schizophrenic amnesia” [13]. Such deficits are commonly viewed as demonstrating hippocampal dysfunction, even though the tasks underlying the composite score can index different aspects of memory and are sensitive to compromise in different brain regions [14]. Instead, these deficits may result from impaired strategic abilities or other executive functions [15], thereby implicating prefrontal or subcortical areas [16].
It is by no means clear that all memory tasks are equally impaired in SZ, or that memory deficits are equivalent at all stages of the disorder. Two commonly used verbal memory tasks are prose recall and paired associate learning, and these differ in administration and in the cognitive constructs that are assessed. Prose recall tests the ability of the subject to organize the presented information and store it for later retrieval. There is also some degree of linguistic redundancy associated with the use of prose; that is, contextual constraint is imposed by virtue of knowing about typical narrative structures (e.g. establishing a context, describing an event, the consequences of the event, and the resolution to the story). Such informational redundancy is usually helpful in recall, but previous research in patients with SZ has shown compromise of this ability [17, 18], which probably implicates more distributed brain networks than the medial temporal lobe alone, and (at least in SZ) has been directly related to the volume of the prefrontal cortex [19].
In contrast, paired-associate learning appears to be more reliant on the temporal lobe. This task requires the forming of arbitrary associations, which involve complex relational learning [14]. The precise neural substrate is critically dependent on the nature of the pairs, such that semantically related (easy) pairs are mediated by lateral temporal neocortex, while semantically unrelated (difficult) pairs are mediated by the medial temporal lobe (most probably the hippocampus [20, 21]). Because hippocampal volume is only mildly reduced in SZ [22, 23], and the associated neuropathology is relatively subtle [24], it is especially important to separate easy from difficult pairs – unfortunately, almost all studies in SZ to date have combined both kinds of pairs to produce a single score.
Another issue is whether there is a significant difference in performance on memory tests between patients with SZ and those with first-episode schizophreniform psychosis (FE), a distinction that is solely reliant on the duration of illness. The present hippocampal volume findings suggest that hippocampus-dependent tasks (such as paired-associates) should be impaired in SZ, but preserved (or only marginally affected) in schizophreniform psychosis [22]. This hypothesis is supported by our cross-sectional study of visuospatial associative learning [25], which found significant impairments in SZ but not in FE patients. These differences were not the result of greater impairment of executive function, suggesting that either visuospatial associative memory declines with continued illness, or that it represents a marker of poor prognosis. In contrast, prose recall is impaired in SZ [26] and FE patients [27], and also prior to psychosis onset in an at-risk cohort who were unimpaired on associative learning [28].
The present study explores whether these findings are specific to the visuospatial domain, and assesses SZ and FE patients on measures of verbal episodic and working memory, as well as potential confounding variables of global cognitive impairment, age and treatment with typical versus atypical neuroleptics.
Method
Participants
Data for patients (n = 65) and controls (n = 47) were selected from a larger database comprising subjects from several ongoing studies being conducted in Melbourne, Victoria. The following subjects were selected for inclusion in the present study.
Established schizophrenia patients
Thirty-two patients with a current DSM-III-R or DSM-IV Axis I diagnosis of SZ (median illness duration = 8.71 years, range = 0.52–24 years; 69% inpatients) were recruited through inpatient and community psychiatric services from the North Western Mental Health Program (NWMHP) in inner Melbourne. Diagnoses were based on one or more of the following: chart review using DSM criteria, the Structured Diagnostic Interview for DSM-III-R (SCID-R/SCID-I) [29] or DSM-IV [30] Axis I disorders and a structured clinical interview for measuring psychopathology (Positive and Negative Syndrome Scale; PANSS) [31]. Where the SCID was unavailable (50% of cases), diagnoses were made by consensus between two consultant psychiatrists (CP and DV). Eleven patients were receiving typical antipsychotic medication at the time of assessment (predominantly via depot), and 21 were treated with atypical medication (mainly clozapine or olanzapine). No patients were receiving both typical and atypical neuroleptics, or anticholinergic medication.
First-episode schizophreniform psychosis
Thirty-three patients were recruited from the Early Psychosis Prevention and Intervention Centre (EPPIC, ORYGEN Research Centre), and also from the NWMHP. These patients were experiencing their first episode of a psychotic illness and received a DSM (III-R or IV) Axis I diagnosis of schizophreniform psychosis. Diagnoses were based on one or more of the following: a structured clinical interview using the PANSS, the Royal Park Multidiagnostic Instrument for Psychosis (RP-MIP) [32] and the SCID-R or SCID-I. Where both the SCID and RP-MIP were unavailable (33% of cases), diagnoses were made by consensus between two consultant psychiatrists (CP and DV). All patients had an illness duration of <6 months at the time of assessment (median = 47 days, range = 3–150 days), and 82% were inpatients. Twenty-five patients were treated with atypical antipsychotics (mainly 2–5 mg risperidone or 10 mg olanzapine), and eight with typical antipsychotic medication; no patient received anticholinergic medication.
Healthy volunteers
Forty-seven healthy volunteers were recruited by approaching ancillary hospital staff and their families, or through advertisements. These participants were recruited from similar sociodemographic areas to patients. Demographic profiles of the experimental groups are presented in Table 1.
Demographic features (mean, SD)
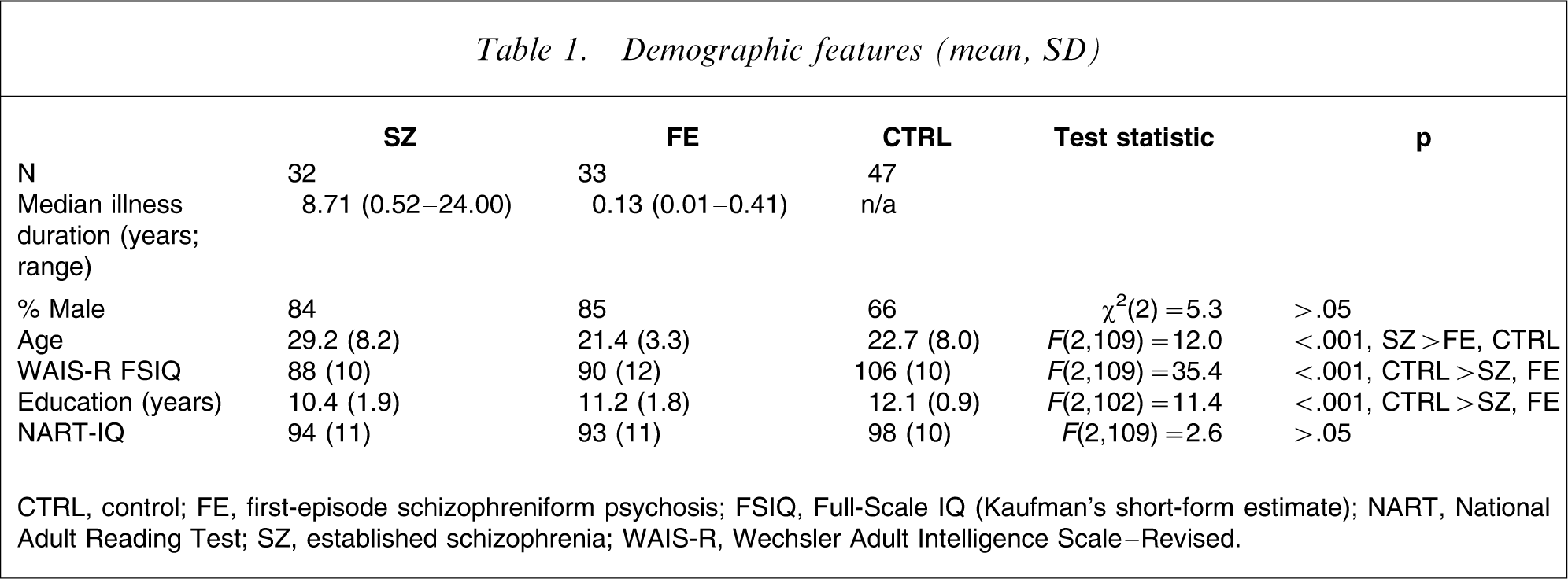
CTRL, control; FE, first-episode schizophreniform psychosis; FSIQ, Full-Scale IQ (Kaufman's short-form estimate); NART, National Adult Reading Test; SZ, established schizophrenia; WAIS-R, Wechsler Adult Intelligence Scale–Revised.
All patients were screened for comorbid medical and psychiatric conditions by clinical assessment and physical and neurological examination. Exclusion criteria for all three groups were history of significant head injury and/or loss of consciousness >1 min, seizures, neurological disease such as epilepsy or cerebrovascular accident, significant hypoxic episode, childhood onset of SZ (<12 years of age), electroconvulsive therapy within 3 months prior to neuropsychological testing, preferred language not English, premorbid IQ <70, known history of a learning disability, known steroid abuse, and a history of substance dependence. Health volunteer (CTRL) subjects with a previous psychiatric disorder or a family history of psychosis were also excluded.
Measures
Each participant was given the following neuropsychological tests: Digit Span from the Wechsler Adult Intelligence Scale–Revised (WAIS-R) [33], and Verbal Paired Associates I (VPA) and Logical Memory I (LM) from the Wechsler Memory Scale–Revised (WMS-R) [34]. Although the Digit Span test includes recital of digits in both forwards and reverse order, only the latter (i.e. Digit Span–backwards; DSp-b) was used to assess verbal working memory function, while VPA and LM were used to assess verbal episodic memory. Participants were also given the National Adult Reading Test [35] to provide an estimate of premorbid intelligence, and a short-form estimate of full-scale IQ (FSIQ) using the Arithmetic, Similarities, Picture Completion and Digit Symbol subtests from the WAIS-R [36] to assess current intelligence.
The eight items comprising the VPA test are divided into four semantically related pairs (designated easy pairs, because the contextual redundancy provides great support for cued recall performances; VPA-e) and four semantically unrelated pairs (designated difficult pairs; VPA-d), and a score is obtained for each type of pair. Performances on the easy and difficult word pairs were analysed separately, because recall of the difficult pairs provides a better measure of dominant hemisphere medial temporal lobe function than does recall of semantically related pairs [20, 21, 37].
Statistical analysis
Statistical tests were performed using SPSS version 11 (SPSS, Chicago, IL, USA) and an alpha level of 0.05 was used. Analyses of between-group differences were defined by a three-level (SZ, FE, CTRL) between-subjects factor. Two analyses were performed; initially, raw scores for DSp-b, LM, VPA-e and VPA-d were examined separately using one-way analyses of covariance (ANCOVA) with age and premorbid IQ entered as covariates. Subsequently, FSIQ was introduced as a covariate in place of premorbid IQ to establish whether any deficits were a result of global cognitive impairment. Age was considered a mandatory covariate for raw scores given that for VPA-e and VPA-d (unlike DSp-b and LM), the WMS-R manual does not provide age-scaled percentiles; age correlated significantly with all four dependent variables. However, examination of the regression slopes between age and the dependent variables showed that there was no difference in the effect of age on performance across the three groups. Post-hoc analyses were performed on the estimated marginal means obtained, using a Bonferroni-adjusted alpha level to control for multiple comparisons. Effect sizes were calculated using Cohen's d [38].
Two-way ANCOVAs on patient data alone were also performed in order to examine any differential effect on cognition due to type of antipsychotic medication. These analyses were defined by a two-level (SZ, FE) group factor and a two-level (typical, atypical) medication type factor, with age and FSIQ entered as covariates.
Results
Tables 2 and 3 show summary data for the four dependent variables; Table 2 shows the raw data, and Table 3 the data expressed as estimated marginal means, after covarying for age and FSIQ.
Raw data for the verbal working memory and verbal episodic memory tasks (mean, SD)
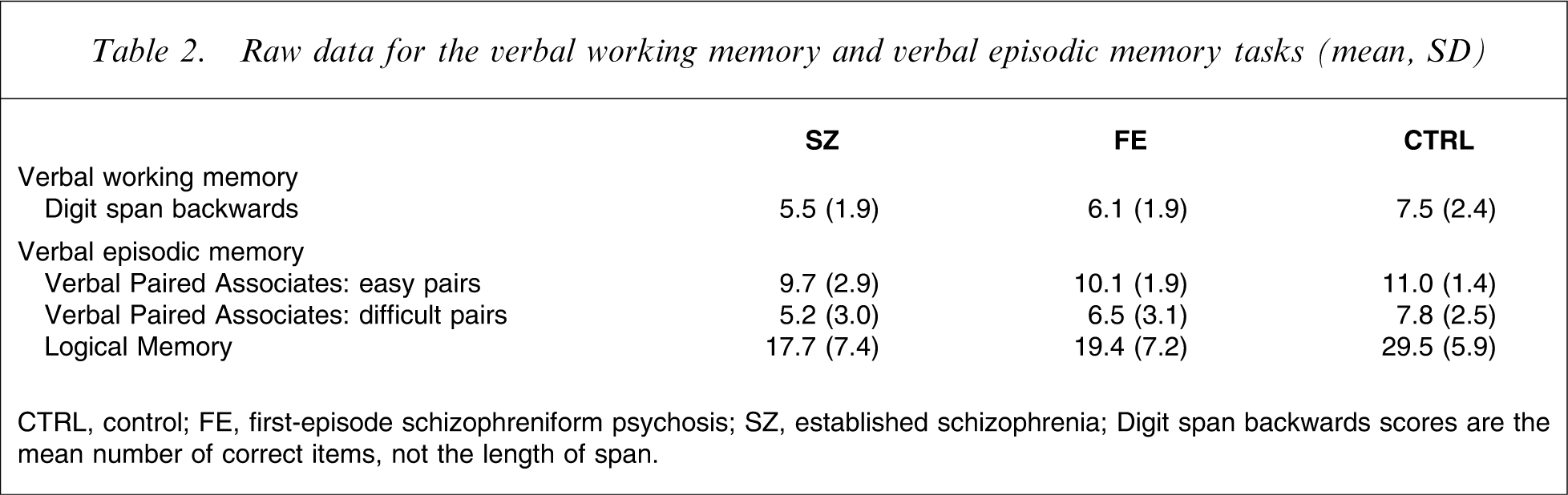
CTRL, control; FE, first-episode schizophreniform psychosis; SZ, established schizophrenia
Digit span backwards scores are the mean number of correct items, not the length of span.
Estimated marginal means on the verbal working memory and verbal episodic memory tasks (mean, 95%CI)
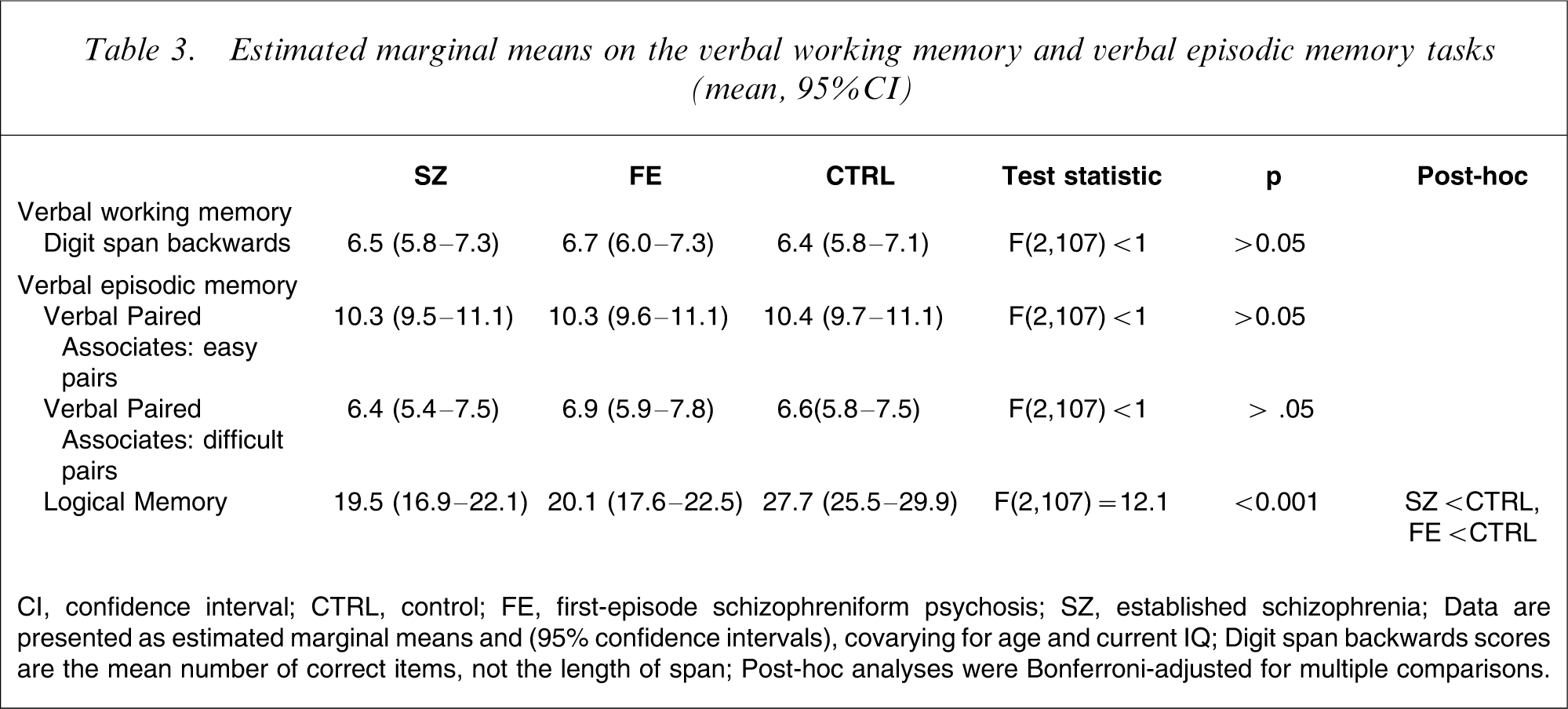
CI, confidence interval; CTRL, control; FE, first-episode schizophreniform psychosis; SZ, established schizophrenia
Data are presented as estimated marginal means and (95% confidence intervals), covarying for age and current IQ
Digit span backwards scores are the mean number of correct items, not the length of span
Post-hoc analyses were Bonferroni-adjusted for multiple comparisons.
Digit span backwards
Performances on this test significantly differed across groups when age and premorbid IQ were covaried (F(2,111) = 4.17, p < 0.05). Post-hoc Bonferroni-adjusted analysis indicated that only SZ patients (p = 0.042) were significantly impaired relative to CTRLs; there was no difference between the SZ and FE groups. When FSIQ replaced premorbid IQ as a covariate, it exerted a strong influence as a covariate (F(1,111) = 39.3, p < 0.001) and there was no longer a significant effect of group (F(2,111) = 0.1, p > 0.1). Effect sizes for the latter analysis were d = 0.05 and d=0.11 for SZ and FE, respectively, in comparison to the CTRL group.
Rerunning these analyses using the age-corrected percentile score instead of covarying for age did not alter the findings.
Verbal paired associates: easy pairs
Performances on this test non-significantly differed across groups when age and premorbid IQ were covaried (F(2,111) = 2.79, p < 0.1). When FSIQ was covaried, this near-significant group effect in recalling the easy pairs was lost (F(2,107) < 1). Again, FSIQ was significant as a covariate (F(1,107) = 10.71, p = 0.001). Effect sizes for the latter analysis were d = 0.04 and d = 0.03 for SZ and FE, respectively, in comparison to the CTRL group.
Verbal paired associates: difficult pairs
ANCOVA incorporating age and premorbid IQ demonstrated a significant effect of group for the difficult pairs (F(2,107) = 3.3, p < 0.05), but post-hoc Bonferroni-adjusted analyses did not demonstrate any significant pairwise differences (the largest difference was between the SZ and CTRL groups). When FSIQ was covaried in place of premorbid IQ, there was no longer a significant group effect (F(2,107) < 1; Cohen's d = − 0.09 and 0.06 for FE and SZ, respectively).
Logical memory
There was a significant group effect when both age and premorbid IQ were covaried (F(2,107) = 28.5, p < 0.001), which was maintained when premorbid IQ was replaced by FSIQ (F(2,107) = 12.10, p < 0.001). In both analyses, post-hoc Bonferroni-adjusted pairwise tests indicated that both SZ (p < 0.001) and FE patients (p < 0.001) were significantly impaired relative to CTRLs; there was no significant difference between the SZ and FE groups. The effect sizes of the difference between CTRL and the SZ and FE groups were d = 1.08 and d = 1.03, respectively.
Rerunning these analyses using the age-corrected percentile score instead of covarying for age did not alter the findings.
Medication type: typical versus atypical neuroleptics
Performance on the memory tasks was compared controlling for age and FSIQ. The only indication that medication affected memory performance emerged in an analysis of performance with difficult paired associates: patients taking typical neuroleptics remembered fewer difficult pairs (mean (SD) = 4.7 (2.8)) than patients on atypical neuroleptics (mean (SD) = 6.4 (2.7); F(1,59) = 4.56, p < 0.05); there was no effect of, or interaction with, patient group (both Fs < 1).
No other main effects or interactions of medication type were significant: for DSp-b and easy paired associates, all Fs < 1; for prose passages there was a non-significant trend for a medication effect (F(1,59) = 3.02, p = 0.08; patients treated with typical medications tended to perform more poorly) and no significant interaction with group (F(1,59) = 1.64, p > 0.05).
Discussion
Using measures of verbal memory from the WMS-R, the current study found evidence for the presence of substantial verbal memory impairment in both SZ and FE patients. There were two important features of the current findings: impairment was evident irrespective of illness chronicity, with no difference in performance found between SZ and FE patients; and selective impairment, over and above the apparent global intellectual deficit, was evident only when patients were required to recall prose passages. Both patient groups were unimpaired relative to CTRLS in terms of working memory and associative learning when age and global intellectual function were controlled; for associative learning, it did not matter whether semantic structure was provided or not. However, associate memory performance (again controlling for age and global intellectual function) was significantly poorer in patients who were on typical antipsychotics when compared to those on atypical antipsychotics, with no interaction with patient group.
The similar magnitude of impairment noted across the patient groups on LM provides some support for the stability of a particular kind of verbal memory impairment across the course of illness. Indeed, impairments on LM have been shown to pre-date the frank onset of psychotic symptoms in an ultra-high-risk group [28]. The finding of no significant impairments with difficult pairs on the VPA stands in contrast with our earlier findings [25], where impaired visuospatial associative learning was found in SZ but not FE patients. The interpretation offered in that study was that progressive decline in associative learning ability may occur over the course of illness, or that poor associative memory is a marker for poor prognosis. The current data do not deny that interpretation but neither do they support it. It may be the case that the spatial associative learning used in our earlier study is more sensitive to dysfunction than the VPA from the WMS-R: it contains greater item power than the VPA, which utilizes only four test items for each of the related and unrelated pair sets.
The finding of verbal memory impairment in SZ and FE patients (when controlling for age and FSIQ) is consistent with a wealth of previous studies, which have typically examined only one of these patient groups [8, 11, 39–43]. The studies of Addington and Addington, Hoff et al. and Saykin et al. are comparable to the present study in that they compared verbal memory function between patients with FE and established SZ [39, 40, 43]. All of these previous studies reported that the magnitude of verbal memory impairment in the two patient groups was equivalent. The current study also found equivalent impairment among SZ and FE patients on LM but this pattern was not observed on VPA-d. In common with the current study, the first-episode sample recruited by Hoff et al. was a first-episode schizophreniform psychosis sample, with illness duration <6 months [40]. However, both the sample recruited by Saykin et al. (median illness duration 2 years) [43] and Addington and Addington (minimum illness duration of 6 months) [39] studied first-episode patients with significantly longer length of illness, which can introduce confounds of illness progression and lengthy medication use. Thus, the present study appears to be one of few studies comparing verbal memory function between established SZ patients and FE patients with very recent illness onset.
Task-specific findings
Working memory
There is general consensus on the presence of verbal working memory impairment in SZ [44]. Surprisingly, SZ and FE patients were not impaired on the DSp-b, a purported measure of auditory–verbal attention and verbal working memory. Importantly for any study of episodic memory, impairment can be due to problems at the level of memory processing per se (i.e. in encoding, storage, and retrieval), but can also be due to problems in the initial stages of information processing, such as poor attention or reduced working memory capacity. Absence of selective working memory problems in the present study allows clearer attribution of the difficulty with connected prose to the formation and/or retrieval of enduring memory traces, rather than to preliminary information processing problems. However, it should be noted that there are psychometric limitations to this test that could affect its ability to detect group differences.
Associative learning with semantically related material
The easy word pairs used in the VPA subtest of the WMS-R contain highly common and well-established associations (e.g. rose–flower). Such contextual redundancy provides great support for cued recall performances: remembering the second element of easy pairs relies less on ability to form a link between the words, and more on ability to remember exactly which highly predictable word was the one to be remembered (e.g. flower, thorn, or perhaps garden when the cue is rose). We might have expected some difficulty to be encountered by patients because there is considerable research evidence that patients with chronic SZ do not benefit from redundancies in information in the performance of cognitive tasks as much as healthy individuals do [17, 18]. It is likely that in this case the task was simply too easy for these differences to have an effect.
Associative learning with semantically unrelated material
In contrast, the conjunctions contained within difficult word pairs are arbitrary. Effective performance requires the subject to maintain the word pairs in working memory and to form new associations in memory. An enormous body of research from animal and human studies suggests that medial temporal lobe (MTL) structures have a dedicated role in forming novel associations, and intact performances on the VPA suggest functional integrity of these regions in schizophreniform illness. The WMS-R VPA subtest is sensitive to MTL dysfunction in left-sided temporal lobe epilepsy [37]; it remains possible that the VPA test is not sensitive to subtle MTL dysfunction. The alternative is the scenario that right-sided MTL dysfunction emerges in the transition from FE to established SZ [25], but left-sided functionality is maintained.
Memory for connected prose
In the LM task two short pieces of highly imageable prose are read to subjects for immediate recall. The task allows for exploitation of informational structure that facilitates subsequent recall. There is some degree of linguistic redundancy associated with the use of prose: that is, contextual constraint is imposed by virtue of knowing about typical narrative structures (e.g. establishing a context, describing an event, the consequences of the event, and the resolution to the story). In contrast with associative learning of arbitrary conjunctions, forming individual links between words plays a less prominent role, and the arbitrary links to be learned operate at a higher level, perhaps linking thematic segments (within which, semantic knowledge might determine content to a great extent, akin to recalling easy pairs). The current finding of impaired story recall in SZ and FE patients is consistent with previous SZ research showing compromised ability to profit from informational redundancy [17, 18].
Impact of typical versus atypical neuroleptics on verbal memory
Investigations of memory dysfunction in SZ may be confounded by treatment with various medications, including antipsychotics, anticholinergics, benzodiazepines, tricyclic antidepressants and lithium. The impact of neuroleptics on memory function in SZ remains unresolved, particularly with respect to long-term treatment effects and whether these medications lead to cumulative changes in brain physiology. Although a meta-analysis did suggest less impact of atypical antipsychotics [45], more recent studies using low doses of typical neuroleptics (approx. 4–6 mg haloperidol) suggest only minimal disadvantage for the older treatments [46, 47]. In contrast, studies examining the influence of anticholinergics, benzodiazepines, tricyclic antidepressants and lithium on memory performance have produced more consistent findings, with the majority suggesting that these agents exert a deleterious effect on memory [48, 49].
The current study investigated the effect of typical versus atypical neuroleptics on verbal memory for a group of patients who were known to be taking only one of these types of medication, while no patient was simultaneously treated with anticholinergic medication. These analyses showed that patients who were receiving typical neuroleptics performed worse than those receiving atypical neuroleptics, but only on the associative learning task with difficult pairs. Sparing of memory with atypical medications is consistent with previously published longitudinal studies of the minimal impact on general cognition with these medications. Although these data do not prove causation, one interpretation of our findings is consistent with a specific detrimental effect of typical medication on the hippocampus, a finding that has also been demonstrated in rats [50]. But it should be borne in mind that some of the FE patients had been prescribed medication for only a very short period of time, and that such prescription is not always independent of cognitive ability (e.g. a patient who cannot remember to take an oral antipsychotic may be switched to a depot injection, which is more likely to be a typical neuroleptic). Furthermore, some of the patients in the SZ group are likely to have been switched between typical and atypical medications in the past. We obtained medication data only for the past month, meaning that we cannot determine whether such switching has an effect on associative memory performance.
Conclusion
The present study has demonstrated that verbal memory impairment in schizophrenia and schizophreniform psychosis is not present for all tasks. Instead, breakdown occurs only when widely distributed and interconnected brain regions are required to process the various associative, grammatical, semantic, prosodic, and discourse level aspects that capture the complexity of a story.
Footnotes
Acknowledgements
This research was supported by project grants (ID numbers: 970598, 981112, 145627) and a programme grant (ID: 350241) from the NHMRC. Professor McGorry was supported by a NARSAD Distinguished Investigator Award and Dr Wood is the recipient of a Clinical Career Development Award from the NHMRC (ID: 359223) and a NARSAD Young Investigator Award. Associate Professor Brewer is supported by the colonial foundation and is th e recipient of a Clinical Career Development Award from the NHMRC.
