Abstract
There has been significant increase in antidepressant use in Australia in the last 15 years, rising from 12.4 defined daily doses per 1000 of the population per day (DDD 1000−1 day−1) in 1990, up to 64.6 DDD 1000−1 day−1 in 2004 [Dudley J: personal communication]. The DDD 1000−1 day−1 provides an overall usage estimate that is equated to a defined (standard) dose independent of the strength of the drug or the quantity supplied [1]. In this instance 64.6 DDD 1000−1 day−1 equates to sufficient supply of the standardized dose to treat 6.5% of the population every day for a year. This increase in antidepressant utilization has been driven largely by the availability of antidepressant medicines and depression awareness campaigns, resulting in increasing recognition and treatment of depression. With increasing use of medicines comes increasing potential for adverse drug events, particularly in the elderly, for whom polypharmacy is common and age-related pharmacodynamic and pharmacokinetic effects can have an impact.
Studies of the prevalence of potential drug interactions with antidepressants are limited and mostly were conducted in the early 1990s, before the large rise in antidepressant use. A 1990 Canadian study found that 1.0% of the elderly population were using two or more tricyclic antidepressants (TCAs) [2], while a 1992 US study using an administrative dataset found that 5.9% of patients had at least one antidepressant drug interaction and 4.9% had duplicate therapy [3]. An Australian study of co-prescribing of TCAs with selective serotonin re-uptake inhibitors (SSRIs) in 1996 found that 5% of the population on antidepressants were on this combination [4]. A separate study in the Australian veteran population in 1996 found an incidence rate of interactions of 0.7% for TCAs with benzodiazepines, and ≤0.3% for TCAs and cimetidine, propoxyphene, verapamil and diltiazem [5]. Tramadol has been recently introduced to the market and a US study found this drug to be commonly co-prescribed with antidepressants, with 19–22% of tramadol users co-prescribed an antidepressant [6].
Since the earlier Australian studies, electronic prescribing software, which includes interaction checking, has been introduced into most general practices in Australia, while pharmacy dispensing software, which was already in existence, has been maintained and updated. In addition, there has been much greater use of the SSRIs and other new agents. Thus, although there has been an increase in antidepressant use, it may be expected that the interaction checking software and the quality use of medicines initiatives in place in Australia, may have minimized potential drug–drug interactions with antidepressants.
The aim of the present study was to assess the prevalence of duplicate antidepressant prescribing and potentially avoidable antidepressant drug interactions in the Australian veteran population.
Method
The Department of Veterans Affairs (DVA) reimburses the cost of health services for all eligible veterans and their dependants. Currently, there is a treatment population of approximately 305 000 veterans, of whom 75% are aged ≥70 years [7]. The pharmacy claims database contains details of all prescription medicines dispensed to veterans for which DVA pay a subsidy. The data file contains 75 million records. The DVA maintain a client file, which includes data on gender, date of birth, date of death and family status. Medicines are coded in the dataset according to the World Health Organization anatomical and therapeutic chemical (ATC) classification [1].
Medicines contraindicated with antidepressants were identified from the Australian Medicines Handbook [8]. A clinical reference group, which included three general medical practitioners, one drug information pharmacist, and two clinical pharmacists, one of whom had specialty practice certification in psychiatry and geriatrics, reviewed the interactions and considered those that were able to be avoided through use of alternative medicines. A number of other potentially significant drug interactions were identified, for example, those that that occur between the SSRI antidepressants and medicines that are inducers, inhibitors or substrates of the cytochrome p450 enzymes. The majority of these interactions can be managed with appropriate monitoring and dosage adjustment. Because the DVA pharmacy claims dataset contains the strength and quantity of the medicine dispensed, but not the dosage prescribed, it is difficult to assess dosage adjustment within the dataset. Hence, these interactions were excluded.
Potentially interacting pairs for which safer alternate therapies were available, and antidepressant duplication, were chosen for analysis. The criteria were (i) dispensings of more than one antidepressant concurrently; (ii) dispensings of an antidepressant (excluding mianserin and reboxetine) and tramadol concurrently; and (iii) dispensings of TCAs or mianserin and anticholinergic drugs (procainamide, quinidine, tiotropium, disopyramide, oxybutynin, pericyazine, pizotifen, promethazine, thioridazine, amantadine, benzatropine, chlorpromazine, clozapine) concurrently. The anticholinergic medicines chosen were those that the Australian Medicines Handbook indicated had significant anticholinergic effects, were used chronically and funded under the Pharmaceutical Benefits Scheme. Topical agents and nebulized ipratropium were excluded.
Study 1
Study 1 examined concurrent dispensings of antidepressants with other antidepressants, tramadol or anticholinergic agents. To avoid identification of patients who had switched therapy and include only patients to whom were dispensed both medicines in the potentially interacting pair, regular concurrent dispensings of both medicines were defined using time intervals. Subjects had to have received at least one dispensing of the antidepressant in the 2 months April–May 2005 and at least one dispensing of the same class of antidepressant in the 2 months June–July 2005. These time periods were used to establish the cohort of regular antidepressant users. Quantities supplied under the pharmaceutical system in Australia generally equate to a 1 month supply. Sixty days was chosen as the dispensing window because analysis of the data indicated that 75% of claims for a repeat dispensing of an antidepressant occurred within 40 days, extending to 49 days when the TCAs were considered alone. For dispensings to be considered an interaction, the interacting medicine had to be dispensed at least once in both equivalent time periods, in which the median dispensing interval for the product was <60 days. When the median dispensing interval was >60 days but <90 days, subjects had to have at least one dispensing of the interacting medicine in the 3 month period March–May 2005 and in the period June–August 2005. The inclusion of dispensings in both time intervals for both medicines ensured that concurrent use was detected.
Study 2: antidepressant dispensing and episodic tramadol dispensings
Tramadol, which is used for pain relief, is used both chronically and episodically. For this reason, episodic tramadol dispensings with regular antidepressant dispensings were also examined. To capture episodic use, the 12 month time period 1 August 2004–31 July 2005 and the following algorithms were used: (i) number of veterans who had tramadol and an antidepressant prescription dispensed on the same day in the 12 month period; (ii) number of veterans with at least eight antidepressant prescriptions in at least 8 different months in the 12 month period and more than one tramadol prescription in more than 1 month in this period (this analysis is not detecting whether the tramadol prescription was in the same month); (iii) number of veterans with at least six antidepressant prescriptions in at least 6 different months in the 12 month period and more than one tramadol prescription in more than 1 month in this period (this analysis is not detecting whether the tramadol prescription was in the same month); (iv) number of veterans with at least two or more occurrences of tramadol and antidepressant dispensings occurring in the same 1 month period over the 12 months; and (v) number of veterans with at least two or more occurrences of tramadol and antidepressant dispensings occurring in the same 2 month period over the 12 months.
Statistical analysis
All analyses were undertaken using SAS version 9.1 (SAS Institute, Cary, NC, USA). Student's t-tests were used to test differences between veterans to whom were dispensed interacting combinations and those to whom were not dispensed the potentially interacting combinations.
Results
Study 1
Overall, 46 859 veterans regularly had an antidepressant dispensed in the period 1 April–31 July 2005. Their mean age at 1 April 2005 was 74.4 years, (SD = 12.6 years), 58% were male and 42%, female.
Overall, 4037 interactions were identified in 3818 veterans (8.1%) to whom were dispensed antidepressants regularly. Antidepressant and tramadol co-prescribing was the most common potential interaction identified (Table 1). Tiotropium and TCA co-prescribing accounted for 57% of the potential interactions between TCAs and anticholinergic drugs. In all, 212 veterans (0.5%) had two or more potentially interacting pairs.
Prevalence of antidepressant co-prescribing and avoidable interactions among veterans
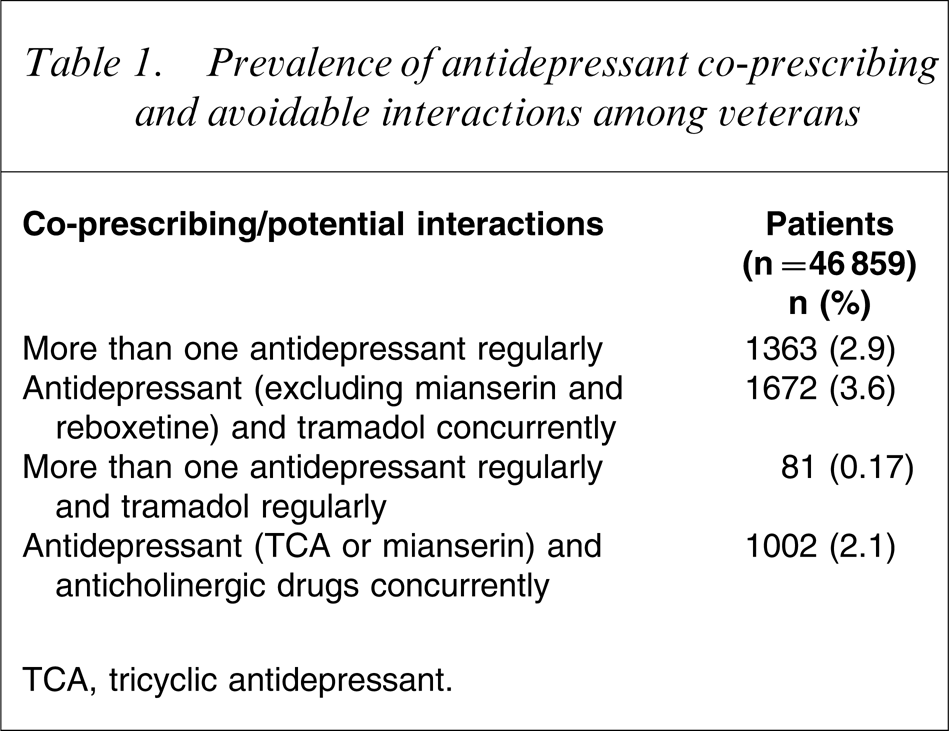
TCA, tricyclic antidepressant.
Veterans in whom a potential interaction was identified, were of similar age (74.5 years, SD = 12.9 years) to those without an interaction (74.4 years, SD = 12.6 years) but more medicines were dispensed to them: on average, 13.9 different pharmaceutical products (SD = 6.6) compared to 9.4 (SD = 5.5; p < 0.05), and on average more prescriptions, 37.9, (SD = 20.2) compared to 23.9 (SD = 14.6; p < 0.05).
Study 2
Analysis showed that 3600 veterans (7.7%), on average, had antidepressants dispensed and had tramadol dispensed in an episodic pattern (Table 2).
Prevalence of antidepressant dispensing and episodic tramadol dispensings by five different algorithms
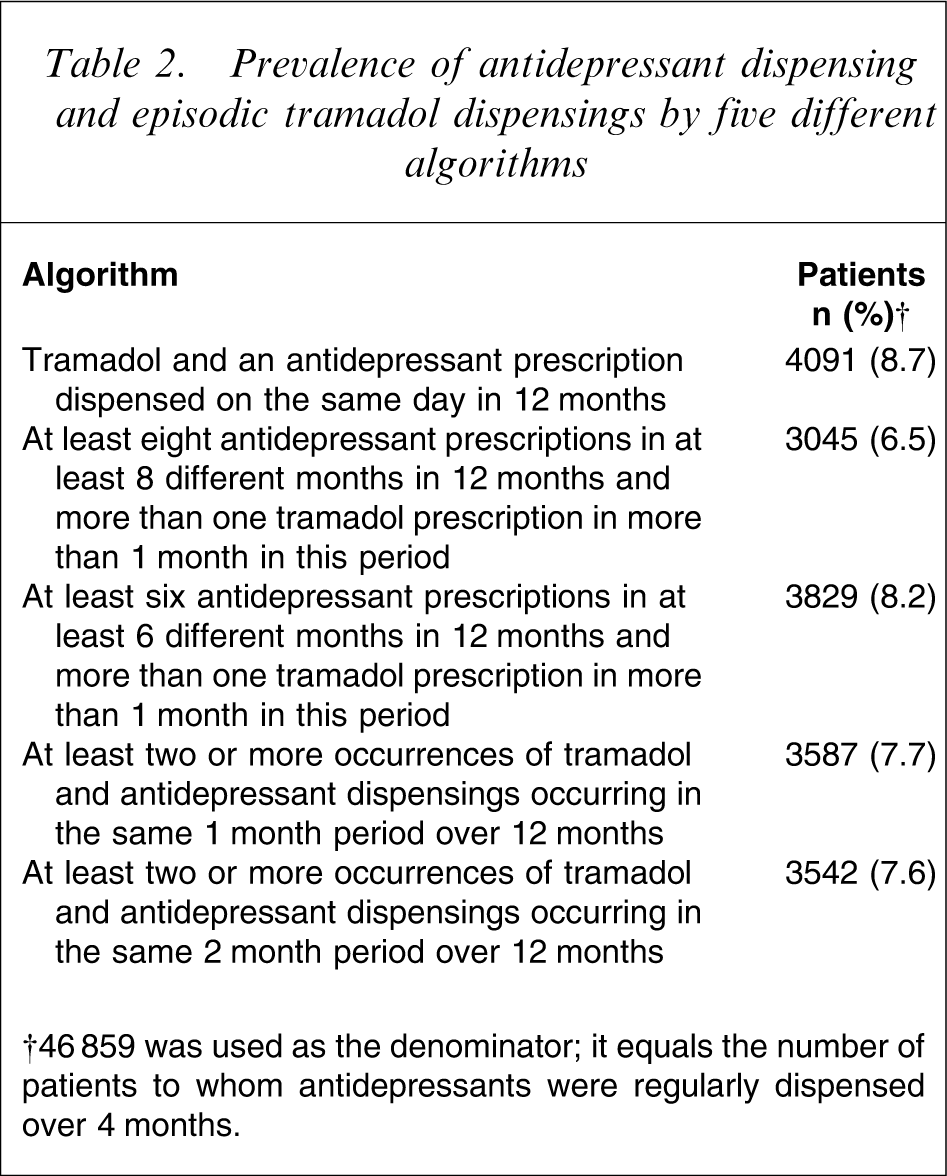
†46 859 was used as the denominator; it equals the number of patients to whom antidepressants were regularly dispensed over 4 months.
Discussion
The present study demonstrates that co-prescribing of medicines with antidepressants that have the potential for interaction and are avoidable is common, occurring in 8% of elderly, regular antidepressant users. Care was taken in designing the present study to exclude all potential cases of switching, by excluding those who had a single dispensing of a medicine. This is likely to have underestimated the prevalence of potential antidepressant interactions. Individuals who had a single dispensing of the interacting medicine may have suffered the consequences, but would not be detected in the dataset. Some individuals who were on both medicines concurrently may have fallen outside the dispensing windows chosen for the study, but again this would be most likely to underestimate the prevalence of the interactions. It should also be noted that this study did not assess the other potentially significant drug interactions with antidepressants that can be managed with dosage adjustments, such as benzodiazepine and antidepressant co-administration, and antipsychotic and antidepressant co-administration. Thus, the overall prevalence of potential antidepressant drug interactions may be much higher.
It would appear that duplicate antidepressant use has declined since the study by McManus et al. [4]. However, the potentially high level of tramadol and antidepressant co-prescribing is noteworthy and recent, given the introduction of this drug in 1998. These interactions increase the risk of serotonin syndrome, which may occur with a high dose of a single drug, but appears more common when serotonergic agents are used together. Serotonin syndrome results from excessive central nervous system and peripheral serotonergic activity. It includes a range of symptoms including changes in mental state, such as confusion, agitation and hypomania. Other symptoms include sweating, diarrhoea, fever, shivering, hypertension, as well as myoclonus, tremor and difficulties with coordination. It can be fatal, although it is reversible if recognized early and suspected contributing medicines ceased quickly [8]. We could not locate any studies providing an estimated incidence of serotonin syndrome from tramadol–antidepressant co-prescribing. The estimated incidence from antidepressants alone is between 0.5 and 1 per 1000 patient months of treatment [9]. A recent case-series also suggests that the interaction of serotonergic antidepressants and opioid analgesics may be unrecognized, particularly for elderly residents of long-term care facilities [10]. Twenty cases of serotonin syndrome associated with tramadol have been reported to Australia's adverse drug reaction advisory committee and in 16 of these, antidepressants or St Johns Wort were co-administered [11]. It would appear that prescribers still need to be cautious in their selection of appropriate analgesia for elderly patients on antidepressant therapy, because other narcotic analgesics that do not contribute to this syndrome are available.
The interaction between TCAs and anticholinergics should be well known. Anticholinergic toxicity is characterized by dry mouth, blurred vision, urinary retention, constipation, mydriasis and dry eyes. It can also cause confusion, memory impairment and hallucinations [8]. The introduction of tiotropium to the Australian market in 2002, an inhaled anticholinergic agent used to treat chronic obstructive pulmonary disease, appears to have contributed to an increased prevalence of interactions between TCAs and anticholinergics. Tiotropium was identified as the interacting anticholinergic agent in >50% of the anticholinergic interactions. Because tiotropium is an inhaled medicine, prescribers may not be aware of its potential for anticholinergic effects, which are significantly greater than ipratropium, the therapy that tiotropium most commonly replaces. A Cochrane meta-analysis of tiotropium for chronic obstructive pulmonary disease found that there was a fivefold increase in the likelihood of dry mouth from tiotropium compared to placebo, and a twofold increase compared to ipratropium [12]. Other anticholinergic side-effects were not reported in the meta-analysis. However, this finding does suggest an increased likelihood of adverse events for the elderly veteran population taking both TCAs and tiotropium, a population who would be particularly vulnerable to anticholinergic toxicity and in whom it may be unrecognized.
The major limitation of the present study is the lack of an assessment of severity or consequence of the interaction. It is unknown how many of these patients were suffering an adverse drug event due to the potential interaction. Despite this, these findings are relevant given the avoidable nature of the interactions assessed. Some insight into the potential for adverse events is gained from the German Drug Safety Program in psychiatry, which found a severe adverse drug reaction rate of 1.4% in patients on antidepressants [13]. Toxic delirium and urinary retention occurred in 0.17% and 0.1%, respectively, of patients taking TCAs. These reactions are associated with the anticholinergic properties of TCAs and that study found that >50% of these adverse drug reactions were due to TCAs interacting with another medicine [13]. Further, serotonin syndrome was reported in 0.04% of SSRI users, with drug interactions accounting for approximately one-quarter of these cases [13].
The results of the present study highlight the ongoing need for monitoring and prevention of drug interactions. All of the potential interactions detected in the present study were considered avoidable because safer alternative therapies were available, either by switching the antidepressant or switching to an alternate non-interacting medicine. In addition, practically all medical practitioners and pharmacies in Australia use electronic prescribing and dispensing software that include drug interaction alerts. Despite these systems, the prevalence of drug interactions remains high. The increasing use of antidepressants, particularly in the elderly, and the results of the present study highlight the necessity of ongoing vigilance, particularly where drug combinations may be avoided.
