Abstract
In recent years, an increasing body of evidence has shown that there are changes in the immune system in psychiatric diseases, especially in depressive disorder. For example, some investigators have found that patients with depressive disorder have abnormalities of immune function [1, 2], and changes in cytokine levels [3, 4]. After Smith proposed the macrophage theory of depression in 1991 [5], and that many cytokines could be produced by macrophage, many studies have focused on the relationship between circumstantial cytokines and depression. These studies demonstrated that major depression is associated with dysregulation of immune mediators, such as the rise in interleukin (IL)-6, tumour necrosis factor-α (TNF-α), IL-1β, IL-2, soluble IL-2R,C-reactive protein and interferon-γ (IFN-γ) [6, 7]. In depressed patients, studies concerning TNF-α levels have consistently indicated that patients with depression have a high level of TNF-α in serum or plasma. However, so far, inconsistent results have been reported for cytokine levels of IL-6 and IL-1β in depression. Unaltered [8–10], increased [11–13], or decreased [14, 15] IL-1β level have been reported in depressed patients compared with normal controls. Similarly, there also have been some studies on IL-6 level in depression, unaltered [3], increased [12, 13, 16], or decreased [17]. Moreover, the reasons for the discrepancy are not known.
Recently it was proposed that leptin plays an important role in major depression, given that weight loss and reduced appetite are typical symptoms of depression. Leptin is a protein synthesized in the adipose tissue and is the product of the obese gene. Leptin is believed to be a messenger from adipose tissue to the brain, and acts by binding to specific receptors in the hypothalamus, decreasing food intake and increasing energy expenditure [18]. Some studies concerning leptin have shown abnormalities of leptin levels in depression. So far, inconsistent results on leptin levels in major depression have been reported, such as unaltered [19], increased [20], decreased [21] or increased only in women [22] and so on.
According to the aforementioned results, cytokines and leptin are involved in the pathophysiology of depression. However, it is difficult to discern whether there are direct effects of cytokines on leptin. To date, little is known about the regulation of adipose tissue production and secretion of IL-6. The clinical significance of adipose-derived IL-6 as well as the relationship between leptin and IL-6 has not yet been well identified, although some studies suggest such a link with leptin from the aspect of TNF-α. Additionally, few clinical studies, till now, have examined the relationship between leptin and immune activity in humans; and the association between leptin and immune function in patients with depression is also not well established. Thus, the aims of the study are: (i) to assess the serum levels of IL-6, IL-1β, TNF-α and leptin in depression, (ii) to clarify whether cytokines are associated with leptin in depression; and (iii) to compare IL-6, IL-1β, TNF-α and leptin levels between male and female study subjects.
Methods
Subjects
A total of 56 subjects participated in the present study, and all subjects provided informed consent. Complete physical, neurological, and laboratory examinations were performed to show that all the subjects were free of significant physical or neurological illnesses, including any acute or chronic infectious, inflammatory, and immune disorders. All subjects agreed to remain free of aspirin and non-steroidal anti-inflammatory drugs (NSAIDs) for at least 2 weeks, and of therapeutic corticosteroids for at least 6 months, prior to testing. Thirty-three subjects (nine male, 24 female; mean age±SE = 42.12±2.27 years; age range = 18–58 years) met DSM-IV criteria for a principal diagnosis of depression and had a 17-item Hamilton Depression Rating Scale (HDRS) total score =20, all of whom were inpatients of the mental hospital of He Nan province (Second Affiliated Hospital of Xinxiang Medical college). Diagnoses and HDRS and Hamilton Anxiety Rating Scale (HARS) evaluation were carried out by two experienced research psychiatrists at vice professor or higher rank through semi-structured interviews in conjunction with all other available medical records. Patients who had been examined and given the same depression diagnoses by the two experienced psychiatrists were recruited into the study.
Patients with schizophrenia or other psychoses, organic mental disorders, substance dependence, obsessive–compulsive disorder, or any other primary psychiatric disorder were excluded. At the time of testing, all subjects were free of major psychotropic drugs for at least 4 weeks (free of fluoxetine for at least 5 weeks). Low-dose benzodiazepines were available for severe agitation and insomnia.
Twenty-three healthy control subjects (seven male, 16 female; mean age±SE = 38.39±1.76 years; age range = 27–58 years) were recruited from the hospital of Rongkang in Luoyang city, all of whom were employees in the hospital. In addition to the medical screening described here, they were also screened for a personal and family (first-degree relative) history of mental disorder by any research psychiatrist using a semi-structured interview, they did not have any disease. Moreover, their HDRS total scores, which were assessed by research psychiatrists, were all under 7.
Procedure
Investigations were carried out on the day after hospital admission. The severity of depressive symptoms was rated according to the Chinese version of the HDRS [23]. The patients were also evaluated with the HARS [23].
Fasting venous blood (5 mL) was withdrawn from ulnar vein with a sterile vacuum tube without additives between 7:00 and 7:30 hours. Serum was separated after being placed for approximately 1 h and stored at under −70°C for future analysis. Once all samples had been collected, the measurements were then done.
Determination of IL-6, IL-1β and TNF-α
IL-6, IL-1β and TNF-α concentrations were determined with enzyme-linked immunosorbent assay (ELISA). We used ELISA kits from Beijing HY-Taik Biomedicine Technique (Sigma, USA) for IL-6, IL-1β and TNF-α (detection range 1–400 pg mL−1). Recombinant cytokines were used as standards. All assays were carried out by the same operator in the same instrument using the recommended buffers, diluents, and substrates. For IL-6, IL-1β and TNF-α, the inter-assay and intra-assay coefficients of variation were 5% and 7.5%, respectively. The serum cytokine assays were carried out in the central laboratory of the Second Affiliated Hospital in Xinxiang Medical College (the mental hospital of Henan province).
Determination of serum leptin
Serum leptin levels were measured using radioimmunoassay kits from Shenzhen Jingmei Biomedicine Technique (Linco research, USA) for leptin (detection range 1–2000 pg mL−1). The inter-assay and intra -assay coefficients of variation were 4.1% and 6.5%, respectively. All assays in patients and controls were carried out at the same time and in the same run by the same operator. The serum leptin assays were completed in the radio-immunity center of Rongkang Hospital in Luoyang city.
Statistical analysis
SPSS for Windows 11.5 was used to analyse the collected data. Data were generally reported as mean±SE. Student's t-test and the χ2 test were applied for comparisons of some demographic data, serum IL-6, IL-1β, TNF-α and leptin values between normal controls and patients. Relationships between the cytokines and leptin levels and clinical variables were evaluated using Pearson's and Spearman's correlation. The level of significance was set at p < 0.05.
Results
Demographic data
Table 1 lists the demographic data and main characteristics of the 56 study subjects. There were no significant differences in the male/female ratio between patients with depressive disorder and normal controls (χ2
Demographic data
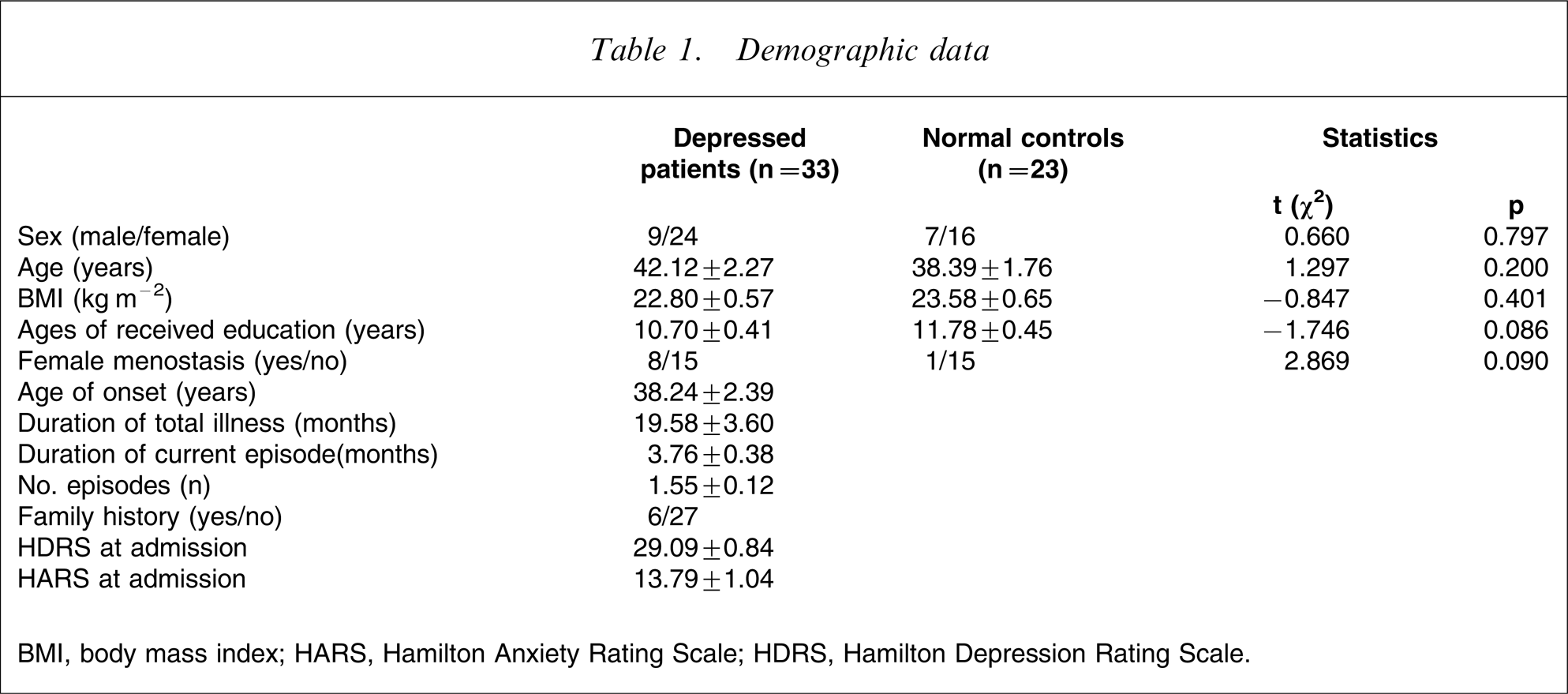
BMI, body mass index; HARS, Hamilton Anxiety Rating Scale; HDRS, Hamilton Depression Rating Scale.
Comparison of serum IL-6, IL-1β,TNF-α and leptin levels in depressive disorder patients and normal controls
All the 33 enrolled patients were included in this part of the study. According to Figure 1, there were no significant differences in IL-1β levels between the depressed patients and the normal controls (t = 0.037, p = 0.971 p > 0.05), but significant differences in IL-6 and TNF-α levels appeared between the two groups (t = 2.333, p = 0.024, t = 2.434, p = 0.019, both p < 0.05), moreover, IL-6 and TNF-α levels were both higher in patients than in controls. In addition, there were significant differences in leptin levels between the two groups (t = − 2.165, p = 0.035, p < 0.05), and leptin levels were lower in patients than in controls.
Comparison of serum interleukin (IL)-6, IL-1β, tumour necrosis factor-α (TNF-α) and leptin levels in the depressive disorder patients and the normal controls. Statistical significance of changes: ∗p < 0.05.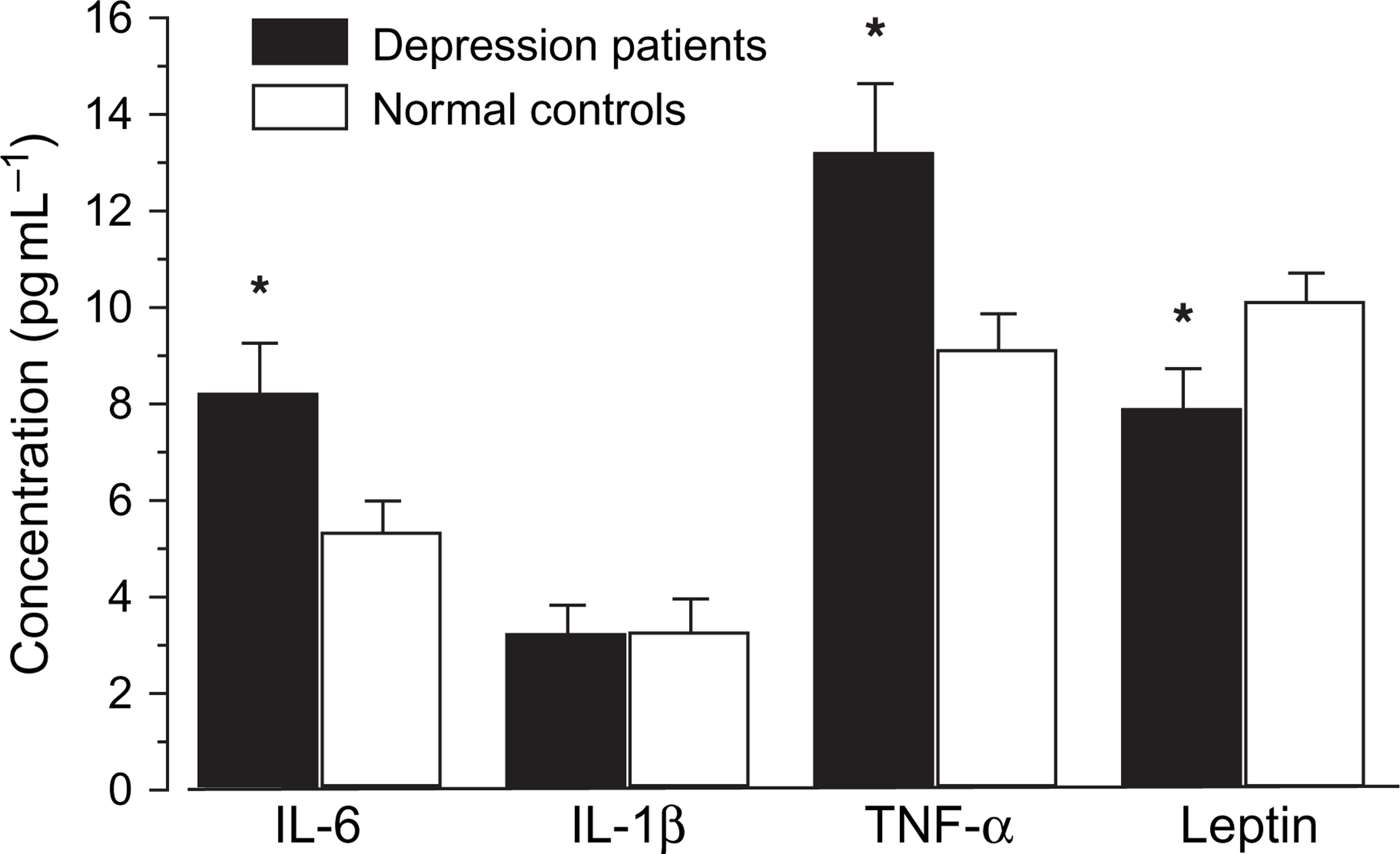
Gender difference in serum levels of IL-6, IL-1β, TNF-α and leptin between depressed patients and normal controls
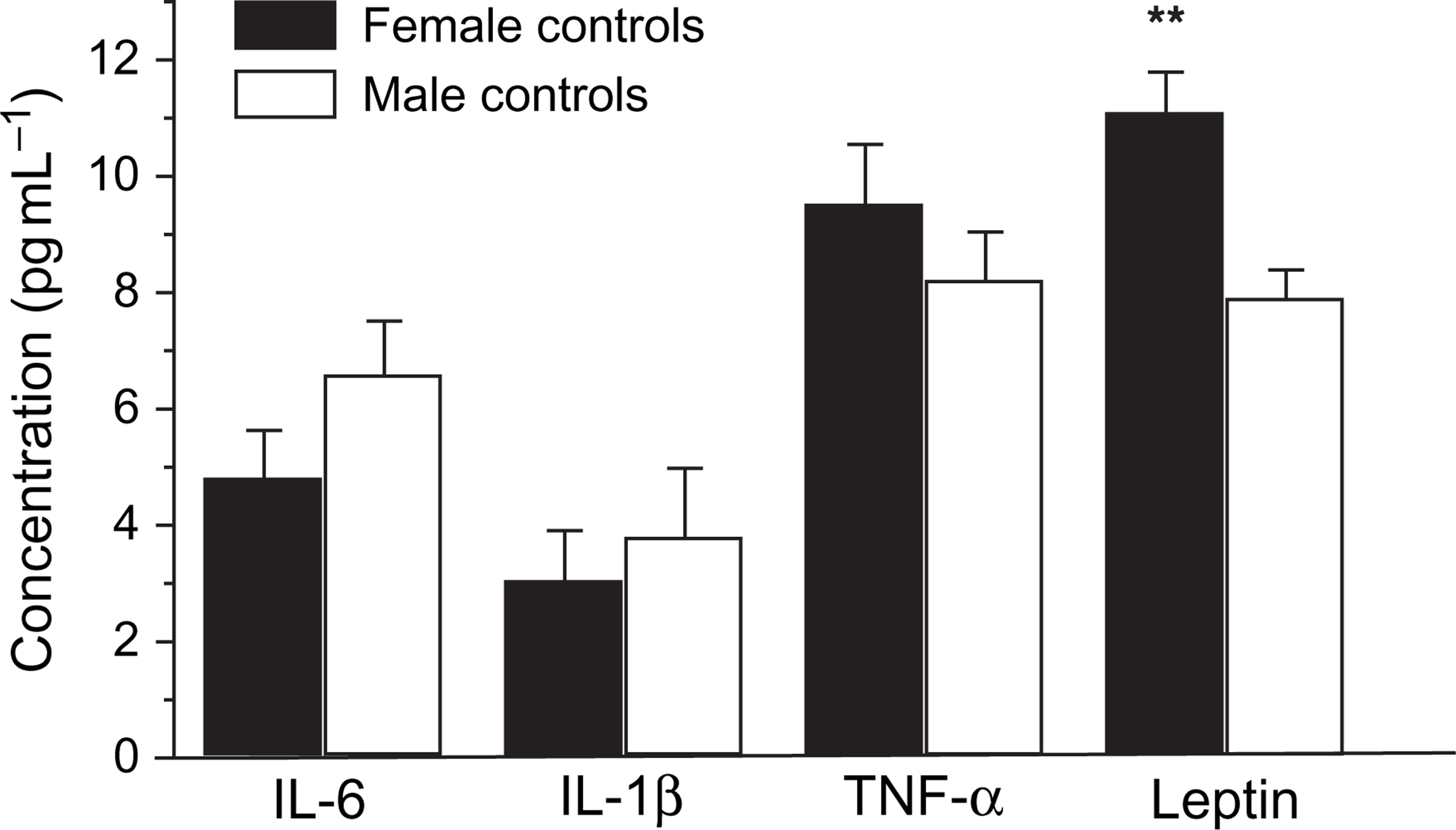
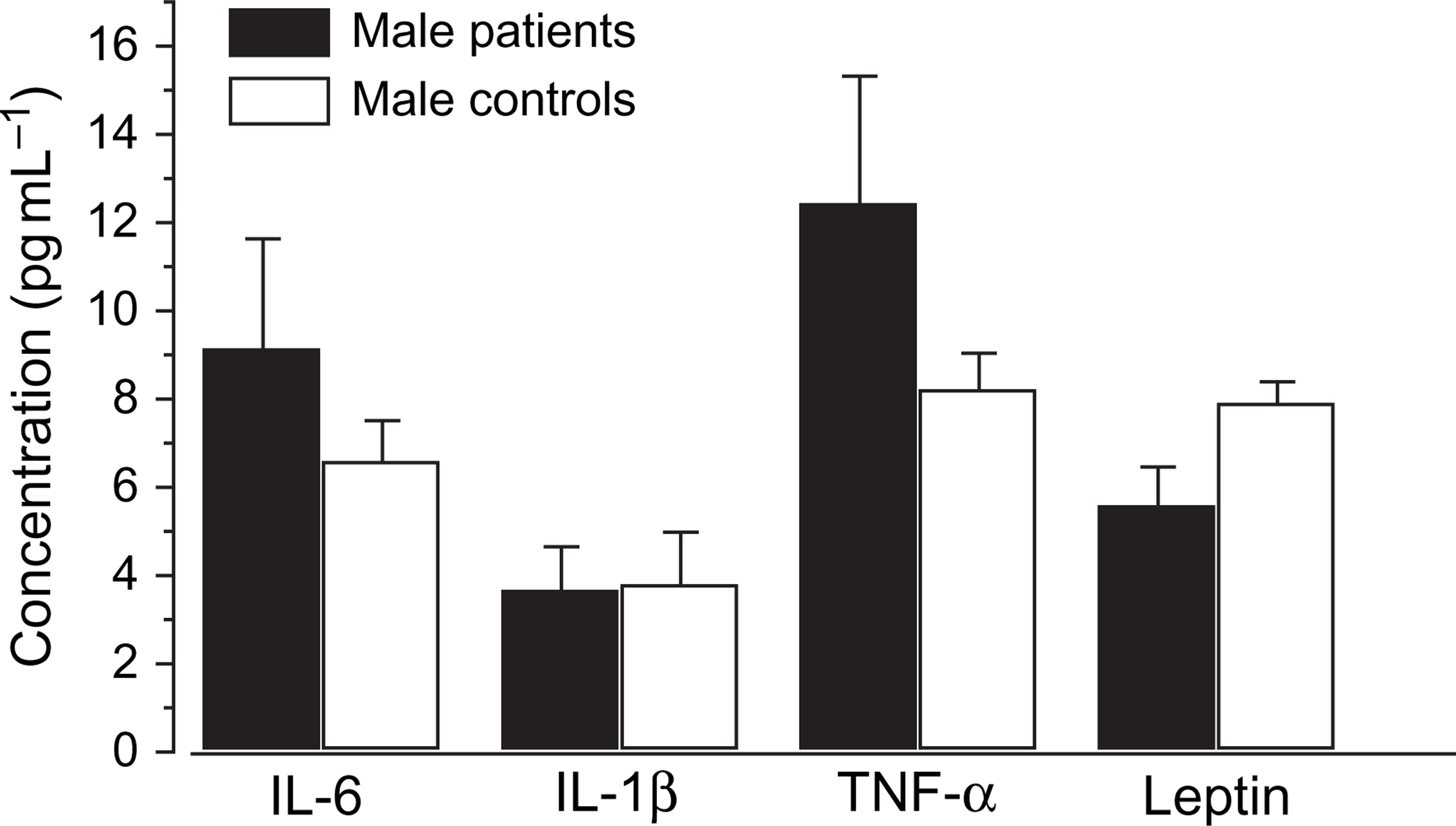
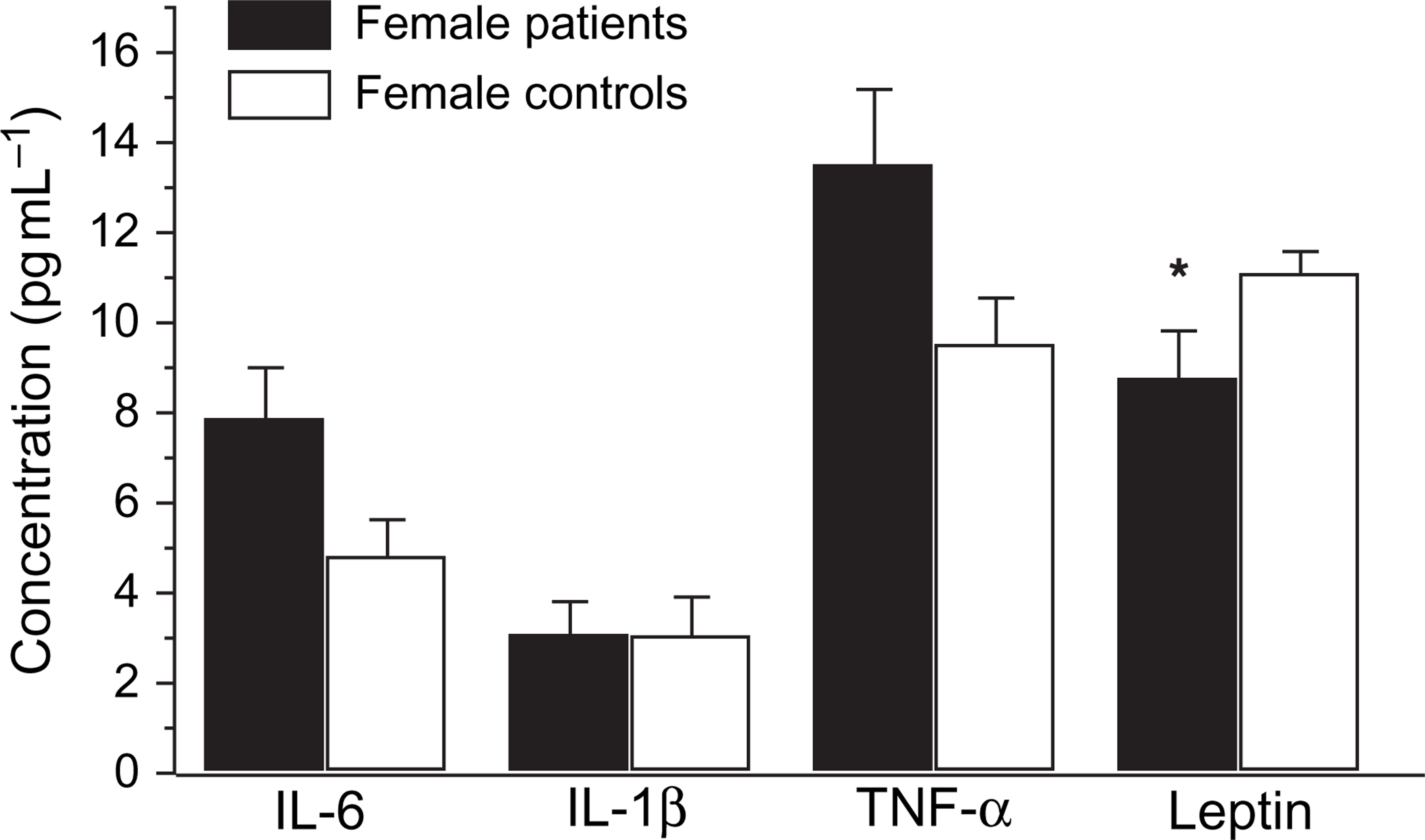
The comparison of cytokines and leptin levels in the male and female patients with corresponding controls were analysed using independent sample T test. There were no significant differences in the cytokine (IL-6, IL-1β and TNF-α) levels between the male and female depressed patients, and no significant difference between male and female normal controls. However, significant differences were found in the leptin levels between male and female patients (Figure 2). Similarly, significant differences appeared between male and female controls (Figure 3). Leptin levels were higher in female than in male patients, and higher in female than in male normal controls. Nevertheless, no significant differences in the cytokines and leptin levels occurred between the male patients and the male controls (Figure 4), or between female patients and female normal controls (Figure 5).
Gender difference in serum levels of interleukin (IL)-6, IL-1β, tumour necrosis factor-α (TNF-α) and leptin between female and male depressed patients Statistical significance of changes: ∗p < 0.05. Gender difference in serum levels of interleukin (IL)-6, IL-1β, tumour necrosis factor-α (TNF-α) and leptin between female and male normal controls. Statistical significance of changes: ∗∗p < 0.01. Gender difference in serum levels of interleukin (IL)-6, IL-1β, tumour necrosis factor-α (TNF-α) and leptin between male patients and male normal controls Gender difference in serum levels of interleukin (IL)-6, IL-1β, tumour necrosis factor-α (TNF-α) and leptin between female patients and female normal controls. Statistical significance of changes: ∗p < 0.05.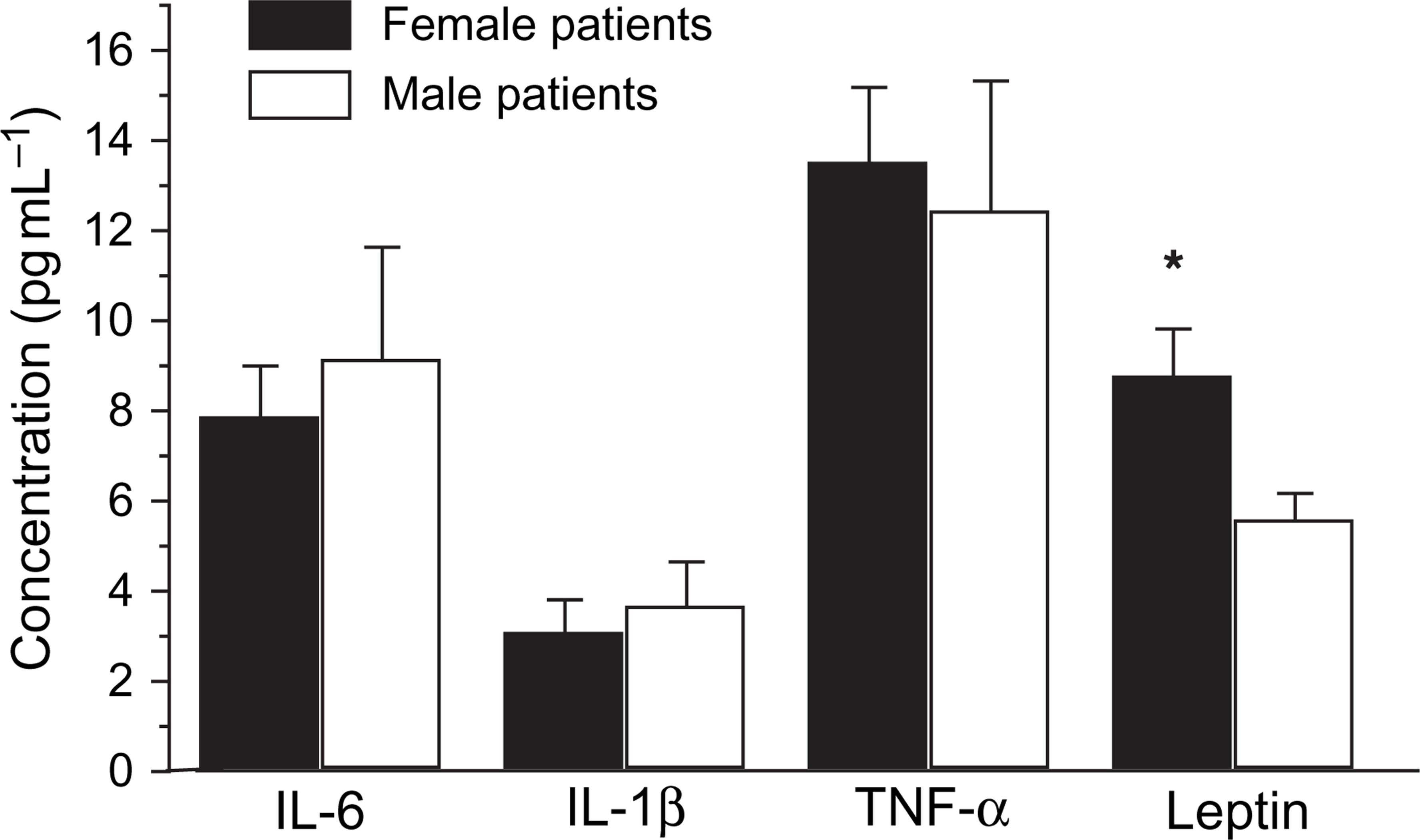
Correlation between cytokines, BMI, and leptin levels and severity of depressive symptoms in depression
We found there were significant correlations between leptin and BMI in depression (r = 0.441, p = 0.010), but leptin levels were not related significantly with BMI in the normal controls (r = − 0.028, p = 0.898). We also found no significant association between leptin and cytokine levels (IL-6, IL-1β and TNF-α), with the coefficients 0.125, 0.335, 0.071 respectively, and p = 0.488, 0.057, 0.676, respectively, in depression. The correlations of IL-6, TNF-α, leptin levels and severity of depressive symptoms were as follows: r = − 0.01, p = 0.958; r = 0.072, p = 0.689; r = − 0.470, p = 0.023, respectively.
Discussion
The main findings of our study were as follows: (i) depressed patients had a high pro-inflammatory cytokine activity; moreover, depressed patients had a higher level than normal controls in terms of serum IL-6 and TNF-α cytokine, while they had the same serum IL-1β cytokine levels as the normal controls; (ii) there were significant differences in the leptin levels between the patients and the controls, and the patients’ leptin levels were much lower than the controls’; (iii) there were significant differences in the leptin levels both between the male and the female patients, and between male and female controls; moreover, the leptin levels were higher in the female than in the male subjects, regardless of whether they were patients or normal controls; and (iv) there was no correlation between IL-6, IL-1β, TNF-α and leptin level in the depressive patients.
The different scientific and methodological approaches, such as the type of quantification of assay and psychopathological response, leading to a variety of results, are not completely conclusive in regard to the clinical course of the disease. Despite this, the majority of studies suggest that an activation of immunological parameters may, indeed, be relevant for the outcome of depressive illness. As far as the issues of methodology are concerned, to minimize the interfering factors, we selected sterile vacuum tubes without additives and the tube with the same batch number in the course of collecting serum samples, so as to avoid endotoxin contamination and to minimize endotoxin. Moreover, all the assays performed on patient and control serum were carried out at the same time and in the same run by the same operator in order to decrease technical variance.
The role that cytokines play in depression has been well documented [12, 24–26], and it has been hypothesized that cytokine hypersecretion might be involved in the pathophysiology of depressive disorders [27]. Our findings that the IL-6, TNF-α cytokine levels in the patients were higher than those in normal controls were in agreement with the previous findings. This demonstrates that depressive disorder is associated with enhanced secretion of the pro-inflammatory cytokines IL-6 and TNF-α. These phenomena may, in part, be explained by the increased activity of immunosuppressive factors, which are produced during the acute phase response and which are frequently increased in major depression, for example, serum cytokines, such as IL-6, TNF-α. Currently there is no evidence for a causal relationship between the presence of an acute phase response (with increased IL-6 production or activity) and the aetiology of depression. However, no significant changes in IL-1β were detected in the present study, compared with previous studies, which could be due to the different methodologies used in the studies. For example, in previous studies, stimulated whole blood and plasma samples were used to analyse cytokine concentrations, whereas serum samples were used in the present study. Gender difference in inflammatory response in different illness conditions has been reported [28, 29]. Moreover, it is also reported in an epidemiological study that age and gender significantly affect plasma cytokine levels in depressed and schizophrenia patients [30]. In contrast, we found no significant differences in gender between depressive disorder and healthy controls in the present study. This could be due to the small sample size used in the present study and that minor depressive disorder was not excluded, while in previous studies only patients with major depression were enrolled. Therefore, the present finding is still in agreement with the hypothesis that IL-6, TNF-α cytokines could be important in the pathophysiology of depression.
In the present study, in serum samples, leptin levels were lower in the depressed patients than in the healthy controls. The result was concordant with previous studies [21, 31]. There is an ambiguity about the effects of depression on leptin levels in the literature. Deuschle et al. reported that leptin plasma concentrations did not differ between the depressed patients and the healthy controls [19], while another study reported that patients with depression had higher nocturnal serum leptin levels [20], and Kraus et al. found that leptin levels were significantly lower in both depression and schizophrenia compared with healthy controls [21]. But Esel et al. reported that leptin levels were higher in depressed patients than normal controls [32]. The reason for this discrepancy may lie in the differences in the clinical features of the patient groups or in the time of blood sampling for leptin.
One of the present findings was that decreased leptin levels were observed in depression patients compared with normal controls. Although a decrease in leptin levels was observed in depressive women compared with healthy women, we found no significant difference between the two groups. In addition, that the severity of the depression was negatively correlated with the leptin levels suggested that depression should have some effects on serum leptin decrease. Therefore, it would be expected that reduced food intake and subsequent weight loss in depression might be associated with decreased leptin levels [32]. However, it has been shown that serum leptin is not significantly correlated with HDRS depression score [32]. This differs from the present study, and at present this discrepancy cannot be explained.
It has been reported that decreased leptin production correlated with the cytokines of inflammatory responses, particularly cytokines IL-6, and TNF-α [34, 35]. IL-6 is a circulating pro-inflammatory cytokine secreted during an acute-phase response by numerous immune cells, including lymphocytes and macrophages. Unlike other cytokines, IL-6 is produced during non-inflammatory conditions in significant amounts by adipose tissue [36, 37]. Like leptin, both serum and adipose IL-6 concentrations are positively correlated with BMI and decrease after weight loss [38]. Baumann et al. concluded that the long form of the leptin receptor exerts signalling actions similar to that of IL-6-type cytokine receptors, including activation of several signal transducers and activators of transcription (STAT proteins) [39]. Limited published studies have provided inconsistent findings regarding the relationship between leptin and IL-6. TNF-α is an acute-phase cytokine, primarily secreted by macrophages in response to pathogens. It is also secreted by other type cells, including adipocytes and T-helper 1 (Th1) cells [40]. The action of TNF-α is believed to be mediated by a complex network of TNF-α-induced cytokines and hormones, including IL-6 and leptin [41]. Leptin and TNF-α, both of which are produced in adipocytes and induce anorectic effects, share many similarities. Obesity is related to elevated expression of both TNF-α and leptin [42]. The results of the present study show that there were no significant correlation of leptin and IL-6, IL-1β and TNF-α. The inconsistent results may be because of the different kinds of research subjects studied; for instance, the previous studies involved healthy, obesity and cancer subjects, whereas the present study consisted of depressed patients and normal controls. Moreover, in the present study the subjects were not divided into subgroups according to BMI. The present study suggests that leptin levels were significantly correlated with BMI in depressive disorder. Nevertheless, in the normal controls we did not find a significant relationship between leptin levels and BMI. This could be explained by the fact that leptin is believed to be a messenger from adipose tissue to the brain, which acts by binding to specific receptors in the hypothalamus, and decreases food intake and increases energy expenditure [18].
Our study also found varying leptin levels due to gender difference both in the depression subjects and the healthy controls, with the leptin levels in female subjects being higher than in male subjects, regardless of whether they were from the patient or the normal control group. The present results are consistent with previous studies. For example, a sexual dimorphism in leptin levels has been consistently reported in that healthy women have higher leptin levels than men [43]. The reason for this gender inequity in terms of leptin remains to be elucidated. A greater amount of subcutaneous and intra-abdominal adipose tissue in women may be an important determinant of the sex difference in leptin concentration [22]. Furthermore, the differences in male and female eating behaviours or upregulated leptin mRNA in proportionally larger adipocytes of female subjects have been suggested for the sexual diergism [44]. Because there were similar results in the two groups, we cannot conclude that only different gender is responsible for difference in the leptin levels in depressed patients.
The present study has some limitations. Because we collected serum samples only in the beginning of therapy, some significant changes in cytokine and leptin levels could have been missed. Moreover, if we had also measured the cytokine and leptin levels in plasma and cerebrospinal fluid, we might have obtained a more comprehensive view regarding the changes. Although we measured the effect of paroxetine on cytokines and leptin levels, in the present study we did not carry out a comparison of cytokine and leptin level between before and after treatment. Also, the sample size was small.
In conclusion, the results of the present study have duplicated that of previous studies and have shown the importance of IL-6, TNF-α cytokine and leptin in the psychoimmunology of depressed patients. There were gender differences in leptin levels in depression. Further studies should be performed in which patients with depressive disorder should be independently divided into female and male subgroups. Moreover, samples sizes of future studies should be increased.
Footnotes
Acknowledgements
The authors thank all doctors and nurses who participated in our study in the Second Affiliated Hospital of Xinxiang Medical College for technical assistance. Moreover, we thank all patients and normal controls for their participation.
