Abstract
Study Design
Case report.
Objective
To report a case of remote cerebellar hemorrhage (RCH) without intraoperative dural tear after revision lumbar spine surgery. RCH is a rare postoperative complication following spine surgery. RCH has previously been reported only in cases with intraoperative dural tear or durotomy.
Methods
Case report and literature review.
Results
A 58-year-old woman underwent removal of L4–S1 posterior spinal instrumented fusion (PSIF) implants and L3–L4 decompressive laminectomy with PSIF. There was no intraoperative dural tear. After doing well initially, the patient developed new neurologic symptoms and was found to have RCH. Lumbar spine magnetic resonance imaging (MRI) demonstrated a large dural defect. After repair of the dura, the patient had dramatic improvement of her neurologic symptoms. At 1-year follow-up, the patient continued to have no neurologic sequelae.
Conclusion
This report demonstrates that RCH can occur without intraoperative dural tear. Although rare, any patient with new onset of declining neurologic symptoms following spine surgery should have a brain MRI and should have RCH on the differential diagnosis.
Introduction
Remote cerebellar hemorrhage (RCH) is a rare complication following spine surgery, and it can be associated with significant permanent disability. Previous cases of RCH after spine surgery have been reported in the setting of intraoperative dural tear or durotomy. 1 , 2 , 3 , 4 , 5 , 6 , 7 , 8 , 9 , 10 , 11 , 12 , 13 , 14 , 15 We present a rare case of RCH following revision lumbar spine surgery without an intraoperative dural tear.
Case Report
A 58-year-old woman after lumbar spine surgery presented with adjacent-level spinal stenosis and spondylolisthesis at L3–L4. She underwent L4–S1 decompressive laminectomies, L4–L5 and L5–S1 posterior lumbar interbody fusion, and posterior instrumented spinal fusion 8 years earlier. She underwent removal of implants with L3–L4 decompressive laminectomy and posterior instrumented spinal fusion. During the procedure, the scar overlying the thecal sac at L4–L5 was removed, but there was no intraoperative dural tear identified.
Postoperatively, the patient was asymptomatic, had decreasing drain output, and was able to stand with physical therapy. After an evening session with physical therapy on postoperative day 2, the patient developed headache and vomiting, and her drain output increased from 100 to 600 mL per shift. On postoperative day 3, she had persistent headache and vomiting, and she developed diplopia and dysarthria. Noncontrast brain magnetic resonance imaging (MRI) demonstrated a bilateral cerebellar parenchymal hemorrhage with mass effect and inferior displacement of the cerebellar tonsils (Fig. 1A, B). A lumbar spine MRI demonstrated a dural defect at the L4–L5 level (Fig. 2). The patient returned to the operating room for irrigation and debridement with repair of the 5-cm dural defect. Postoperatively, her dysarthria and diplopia improved, and the patient was discharged.
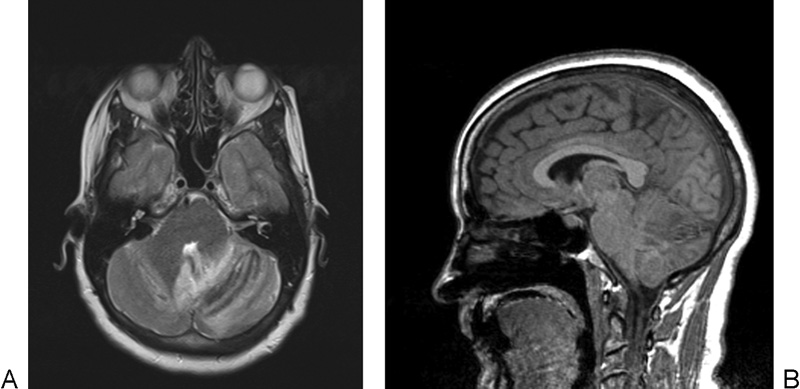
(A) Axial T2-weighted brain magnetic resonance imaging (MRI) demonstrating bilateral cerebellar hemorrhage, left greater than right. (B) Sagittal MPRAGE (magnetization-prepared rapid gradient-echo) brain MRI demonstrating inferior displacement of cerebellar tonsils and mass effect on the fourth ventricle.
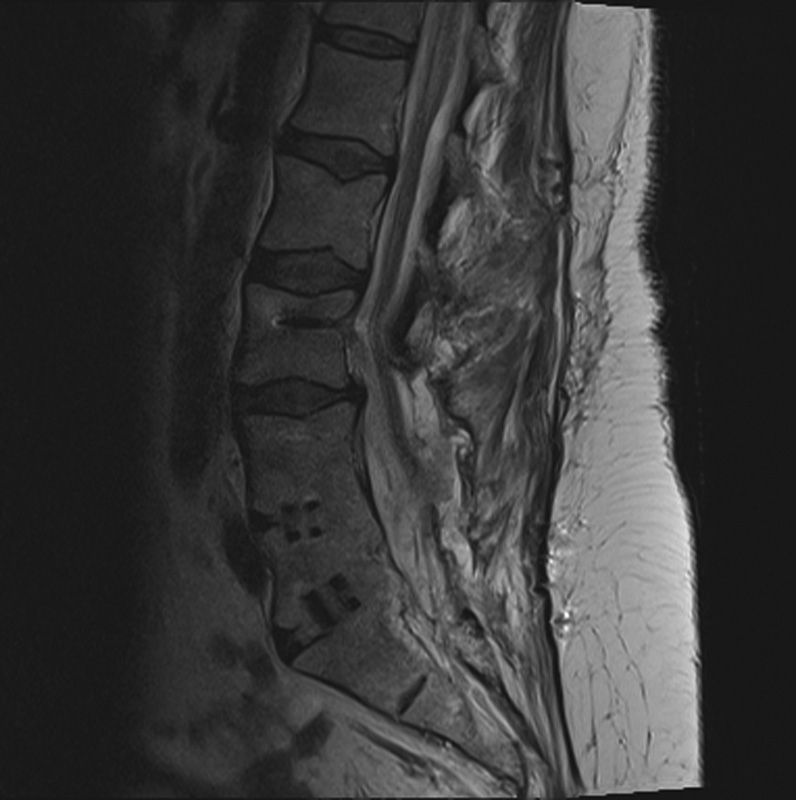
Sagittal T2 magnetic resonance imaging demonstrating fluid collection consistent with cerebrospinal fluid dorsal to the thecal sac at the L4–L5 level with large dural defect of ∼5 cm.
At 6-week follow-up, the patient continued to have some intermittent dizziness and visual changes, but her dysarthria had resolved. At 3 months postoperatively, the patient no longer had any residual neurologic sequelae. At 1 year postoperatively, the patient continued to have no residual sequelae, she had full strength in her lower extremities, and her initial back complaints had resolved.
Discussion
RCH remains a rare complication following spine surgery that should be considered for any patient undergoing an unexplained neurologic change after surgery.
The predominant theory on the development of RCH involves the cerebral venous system. It has been proposed that significant loss of cerebrospinal fluid could result in downward cerebellar displacement. This caudal cerebellar sag could lead to stretch or rupture of the bridging cerebellar veins causing hemorrhagic infarction or direct hemorrhage. 7 , 14 , 16 , 17 This theory is supported by most cases, including the current case, reporting bilateral cerebellar hemorrhage; an arterial bleed would typically be unilateral. Additionally, the previous case reports have noted that the cerebellar hemorrhage is typically located in the upper vermis and cerebellar sulci where cerebellar venous drainage occurs. 7 The current case report supports this theory as the patient had bilateral cerebellar hemorrhage located in the upper vermis.
The prior cases of RCH after spine surgery have been in the setting of intraoperative durotomy or dural tear. 1 , 2 , 3 , 4 , 5 , 6 , 7 , 8 , 9 , 10 , 11 , 12 , 13 , 14 , 15 However, the current case had no intraoperative dural tear. Although it is entirely possible that there was an unnoticed intraoperative dural tear, the patient's hospital course does not support this theory. The patient had no symptoms initially after surgery. Our proposed theory is that during the revision surgery, removal of the overlying fibrous tissue to effectively decompress the thecal sac resulted in iatrogenic dural ectasia. During the therapy session, the patient may have exerted herself enough to rupture the thecal sac resulting in a change in the drainage output and appearance. The current case further supports the theory that RCH can occur following injury to the dura during spine surgery.
In several previous case reports of RCH after spine surgery, patients had residual neurologic deficits. 5 , 6 , 14 Residual neurologic sequelae have been thought to be related to the extent of bleeding, location within the cerebellum, presence of hemorrhage in other cerebral locations, amount of time between onset of symptoms and intervention, and presence of other complications. Our report demonstrates that complete neurologic recovery is possible following RCH and that full neurologic recovery may take several months.
RCH is a rare complication following spine surgery that can occur without intraoperative dural tear. RCH should remain on the differential diagnosis of any patient with new neurologic decline following spine surgery.
Disclosures
Justin M. Haller, none
Graham Calvert, none
William R. Spiker, none
Darrel S. Brodke, none
Brandon D. Lawrence, none
This case report is a helpful reminder of the frailty of patients with spine injuries in the perioperative period. Fortunately, persistent clinical vigilance and directed intervention avoided greater damages in this patient. Although we might find some reassurance in the apparent rarity of such a complication, the contiguous nature of the cerebrospinal space invites for some complications to present with symptoms away from the surgical site. The nature of some central nervous system symptoms may also be unfamiliar to some surgeons. As this report shows impressively, picking up on changes in neurologic status is clinically important for many reasons. For instance, it can be unclear when the described symptoms of headaches, vomiting, diplopia, and dysarthria are detected in a patient heavily medicated with opiates and muscle relaxants. Rapid postoperative mobilization where possible (an original Arbeitsgemeinschaft für Osteosynthesefragen principle first formulated in 1959) and increased efforts at minimizing oversedation (for instance, by applying multimodal pain management in patients after spine surgery) appear to be most desirable practices in the best interest of patient safety. There also seems to be a lingering uncertainty as to the management of persistent dural tears relative to postoperative management, a topic to perhaps explore in greater detail in the future. How long do we keep a patient with dural deficiency at bed rest, under what circumstances do we mobilize such a patient, what is the role of drains, and what form of troubleshooting interventions are effective?
